Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Recent immunohistochemical findings have assisted in the histogenetic classification of many of the soft tissue tumors. However, because vimentin is positive in many tumors, it is of no value in the differential diagnosis of tumors discussed in this chapter. On the other hand, the “fibrohistiocytic” tumors are still largely enigmatic with respect to their histogenesis, and the diagnosis is largely dependent on hematoxylin and eosin (H&E) inspection. In addition to the fibrohistiocytic tumors, this chapter includes those entities that have traditionally been grouped together on the basis of collagen production and/or the presence of fibroblasts or fibroblast-like cells forming an integral component of the tumor. It also includes tumors of presumptive origin from dermal dendrocytes and myofibroblasts.
Dermal dendrocytes are bone marrow–derived cells found in different parts of the dermis. They are closely related to mast cells in the perivascular space. They express factor XIIIa and von Willebrand factor receptor, suggesting a possible role in tissue repair and hemostasis. An antigen-processing function has also been proposed. The subset of dendrocytes expressing factor XIIIa is found in some of the acral angiofibromas and in dermatofibromas. They are also increased in many other situations. Another subset of dendrocytes, comprising 10% to 30% of all interstitial cells in the reticular dermis, expresses CD34. This antigen is expressed in vascular endothelial cells, some perivascular and interstitial dendritic cells in the dermis (as mentioned previously), and spindle cells in the basement membrane zone of eccrine ducts and the bulge area of the hair follicle. It is also expressed in a wide range of tumors. The two subsets of dendrocytes appear to interact in many situations. A group of CD34 + tumors that could loosely be categorized as CD34 + dendrocytomas is discussed next.
A hamartoma composed of CD34 + cells was reported in 1995 as a “ dermal dendrocyte hamartoma. ” It presented in a 1-week-old infant as a deep red pedunculated nodule, soft with fine wrinkles and white, stubby hairs on the surface. Three congenital lesions reported as “ medallion-like dermal dendrocyte hamartoma ” were reported in 2004 by Rodriguez-Jurado et al. There have now been 14 papers published about similar lesions, usually referred to as “medallion-like” but also described as polypoid or plaque-like. They have been be either congenital or acquired, and typically present as slow-growing, flat, well-demarcated plaques that can be indurated and of erythematous to brownish color. There is no particular site predilection, having been reported in the head and neck, trunk and extremities. They have a benign clinical course and usually do not recur once removed. Kutzner et al. concluded that this lesion is of fibroblastic rather than dendrocytic lineage, and they prefer the descriptive term plaque-like CD34 + dermal fibroma instead of medallion-like dermal dendrocytic hamartoma . Other variants of this lesion include a granular cell type, presenting as multiple papules and plaques in an infant and called granular cell dendrocytosis , a posttraumatic myxoid tumor of the thumb called a myxoid dermatofibrohistiocytoma , a CD34 + eruptive fibroma , consisting of multiple papules composed of CD34 + spindle cells on the neck and upper chest of a female teenager, and a CD34-reactive myxoid dermal dendrocytoma that presented as a flesh-colored papule on the palm of a 66-year-old woman.
The connecting link among all of these lesions is the predominance of CD34 + cells, which have been described as fibroblast-like spindled to dendritic cells with elongated nuclei, inconspicuous nucleoli, and little or no mitotic activity. The arrangements of these cells can be loosely storiform or fascicular, sometimes with slight zonation, with an orientation perpendicular to the epidermis in the superficial portions of the lesion and a storiform arrangement in deeper portions in plaque-like or medallion-like lesions ( Fig. 35.1 ), or in a thin, band-like horizontal array in the more hypocellular lesions. They sometimes concentrate in a perivascular, perineural, and/or periadnexal array, though this was not the case in the CD34 + eruptive fibroma. The subcutis is not generally involved, nor is fat-entrapment identified. Other specific findings seen in individual cases have included short, malformed follicles, some resembling primary epithelial germ in dermal dendrocytic hamartoma, a richly vascularized stroma in the plaque-like or medallion variety, granular cells with CD34 staining of the cell membranes and cytoplasm between the granules, and a myxoid stroma. The latter two reported lesions were both acral, and there are enough similarities to suggest that these lesions may represent variants of superficial acral fibromyxoma or cellular digital fibroma (see later).
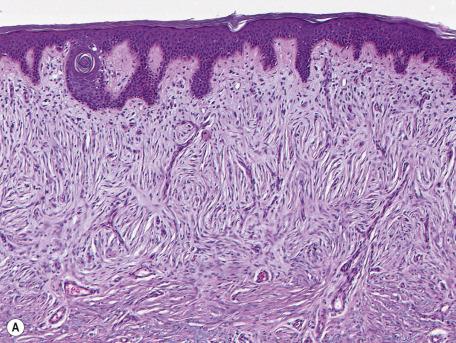
Immunohistochemically, in addition to CD34 expression, these cells are positive for fascin and vimentin, Factor XIIIa expression has been either negative or “inconsistent.” However, factor XIIIa + cells may be present in papillary dermal dendrocytes, and in the case of myxoid dermatofibrohistiocytoma, factor XIIIa + cells with elongated dendritic processes surrounded the negatively staining tumor cells. Tumor cells are negative for S100, smooth muscle action, MAC387, HHF-35, myelin basic protein, muscle-specific action, HMB45 or CD68.
Ultrastructural study of the dermal dendrocyte hamartoma of infancy with malformed hair follicles showed spindle shaped mesenchymal cells with 2 to 3 elongated dendrites, elongated and thinned nuclei with prominent heterochromatin and dark pyknotic euchromatin, and cytoplasm with well-developed rough endoplasmic reticulum. No dense bodies, aggregates of microfilaments, or basal lamina material was identified around the spindle cells. The case of granular cell dendrocytosis showed cells with numerous phagolysosomes containing variously sized and shaped electron-dense granules surrounded by halos, with rare osmiophilic fat vacuoles. Pseudopod-like projections were detected in granular cells lacking basement membranes.
The main differential diagnostic consideration is superficial, plaque-like dermatofibrosarcoma protuberans (DFSP). Although there are some morphological differences, definitive differentiation can be achieved by using reverse transcriptase–polymerase chain reaction (RT-PCR) or fluorescence in situ hybridization (FISH) analysis, the latter being preferred for archival formalin-fixed, paraffin-embedded material (see Dermatofibrosarcoma Protuberans , Differential Diagnosis , p. 1052 ). Fibroblastic connective tissue nevus involves the entire dermis with entrapped appendages and adipocytes; CD34 expression is patchy and weak. Dermatomyofibroma is CD34 negative and shows horizontally arranged bundles of slender myofibroblasts that may be positive for smooth muscle actin. Children with adenosine deaminase–deficient severe combined immunodeficiency have developed lesions resembling plaque-like CD34 + dermal fibroma, though they are relatively hypocellular and they regularly display the COL1A1–PDGFB fusion characteristic of DFSP (see later).
Tumors of myofibroblasts are also considered in this chapter. They often coexist with ordinary fibroblasts in many of the lesions. There is still a lack of consensus regarding their exact role in the formation of the soft tissue tumors usually attributed to them. It has even been suggested that myofibroblasts are merely a functional stage of fibroblasts, smooth muscle cells, or pericytes, and that their identification requires the ultrastructural recognition of the so-called fibronexus (microtendons). In addition to an origin from fibroblasts, myofibroblasts have been derived experimentally from microvascular endothelial cells by the action of inflammatory cytokines. Neoplastic myofibroblasts express vimentin, muscle actin, α smooth muscle actin, and/or desmin, although the specificity of desmin has been questioned. Myofibroblasts are best defined ultrastructurally because their immunohistochemical profile is not specific.
Various attempts have been made to simplify this difficult subject, including an algorithmic approach based on colors and the application of molecular genetics.
Acral angiofibromas are a clinically diverse group of entities that share distinctive histological features. They are thought by some to represent hyperplasias of the papillary and/or periadnexal dermis (the adventitial dermis). Immunohistochemical studies have shown that the large stellate fibroblast-like cells that characterize these tumors express factor XIIIa. They are not mesenchymally derived fibroblasts. Factor XIIIa appears to be important in the promulgation of fibroplasia.
Tumors derived from the perifollicular mesenchyme—the perifollicular fibroma, trichodiscoma, and fibrofolliculoma—are usually considered separately from the acral angiofibromas. They are discussed with the tumors of the hair follicle (see p. 969 ).
The following clinical conditions are discussed:
Adenoma sebaceum (tuberous sclerosis)
Angiofibromas in other syndromes
Fibrous papule of the face
Pearly penile papules
Acral fibrokeratoma
Familial myxovascular fibroma.
