Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
![]() Access video lecture content for this chapter online at Elsevier eBooks+
Access video lecture content for this chapter online at Elsevier eBooks+
Breast augmentation is one of the most commonly performed aesthetic surgery procedures worldwide, with breast implants being a staple of reconstructive and aesthetic surgery.
The ability of the human body’s tissue to expand when applying constant pressure to it has been used by plastic surgeons to treat and reconstruct a multitude of defects and malformations. For tissue expansion an (inflatable) expander is temporarily placed under the body’s tissue and constantly inflated gradually. This constant pressure leads to an increase in skin, along with increased vascularity. Depending on the expander’s position, lengthening of muscles, bones, and nerves can additionally be achieved. In some reconstructive breast patients, permanent expanders may be used with the advantage of avoiding expander to implant exchange.
Breast implants can be inserted immediately or shortly after the expansion of the overlying tissue with a temporary expander.
Tissue expansion as well as breast implantation require precise patient selection and planning with meticulous attention to detail.
Mechanotransduction is the conversion of mechanical forces into biochemical signals and an integral part of tissue expansion. Skin that is stretched beyond its physiological limits (mechanical stress) activates mechanotransduction and a multitude of pathways increasing mitotic activity, collagen synthesis, cell growth, and ultimately resulting in the formation of new tissues and skin surface area with a significantly higher vascularity of expanded tissue.
Multiple findings have reported that fast changes in extension, alignment, and collagen adapt to mechanical expansion like strain or stretch. Both elastin and collagen realign in a parallel fashion in response to expansion. Elongation occurs to the direction of mechanical stretching. Strain beyond physiological limit leads to changes of cell function. This feature serves as the basis for the clinical implications of tissue expansion.
Several studies have focused on cellular and molecular mechanisms, including growth factors, cascades, cytoskeleton, protein kinases, DNA synthesis, and gene expression. These mechanisms are all involved in the expansion of skin. Mechanical stress activates mechanosensitive ion channels, G-proteins, protein kinases, integrin-matrix interactions, and many more which helps to convert physical cues to biologic responses. Fibroblasts prepare to proliferate in response to mechanical stretch. The extracellular matrix is essential for strain-induced cell proliferation. Extracellular forces transmitted lead to the deformation of the matrix, followed by alteration of membrane and adhesion complexes.
As a conclusion, mechanical stimulation is capable of activating highly integrated signaling cascades. For tissue expansion this characteristic helps enlarging skin.
Tissue expansion has been shown to induce angiogenesis and ischemia on the overlying skin.
Studies have shown that the level of gene expression, vascular endothelial growth factor (VEGF), platelet-derived growth factor (PDGF), and basic fibroblast growth factor (bFGF) is significantly higher expressed in expanded than in non-expanded tissue, leading to increases in the flap´s robustness and the possibility of harvesting a larger flap.
For centuries, the concept of applying external forces to initiate tissue expansion has been well known and was first reported for bone elongation by Codvilla in 1905. Codvilla is thought to be one of the trailblazers in bone tissue regeneration and was followed by many more, including Ilizarov. In 1957, Neumann was the first to achieve soft-tissue growth by subcutaneously implanting a latex balloon for reconstructing part of an external ear. Later, Radovan used tissue expansion with two valves to cover an arm defect, while in 1977 Austad and Rose used a salt-osmotically driven self‑inflating silicone expander manufactured by Dow Corning Medical Products.
The first recognized attempt of reconstructive breast augmentation was performed in the nineteenth century by Czerny (1895) where he transplanted an autologous lipoma into a patient’s breast to fill a defect. In 1904, paraffin injections were used resulting in serious foreign body responses like formation of hard masses or paraffinomas, as well as inflammatory reactions, tissue necrosis, pulmonary embolism, and cerebral embolism. After failed attempts of injecting liquid silicone, Cronin and Gerow (in conjunction with the Dow Corning Center for Aid to Medical Research) developed the first silicone-filled implant where the silicone gel was confined in a synthetic, separate elastomer shell (1962). In 1963, this model was commercially manufactured by Down Corning and introduced the age of breast implants in plastic surgery. Almost simultaneously, in 1964, the first saline-filled breast implant was manufactured in France. However, in 1992, the Food and Drug Administration (FDA) announced a moratorium for silicone-gel breast implants in the US because of a potential connection between silicone implants and various systemic diseases which made only saline-filled implants available for aesthetic breast augmentation. About 14 years later, after multiple evidence-based studies had been performed, the FDA lifted the ban for silicone-gel implants for breast reconstruction as well as aesthetic breast augmentation in 2006.
Since their development, breast implants have undergone a myriad of changes adjusting and improving the surface, filling, and coating of implants with the available to-date fifth generation of silicone-gel implants containing a highly cohesive silicone gel.
Although critically judged in the first place, expanders and breast implants are a mainstay of modern medicine and play a key role in the armamentarium of plastic and reconstructive surgery.
Tissue expanders are a staple of reconstructive surgery for the correction of soft-tissue defects. Because living tissue responds dynamically to mechanical forces, lost tissue can be replaced by expanding neighboring tissue of similar color, texture, sensation, hair, and thickness. Additionally, a remote donor site can be avoided.
