Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Tuberculosis (TB) is a granulomatous disease caused by the slow-growing, acid-fast bacillus Mycobacterium tuberculosis . Prior to the emergence of severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2; Chapter 337 ), TB was the leading infectious disease cause of death worldwide. People become infected with M. tuberculosis by inhaling infectious organisms through aerosol transmission from a person who has active TB.
The M. tuberculosis complex is a group of highly genetically related species that cause tuberculosis in humans, livestock, and wildlife. M. tuberculosis and Mycobacterium africanum , which cause TB in humans, are obligate human pathogens with no known environmental reservoir. Other members of the M. tuberculosis complex cause infection in distinct mammalian hosts, including Mycobacterium bovis in cattle, Mycobacterium caprae in goats and sheep, Mycobacterium pinnipedii in sea lions, and Mycobacterium microti in voles. Although humans with active TB can infect animals, subsequent transmission of M. tuberculosis from animal to animal or back to humans is extremely rare. Animal-adapted members of the M. tuberculosis complex can also infect humans, most often when M. bovis (or Mycobacterium orygis in Africa and Southeast Asia) is retransmitted from infected cattle or unpasteurized dairy products, but subsequent human-to-human transmission is rare. Members of the M. tuberculosis complex likely arose from a common ancestor that resembles Mycobacterium canettii , which is a species that is phylogenetically positioned between mammalian pathogens and environmental mycobacteria and that has been isolated in rare cases of human tuberculosis from the Horn of Africa.
M. tuberculosis is a nonmotile, non-spore-forming, facultative anaerobe that is rod-shaped and approximately 2 to 4 μm in length. Unique features of M. tuberculosis include its “waxy” cell envelope, extremely slow growth, and ability to replicate intracellularly. M. tuberculosis doubles every 18 to 24 hours, so culturing the bacilli from clinical specimens may take weeks. The waxy coat prevents decolorization of carbol fuchsin stain after washing with strong acids, thereby leading to the characteristic “acid-fast” staining. A plasma membrane, peptidoglycan layer, arabinogalactan layer, outer membrane, and capsule comprise the cell envelope. The outer membrane contains mycolic acids and a complex array of unique lipids, such as phthiocerol dimycocerosate, phenolic glycolipids, trehalose dimycolates, and sulfolipids. This complex cell envelope, which is important for pathogenesis and transmission, also creates a formidable barrier to the entry of antibiotics. The ability of M. tuberculosis to withstand adverse conditions and disrupt host immunity depends upon this unique cell envelope.
M. tuberculosis is classified into distinct phylogenetic lineages that are thought to have originated in the Horn of Africa and spread to other parts of the world in association with human migration. The more ancestral strains (lineages 1 and 5 to 7) are geographically restricted. Lineages 5 and 6 are M. africanum lineages found almost exclusively in West Africa. The “modern” M. tuberculosis lineages (lineages 2 to 4) are responsible for the global epidemic of TB. Although all lineages cause the same spectrum of disease, lineage appears to impact pathogenesis and epidemiologic spread. For example, certain lineage 2 strains are more likely to cause severe pulmonary infection, to develop drug resistance more rapidly, and to be more transmissible. Lineage identification is not part of routine clinical care, but more detailed strain typing is useful in investigations of outbreaks to establish whether cases are epidemiologically linked. Strain typing can be performed by restriction fragment length polymorphism of the insertional element IS6110, spoligotyping, mycobacterial interspersed repetitive unit analysis, and whole-genome sequencing. In addition, genotyping can be used to distinguish relapse from reinfection in high burden settings and in relatively rare instances of laboratory cross-contamination of diagnostic specimens.
The World Health Organization (WHO) estimates that 1.7 billion people, a quarter of the world’s population, are latently infected with M. tuberculosis . In 2018, an estimated 10 million new active cases of TB and 1.2 million deaths occurred worldwide, with the majority occurring in low- and middle-income countries. As a result, TB was the leading infectious disease killer prior to being overtaken by SARS-CoV-2 during the coronavirus disease 2019 (COVID-19) pandemic. Worldwide, 35% of cases occur in women, and the prevalence of disease peaks in young adults. HIV has a profound impact on the epidemiology of TB: 10% of TB cases worldwide occur in people with HIV infection, and TB is the leading cause of death among persons living with HIV ( Chapter 358 ).