The entity reported as linear papular ectodermal–mesodermal hamartoma has some features of this group.
“Adenoma sebaceum” is the misnomer (there is no adenomatous proliferation of sebaceous glands as the name implies) used for the angiofibromatous lesions found in most patients with tuberous sclerosis (OMIM 191100 ), an autosomal dominant neurocutaneous syndrome in which learning disability and epilepsy are often present. Major reviews of the tuberous sclerosis complex have been published in recent years. Other organ systems are often involved. Other cutaneous angiofibromatous lesions may accompany adenoma sebaceum, and these include plaque-like lesions of the forehead and scalp and ungual fibromas (see acral fibrokeratomas on p. 1022 ). “Shagreen patches,” with the histology of connective tissue nevi, are commonly found in tuberous sclerosis. They are usually present by puberty. Hypopigmented macules are a common finding. Molluscum pendulum is less common. Oral fibromas, mostly gingival in location, and dental pits are common findings in the mouth. Genetic linkage studies initially indicated that approximately half of all families with tuberous sclerosis showed linkage to chromosome 9q34 (TSC1) and the remainder to chromosome 16p13 (TSC2) . Subsequent studies have shown that TSC1 mutations account for only 10% to 30% of the families identified with tuberous sclerosis complex. In sporadic cases, there is an even greater excess of mutations in TSC2 . This latter group is usually associated with more severe disease. No identifiable mutations can be found in 15% to 20% of patients meeting the clinical criteria of tuberous sclerosis. Hamartin is encoded by TSC1 , and tuberin, a tumor suppressor, is encoded by TSC2 . There is a wide spectrum of mutations. More than 200 TSC1 and nearly 700 TSC2 unique allelic variants have been reported. Approximately two-thirds of all cases are sporadic and assumed to result from new mutations, many of which are in TSC2 . Other cases are inherited as an autosomal dominant trait. Mutation screening in tuberous sclerosis is labor-intensive and expensive. It is now available commercially. Prenatal or preimplantation genetic testing is becoming more widely available. A study of cultured fibroblasts from cutaneous tumors of patients with tuberous sclerosis demonstrated second-hit mutations of TSC2 of the CC>TT type, which are considered “ultraviolet signature” mutations; this suggests that restriction of ultraviolet exposure in children with tuberous sclerosis might reduce the number and severity of facial angiofibromas.
Adenoma sebaceum consists of several or multiple papules and nodules, sometimes grouped, with a predilection for the butterfly area of the face, particularly the nasolabial groove. They appear in early childhood as pink-red to yellow-brown lesions, and their growth is usually progressive until adult life. A giant angiofibromatous plaque and a cluster growth of large nodules have been reported. Unilateral facial involvement is another clinical variant. It probably represents mosaicism.
Facial angiofibromas in tuberous sclerosis have been treated with a scanning carbon dioxide laser. The benefits of therapy should be weighed against both early morbidity and the risks of long-term complications such as scarring and hypopigmentation. Erbium lasers have also been used. Clinical trials using sirolimus (rapamycin), an mTOR inhibitor, have shown promise. mTOR has a central role in the control of cell growth and proliferation. Its activation is influenced by heterodimers of TSC1 and TSC2 . Topical treatment with a rapamycin–tacrolimus ointment resulted in significant improvement of these skin lesions. It has been suggested that long-term therapy with sirolimus may increase the risk of malignant tumors in these patients. The finding of increased levels of certain matrix metalloproteinases (MMPs) in lesional skin raises the possibility of antiprotease treatments in the future or the use of retinoids, which inhibit the production of MMPs.
The lesions vary from rounded elevations to raised pedunculated growths ( Fig. 35.2 ). The epidermis shows some flattening of rete ridges with patchy melanocytic hyperplasia and also mild overlying hyperkeratosis. The dermal component consists of a network of collagen fibers, often oriented perpendicular to the surface in the subepidermal zone and having an onion-skin arrangement around follicles and sometimes blood vessels ( Fig. 35.3 ). There is an increase in “fibroblastic” cells, which are plump, spindle shaped, stellate, or even multinucleate. There is often a sparse inflammatory infiltrate that includes mast cells. The blood vessels are increased in number, and some are dilated with an irregular outline. It has been suggested that a functional loss of tuberin may stimulate vascular growth. Sporadic angiofibromas do not show loss of tuberin or hamartin. Follicles may show epithelial proliferation, and there may be primitive small follicles. Elastic tissue is absent from the stromal fibrous tissue. The extracellular glycoproteins fibronectin and tenascin are increased in the stroma. There is also overexpression of MLH-1 and psoriasin genes in the cutaneous hamartomas. Staining for CD31 confirms the increased vascularity of these lesions.


Ultrastructural examination has shown large numbers of microvilli on the luminal surface of the endothelial cells of the vessels. The stroma contains many banded structures. No myofibroblasts have been seen.
Facial angiofibromas, both unilateral and bilateral, have already been mentioned as an important manifestation of tuberous sclerosis. They have also been described in a patient with neurofibromatosis 2 (NF-2; OMIM 101000 ) as a cluster of small papules on the ear. Multiple facial angiofibromas are seen quite often in patients with multiple endocrine neoplasia (MEN) type 1 (OMIM 131100 ). They tend to present in adult life. Other cutaneous tumors in this syndrome include collagenomas and lipomas. There may also be café-au-lait macules and confetti-like hypopigmented macules. The tumors show allelic deletion of the MEN1 gene. It encodes a protein called menin, which is presumed to act as a tumor suppressor. Basic fibroblast growth factor (FGF) is elevated in many patients and may be responsible for the formation of the cutaneous tumors.
Angiofibromas (often reported as perifollicular fibromas) have been reported in the Hornstein–Knickenberg syndrome , which appears to be a slightly different phenotypic expression of the Birt–Hogg–Dubé syndrome (see p. 969 ). Multiple facial angiofibromas have also been reported in the Birt–Hogg–Dubé syndrome (OMIM 135150 ).
Multiple eruptive angiofibromas of the trunk have been reported in the absence of any underlying disease state. Unilateral facial angiofibromas were present in a teenage boy, with no signs of a systemic syndrome.
Fibrous papules of the face are usually solitary, dome-shaped papules, measuring 3 to 5 mm in diameter, found particularly on the nose of middle-aged adults. They are flesh colored and usually asymptomatic, although some may bleed after minor trauma. They were originally regarded as fibrosed dermal nevi, a proposition that has been disproved by electron microscopy and immunohistochemistry. The presence of factor XIIIa in the spindle cells and in some stellate cells suggests that fibrous papule is a proliferative reactive process consisting mainly of dermal dendritic cells.
The changes are similar to those described for adenoma sebaceum . However, the vessels are sometimes more ectatic and less likely to show concentric fibrosis than in adenoma sebaceum ( Fig. 35.4 ). Furthermore, the bizarre cells in the dermis are usually more numerous and the basal melanocytic hyperplasia more prominent in fibrous papule of the face. Rarely, the stromal cells may contain coarse cytoplasmic granules leading to a granular-cell appearance. Another rare pattern involves the presence of numerous fibroblasts/histiocytes/dendrocytes with clear vacuolated cytoplasm embedded in a dense sclerotic and hyalinized stroma. A few multinucleate “floret”-like cells may be present. Only a few cells stain for factor XIIIa and CD68. This lesion, clear cell fibrous papule , may eventually prove to be unrelated to fibrous papule, although the cases reported were all on the face, predominantly the nose ( Fig. 35.5 ). These lesions show aggregates of cells with small round nuclei and cytoplasm containing large vacuoles or fine vacuoles with scalloped nuclei. The cells express vimentin, CD68, factor XIIIa and NKI/C3; PAS and mucicarmine stains are negative, as are S100 (usually), cytokeratin AE1/AE3, EMA, CEA, and HMB-45. Rare variants include hypercellular , pigmented , pleomorphic , inflammatory , and epithelioid fibrous papules. The morphological features of isolated oral fibromas of the tongue closely resemble those of fibrous papule, and as is the case for many fibrous papules, these lesions tend to lack concentric fibrosis around vessels.


The spindle and stellate cells in fibrous papule of the face contain vimentin and factor XIIIa (discussed previously) but not S100 protein. α 1 -Antitrypsin and lysozyme were detected in one study, although this has not been confirmed subsequently. A case with CD34 + cells has been reported. The cells of the epithelioid fibrous papule are reactive for procollagen and are negative for NKI/C3, unlike previously described clear cell variants.
Ultrastructural studies suggest that the stellate cells are fibroblastic or fibrohistiocytic.
