Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
This chapter provides an overview of the principles of newborn skin care followed by a summary of the many diseases of the newborn presenting with primary or secondary cutaneous manifestations. A basic understanding of the structural development of the skin as well as the multiple functions subserved by the skin during transition to extrauterine life will be highlighted. The caregiver will be able to distinguish benign and transient lesions of newborn skin, such as erythema toxicum, from potential life-threatening diseases such as herpes simplex neonatorum. In addition, the chapter highlights the fact that many common cutaneous findings in the newborn, such as sebaceous gland hyperplasia and neonatal pustular melanosis, have an intrauterine etiology. The advantage of the accessibility of the skin to physical examination is counterbalanced by the extreme structural and functional diversity of this organ.
The skin is a critical interface between the body and environment. This layer differentiates between “self” and “nonself.” At birth, the first response in resuscitation includes drying the baby to prevent heat loss and an assessment of skin color, perfusion, and integrity. Pathologic processes visible on the skin surface range from general signs of systemic dysfunction (cyanosis, pallor, jaundice) to clinical evidence of specific diseases (vesicles, petechiae). Cutaneous characteristics are routinely used as determinants of gestational age (breast buds, plantar creases, desquamation). The definitive organ of mammals, the mammary gland, similar to the epidermis, is an ectodermally derived cutaneous structure. Maternal–infant bonding is, in large part, a complex dynamic interaction between skin surfaces.
At birth, the skin of the term infant must perform multiple functions critical for survival in an extrauterine environment ( Box 94.1 ). Many of these functions depend on structural development of the skin during the last trimester of pregnancy. The development of a competent epidermal barrier, for example, is essential for temperature regulation, maintenance of fluid homeostasis, infection control, and prevention of penetration of environmental toxins and drugs. Similarly, the acquisition of the skin microbiome is important in regulating keratinocyte homeostasis to locally produce hormones, neurotransmitters, and cytokines, which in turn maintain epidermal integrity, enhance innate immunity, and inhibit pathogenic bacteria.
Barrier to water loss
Thermoregulation
Infection control
Immunosurveillance
Acid mantle formation
Antioxidant function
Ultraviolet light photoprotection
Barrier to chemicals
Tactile discrimination
Attraction to caregiver
The epidermal permeability barrier primarily resides in the outermost layer of the epidermis, the stratum corneum. This layer, approximately one-fourth the thickness of a sheet of paper, develops in utero during the third trimester of pregnancy in conjunction with a protective mantle of vernix caseosa. Extremely low birth weight preterm infants (<1000 g birth weight) lack a well-developed stratum corneum, which poses special problems for newborn care. The stratum corneum is necessary for the adherence of thermistors, cardiorespiratory monitors, and endotracheal tubes, and forms the primary environmental interface with caregivers and parents. Box 94.2 gives a summary and consensus of general principles of skin care.
Provide immediate drying and tactile stimulation.
Remove blood and meconium.
Leave vernix intact and spread to allow absorption.
Limit frequency.
Use neutral pH cleansers.
Use only water for infants weighing less than 1000 g.
Avoid antibacterial soaps.
Avoid petrolatum-based ointments in infants with very low birth weight during the first week of life.
Use emollients as needed for dryness in older infants.
Use chlorhexidine, and remove excess after procedure.
Use of isopropyl alcohol and alcohol-based disinfectants is discouraged in infants with very low birth weight until the stratum corneum has formed.
Minimize use of adhesives.
Use hydrogel electrodes.
Avoid solvents and bonding agents.
Counsel patience during tape removal.
For infants younger than 30 weeks’ gestation, select high incubator humidity (>60%) and transparent, semipermeable dressings to reduce evaporative water and heat loss.
Use incubators and supplemental conductive heat to avoid drying effects of radiant warmers.
Measure humidity routinely.
Allow natural drying.
Avoid the routine use of isopropyl alcohol.
Human skin has two distinct but interdependent components—the epidermis and the dermis ( Fig. 94.1 ). The epidermis has marked regional variations in thickness, color, permeability, and surface chemical components. It consists of a highly ordered, compact layering of keratinocytes and melanocytes. Traditionally, the epidermis is segmented into distinct structural and functional compartments called the stratum germinativum (basale), stratum spinosum, stratum granulosum, and stratum corneum (see Fig. 94.1 ). Intermixed is a third distinct cell type, the Langerhans cell, which is derived from bone marrow precursors and migrates into the primitive epidermis.