The risk of TB depends upon the country of origin. The prevalence of latent TB infection is estimated to be 4% in the United States but exceeds 20% in Southeast Asia, the Western Pacific, and Africa. The same geographic disparity is seen for active disease. In Southeast Asia, the incidence of TB is 220 per 100,000 persons per year, whereas most high-income countries have an incidence of less than 10 per 100,000 persons per year ( Fig. 299-1 ). In high-burden settings with substantial ongoing transmission, active TB most often occurs because of progression during the first 2 years after initial infection. In low burden settings, however, active TB is more likely to result from reactivation of more long-standing latent infection. Although most people develop an adaptive immune response that controls infection, this response does not prevent reinfection. In high-burden settings, individuals previously treated for TB are at increased risk of being infected with a new strain of M. tuberculosis compared with individuals without a TB history.

For over 100 years, the incidence of TB has declined in the United States, with the notable exception of 1984 to 1992, when the rise of the HIV epidemic led to an increasing incidence. In 2019, the 8920 reported active cases (2.7 per 100,000 persons) were the lowest since reporting began in the early 1950s but still far from the goal of eliminating TB, which is generally defined as an incidence below one case per million people per year. In the United States, the majority (about 70%) of TB cases occur in immigrants from high-incidence countries. This elevated risk of TB disease persists in immigrants from high-prevalence countries long after their arrival in the United States; for example, approximately 45% of new TB cases among foreign-born residents were diagnosed 10 or more years after they first arrived in the United States. Other risk factors for TB infection and progression to active disease include having household contact with active pulmonary TB, HIV disease or other causes of immunosuppression, residence in a homeless shelter or correctional faculty, diabetes mellitus, and underlying lung disease ( Table 299-1 ). In general, people who are at highest risk for active TB disease are individuals who were recently infected and who are immunosuppressed. These risk factors also inform the interpretation of tuberculin skin testing and treatment strategies for latent TB (see later).
| RISK FACTOR | INCREASED RISK OF RECENT INFECTION ∗ | INCREASED RISK OF PROGRESSION TO ACTIVE DISEASE | DEFINITION OF A POSITIVE TB SKIN TEST |
|---|---|---|---|
| Household contact of active pulmonary TB | X | ≥5 mm | |
| Solid organ transplant recipient, immunosuppressive treatment (TNF-α inhibitors, prednisone >15 mg/day for >1 month), fibrotic lesion on chest radiograph consistent with prior TB | X | ≥5 mm | |
| HIV infection | X | ≥5 mm | |
| Birth or extended residence in higher-prevalence countries; persons using nonprescription injectable drugs who are known to be HIV-negative; children under 5 years old; children and adolescents exposed to adults in high-risk categories | X | ≥10 mm | |
| Residents or employees in hospitals, homeless shelters, correctional facilities, nursing homes, residences for HIV-infected | X | ≥10 mm | |
| <90% IBW or BMI ≤20; cigarette smoking; silicosis; diabetes mellitus (particularly insulin-dependent or poorly controlled); end-stage renal disease; post-gastrectomy or jejuno-ileal bypass; carcinoma of the head and neck; lung cancer; lymphoma; leukemia | X | ≥10 mm | |
| None | ≥15 mm |
∗ Recent infection itself also increases the risk of progressing to active disease.
The worldwide problem of TB is compounded by drug resistance. In 2019, about 3% of new TB cases and 18% of previously treated cases had drug-resistant TB. Multidrug-resistant TB (resistant to both isoniazid and rifampin) exists worldwide, with the majority of cases occurring in India (27%), China (14%), and Russia (8%). Extensively drug-resistant (XDR) TB, which is resistant to isoniazid, rifampin, fluoroquinolones, and at least one of the three second-line injectable agents, is estimated to occur in about 10% of patients with multidrug-resistant TB. The epidemic of drug resistance is driven both by transmission of drug-resistant isolates and the acquisition of drug resistance due to inappropriate treatment.