Pearly penile papules are persistent asymptomatic pearly white papules, 1 to 3 mm in diameter, occurring in groups or rows on the coronal margin and sulcus of the penis. Rarely, they may be found on the penile shaft or glans. They are found in 10% to 30% of young adult men, and they are more common in black people and in the uncircumcised. They may be misdiagnosed as warts, but there is no causative role for human papillomavirus (HPV) in their genesis.
Treatment with cryotherapy has been effective in some studies but not in all. Carbon dioxide laser has also proved effective.
There is a rich vascular network, surrounded by dense connective tissue containing an increased number of plump and stellate “fibroblasts” ( Fig. 35.6 ). They resemble other lesions in this group, except for the absence of pilosebaceous follicles.

Included in the acral fibrokeratoma group are lesions reported as acquired digital fibrokeratoma, acquired periungual fibrokeratoma, “garlic clove fibroma,” and the subungual and periungual fibromas of tuberous sclerosis. This unifying concept is an attempt to overcome the needless proliferation of terms, and it gives recognition not only to the common histopathological features but also to the fact that occasional lesions have been reported in sites other than digits.
The lesions are usually solitary, dome-shaped, or elongated thin horns, 1 to 3 mm in diameter and up to 15 mm in height. A giant variant measuring almost 4 cm in diameter has been reported. There is sometimes a history of trauma. The ungual fibromas of tuberous sclerosis are often multiple, sometimes in clusters, and develop around the time of puberty. They are found in approximately half the patients with tuberous sclerosis.
Some fibrokeratomas originate from the dermal connective tissue, whereas others appear to originate from the proximal nail fold. An invaginated variant has been reported in relation to the nail apparatus. This difference in the site of origin may account for the heterogeneous features observed in this entity.
Multiple acral fibromas with a myxoid but poorly vascularized stroma have been reported in a patient with familial retinoblastoma, leading to the suggestion that multiple acral benign tumors with a fibrous component might be a cutaneous marker of tumor suppressor gene germline mutation. Lesions reported as familial multiple acral mucinous fibrokeratomas and familial myxovascular fibroma (see later) are probably further examples of this hypothesis.
The epidermal covering usually shows hyperkeratosis and sometimes acanthosis. There is a core of thick collagen bundles that are oriented predominantly in the vertical axis ( Fig. 35.7 ). Stellate fibroblasts are often present. There is sometimes prominent cellularity and a rich vascular supply. These latter two features have not been prominent or have been specifically excluded in some of the reports, suggesting that some of the lesions might best be regarded as fibromas rather than angiofibromas. The rare invaginated variant is characterized by a deep epithelial invagination proximal to the normal matrix. A pseudo–nail plate is produced.

There are usually sparse elastic fibers, few inflammatory cells, and no hair follicles. Neural tissue is not present, unlike the clinically similar entity of supernumerary (rudimentary) digits. The stromal cells express varying amounts of factor XIIIa.
Mention is made here of the cellular digital fibroma, which is composed of intersecting fascicles of thin, delicate spindle cells in the superficial reticular dermis with a fibrotic and slightly myxoid stroma. It may be histogenetically distinct from other angiofibromas and digital fibrokeratomas because the constituent cells in cellular digital fibromas stain strongly for CD34, with only scattered stromal cells expressing factor XIIIa. This entity needs to be distinguished from DFSP. It has been suggested subsequently that cellular digital fibroma is a variant of, if not the same as, superficial acral fibromyxoma (see p. 1064 ). There is certainly considerable histopathological and immunohistochemical overlap of the two conditions, a difference being that the CD34 + cells in cellular digital fibroma are CD99–, whereas in superficial acral fibromyxoma they are reportedly CD99+.
On dermoscopy, acral fibrokeratoma shows a white, scaly collarette, peripheral to which is a zone of erythema with globular vessels.
Three kindreds have been reported in which multiple verrucous papules developed on the fingers and hands. On histological examination, the papules showed a fibrovascular proliferation of the papillary dermis, with variable myxoid change and overlying epidermal acanthosis and hyperkeratosis.
Larger examples of angiofibroma have features in common with connective tissue nevi, and these may in fact be closely related entities. As noted previously, a major differential diagnostic consideration is the supernumerary digit (rudimentary polydactyly), which can closely resemble digital fibrokeratoma clinically. However, supernumerary digits characteristically occur at the base of the fifth finger, are present at birth, may be bilateral, and show prominent nerve bundles in the deep dermis, with a configuration comparable to neuromas of other types. These are in fact believed to occur as the result of autoamputation of a true accessory digit. As supporting evidence of this concept, the editor has seen an example of an accessory digit, to which was attached a neuroma with features of a small supernumerary digit. The connective tissue changes of onychomatricoma can resemble those of fibrokeratoma of the nail bed, especially if the nail plate has been removed. A distinction can be made by recognizing in onychomatricomas the characteristic epithelial-lined invaginations surrounding optically empty cavities—negative images of the keratogenous zones that comprise the ungual spurs on the inferior border of the involved nail plate. There is some overlap of familial myxovascular fibroma with the lesions included within the concept of cutaneous myxomas (see p. 450 ).
This is a recently described soft tissue tumor that often occurs in the lower extremity, adjacent to a joint. However, it has also been found on the back, abdominal wall, pelvic cavity and breast, and recently presented as a subcutaneous mass on the left cheek. Therefore, it may be of occasional dermatopathological significance. There is a wide age range of incidence, and females are more commonly affected. While there have been local recurrences, metastases have not been reported. Microscopically, the lesions are described as lobulated and well circumscribed but not encapsulated. There are alternating myxoid and collagenous areas, variations in cellularity, and evenly distributed branching vessels with thick walls. Short spindle cells with pale cytoplasm and nuclei with fine chromatin are observed, and there are occasional multinucleated cells and giant cells. Yamada et al additionally described amianthoid fibers (resembling fine, silky asbestos), extravasated erythrocytes and hemosiderin deposition, cystic changes, necrosis, and aggregates of foamy histiocytes. Immunohistochemical findings vary somewhat among reports, but the spindle cells may express epithelial membrane antigen (EMA), desmin, CD163, CD68, estrogen receptor, progesterone receptor, and signal transducer and activator of transcription 6 (STAT6). They have been negative for smooth muscle actin (SMA), muscle-specific actin (MSA), S100, CD34, CD31, and pan-cytokeratin. There is low Ki-67 expression. A key to the diagnosis is the recognition of AHRR – NCOA2 or GTF2I–NCOA2 gene fusions, which demonstrate the role of NCOA2 in the development of soft tissue angiofibromas. Additional gene fusions have recently been reported in this tumor.
This heterogeneous group of lesions forms a histological spectrum, at one end of which is the fibroma and at the other end the fibrosarcoma. In between are the fibromatoses, which have been defined as a “group of nonmetastasizing fibrous tumors which tend to invade locally and recur after surgical excision.”
The fibromatoses include entities such as palmar and plantar fibromatosis, extra-abdominal desmoid, knuckle pads, pachydermodactyly, Peyronie's disease of the penis, and various juvenile fibromatoses such as juvenile aponeurotic fibroma, fibrous hamartoma of infancy, digital fibromatosis of childhood, and infantile myofibromatosis. The tumors in this latter category usually contain an admixture of fibroblasts and myofibroblasts. This is the explanation for their inclusion also as tumors of myofibroblasts (see later).
Plantar fibromatosis is a benign, but sometimes locally aggressive, proliferation of fibrous tissue involving the deep subcutis and fascia of the plantar surface of the foot. Familial cases occur. In a study, from the Armed Forces Institute of Pathology (AFIP), of 56 cases of palmar–plantar fibromatosis in children and preadolescents, there was a high incidence of local recurrence of their fibromatosis. Most cases had been initially managed by local excision, and in most cases, there were positive margins. Most cases of plantar fibromatosis contain multinucleated giant cells, but the number of these cells is quite variable. They also occur in palmar fibromatosis. A variant with distinct nodules occurs. Peyronie's disease of the penis, nodular and diffuse fibrous proliferations of the penis and tunica vaginalis, and Dupuytren's disease of the palmar fascia are similar fibromatoses. There is a high incidence of chronic liver disease in patients with Dupuytren's disease. The desmoid tumor (a clonal process) and extra-abdominal desmoid tumor are usually regarded as belonging to the domain of soft tissue tumors. They are not considered further. Platelet-derived growth factor (PDGF) receptors and ligands are upregulated in fibromatoses; they may play a role in the growth of these tumors. The nuchal fibroma (see p. 401 ) and the nuchal fibrocartilaginous tumor (see p. 465 ) are considered elsewhere. The nuchal-type fibromas that arise in association with colonic polyps may be multiple and occur in different locations. They have been called Gardner fibromas. They are discussed later in this chapter (see p. 1025 ).