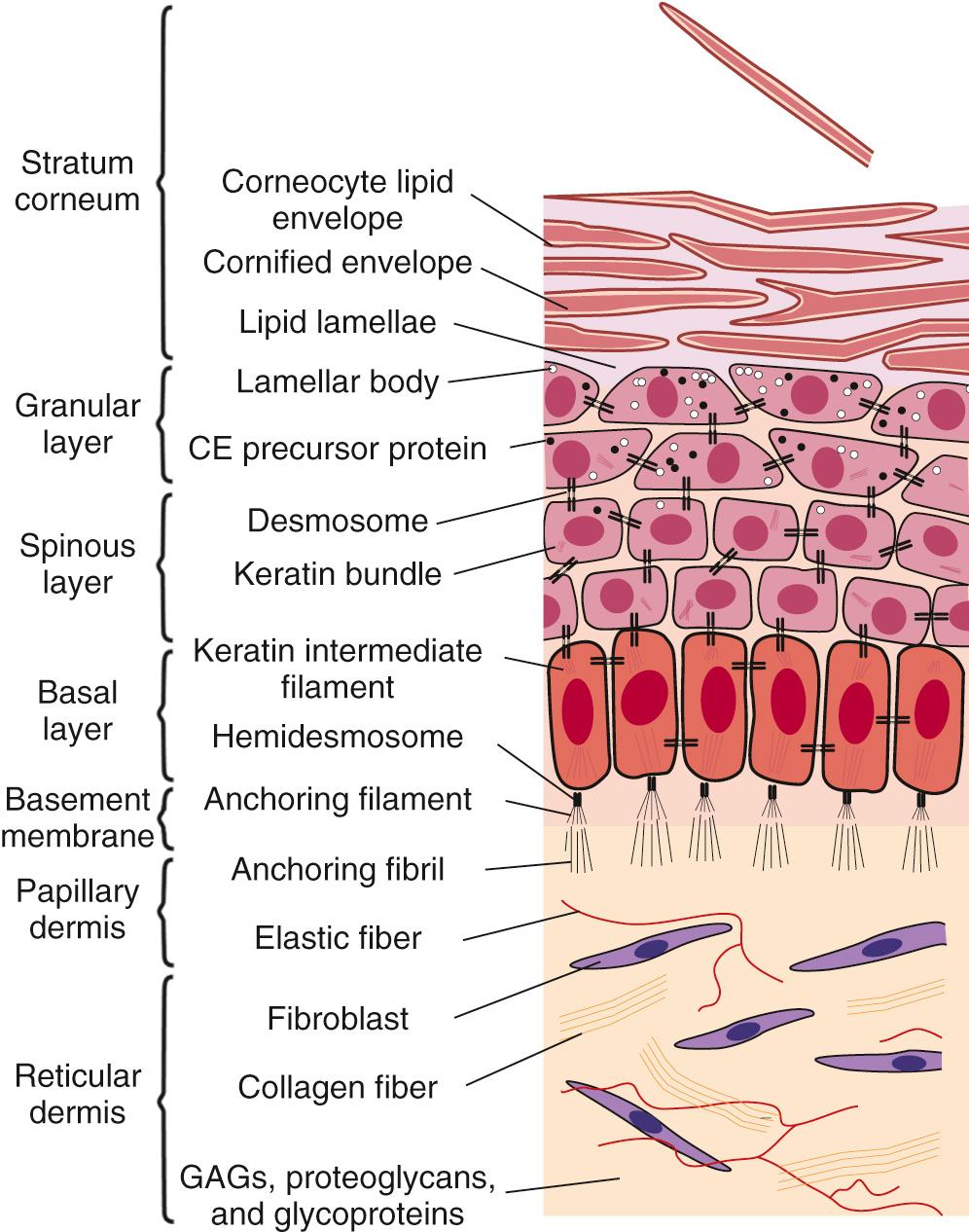
The process of cutaneous morphogenesis can be divided into embryonic and fetal periods ( Fig. 94.2 ). This transition, which occurs at approximately 2 months, is an important time in skin development, because many critical morphogenetic events occur during this transitional period. Between 30 and 40 days of development, the embryonic skin consists of a two-layered epidermis: the basal layer, associated with the basal lamina, and the periderm, which serves as a cover and a presumptive nutritional interface with the amniotic fluid. The basal layer includes cells that give rise to the future definitive epidermis, whereas the periderm is a transient layer that covers the embryo and fetus until the epidermis keratinizes at the end of the second trimester. Basal cells join with each other and peridermal cells by a relatively few desmosomes but do not yet form hemidesmosomes with the basement membrane. Small numbers of keratin intermediate filaments are associated with these junctions. Matrix adhesion of the embryonic epidermis is likely mediated by actin-associated α6β4 integrin.
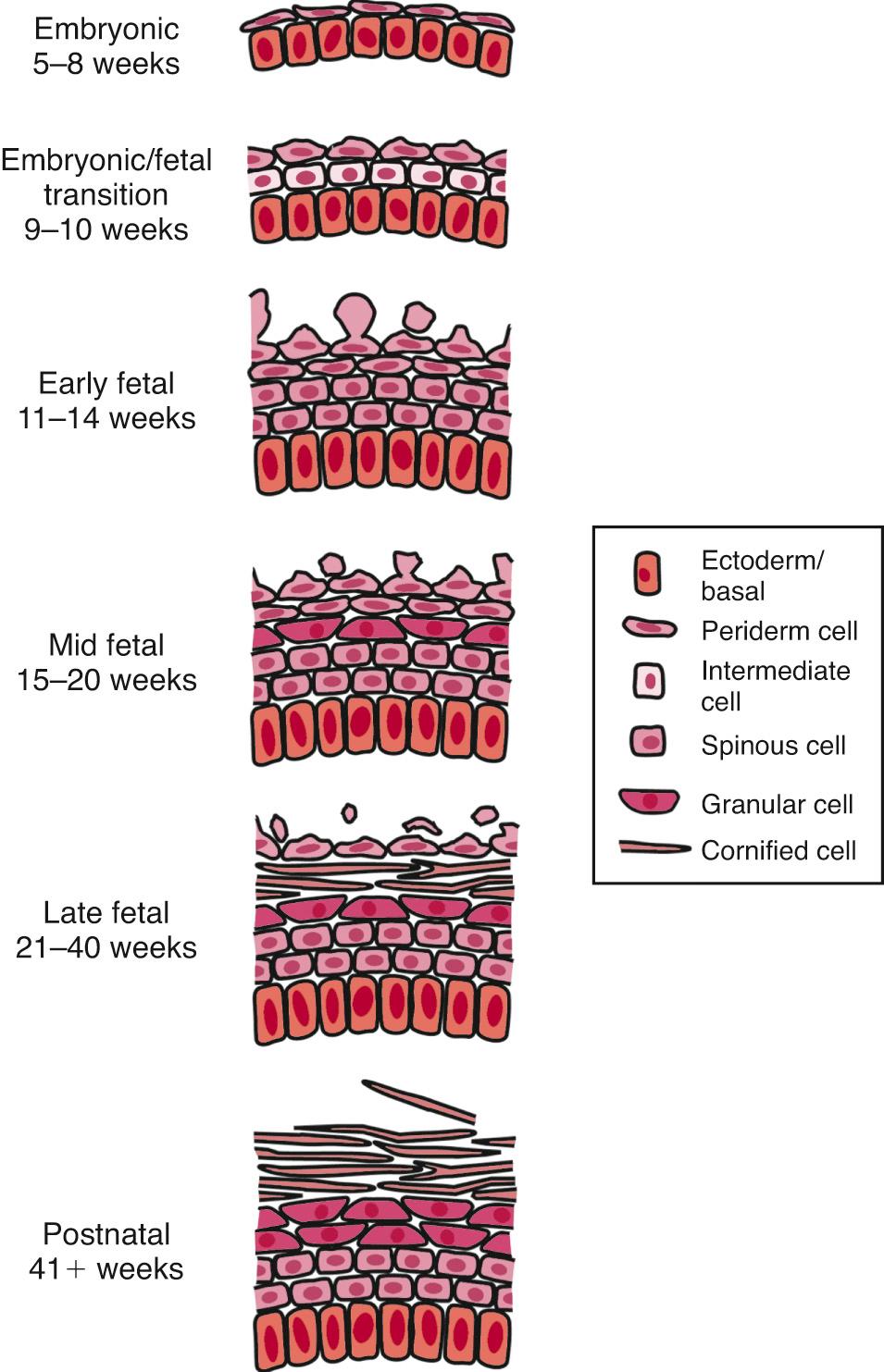
Langerhans cells and melanocytes migrate into the embryonic epidermis and are identifiable at 40 and 50 days of gestation, respectively. At this stage, the Langerhans cells do not express CD1 antigen on their cell surfaces, nor are Langerhans cell granules identifiable. Likewise, at this time, the melanocyte lacks the characteristic cytoplasmic organelle, the melanosome. A third immigrant cell, the Merkel cell, does not appear to be present in embryonic epidermis and may differentiate at a later stage from keratinocytes in situ.
At the time of embryonic–fetal transition (60 days’ gestation), the epidermis begins to stratify, forming an intermediate layer of cells. These young keratinocytes still contain a high volume of glycogen in their cytoplasm and produce large amounts of intermediate filaments in association with the desmosomes. At this time, new keratins are identifiable as markers of differentiation, as is the pemphigus antigen, which is detectable on the cell surface. These cells, unlike adult spinous cells, remain proliferative and continue to express epidermal growth factor receptors on their surfaces. Two to three additional layers of intermediate cells are added during the second trimester. These cells show a progressive increase in the number of keratin filaments but do not further differentiate until the onset of keratinization in the interappendageal epidermis around 22-24 weeks’ gestation.
The Langerhans cells in the embryonic epidermis begin to express CD1 antigen and contain Langerhans granules after the embryonic–fetal transition. The numbers of Langerhans cells increase significantly during the third trimester; however, the function of these cells in fetal skin remains unknown. Between 80 and 90 days’ gestation, melanocytes also increase in density at a time when keratinocytes in the epidermis are forming appendages, and melanin synthesis begins at the end of the first trimester. Transfer of melanosomes to keratinocytes occurs in the 5th month of gestation.