In response to the rise in HIV/TB coinfections and multidrug-resistant M. tuberculosis in 2013, the WHO declared TB a global public health emergency and developed the End TB Strategy. Unfortunately, the COVID-19 pandemic has interfered with these efforts. TB services have been disrupted by worldwide lockdowns, overwhelmed health care systems, and the reallocation of health care workers and resources to care for COVID-19 patients. In 2020, 1.4 million fewer TB cases were reported worldwide than in 2019, almost certainly owing to underreporting rather than a true reduction in incidence. The reduction in detection and, hence, in prompt treatment in 2020 is projected to result in 0.5 million excess TB deaths, with worldwide TB mortality climbing back to levels last seen in 2010.
M. tuberculosis effectively disarms the innate immune response, persists in the face of adaptive immunity, and disseminates, thereby leading to a wide spectrum of infectious manifestations. The infection cycle begins when M. tuberculosis is transmitted from a person with active pulmonary TB through aerosolized particles that are small enough to reach the alveoli (1 to 5 μM; Fig. 299-2 ). The degree of infectiousness depends upon the severity of cough, which is promoted by sulfolipid in the mycobacterial cell envelope, and the extent of bacterial burden. Patients with cavitary lesions, which can have 10 7 to 10 9 bacilli, are the most infectious. Patients who do not have visible acid-fast bacilli on sputum smears are much less likely to transmit, but they still can infect others. Patients with HIV are less likely to develop cavitary disease, to be smear positive, or to transmit infection. On average, infectious individuals will infect 3 to 10 people per year. In household settings, where exposure may be intense and prolonged, 25 to 50% of contacts become infected.
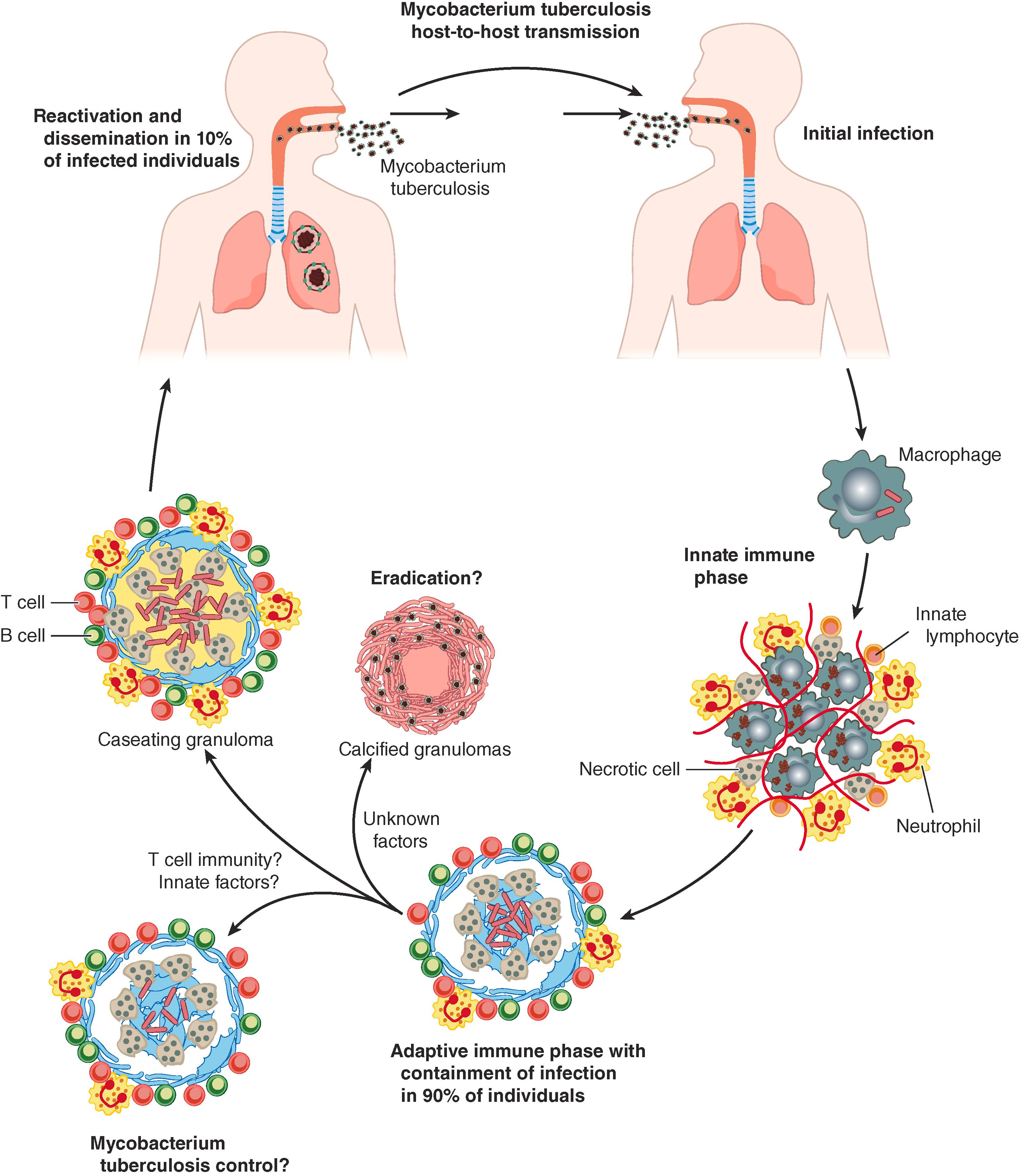
A central feature of the pathogenesis of M. tuberculosis is its ability to survive and grow in host phagocytic cells that normally clear infection. When M. tuberculosis bacilli reach the distal alveoli, they are first taken up by alveolar macrophages, which normally kill microbes and clear debris, but they are poorly equipped to handle M. tuberculosis ( Fig. 299-2 ). Over time, monocyte-derived macrophages, interstitial macrophages, and neutrophils are recruited to the site of infection, but they also fail to control M. tuberculosis replication. Infected dendritic cells travel to the draining lymph node, where presentation of M. tuberculosis antigens leads to priming and expansion of antigen-specific T cells, which return to the infected lung. Once an adaptive immune response develops, T cells, B cells, and activated macrophages form the characteristic granuloma, and bacterial control is established. The interval from the initial infection to the development of a positive tuberculin skin test and/or IFN-γ release assay (IGRA) is usually between 2 and 8 weeks. This delay in the adaptive immune response allows M. tuberculosis to establish infection, which is often asymptomatic, and grow relatively unhindered for weeks.