Tumors of myofibroblasts include nodular fasciitis, fibrous hamartoma of infancy, digital fibromatosis of childhood, postoperative spindle cell nodule, dermatomyofibroma, infantile myofibromatosis, inflammatory myofibroblastic tumor, plexiform fibrohistiocytic tumor, and myofibroblastic sarcoma. Partial myofibroblastic differentiation is seen in low-grade fibromyxoid sarcoma and angiomyofibroblastoma of the vulva. Plexiform fibrohistiocytic tumor is considered with the fibrohistiocytic tumors (see p. 1048 ) because of the dual population of cells. Congenital–infantile fibrosarcoma is usually included as a myofibroblastic lesion. The case with myofibroblastic proliferation confined to the skin of the neck defies classification.
Electron microscopy is of some value in distinguishing between the various spindle cell tumors of the skin and soft tissues. Cytogenetic analysis of tumors adds further information; some tumors have specific karyotypic aberrations.
Skin tags (soft fibromas, acrochordons, fibrolipomas, fibroepithelial polyps) are common cutaneous lesions that have received little attention in the dermatological literature because hitherto they have been regarded as being of little consequence. However, there have been several reports suggesting an association between the presence of skin tags and underlying diabetes, abnormal lipid profile, colonic polyps, or acromegaly. The association with colonic polyps is controversial, and several studies have failed to confirm its existence. HPV types 6/11 were detected in a significant number of cases in one study. This finding remains to be confirmed.
Skin tags have a predilection for the axilla, neck, groin, eyelids, and beneath pendulous breasts. They are more common in obese females, and they may develop in pregnancy. In one autopsy study, they were present in 64% of individuals older than the age of 50 years, whereas in a more recent study they were present in 46% of 750 individuals examined. There are three clinical types : furrowed papules approximately 2 mm in width and height; filiform lesions, approximately 2 mm in width and 5 mm in height; and large bag-like protuberances, usually on the lower trunk. These larger lesions are very occasionally multiple. The term fibroepithelial polyp is sometimes used for this latter variant. Experimentally, these polyps show downregulation or loss of tuberin and/or hamartin expression that may promote collagen formation, leading to their formation and growth.
Vestibular papillae of the vulva are skin tag–like smooth projections of the vestibular mucosa that appear to be normal anatomical variants. They are not related to HPV infection, as previously suggested.
Fibroepithelial polyps of the anus are relatively common lesions, some of which are thought to arise from enlargement of anal papillae. They should be distinguished from the much smaller infantile perianal (perineal) pyramidal protrusion, which occurs predominantly in young girls in the midline, anterior to the anus. Sometimes they are posterior to the anus. Underlying constipation and anal fissures are often present. A variant associated with lichen sclerosus may also occur. They are edematous, flesh-colored, sessile protrusions measuring 1 or 2 cm in length. They should not be mistaken for condylomas. One case has been successfully treated with topical steroid applications.
The lymphedematous fibroepithelial polyp of the penis is a rare polyp of the glans penis or prepuce associated with long-term condom catheter use. On rare occasions, it has been associated with chronic phimosis. The lesions are gray-white polyps measuring 2 to 7.5 cm in diameter. The majority of lesions affect the ventral surface of the glans near the urethral meatus. It has been postulated that the lesions represent a reactive hyperplastic process involving the subepithelial stroma. A related lesion is the polyp of the glans penis that developed in a man who practiced genital hanging kung fu.
The histological features vary with the clinical type. The furrowed papules show epidermal hyperplasia and sometimes horn cyst formation. These lesions, with seborrheic keratosis-like surface changes, are most common on the neck and eyelids ( Fig. 35.8A ). The filiform lesions are covered by an epidermis that shows only mild acanthosis. Pagetoid dyskeratosis (see p. 336 ) is sometimes an incidental finding in the overlying epidermis. The connective tissue stalk is usually composed of well-vascularized, loosely arranged collagen. Elastic fibers are present in normal amounts. A few fat cells and, sometimes, nevus cells may be present. The larger, bag-like lesions (fibroepithelial polyps, fibrolipomas) usually have a stroma composed of loosely arranged collagen and a central core of adipose tissue ( Fig. 35.8B ). It has been suggested, and subsequently challenged, that there is little utility in submitting these lesions for histological examination.
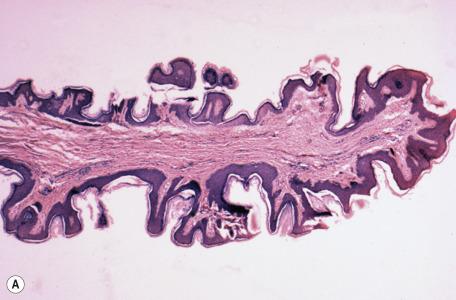
Vestibular papillae of the vulva consist of connective tissue projections covered by stratified squamous epithelium. Sometimes the epithelium is glycogen rich, simulating koilocytes.
An unusual cutaneous polyp with bizarre stromal cells, thought to represent a degenerative phenomenon, has been reported as a “ pseudosarcomatous polyp .” Subsequent correspondence raised the possibility that the lesion may have been a dermal spindle cell lipoma or “ancient” (degenerative) change in a polyp.
The fibroepithelial polyp of the anus has a myxoid and/or collagenous stroma covered by stratified squamous epithelium that may show some swollen cells with vacuolation near the surface. The stroma sometimes contains atypical cells showing fibroblastic and myofibroblastic differentiation. Hyalinized vascular changes may be present near the base of the polyps. There is an increase in CD34 + stromal cells. The infantile perianal pyramidal protrusion reveals epidermal acanthosis, marked edema in the upper dermis, and a mild inflammatory cell infiltrate.
The lymphedematous fibroepithelial polyp of the penis has a polypoid configuration covered by keratinizing squamous epithelium. The stroma is edematous with dilated vessels and focal proliferation of capillary-sized vessels with hyalinization. The focally myxoid stroma contains small spindle-shaped cells with ill-defined, pale eosinophilic cytoplasm. There are abundant bizarre multinucleated giant cells. A mild infiltrate of lymphocytes, plasma cells, and mast cells is present in the stroma.
Other types of lesions can arise within, or have clinical features of, fibroepithelial polyps, including basal cell carcinomas or cutaneous pseudosarcomatous polyps. Polypoid melanomas also exist, although lesions tend to be larger than the average fibroepithelial polyp; these can be amelanotic. The “acrochodrons” of Birt–Hogg–Dubé syndrome usually show evidence of fibrofolliculoma or trichodiscoma when lesions are appropriately oriented and sectioned. There are two recent cases of well-differentiated liposarcomas that presented clinically as skin tags; diagnosis was supported by FISH analysis, showing amplification of murine double-minute type 2 (MDM2) gene and the cyclin-dependent kinase-4 (CDK4) gene. Findings such as these, although uncommon, support the argument that at least some fibroepithelial polyps, particularly those with unusual clinical features, should be submitted for histopathological study.
Prepubertal vulvar fibroma was the term used by Iwasa and Fletcher in 2004 for a hitherto unrecognized mesenchymal tumor of the vulva in 11 prepubertal girls. These authors regarded them as tumors on the basis of recurrences in several cases after excision. The following year, Vargas and colleagues reported 14 cases, also from Boston, of a similar lesion that presented with enlargement of one, or occasionally both, labia majora. On the basis of its occurrence at an age roughly coincident with the time of breast budding, its capacity for spontaneous regression, and its composition of elements native to the vulva, the authors concluded that the entity represented an asymmetrical physiological enlargement in response to hormonal surges of pre- and early puberty.
The girls were aged 4 to 13 years. The lesions were ill-defined and consisted of fibro-fatty tissue ranging in size from 2 to 8 cm in greatest dimension. As previously mentioned, several cases recurred after initial excision.
The tumors were poorly marginated, hypocellular masses in the dermis and subcutaneous tissue. They were composed of the usual constituents of vulvar soft tissue, with expansion of the fibrous component. There were bland spindle-shaped cells in a variably collagenous to edematous or myxoid stroma. The interconnected fibrous bands encircled lobules of fat, blood vessels, and nerves. The fibroblastic cells were immunoreactive for CD34 ; they also expressed estrogen and progesterone receptors.