Melanin production is low in the newborn, who is not as pigmented as the older child and is more sensitive to sunlight, but there is no significant difference in the number of melanocytes in light, medium, or deeply pigmented skin or in infant or adult skin. Difference in color is the result of the shape, size, chemical structure, and distribution of melanosomes and the activity of individual melanocytes.
The appendages derive from embryonic invaginations of epidermal germinative buds into the dermis. They include hair, sebaceous glands, apocrine glands, eccrine glands, and nails. The arrector pili muscle is attached to the hair follicle. Lanugo (i.e., fine, soft, immature hair) frequently covers the scalp and brow of the premature infant. The scalp line may be poorly demarcated, and lanugo also may cover the face. Scalp hair is usually somewhat coarser and matures earlier in dark-haired infants.
The growth phases of the hair follicle are usually synchronous at birth. Eighty percent of the follicles are in the resting state. During the first few months of life, the synchrony between hair loss and regrowth is altered so that 20% of scalp hairs are growing in the same phase at the same time. Hair may become coarse and thick, acquiring an adult distribution, or there may be temporary alopecia.
There are sex differences in hair growth; boys’ hair grows faster than girls’ hair. In both sexes, scalp hair growth is slower at the crown. The normal pattern of hair growth is disturbed in many chromosomal disorders. Evidence supports a genetic link between the formation of clockwise posterior parietal hair whorls and specific (right) handedness of the individual. This finding supports an unexpected association between gene-based hair patterning and human brain development that warrants further substantiation. Supporting the significance of the skin–brain connection, evidence has shown the human hair follicle can synthesize cortisol de novo and is a functional equivalent of the hypothalamic-pituitary-adrenal axis.
The eccrine sweat glands are coiled structures that form during the first trimester as a downgrowth from epidermal cellular clones. No new glands are formed after birth. They are distributed over the entire body surface and are innervated by sympathetic cholinergic nerves. During the first 24 hours of life, term infants usually do not sweat; on approximately the 3rd day, sweating begins on the face. Palmar sweating begins later. Thermal sweating as a function of body or environmental temperature should be distinguished from emotional sweating associated with crying or pain. The latter is best assessed on the palms and soles.
The sebaceous glands differentiate primarily from the epithelial portion of the hair follicle at approximately 13-15 weeks of life and almost immediately produce sebum in all hairy areas. The glands develop as solid outpouchings from the upper third of the hair follicle. These solid buds become filled with liquid centrally where the cells disintegrate; acini and ducts develop, opening most frequently into the canals between the hair follicle wall and hair shaft. Ectopic glands occur occasionally on the lips, buccal mucosa, esophagus, and vagina.