Some individuals may sterilize the initial infection by virtue of their innate immune response by clearing bacilli before an adaptive immune response has time to develop. Individuals who are highly exposed to M. tuberculosis and appear to resist infection, as evidenced by persistently negative tuberculin skin test and/or IGRA, appear to have increased expression of metabolic pathways that respond to free fatty acids as well as increased M. tuberculosis– specific antibody responses.
Granulomas, which are the histopathologic hallmark of M. tuberculosis infection, are thought to be important in preventing progressive disease after infection is established. Granulomas are an organized collection of macrophages, neutrophils, and lymphocytes. Macrophages within the granuloma can undergo an epithelioid transformation, form multinucleated giant cells, or become lipid-laden and “foamy” in appearance. Characteristically, the granulomas become necrotic and have a caseating appearance (like cheese). Over time, these granulomas calcify. CD4 T cells and TNF-α are critical in the formation of granulomas and to host protection. CD4 T cells secrete cytokines, such as IFN-γ, which promote the microbicidal activities of macrophages. Patients with CD4 T-cell dysfunction from HIV infection or patients treated with TNF-α inhibitors fail to form well-organized granulomas, and they are at high risk for developing active TB and disseminated infection. Similarly, patients who have inborn errors of IFN-γ signaling or who acquire IFN-γ neutralizing antibodies are highly susceptible to active disease and have a greater likelihood of developing disseminated and recurrent disease. Although granulomas prevent progressive infection, they may also facilitate bacterial persistence. Granulomas may enhance bacterial spread between aggregated macrophages, create an immunosuppressive environment, and keep CD4 T cells peripherally located, away from the more central, infected macrophages, thereby obstructing key immune cell interactions. Even within an infected individual, different granulomas establish different degrees of immune control.
Once infected, some patients will develop active disease over the first year of infection (termed progressive primary infection). However, most often, the initial infection, while disseminated, is controlled, thereby resulting in latent tuberculosis infection. Although the distinction between latent tuberculosis infection and active disease is extremely important clinically, the classification oversimplifies what is a more complex spectrum of infectious stages that include bacterial elimination and subclinical disease ( Fig. 299-3 ). Some patients diagnosed with latent tuberculosis infection may sterilize the infection, but it is impossible, with current diagnostics, to tell whether the tubercle bacilli have been cleared by the adaptive immune response or are still viable but controlled.