The use of tissue expanders in reconstructive surgery is widespread and can be applied to almost any part of the human body. Tissue expanders’ main usage is in reconstructive breast surgery. The growth of tissue while expanding is permanent, but will retract to some degree once the expander is removed.
Tissue expanders are expandable, balloon-like medical devices that allow the body to “grow” additional skin through applying controlled mechanical stretch beyond its physiological limits (mechanotransduction). These expanders are made of silicone and fillers and can be placed temporarily or permanently.
Modern tissue expanders have undergone several improvements over the decades. Nowadays, most of them contain just one remote valve which is connected by a flexible filling tube to the expander. The tubing is especially beneficial when the breast pocket is too firm to safely keep an expander with an integrated valve or when the overlying skin is very thin and fragile. The remote position of the valve can easily be palpated under the skin. Thanks to the remote valve and Silastic filling tube, inflating the expander via saline injection can be done without the risks of puncturing the balloon.
Additionally, there are expanders with an integrated valve system on their surface where the inflation reservoir is integrated within the expander. Even though these expanders avoid the risk of mechanical problems or potential bacterial colonization of the filling tube, the risk of accidentally piercing the expander during inflation is higher than with remote valves. Magnetic metal-finding or ultrasonic devices have been proven helpful to properly locate the position of the valve.
In addition to these types, there are self-inflating expanders. These hydrogel (vinyl pyrrolidone and methyl methacrylate) expanders were developed for decreasing pain and the number of in-patient visits. Nowadays, self-inflating expanders consist of a plastic shell to lower uncontrolled expansion. The active hydrogel of these expanders absorbs extracellular fluid via osmosis resulting in enlargement of the expander within 6–8 weeks. In addition to the osmotic hydrogel expander there is a self-inflating expander which uses carbon dioxide for inflation by a remote radio-control. One disadvantage of self-inflating expanders is possible continued expansion beyond vital tissue vascularity.
Common tissue expanders come in circular, rectangular or crescentic shapes with volumes ranging from 50 to 1000 cc.
Differential expanders are designed to produce more expansion in one part than in the other around the surface area of the same expander. This feature is frequently used in breast reconstruction to improve the lower pole of the breast, for example an anatomical expander which best mimics the natural breast shape. Once the skin expansion is sufficient, these temporary anatomic expanders are replaced by permanent breast implants. Besides the aforementioned expanders, the use of a permanent breast expander can be of advantage over a temporary expander or a fixed-volume implant in some patients. The Becker expander‑implant contains a double-lumen two-chamber system ( Fig. 26.1 ). Here, the expander’s inner chamber is filled with saline during the expansion while its outer chamber is already prefilled with gel similar to an implant. Thus, skin expansion can be pursued until skin extent is adequately completed with this expander being left as a permanent breast implant. The Becker system also has a remote valve with a filling tube which can be detached by traction at the end of the expansion. This allows automatic sealing of the saline‑filled chamber and functions as a permanent implant thereafter.
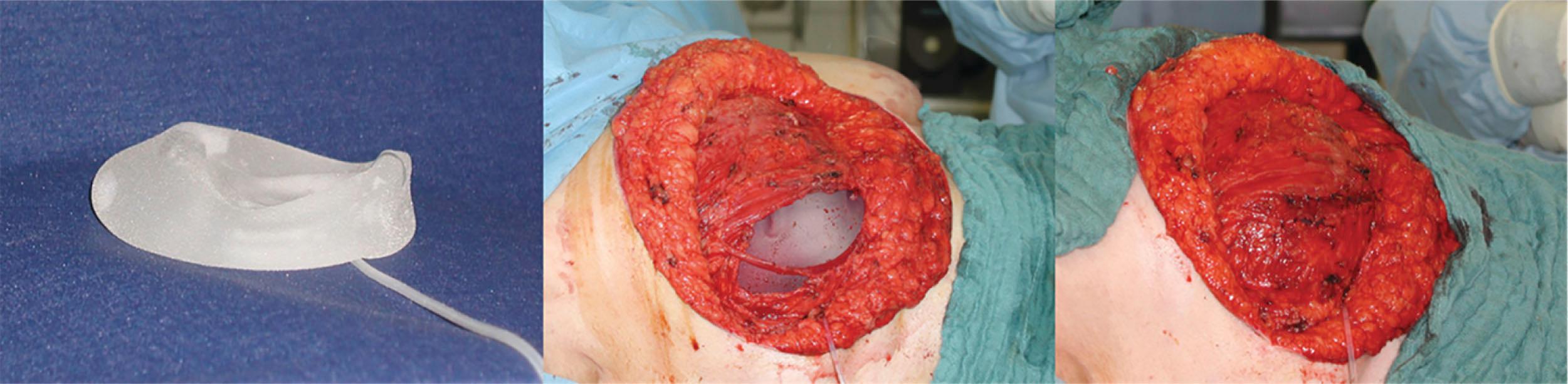
For difficult and uncommon defects, custom‑built expanders can individually be produced with specifications related to volume, width, length, and projection.
Since the early 1960s, there have been five generations of breast implants based on characteristics of shell, filler, shape, and surface configuration. The clinical behavior of a given implant may be based on its respective generation.
Most commercially available breast implants feature some degree of elastomer surface modifications to increase surface roughness. However, manufacturing processes of breast implants differ from company to company, including a differently used taxonomy, complicating valid scientific comparisons between breast implants of different companies.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here