Surprisingly little is known about the regulation of the rapid growth and activity of sebaceous glands up to and immediately after birth. Hypothetically, secretion by the fetal adrenal gland of weak androgens such as dehydroepiandrosterone (DHEA) is followed by intra-sebocyte conversion to testosterone and production of products of the pilosebaceous unit such as vernix caseosa. This hypothesis remains to be fully tested, but it has the singular advantage of functionally integrating two disparate and unexplained facts; that is, the mutual hyperplasia of the sebaceous gland and the adrenal cortex during the latter half of gestation in the human fetus. Androgens are the only hormones that unequivocally have a stimulating effect on the sebaceous glands; estrogens depress their growth. Between 2 months and 2 years, the sebaceous glands of the normal infant begin a period of quiescence that lasts until puberty.
The apocrine glands are relatively large organs that originate from and empty into a hair follicle. Embryologically, they develop somewhat later than the eccrine glands. Apocrine development is advanced by 7 or 8 fetal months, when the glands begin to produce a milky-white fluid containing water, lipids, protein, reducing sugars, ferric iron, and ammonia. In the newborn, the acini are well formed. The biologic function of the gland is unknown in infants, but it is related to pheromone production in other animal species.
Vernix caseosa, a unique material that coats the fetal skin surface during the last trimester of pregnancy, contains lipids of sebaceous origin as well as numerous water-laden fetal corneocytes. A growing body of evidence supports the hypothesis that vernix caseosa participates in regionally “waterproofing” the skin surface, thereby allowing cornification to occur, initially around the hair follicles and then over the interfollicular skin. A better understanding of vernix caseosa and fetal sebaceous gland physiology is biologically relevant and may be applicable to care of the very low birth weight infant and to clinical situations in which the epidermal surface is inadequately developed, burned, or traumatized.
Physiologically, vernix is a hydrophobic material containing wax esters, cholesterol, ceramides, and squalene in addition to other minor lipid components. Vernix has high water content (≈80%), with the water primarily distributed within flat, polygonal, cornified squames. As term approaches, vernix detaches from the fetal skin surface under the influence of pulmonary surfactant and is swallowed by the fetus. In close analogy to breast milk, vernix contains multiple molecules associated with the innate immune system, including lysozyme, lactoferrin, and defensins, as well as antioxidants such as alpha tocopherol. Vernix possesses both emollient (moisturizing) and cleansing functions. Thus, at birth, the human infant is covered with a complex material possessing endogenous anti-infective, antioxidative, moisturizing, and cleansing capabilities. Extreme prematurity, as well as postmaturity, is associated with diminished vernix on the skin surface at birth.
The fetal dermis acquires some of the characteristics of adult dermis around 20 weeks’ gestation. However, it is only in the more mature fetus that the true structural and biochemical qualities of the newborn dermis are detectable. The dermis contains three identifiable zones: the region adjacent to the basement membrane, the papillary dermis, and the reticular dermis (see Fig. 94.1 ). The superficial dermis has finer collagen fibers and is biochemically more active than the deeper zone. The papillary dermis also is more susceptible to light injury and elastic tissue degeneration than the reticular dermis. The dermis has a symbiotic relationship with, and may exert a controlling influence on, the epidermis. It is a metabolically active tissue that contains fibrous elements, amorphous ground substance, free cells, nerves, blood vessels, and lymphatics.
The fibrous elements are collagen and elastic tissue. Collagen types I, III, V, and VI comprise the dermal collagens, whereas type IV is present in the basement membrane of the dermal–epidermal junction and dermal vessels, and type VII occurs in anchoring fibrils and basal keratinocytes. With increasing age, collagen becomes progressively less soluble, and thicker bundles predominate. The morphologic characteristics and chemical properties of cutaneous elastic fibers differ from those of collagen. Elastic fibers are first detectable in the skin by histochemical means and electron microscopy at around 20 weeks’ gestation.
The fibroblasts are the most numerous cells in the dermis. They produce collagen and the glycosaminoglycans of the ground substance. Mast cells (which produce heparin and histamine), histiocytes, macrophages, lymphocytes, neutrophils, and an occasional plasma cell and eosinophil are also present in the dermis. The major glycosaminoglycans in the ground substance of skin are hyaluronic acid and dermatan sulfate (i.e., chondroitin sulfate B). An increase in age brings a shift from hyaluronic acid toward dermatan sulfate and a decrease in water content of the dermis.
The vascular network supplying the skin develops in early embryonic life from the mesoderm. In 60- to 70-day-old fetuses, there are two vascular networks: a superficial one that appears to correspond to the subpapillary plexus of adult skin and a deeper network that represents the deep reticular vascular network of adult skin. New capillary beds organize to supply the developing epidermal appendages from the 5th month of gestation. These two networks persist into the newborn period; however, the arrangement of vessels remains unstable for the first year of life, and further organization of the vascular network occurs postnatally.
Vasomotor tone is controlled by a delicate and complex series of neural and pharmacologic mechanisms that involve the sympathetic nervous system, norepinephrine, acetylcholine, and histamine, and may involve serotonin, vasoactive polypeptides, corticosteroids, and prostaglandins. The physiologic requirements of skin vary considerably; skin blood flow ranges from 0.1-150 mL/mm 2 of tissue per minute.
The nerve networks in the dermis develop at a very early embryologic age and appear to be distributed in a random fashion. The most superficial nerves have the smallest diameter and are the least myelinated. In addition to the dermal nerve network, which may show considerable regional variation, nerve fibers may serve particular regions or structures such as hair follicles, eccrine glands, arrector pili muscles, and the subepidermal zone. Superficial nerves conduct mechanosensory stimuli from the specialized Merkel cells within the epidermis. The sebaceous glands are not innervated.