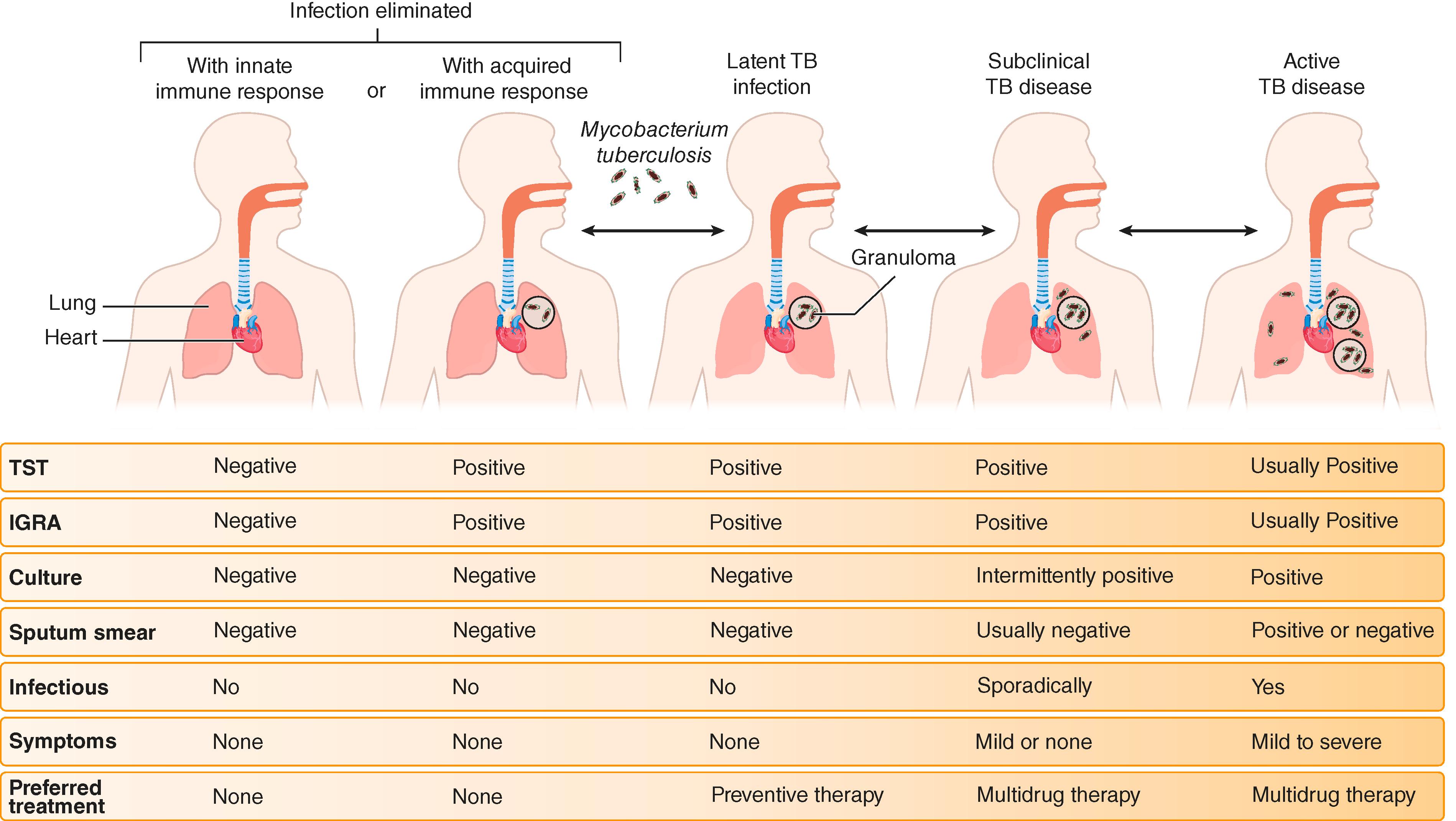
Recent infection and a variety of conditions (see Table 299-1 ) increase the risk of active disease. HIV infection ( Chapter 358 ) is the strongest known risk factor, even among individuals with relatively preserved CD4 T-cell counts. The majority of patients with active TB have no identified immunologic risk factor, but neutrophil-driven inflammation and a type I IFN response seem to promote progressive disease.