Gardner fibroma is a benign lesion of the superficial and deep soft tissues, presenting most commonly in childhood and adolescence with an association with familial adenomatous polyposis and desmoid fibromatosis. Sporadic cases also occur. The majority of cases involve the back and paraspinal region, followed by the head and neck, and the extremities. Almost 20% of patients have concurrent or subsequent desmoid tumors. There is a strong association with familial adenomatous polyposis or adenomatous polyposis coli, but the proportion of sporadic cases that have the APC mutation remains to be elucidated. The Gardner fibroma can be the presenting feature of familial adenomatous polyposis.
There are some overlap features with nuchal fibroma (see p. 401 ), but the predilection of nuchal fibroma for middle-aged men, its location on the posterior neck, and its association in some patients with diabetes mellitus—but not familial adenomatous polyposis—usually allows a distinction to be made.
There is a bland, hypocellular proliferation of haphazardly arranged coarse collagen fibers with inconspicuous spindle cells. There are small blood vessels and a sparse mast cell infiltrate. The collagen forms around adipose tissue lobules. There is no increase in small nerve bundles, as seen in nuchal-type fibroma (see p. 401 ).
In one study, 64% of cases tested showed nuclear reactivity for β-catenin, and 100% of cases showed nuclear reactivity for both cyclin D1 and c- myc .
Pleomorphic fibroma of the skin was described by Kamino et al. in 1989. It presents as a slow-growing lesion, clinically indistinguishable from a polypoid skin tag. A subungual lesion has been reported on several occasions.
The lesion is usually a dome-shaped nodule with variable cellularity. The spindle-shaped cells show striking nuclear pleomorphism with rare mitotic figures ( Fig. 35.9 ). A variant with myxoid stroma occurs. The cells express vimentin and CD34 but not desmin, Ki-M1p, or S100 protein, suggesting a possible origin from dendrocytes rather than myofibroblasts as originally thought. Staining for factor XIIIa has been moderate in some studies but negative or patchy in others. The nuclear atypia is similar to the “degenerative” changes seen in a number of benign mesenchymal tumors.
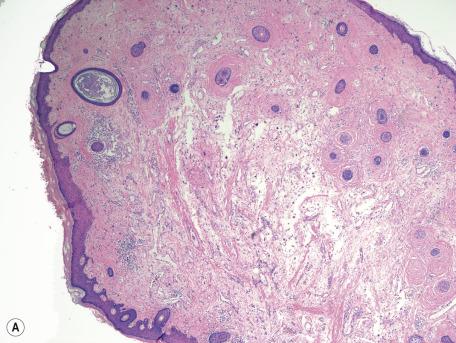
The morphological features of pleomorphic fibroma can resemble those of dermal atypical lipomatous tumor/well-differentiated liposarcoma. A clue to the diagnosis of pleomorphic fibroma is that any adipocyte foci that may be encountered lack cytological atypia and may simply represent entrapped adipose tissue within the lesion. The importance of this finding is emphasized in the case of a cutaneous polyp that had some microscopic features of pleomorphic fibroma but also showed an area of adipocyte differentiation with pleomorphic lipoblasts and S100 positivity among tumor cells, resulting in a diagnosis of pleomorphic liposarcoma. Immunohistochemical and molecular analysis may be helpful in difficult cases; one study searching for 12q15/ MDM2 amplification by FISH analysis, and MDM2 expression using immunohistochemistry, found negative results in all 15 cases of pleomorphic fibroma. Another recent study found nuclear MDM2 immunoreactivity in a case of pleomorphic fibroma in the absence of MDM2 gene amplification, indicating the importance of FISH analysis for accurate diagnosis, especially when attempting to exclude atypical lipomatous tumor, which is typically associated with MDM2 amplification. Hinds et al. demonstrated recurrent loss of RB1 on chromosome 13 in pleomorphic fibroma by both retinoblastoma (Rb) protein expression loss and loss of 13q by array comparative genomic hybridization—thereby showing that this tumor shares the same genetic abnormalities as spindle cell and pleomorphic lipomas.
Sclerotic (“plywood”) fibroma is an uncommon fibrocytic neoplasm that occurs sporadically as a solitary lesion and also in a multifocal form in patients with Cowden's disease. A solitary lesion may be the presenting feature of this syndrome. The terms hypocellular fibroma and circumscribed, storiform collagenoma have also been used for this entity. The lesions are flesh-colored papules or nodules measuring 0.5 to 3 cm in diameter. Sporadic cases, unassociated with Cowden's disease, have been reported in the oral mucosa.
Although it was initially regarded as an involutional lesion, one study demonstrated ongoing type I collagen synthesis and deposition, suggesting that the lesion is a proliferating neoplasm. Local recurrence has been reported.
The lesions are circumscribed, unencapsulated dermal nodules, often with an attenuated overlying epidermis. They are composed of thickened and homogenized eosinophilic collagen bundles arranged in a laminated manner with intervening prominent clefts. Vaguely storiform or whorled patterns of collagen may be present ( Fig. 35.10 ). The lesions are of low cellularity. The nuclei are tapered to stellate. Elastic fibers are absent from the lesion, but there is often some stromal mucin. Similar collagenous changes have been seen as a focal phenomenon in the vicinity of inflammatory lesions, such as folliculitis, and in dermatofibromas, nevi, angiofibromas, erythema elevatum diutinum, fibroadenoma of axillary accessory breast tissue, and neurofibromas where the sclerosis may be more extensive. This has led to the view that sclerotic fibroma-like change may represent a common reaction pattern in the skin.
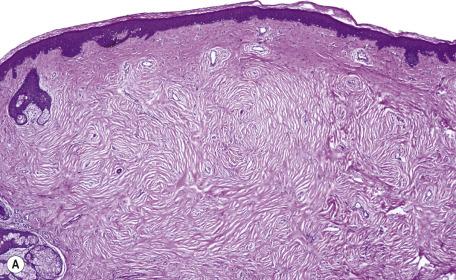
The spindle cells stain for vimentin and factor XIIIa. CD34 positivity occurs focally, with no consistent localization. The finding of diffuse CD34 and CD99 positivity in both sclerotic fibromas and pleomorphic fibromas has led to the suggestion that these tumors may be linked in some way. Both proliferating cell nuclear antigen (PCNA) and Ki-67 immunoreactivity have been detected, as would be expected in a growing neoplasm. The proliferating index (MIB-1) was very low in another series.
The lesion reported as a pacinian collagenoma is probably a variant. It was composed of paucicellular collagen fibers arranged in concentric lamellations giving an “onion-skin” appearance. The cells stained for CD34. It had some resemblance to the perineurioma (see p. 1098 ).
Cases resembling sclerotic fibroma but with variable numbers of bizarre, multinucleated cells, often with a foamy cytoplasm, have been reported as pleomorphic sclerotic fibroma , giant cell collagenoma , and cellular storiform collagenoma.
The widened collagen bundles contain tightly packed collagen fibrils, only 50 nm in diameter. A recent ultrastructural study found that spindle cells with myoid features had proliferated around blood vessels.
In addition to the other lesions that can occasionally show changes of sclerotic fibroma, noted previously, differential diagnostic considerations include two uncommon neural tumors: sclerotic perineurioma and a variant of pacinian neurofibroma, which has also been termed fibrolamellar nerve sheath tumor. The sclerotic perineurioma features dense connective tissue but also epithelioid and spindled cells in trabecular or whorled arrangements; these cells are positive for epithelial membrane antigen, as is typical for cells of the perineurium. The pacinian neurofibroma, or fibrolamellar nerve sheath tumor, is said to closely resemble sclerotic fibroma but may feature increased amounts of mucin (which, however, is also found in sclerotic fibromas), scattered cells with small nuclei, some of which can be S100 + , and occasional pigmented dendritic melanocytes. A recent report described similarities between sclerotic fibroma and solitary fibrous tumor of the oral cavity. The distinction appeared to rest on the increased cellularity of the latter. The constituent cells of both were positive for vimentin, CD34, and CD99; bcl-2 positivity was evident only in the example of solitary fibrous tumor.
Collagenous fibroma, a recently described tumor, may arise in the subcutaneous tissue or muscle. Dermal involvement is rare. The tumors are firm and nontender. Most measure 2 or 3 cm in diameter, but larger lesions have been reported. There is a predilection for adult men. Any part of the body may be involved. Cases involving the oral cavity have been reported.
Few cases have been subject to cytogenetic analysis. It appears that a specific breakpoint of 11q12 occurs, combined in several cases with a reciprocal translocation. A recent report has shown a t(2;11) translocation in a case of collagenous fibroma, whereas another found trisomy 8 as the only cytogenetic abnormality.
Surgery is the treatment of choice, with no reports of recurrence.