Special neurologic structures include a dense perifollicular nerve network with exquisite tactile sensory properties and mucocutaneous end organs highly concentrated in erogenous zones. Meissner tactile organs are found in newborn skin as undeveloped structures that mature after birth. Merkel corpuscles probably govern two-point tactile discrimination on palms and fingertips; they are disk-shaped terminals that are seen during the 28th week of fetal life. After birth, these receptors undergo little alteration. Vater-Pacini corpuscles are found around the digits, palms, and genitals; they are fully formed and numerous at birth.
The arrector pili muscles are innervated by sympathetic nerves, and norepinephrine acts as the neurotransmitter. The eccrine glands are innervated by sympathetic fibers, but acetylcholine acts as the neurotransmitter. Parasympathetic fibers may accompany the sensory nerves in the vessel walls and cause vasodilation. The axon reflex is poorly developed in the newborn; in the neonate of low birth weight, axon reflex sweating may be difficult to elicit.
This section provides a cursory synopsis of normal and abnormal aspects of newborn skin. An accurate description of primary and secondary skin lesions forms the basis for the diagnosis of skin pathology. Tables 94.1 and 94.2 describe the basic lesional morphology of infant skin with specific examples. More definitive treatises on normal skin structure, function, development, and specific disease states are available. The remainder of this chapter delineates specific conditions of newborn skin ranging from transient lesions to definitive cutaneous diseases and diagnostic categories.
| Type | Description | Clinical Examples |
|---|---|---|
| Macule | A circumscribed, flat lesion with color change, up to 1 cm in size; by definition, they are not palpable | Ash leaf macules, café-au-lait spots, capillary ectasias |
| Patch | Same as macule but >1 cm in size | Nevus depigmentosus, nevus simplex, mongolian spots |
| Papule | A circumscribed, elevated, solid lesion, up to 1 cm in size; elevation may be accentuated with oblique lighting | Verrucae, milia, juvenile xanthogranuloma |
| Plaque | A circumscribed, elevated, plateau-like, solid lesion, >1 cm in size | Mastocytoma, nevus sebaceous |
| Nodule | A circumscribed, elevated, solid lesion with depth, up to 2 cm in size | Dermoid cysts, neuroblastoma |
| Tumor | Same as a nodule but >2 cm in size | Hemangioma, lipoma, rhabdomyosarcoma |
| Vesicle | A circumscribed, elevated, fluid-filled lesion up to 1 cm in size | Herpes simplex, varicella, miliaria crystalline |
| Bulla | Same as a vesicle but >1 cm in size | Sucking blisters, epidermolysis bullosa, bullous impetigo |
| Wheal | A circumscribed, elevated, edematous, often evanescent lesion, caused by accumulation of fluid within the dermis | Urticaria, bite reactions, drug eruptions |
| Pustule | A circumscribed, elevated lesion filled within purulent fluid, <1 cm in size | Neonatal pustular melanosis, erythema toxicum neonatorum, infantile acropustulosis |
| Abscess | Same as a pustule but >1 cm in size | Pyodermas |
* Lesions arise de novo and are therefore most characteristic of the disease process.
| Type | Description | Clinical Examples |
|---|---|---|
| Crust | Results from dried exudates overlying an impaired epidermis. Can be composed of serum, blood, or pus. | Epidermolysis bullosa, impetigo |
| Scale | Results from increased shedding or accumulation of stratum corneum as a result of abnormal keratinization and exfoliation. Can be subdivided further into pityriasiform (branny, delicate), psoriasiform (thick, white, and adherent), and ichthyosiform (fish scalelike). | Ichthyoses, postmaturity desquamation, seborrheic dermatitis |
| Erosion | Intraepithelial loss of epidermis. Heals without scarring. | Herpes simplex, certain types of epidermolysis bullosa |
| Ulcer | Full thickness loss of the epidermis with damage into the dermis. Will heal with scarring. | Ulcerated hemangiomas, aplasia cutis congenita |
| Fissure | Linear, often painful break within the skin surface, as a result of excessive xerosis. | Inherited keratodermas, hand and foot eczema |
| Lichenification | Thickening of the epidermis with exaggeration of normal skin markings caused by chronic scratching or rubbing. | Sucking callus, atopic dermatitis |
| Atrophy | Localized diminution of skin. Epidermal atrophy results in a translucent epidermis with increased wrinkling, whereas dermal atrophy results in depression of the skin with retained skin markings. Use of topical steroids can result in epidermal atrophy, whereas intralesional steroids may result in dermal atrophy. | Aplasia cutis congenita, intrauterine scarring, and focal dermal hypoplasia |
| Scar | Permanent fibrotic skin changes that develop as a consequence of tissue injury. In utero scarring can occur as a result of certain infections or amniocentesis or postnatally from a variety of external factors. | Congenital varicella, aplasia cutis congenita |
* Lesions arise as characteristic modifications of primary lesions through environmental interaction (e.g., drying) or subject interaction (e.g., scratching).
A number of benign and transient lesions of the skin are commonly observed in a normal nursery population. It is important for the infant caregiver to distinguish such ephemeral lesions from significant life-threatening diseases with cutaneous manifestations.
Approximately 8% of infants have multiple, white, 1-mm cysts (i.e., milia) scattered over the cheeks, forehead, nose, and nasolabial folds. These milia may be few or numerous, but they frequently occur in clusters. Milia are often confused with the more common condition of sebaceous gland hyperplasia (43% of infants), which manifests as smaller, flatter, more yellow dots seen on the midface as well ( Fig. 94.3 ). Histologically, milia are keratogenous cysts similar to Epstein pearls, which are distributed along the midpalatal raphe. Bohn nodules are cysts that occur on the palate and the buccal and lingual aspects of the dental ridges and represent remnants of mucous gland tissue. Intraoral lesions are found in 75%-80% of newborns. Because all these cysts exfoliate or involute spontaneously within the first few weeks of life, no treatment is required.