Most cases of primary TB go clinically unrecognized and resolve without treatment. Individuals may develop fever, shortness of breath, nonproductive cough, and, rarely, erythema nodosum. Chest radiographs may show a small area of patchy opacity in the mid-lung fields (called a Ghon focus) with associated hilar lymphadenopathy (together called a Ghon complex) ( Fig. 299-4A ). Patchy, linear, or nodular parenchymal opacities may be seen. Lymphadenopathy, which is often unilateral, involves the hilum and right paratracheal nodes. Enlarged lymph nodes can cause bronchial compression, thereby leading to upper or middle lobe collapse. A transient pleural effusion may develop ( Fig. 299-4B ).

In most cases, the outcome of primary TB is latent tuberculosis infection. However, particularly in the absence of an effective adaptive immune response, primary infection can progress to pulmonary and extrapulmonary disease, including TB meningitis and miliary TB. Progressive primary infection is more likely to occur in children, the elderly, and the immunocompromised.
Most often, the immune response controls primary TB, thereby leading to clinically silent infection. Diagnosis of latent tuberculosis infection is made by demonstrating an immunologic response to M. tuberculosis antigens. Chest radiography may show evidence of prior infection; the Ghon complex may leave a calcified Ghon lesion in the mid-lung fields or calcified hilar nodes (when found together this is referred to as a Ranke complex). However, these radiographic findings are nonspecific and can also occur in other granulomatous infections, such as healed histoplasmosis ( Chapter 308 ).
Active TB disease most often involves the lungs. When pulmonary TB occurs within the first year of infection, it is called progressive primary infection. Pulmonary TB that occurs more distant to the initial infection is considered reactivation of latent tuberculosis infection (also termed “post-primary disease”). The most common symptom of pulmonary TB is a persistent cough, which can be productive or nonproductive and accompanied by fever, chills, night sweats, and weight loss; hemoptysis may occur in advanced disease. However, up to 25% of persons with culture-confirmed pulmonary TB do not report cough.
Physical examination findings are not specific. There may be abnormal breath sounds, such as post-tussive rales in the upper lung zones. Amphoric breath sounds can indicate a cavity. Lymphadenopathy is not common in immunocompetent adults with pulmonary TB.
Leukocytosis and anemia may be present. Progressive primary disease typically results in dense consolidation on chest radiography, with a predominant involvement of the middle and lower lobes ( Fig. 299-4C,D ), but the clinical and radiographic features of progressive primary and reactivation disease overlap. Reactivation pulmonary TB usually manifests as cavitary disease involving the apical and posterior segments of the right upper lobe and the apical-posterior segment of the left upper lobe, distant from the site of the primary infection ( Fig. 299-5 ). Other radiographic findings can include fibrotic scars, loss of lung volume, and calcification. Endobronchial spread to lower portions of the lungs can lead to radiographic tree-in-bud opacities. Erosion into blood vessels or lymphatics can lead to dissemination and a miliary pattern.