Collagenous fibroma is usually a well-demarcated tumor in the subcutis, although some infiltration is often present at the periphery. It is hypocellular and composed of large, stellate, or spindle cells set in a densely collagenous or fibromyxoid stroma. There are usually no mitoses. There are inconspicuous small vessels. The cells express vimentin. They are focally positive for α-SMA and muscle-specific actin. Factor XIIIa was present in one case. S100 protein and CD34 are not expressed. Scattered cells may show a myofibroblastic immunophenotype.
One important differential diagnostic consideration is fibromatosis (see later discussion), a term applied to a group of tumors that tend to recur after local excision. In contrast to collagenous fibroma, the lesions of fibromatosis are typically more cellular, have fascicular arrangements of cells, and show greater degrees of peripheral infiltration. Nuclear β-catenin staining is also commonly present in forms of fibromatosis but has been reported to be negative in examples of collagenous fibroma. There can be a close resemblance between desmoplastic fibroblastoma and fibroma of tendon sheath . In a recent study, Kato et al. found that all of their cases of desmoplastic fibroblastoma showed diffuse, strong FOSL1 nuclear immunoreactivity, whereas none of the fibromas of tendon sheath were positive. However, chromogenic in situ hybridization failed to reveal FOSL1 rearrangements in 7 tested desmoplastic fibroblastomas, suggesting that the immunohistochemical finding may not be a direct result of FOSL1 gene rearrangement (FOS proteins are considered regulators of cell proliferation, differentiation and transformation).
Knuckle pads (discrete keratodermas over the knuckle and finger articulations) are well-formed skin-colored nodules overlying the interphalangeal and metacarpophalangeal joints of the hands. They are usually multiple. There are several clinical variants, including a familial group, an occupational or recreational-related group, and an acquired idiopathic group. They have followed the prolonged playing of video games. An association with knuckle cracking and with pseudoxanthoma elasticum has been reported. Of historical interest is the prominent knuckle pad on the right thumb of Michelangelo's statue of David.
Inherited knuckle pads of the feet have been described in association with leukonychia and deafness, with and without keratosis palmoplantaris, including epidermolytic palmoplantar keratoderma. Genetic abnormalities found in the knuckle pad–palmoplantar keratoderma association include a mutation in the 2B rod domain of keratin 9 and a G59A mutation in the GJB2 gene, encoding the gap junction protein connexin. They have also been associated with acrokeratoelastoidosis. They are well-defined plaques on the dorsal aspect of the feet. Acquired lesions of the feet have been associated with repetitive friction from athletic gear. An example of granuloma annulare mimicking knuckle pads has been described.
There appear to be at least three histological types. The usual lesions show prominent hyperkeratosis, hypergranulosis, and epidermal acanthosis. There is minimal thickening of the papillary dermis. Another type has macronodules of swollen collagen fibers surrounded by thickened elastic fibers. A third variant with prominent subcutaneous fibrosis, belonging to the fibromatoses, has been documented.
Pachydermodactyly is characterized by fibrous thickening of the lateral aspects of the proximal interphalangeal joints of the fingers, usually in males. The thumbs and fifth fingers are usually not involved. More extensive lesions have been reported. A case of unilateral pachydermodactyly transgrediens (involving the hypothenar side of the left hand as well as the joints) has been described. It has been reported in two siblings.
Pachydermodactyly is regarded as a localized form of superficial fibromatosis. Trauma, such as finger rubbing, has been implicated in the etiology. In fact, repetitive trauma (e.g., from sporting or occupational activities or related to tic-like behaviors, obsessive–compulsive disorders, or Asperger syndrome) has been considered a precipitating factor in at least one-third of cases. It has also been suggested that this entity might be related to knuckle pads (discussed previously).
The overlying epidermis shows hyperkeratosis and acanthosis. There is thickening of the dermis, with coarse collagen bundles in haphazard arrangement and a mild proliferation of fibroblasts that prove to be CD34 + . Increased fibroblastic activity and collagen deposition around sweat glands have been noted. Deposits of mucin of varying degrees are sometimes present in the interstitium, and elastic fibers are reduced. There is no inflammation. Types III and V collagens are mainly found.
Self-healing juvenile cutaneous mucinosis can present with fibromucinous nodules in a periarticular distribution on the hands that could be confused with pachydermodactyly. However, fusiform swelling of the lateral digits is not observed in the former condition, and patients often have lesions in other locations, including the head, face, and trunk. Microscopically, the lesions of self-healing juvenile cutaneous mucinosis are quite different in that they feature considerable dermal mucin deposition with mild perivascular inflammation. In subcutaneous lesions, there may be large mucinous lobules, sometimes accompanied by stellate, rhabdoid, or ganglion-like giant cells, creating a resemblance to proliferative fasciitis.
Nodular fasciitis is a reactive proliferation of myofibroblasts with a predilection for the subcutaneous tissues of the forearm, upper arm, and thigh of young and middle-aged adults. Dermal and intravascular variants have been reported. Intradermal variants tend to occur in young adults and arise in the extremities or trunk, and they show a similar clinical course and biological behavior to traditional nodular fasciitis. It usually grows quite quickly to reach a median diameter of 1.5 cm. Multiple lesions have been reported. Recurrences are rare, even after incomplete surgical removal, and their occurrence should lead to a reappraisal of the original histological diagnosis. The diagnosis can usually be made readily by fine needle aspiration. Regression of the lesion has followed this procedure. Regression may also occur after the use of intralesional corticosteroids. Despite having high activity for PCNA, the cells do not express p53 or show aneuploidy. It is polyclonal and a reactive process. Recent work found that a high percentage of cases of nodular fasciitis demonstrate fusion of the myosin heavy chain 9, nonmuscle (MYH9) promoter region to the coding region of ubiquitin carboxyl-terminal hydrolase 6 (USP6), with the latter being involved in another translocation associated with aneurysmal bone cysts. The authors of this study propose that this may serve as a model of transient neoplasia.
Cranial fasciitis of childhood is a distinct clinical variant arising in the deep soft tissues of the scalp, with involvement of the underlying cranium. Conservative surgical excision is curative. Intralesional corticosteroids were used in one case with a successful outcome.
Nodular fasciitis is composed of a proliferation of spindle-shaped to plump fibroblasts that may be arranged in haphazard array (“tissue culture appearance”) or in bundles that form S-shaped curves ( Fig. 35.11 ). A vague storiform pattern is sometimes present focally. Mitoses are frequent, but atypical forms are rare. Cleft-like spaces may be seen between the fibroblasts. There is a variable amount of myxoid stroma and extravasated erythrocytes. Collagen is usually sparse. Capillaries with plump endothelial cells are common. Scattered lymphocytes are dispersed throughout the lesion.
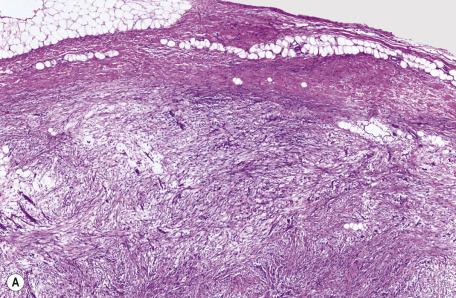
Intravascular fasciitis, in which the proliferation occurs within small and medium-sized arteries and veins, appears to represent an origin from myofibroblasts within vessel walls rather than extension from the extravascular component that is often present. It is not associated with aggressive growth or metastasis. Vascular involvement results in a plexiform appearance.
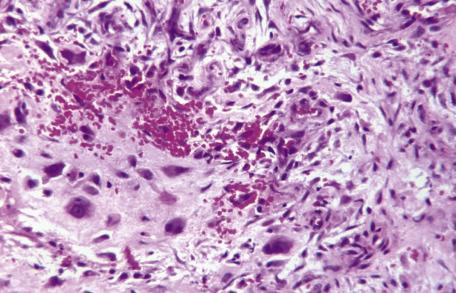
Less common findings include the presence of bone and cartilage; osteoid is not uncommon in the variant known as cranial fasciitis. Scattered multinucleate fibroblasts and osteoclast-like giant cells are often present, and, occasionally, the latter cells are quite numerous. The term ossifying fasciitis has been used if there is a significant component of osteoid or bone. A variant with numerous cells resembling ganglion cells, akin to those seen in proliferative myositis, may be separated as a distinct entity— proliferative fasciitis ( Fig. 35.12 ). Several examples of intradermal proliferative fasciitis have now been reported.
Various histological subtypes of nodular fasciitis have been proposed based on the cellularity, the amount of myxoid stroma, and the presence of collagen or other histological features, such as osteoclast-like cells, ganglion-like cells, or bone. Some of the proposed subtypes merely reflect changes in the histological composition during the evolution of the lesion.