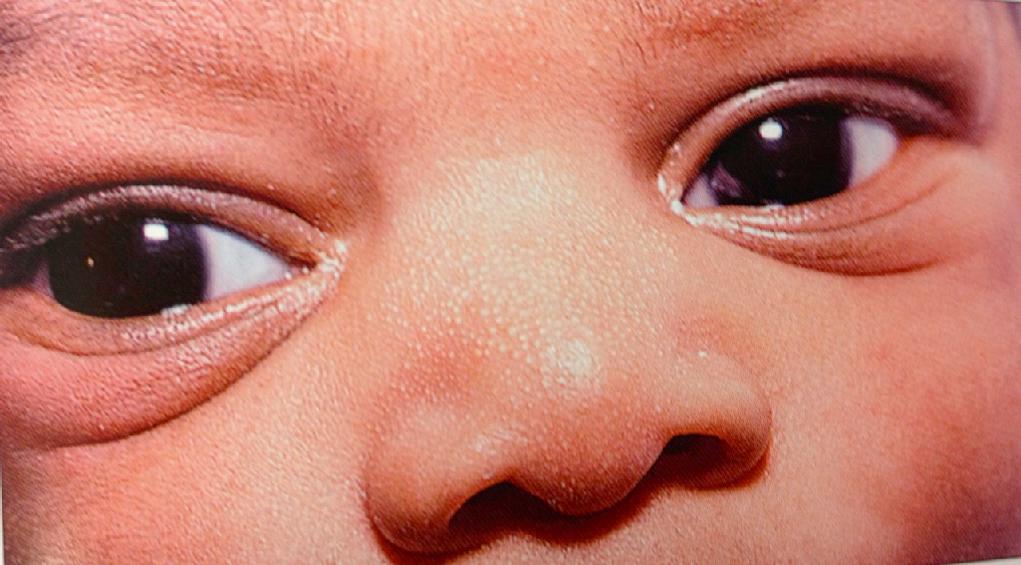
The most frequently encountered pigmented lesion is the mongolian spot (dermal melanosis), which occurs frequently in African-American, Asian, and Native-American infants and infrequently in white infants. Although most of these lesions are found in the lumbosacral area, occurrence at other sites is not uncommon. The pigmentation is macular and gray-blue, lacks a sharp border, and may cover an area 10 cm or larger in diameter. Pigmentary lesions result from delayed disappearance of dermal melanocytes.
Most of these lesions gradually disappear during the first few years of life, but aberrant lesions in unusual sites are more likely to persist. Because of their distinctive color and morphology, mongolian spots are not easily confused with congenital pigmented melanocytic nevi or café au lait spots, which often have their onset later in infancy or in early childhood, although they may be present at birth.
Rarely, extensive mongolian spots may be associated with inborn errors of metabolism (Hurler syndrome, GM1 gangliosidosis, mucolipidosis) and neurocristopathies.
The nevus simplex is the most common neonatal cutaneous lesion and is present in up to 80% of normal newborns ( Fig. 94.4 ). These lesions blanch when compressed and are usually centrally located and symmetric, appearing on the nape, the eyelids, and the glabella. In a prospective study of affected infants, most of the facial lesions had faded by 1 year of age, but those on the neck were more persistent. Surveys of adult populations confirm the persistence of the nuchal lesions in approximately one-fourth of the population.
Harlequin color change is a transient phenomenon observed in the immediate neonatal period and common in the low birth weight infant. The dependent side of the body becomes intensely red, and the upper side pales, with a sharp midline demarcation. The peak frequency of attacks in one series occurred on the 2nd, 3rd, and 4th days, but episodes were observed during the first 3 weeks of life. These episodes are of no pathologic significance. They have been attributed to a temporary imbalance in the autonomic regulatory mechanism of the cutaneous vessels; there are no accompanying vital sign changes.
Erythema toxicum is a benign and self-limited eruption that usually develops at 24-72 hours of age, but new lesions may appear until 2-3 weeks of age. The disorder is more common in term than premature infants, which suggests that it may represent an inflammatory reaction requiring mature skin. These lesions may vary considerably in character and number; they may be firm, 1-3 mm, pale yellow to white papules or pustules on an erythematous base resembling flea bites or erythematous macules as large as 3 cm in diameter. Individual lesions are evanescent, often lasting only a matter of hours. They may be found on any area of the body but occur only rarely on the palms and soles. They are asymptomatic with no related systemic involvement and the cause is unknown, although a variety of specific cytokines have been implicated in the pathogenesis. A microscopic examination of a Wright- or Giemsa-stained smear of the pustule contents demonstrates numerous eosinophils; Gram stains are negative for bacteria and cultures are sterile. No treatment is necessary because spontaneous resolution occurs in 6 days to 2 weeks.
Transient neonatal pustular melanosis is a distinctive eruption that heals with brown pigmented macules. It consists of three types of lesions: first-stage lesions are small superficial vesiculopustules with little or no surrounding erythema. These rapidly progress to the second stage, which consists of collarettes of scale or scale crust surrounding a hyperpigmented macule (third stage) ( Fig. 94.5 ). All three types of lesions may be present at birth, but the macules are observed more frequently. The lesions may be profuse or sparse and occur on any body surface, including the palms, soles, and scalp. Sites of predilection are the forehead, submental area and anterior neck, and lower back. When intact pustules rupture, a pigmented macule often is discernible central to the collarette of scale, which represents the margin of the unroofed pustule. Presumably, the macules result from postinflammatory hyperpigmentation, and those present at birth may be the sequelae of in utero pustular lesions.