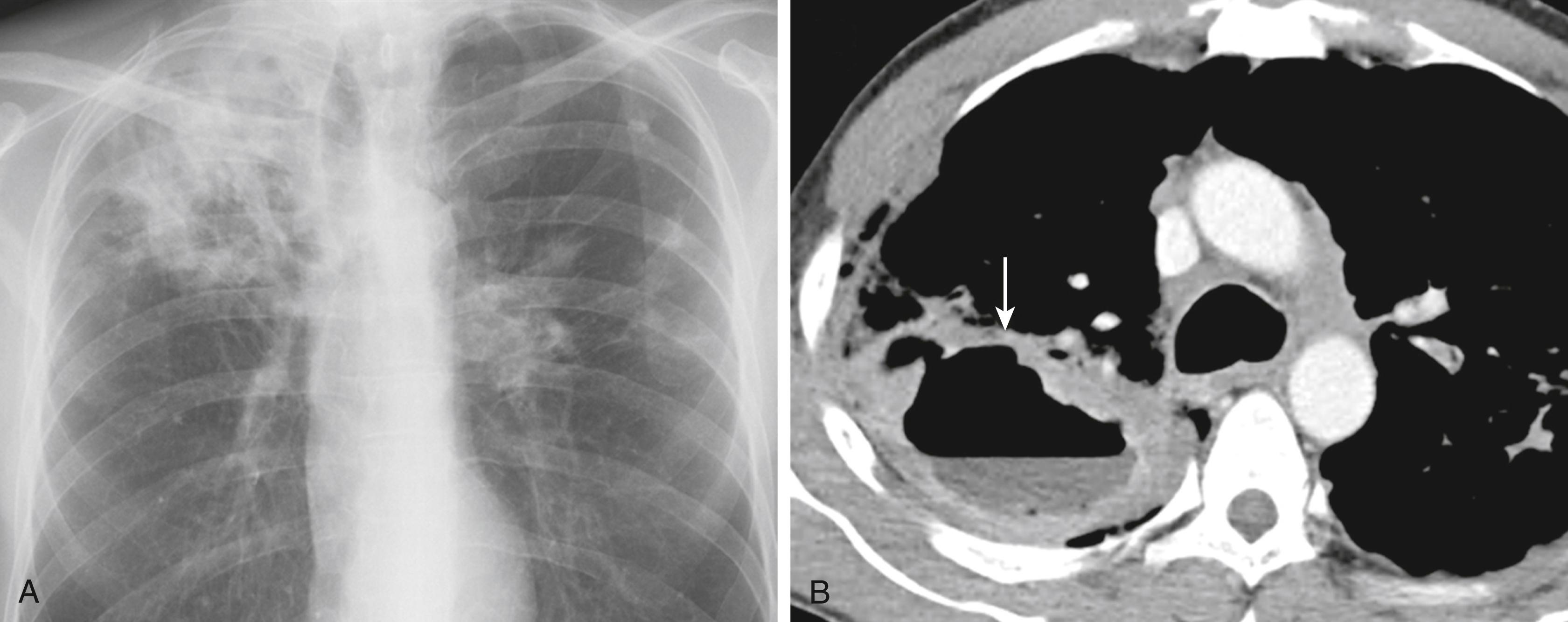
In patients coinfected with HIV ( Chapter 358 ), the clinical manifestations depend upon the degree of immune dysfunction. Disease in HIV-infected patients with relatively preserved immune function may be indistinguishable from disease in immunocompetent hosts, but as the degree of immunosuppression increases, the disease manifestations become more atypical. Persons living with HIV are less likely than immunocompetent individuals to be sputum-smear positive, less likely to have cavitary disease, and more likely to develop disseminated infection. Although most patients with pulmonary TB have abnormal chest radiographs, persons with HIV may have a normal chest radiograph despite smear-positive disease; a computed tomographic (CT) scan is more sensitive in these cases.
Because primary infection is a disseminated process prior to immunologic control, extrapulmonary disease can affect nearly any organ system. Extrapulmonary disease can also mimic a wide array of other disorders, and it can be difficult to make a microbiologic diagnosis, since the bacterial burden may be relatively low and the site of infection may be difficult to access.
Involvement of the pleural space can occur during primary infection or reactivation. Most often TB pleuritis occurs when a small number of organisms gain access to the pleural space and elicit an inflammatory response. During primary infection, TB pleuritis may be clinically silent and resolve spontaneously, or it can result in cough, pleuritic chest pain, and systemic symptoms, with associated exudative pleural effusions ( Fig. 299-4B ). Pleural involvement most often occurs 3 to 6 months after primary infection. In some cases, effusions can be large and cause dyspnea, but most often they are small and unilateral. More rarely, TB can cause an empyema when a large number of bacilli erode into the pleural space from a cavity or parenchymal focus.