Immunoperoxidase studies demonstrate HSP47 (a useful marker of skin fibroblasts), smooth muscle actin, muscle-specific actin, calponin, vimentin, and KP1, but not desmin, CD34, cytokeratin, or S100 protein. A similar staining pattern is seen in proliferative fasciitis, although the ganglion cells express only vimentin. The multinucleate cells in cranial fasciitis stained for CD68 in one reported case. Ki-67 expression can be high in nodular fasciitis, but this does not imply a likelihood of recurrence. In one study of nodular fasciitis of the head and neck, there was consistent positivity for smooth muscle actin, calponin, CD10, and PG P9.5; CD34 and CD56 were negative in all cases. The recently reported cases of intradermal proliferative fasciitis have shown an absence of SMA staining among ganglion-like cells but, in some cases, expression of factor XIIIa, suggesting fibroblastic rather than myofibroblastic lineage.
Few ultrastructural studies have been carried out, but in one study of eight cases, myofibroblasts were the predominant cell present. Cells with features of fibroblasts or histiocytes are also seen. Myofibroblasts and fibroblasts are also present in cranial fasciitis.
Nodular fasciitis must be distinguished from fibrosarcoma and malignant fibrous histiocytoma (MFH). In addition to its characteristic subcutaneous location and rapid growth, nodular fasciitis lacks the dense cellularity, herringbone pattern, and marked mitotic activity expected in fibrosarcoma or the marked pleomorphism seen in many examples of MFH. The interconnecting fascicles of bland-appearing fibroblasts, dense collagen, and pronounced skeletal muscle infiltration associated with forms of fibromatosis help distinguish this group of lesions from nodular fasciitis. Deep fibrous histiocytomas can often be distinguished by whorled or “curlicue” arrangements of cells, dense collagen, and sometimes by the presence of hemosiderin, lipid-laden macrophages, and Touton-like giant cells. Based on the study of head and neck nodular fasciitis by Morgen et al., CD10 positivity would argue against leiomyosarcoma (usually CD10 – ); calponin expression against myofibroblastic sarcoma; PGP 9.5 positivity (and CD34 negativity) against DFSP; and CD56 negativity against malignant peripheral nerve sheath tumor, schwannoma, leiomyoma, or leiomyosarcoma.
Atypical decubital fibroplasia (ischemic fasciitis) is a pseudosarcomatous proliferation, found in immobilized or debilitated patients, that is different from a decubitus ulcer. The affected area measures 1 to 9 cm in diameter. It involves the shoulders, ribs, sacrococcygeal region, and the tissues overlying the greater trochanter. There is involvement of the deep dermis, subcutis, and deep fascia with a zonal arrangement of fibrinoid necrosis, reactive fibrosis, neovascularization, fat necrosis, and ectatic vessels. Atypical fibroblasts are usually present. Microscopic changes resembling ischemic fasciitis have been found in the region of an intra-arteriolar cholesterol embolus.
A study of 44 cases by Liegl and Fletcher found that there was an inconsistent association with immobility or debilitation.
Postoperative spindle cell nodule is a rare lesion that develops soon after a surgical procedure in genital skin or the genitourinary system. Lesions have also been reported at other sites. It is composed of spindle-shaped cells in a pattern of interlacing fascicles ( Fig. 35.13 ). There are occasional mitotic cells. Vascular proliferation, hemorrhage, and inflammation accompany the process. The cells express vimentin; desmin and muscle-specific actin have been present in some cases. They do not express keratin. A study of the cytology of this lesion showed bland spindled cells with eosinophilic cytoplasm, oval nuclei with inconspicuous nucleoli, and mitotic activity but without atypical mitoses.
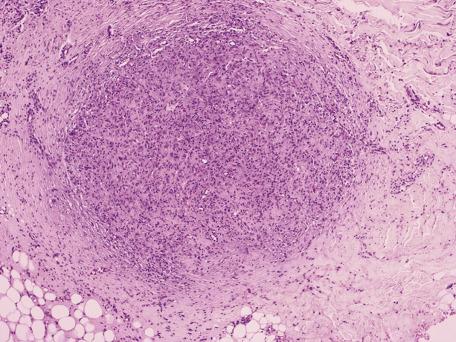
Solitary fibrous tumor was first reported by Klemperer and Rabin in 1931. It has been well-known to surgical pathologists as a tumor of the pleura or peritoneum that must be distinguished from mesothelioma. In recent years, it has been reported in a variety of other organs, including the orbit, oral mucosa, soft tissue, and skin, and therefore may be encountered by dermatopathologists. Skin lesions typically present as nodules and may be mistaken for cysts. Most cases have been on the head and neck; vaginal involvement has also been reported. Most patients with solitary fibrous tumor, both in skin and elsewhere, have been middle-aged to older adults, but children have developed the tumor and the editor has observed one case in an infant. Large pleural lesions may produce pulmonary symptoms or hypertrophic osteoarthropathy, and hypoglycemia may occur in a few cases as the result of secretion of insulin-like growth factors. These symptoms have not been reported with cutaneous lesions to date.
Classically, the solitary fibrous tumor is benign, responding well to excision or wedge resection in the case of pleural tumors. However, 10% to 20% of lesions in other organs have been malignant. Cutaneous tumors have thus far behaved in a benign manner; one occipital lesion did show invasion of the underlying calvarium, but it responded to local excision with no recurrence in 6 months of follow-up.
The tumor cells are predominantly fibroblastic, although focal myofibroblastic differentiation is often seen. This tumor was originally included with the hemangiopericytomas. The “driver” mutation of solitary fibrous tumor appears to be an NAB2–STAT6 gene fusion; upregulation of GRIA2 has also been described.
Treatment of cutaneous cases is surgical excision of the lesion.
Solitary fibrous tumors are well circumscribed and found in the dermis and subcutis. They are composed of short-spindled and ovoid cells with scant cytoplasm that are best known for forming a “patternless pattern,” although the cells may also be arranged in short fascicles or storiform configuration ( Fig. 35.14 ). Cellular areas are interspersed with less cellular zones of thick, hyalinized collagen, occasionally with myxoid foci. Vessels may be numerous, and they may take on “staghorn” contours, thereby mimicking hemangiopericytoma. Mast cells are not frequent in these tumors. The constituent spindled cells usually have a bland appearance, and mitotic figures are rare. Malignant variants (not yet reported in skin) feature increased cellularity, pleomorphism, and mitotic activity. A value greater than four mitoses per 10 high-power fields has been used as a criterion to define malignant solitary fibrous tumors. A histopathologically malignant solitary fibrous tumor has been reported on the scalp. It featured nuclear crowding, pleomorphism, geographical necrosis, and a mitotic rate of 8 per 10 high-power fields; however, no recurrence or metastasis had been reported with an 18-month follow-up period.
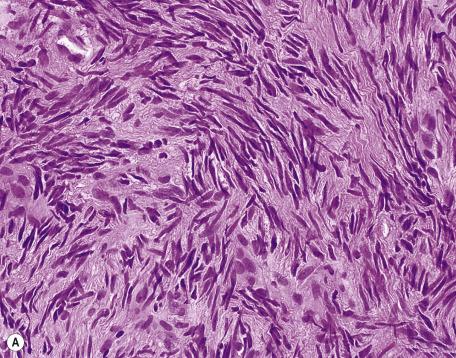
The giant cell angiofibroma, originally described in the orbit and subsequently in other locations, including the skin, appears to be a giant cell-rich variant of solitary fibrous tumor.
Immunohistochemical staining regularly shows CD34 expression by spindle cells, and this feature has come to represent part of the definition of solitary fibrous tumor. Vimentin is also usually positive. Staining for bcl-2 is often observed. Focal staining for CD99 also occurs. Interspersed dermal dendritic cells are positive for factor XIIIa, and factor XIIIa + dendrocytes were present in an oral lesion. Staining for desmin is usually negative, and negative results are obtained when staining for keratin and epithelial membrane antigen. The mesothelial markers calretinin and HBME-1 have not been detected in five dermal cases.
Other markers that have been reported to be negative include cytokeratin AE1/AE3, ERG, S100 and SMA. In connection with molecular findings, STAT6 nuclear staining is considered a good immunomarker for the NAB2–STAT6 gene fusion, and similarly, immunostaining for GRIA2 can reflect the upregulation of the GRIA2 gene.
On ultrastructural examination, there are cells with both fibroblastic and myofibroblastic features.