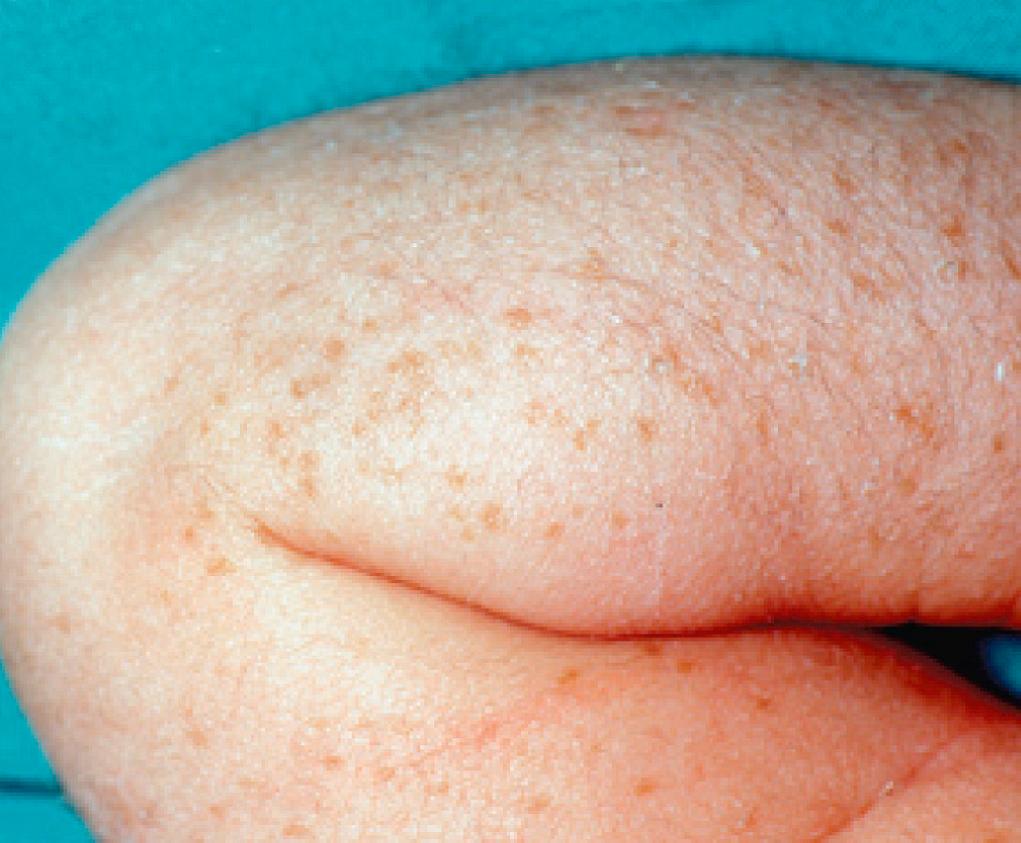
Pustular melanosis may be confused with erythema toxicum, congenital cutaneous candidiasis, or staphylococcal pyoderma. Cultures and Gram stains of smears prepared from intact pustules are devoid of organisms; Wright stains of intralesional contents demonstrate cellular debris, polymorphonuclear leukocytes, and a few or no eosinophils, in contrast to those of erythema toxicum. Although the pustules disappear in 48 hours, the hyperpigmented macules may persist for as long as 3 months. These lesions require no therapy as the disorder is benign and transient.
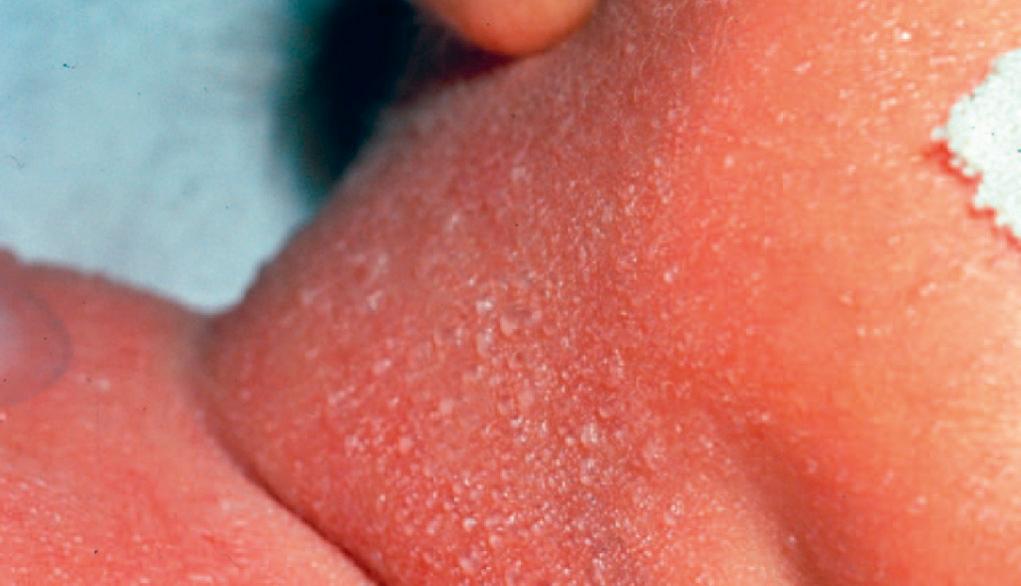
Miliaria is an eruption characterized by crops of superficial vesicles resulting from eccrine sweat gland duct obstruction leading to sweat retention. The three types of lesions are superficial thin-walled vesicles without inflammation (i.e., miliaria crystallina, Fig. 94.6 ); small, erythematous, grouped papules (i.e., miliaria rubra); and nonerythematous pustules (i.e., miliaria pustulosis or profunda). The eruption most frequently develops in the intertriginous areas and over the face and scalp. It is exacerbated by exposure to a warm and humid environment. Miliaria sometimes can be confused with erythema toxicum; rapid resolution of the lesions when the infant is placed in a cooler environment differentiates them from other causes of infectious pustulosis. A Wright-stained smear of vesicular lesions demonstrates only sparse squamous cells or lymphocytes, permitting exclusion of infectious vesicular eruptions. No topical therapy is indicated.