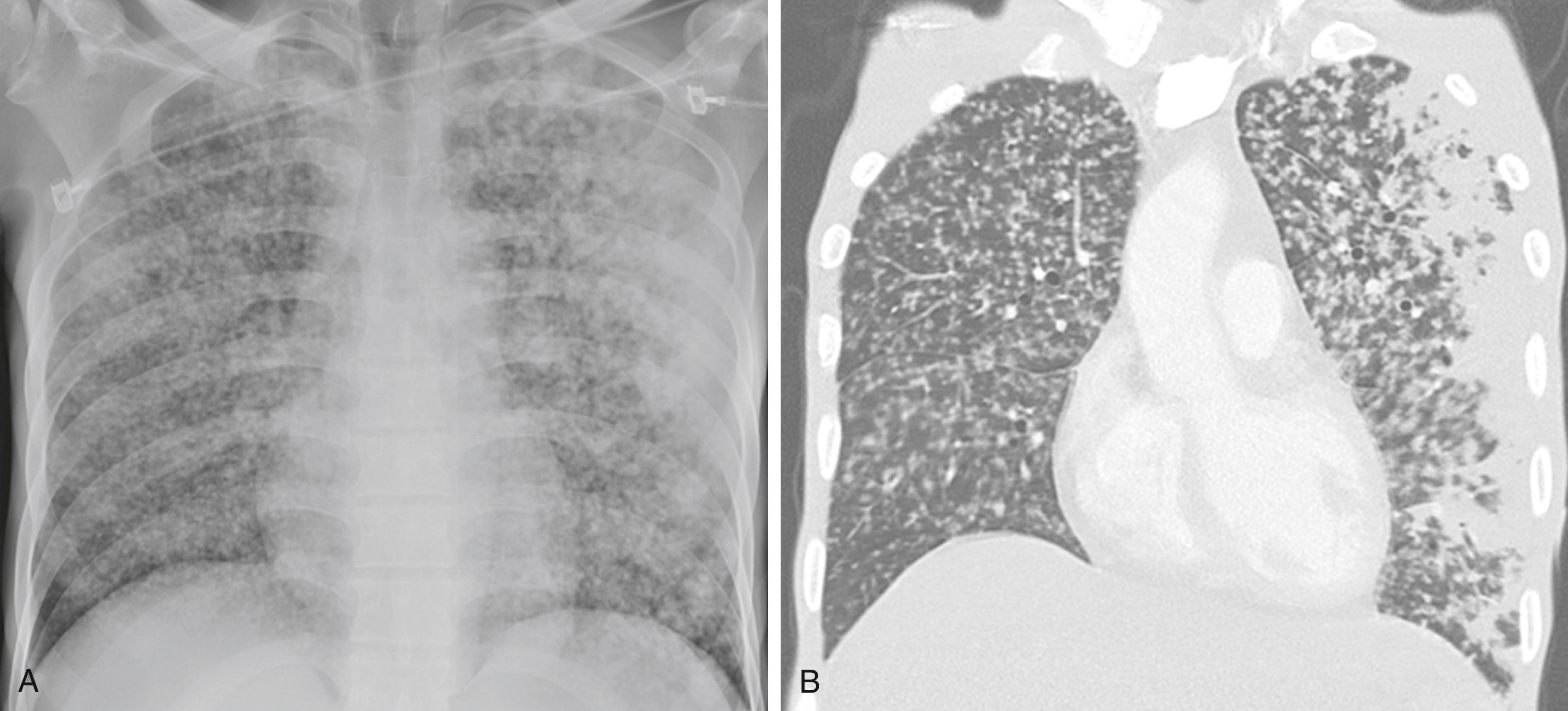
Miliary TB is a multisystem form of TB in which 1- to 2-mm granulomatous nodules are found in the lungs and other organ systems. The most commonly involved organs are lungs, liver, bone marrow, kidney, adrenal glands, and spleen. Miliary TB usually presents with systemic symptoms (including fever, weight loss, and night sweats), often in the absence of localizing symptoms. The physical examination also may reveal choroidal tubercles (white-yellow plaques on fundoscopic exam), lymphadenopathy, and hepatomegaly. Chest radiographic abnormalities may initially be subtle, with the classic miliary pattern seen in about 85% of patients ( Fig. 299-6 ). Patients also may have other chest radiographic abnormalities, such as cavities, pleural effusions, or pericardial effusions. Since HIV-infected patients and patients who are being treated with TNF-α inhibitors may not form well-circumscribed granulomas, their findings on plain chest radiography may be absent or subtle, and a CT scan is more sensitive. In 20 to 40% of patients with advanced AIDS and miliary TB, blood cultures are positive for M. tuberculosis .
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here