Spindle cell lipoma is potentially a close mimic of solitary fibrous tumor, as noted by Sigel and Goldblum. However, features of spindle cell lipoma that differ from those of solitary fibrous tumor include overwhelming occurrence in middle-aged to elderly men on the posterior neck or shoulder, interspersed mature lipocytes, and numerous mast cells. Hemangiopericytoma shares with solitary fibrous tumor both a similar morphology and immunohistochemistry, suggesting that these could be variants of the same entity. However, the spindled cells as well as their arrangements in solitary fibrous tumor differ from those of classic hemangiopericytoma. In contrast to solitary fibrous tumor, hemangiopericytomas lack hyalinized collagen and do contain mast cells. Furthermore, hemangiopericytoma-like foci are seen in a wide variety of tumors, some of which may actually represent myofibromas. Dermatofibromas often feature epidermal hyperplasia and xanthomatization, and CD34 expression is typically either absent or focal. Dermatofibrosarcoma protuberans (DFSP) is poorly circumscribed, usually has a distinctive storiform pattern, and lacks either thick, hyalinized collagen or prominent vasculature. The cells of monophasic synovial sarcoma of the spindle cell variety express keratin and EMA and are negative for CD34, in contrast to solitary fibrous tumor. About 75% of cases of DFSP show positive GRIA2 reactivity, which is also found in solitary fibrous tumor. However, all reported cases of DFSP so far have been negative for STAT6, which therefore may be a useful differentiating marker.
Fibrous hamartoma of infancy is an uncommon fibroproliferative lesion of the subcutaneous tissue that is present at birth or develops in the first 2 years of life. It most commonly occurs around the shoulder, axilla, and upper arms, but cases involving the scalp, inguinal region, scrotum, vulva, perianal area, and lower extremities have been reported. Two recent cases have involved the eyelid. There have been examples of giant (10.5 × 8.5 × 4.5 cm) and/or multifocal lesions. There is a male predominance of 3 : 1. The clinical course is benign, despite its infiltrative appearance and tendency to local recurrence. It has been reported in an infant with Williams’ syndrome (see p. 433 ). A reciprocal translocation [t(2;3)(q31;q21)] was present in one case. Another study showed complex translocations that involved chromosomes 1, 2, 4, and 17. Complex chromosomal abnormalities were also found in two cases of fibrous hamartoma of infancy with sarcomatous areas. Epidermal growth factor receptor (EGFR) exon 20 insertion/duplication mutations have now been reported in at least five cases of fibrous hamartoma of infancy.
The subcutaneous tissue of excised lesions has a glistening gray-white appearance interspersed with fatty tissue. The involved area measures 2 to 8 cm in maximum diameter.
This tumor should be treated by complete excision; an aggressive approach should be avoided because the overall prognosis is excellent. Recurrences, when they happen, take place within the first year after surgery. However, a recent report described a case that recurred 14 years after the primary surgery.
Fibrous hamartoma of infancy has poorly defined margins. It is centered on the subcutis. There are three different tissue components : interlacing trabeculae of fibrocollagenous tissue, interspersed mature fat, and small nests of loosely arranged mesenchymal cells ( Fig. 35.15 ). The fibrous trabeculae vary in thickness and arrangement and contain spindle-shaped cells; they express vimentin, SMA, muscle-specific actin and CD34 but not S100 protein. The mature adipose tissue is positive for S100 protein, and the Immature mesenchymal cells express CD34 and, in one study, bcl-2. There is negative staining for desmin, neuron-specific enolase, β-catenin and Ki-67. An additional finding that has been recently emphasized is that of densely collagenized, hypocellular areas with pseudoangiomatous slit-like spaces lined by spindle cells resembling those within mesenchymal nests; these areas have been variously described as “pseudoangiomatous” and “giant cell fibroblastoma-like” foci—the latter partly a result of the presence of mesenchymal cells with multilobated nuclei. In one study, FISH analysis for PDGFB gene rearrangements was negative in the five tested cases. Sparse lymphocytes may be present in the stroma.
The overlying skin often shows eccrine changes that include hyperplasia, duct dilatation, intraluminal papillary formations, and squamous syringometaplasia. Primitive mesenchymal cells may replace the normal eccrine gland stroma. Increased numbers of terminal hair follicles, microfollicles, abortive hair follicles, and epidermal basaloid follicular hyperplasia have also been reported. Another case that presented with clinical dimpling and small white nodules showed, on microscopic examination, partial dermal thinning, a follicular unit with features of folliculosebaceous cystic hamartoma, smooth muscle aggregates, and keratinous cysts.
The constituent cells have the features of myofibroblasts, although some fibroblasts are also present. Fibroblasts alone were present in one reported case. Primitive mesenchymal cells are present in the immature-appearing areas.
Digital fibromatosis of childhood (infantile digital fibromatosis, recurring digital fibrous tumor of childhood, inclusion body fibromatosis ) is a rare benign tumor of myofibroblasts with characteristic cytoplasmic inclusion bodies. It presents as a dome-shaped firm nodule, up to 1 cm in diameter, usually on the digits. The thumbs and great toes are spared. Lesions are usually solitary, but a second tumor is sometimes noted at the time of presentation or develops subsequently. The tumors may be present at birth or appear in the first year of life. Onset in late childhood or adult life is rare. A history of preceding trauma has been reported. There is a rare syndrome consisting of recurrent digital fibroma, focal dermal hypoplasia, and limb malformations.
A viral etiology was originally suspected because of the characteristic inclusion bodies, but all cultures have been negative, excluding this hypothesis. The inclusions are now known to be filamentous aggregations composed largely of actin.
The tumor often recurs after local excision; very occasionally, it may regress spontaneously, but this may be accompanied by functional impairment. Spontaneous regression increases after long-term follow-up, casting some doubt on the benefits of surgical excision with its high (up to 60%) recurrence rate. Intralesional fluorouracil has also proven effective in the treatment of this entity.
The tumor is nonencapsulated and extends from beneath the epidermis, through the dermis, and usually into the subcutis. It is composed of interlacing bundles of spindle-shaped cells and collagen bundles. There may be some vertical orientation of the cells and fibers superficially ( Fig. 35.16 ). The appendages become incorporated within the tumor. The nuclei of the cells are oval or spindle shaped, and some stellate forms may be present. There are only occasional mitoses. The cytoplasm of the cells merges imperceptibly with the collagen. There are characteristic small eosinophilic inclusion bodies within the cytoplasm of the tumor cells, often in a paranuclear position ( Fig. 35.17A ). A clear halo is sometimes discernible in well-stained sections. These bodies measure 2 to 10 µm in diameter, and they may be mistaken for red blood cells. They stain red with the Masson trichrome stain and deep purple with the PTAH method ( Fig. 35.17B ). They are periodic acid–Schiff (PAS) negative but actin positive. In addition, the inclusion bodies stain with antibodies to calponin 1 using an enzymatic antigen retrieval method.
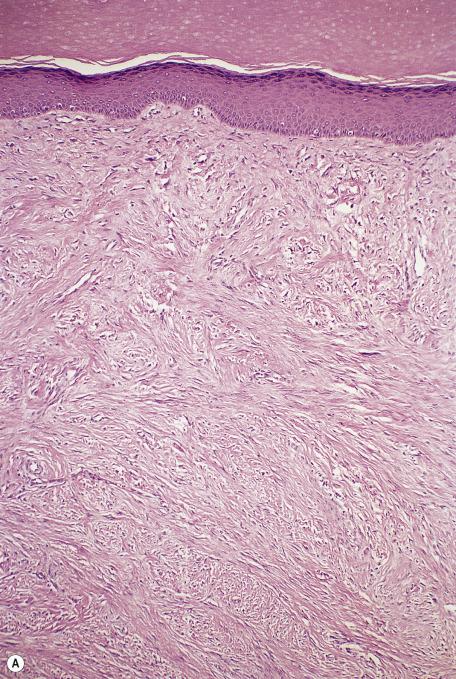
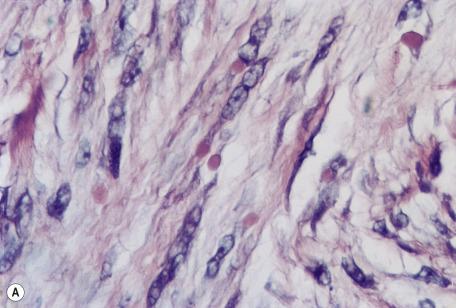
There are small capillaries and a few scattered elastic fibers in the stroma. Lymphocytes are variable in number. The overlying epidermis usually shows flattening of the rete ridges. Ulceration is rare.
The immunocytochemical localization of vimentin and muscle-specific actin in the proliferating cells confirms their myofibroblastic nature. The cells also express desmin.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here