Neonatal acne occurs in up to 20% of newborns and is more commonly seen in boys. Increased sebaceous secretions secondary to maternal and neonatal androgens and colonization of the sebaceous glands by the yeast Malassezia furfur are implicated in its pathogenesis and are characterized by small red papules and pustules on the face during the first weeks of life. Unlike in adolescents, comedones and cysts are usually absent. The lesions are asymptomatic and resolve spontaneously without scarring over several weeks. The clinical significance lies in differentiating this rash from infections, excluding virilization as its underlying cause and potential implication of severe acne in adolescence.
Infantile acropustulosis is characterized by recurrent crops of pruritic, sterile vesiculopustules with a predilection for the palms and soles. The lesions appear in crops every 2-3 weeks and last 7-10 days. They involve the hands and feet primarily, but occasionally may involve the limbs as well. Because of the distribution, it is often confused with scabies. It is more common in African-American and male infants. The condition resolves spontaneously. A subcorneal pustule filled with neutrophils is seen on skin biopsy. Traditional therapy consists of topical steroids and antihistamines, although one report indicates successful treatment with topical maxacalcitol, an active form of vitamin D3. A recessive mutation in the gene coding interleukin 1 receptor antagonist has been described in a case of infantile pustulosis treated successfully with recombinant human interleukin 1 receptor antagonist anakinra.
The most common and benign causes of excessive scaling are due to physiologic desquamation (normal term infants) and dysmaturity (post-mature and small for gestational age infants), neither of which have long-term sequelae. Less common causes include the congenital ichthyoses and the ectodermal dysplasias (particularly hypohidrotic ectodermal dysplasia), all of which are chronic, heritable disorders.
The gestational age of a newborn with accentuated physiologic desquamation usually is 40-42 weeks; peak shedding occurs near the 8th day of life. These infants are otherwise normal in physical appearance and behavior. In contrast, the dysmature infant (see Chapter 22 ) has distinctive characteristics. The body is lean with thin extremities and decreased subcutaneous fat. The ponderal index is low with diminished body weight in relation to length. The skin is often scaly with parchment-like desquamation, especially of the distal extremities. There is often meconium staining of the skin as well as the nails and umbilical cord. The hair is abundant and the nails are abnormally long. In the normal infant with accentuated physiologic scaling and the dysmature infant, desquamation is a transient phenomenon, and the integument continues to serve its intended protective function. In contrast, the infant with congenital ichthyosis may have serious difficulties early in life because of impaired barrier function and the subsequent risks of secondary infection.
The term ichthyosis derives from the similarity of the skin condition to the scales of a fish. It refers to a complex and often confusing plethora of conditions characterized by disorders of cornification with or without systemic symptoms. In 2010, a consensus conference of experts developed a new classification that was clinically based and easily comprehensible by clinicians. It was broadly divided into nonsyndromic (restricted to skin) and syndromic (skin plus other organs). Ichthyosis in the newborn period may manifest as scaling only, scaling and erythroderma, a collodion membrane, or the thickened plates of harlequin ichthyosis. Although rare, it is important for the neonatal caregiver to recognize the patient with ichthyosis as the barrier function of the skin is often compromised. Prompt management and diagnosis can be lifesaving. Selected conditions with particular relevance to the newborn are highlighted below, followed by a general overview of treatment strategies. Table 94.3 lists the major clinical phenotypes of the ichthyoses in the newborn period.
| Phenotype | Type of Ichthyosis |
|---|---|
| Erythroderma | Netherton syndrome, KID syndrome, CIE Also consider: Omenn syndrome, SCID, ectodermal dysplasia, erythrodermic psoriasis |
| Generalized scaling | XLI, Netherton syndrome, Sjögren-Larsson syndrome, trichothiodystrophy, neutral lipid storage disease |
| Collodion baby | Lamellar ichthyosis, CIE, self-healing collodion baby, harlequin ichthyosis (mild), Sjögren-Larsson syndrome, trichothiodystrophy, neutral lipid storage disease Also consider: ectodermal dysplasias, Gaucher disease |
| Vernix-like hyperkeratosis | Harlequin ichthyosis, KID syndrome |
| Constrictive scales with deformities | Harlequin ichthyosis |
| Blisters/erosions | Epidermolytic ichthyosis, ichthyosis bullosa of Siemens Also consider: epidermolysis bullosa, TEN, SSSS, immunobullous disease, mastocytosis |
X-linked ichthyosis is the most common form of ichthyosis in the newborn period, affecting approximately 1 in 2500 male babies. Genetic mutations of the STS gene lead to deficiency of steroid sulfatase. Female carriers of the gene for X-linked ichthyosis, when pregnant with an affected male fetus, have a deficiency of placental steroid sulfatase reflected by low maternal urinary estriol excretion and a difficult or prolonged labor that often requires intervention. Clinical manifestations of X-linked ichthyosis in males include alterations in the skin, eye, and testes of affected infants. Most patients have cutaneous findings at birth, and 80%-90% show scaling by 3 months of age. Rarely, a collodion membrane may be present. The hyperkeratosis is variable, but the scales typically are large, thick, and dark and are prominent over the scalp, neck, anterior trunk, and extensor extremities. Sparing of palms and soles and partial sparing of the flexures are helpful diagnostic features. Systemic manifestations are generally absent, and complications are rare. Skin biopsy is helpful (although the histologic pattern resembles that of lamellar ichthyosis), showing hyperkeratosis, a well-developed granular layer, hypertrophic epidermis, and a perivascular lymphocytic infiltrate. This form of ichthyosis is clinically apparent in affected homozygous males and not in heterozygous females. Cryptorchidism is seen in approximately 25% of affected males; these patients may be at increased risk for testicular cancer. Deep stromal corneal opacities have also been found in about 50% of male infants with the disorder and in a smaller percentage of female carriers, but they do not affect vision.
Harlequin ichthyosis and harlequin fetus are rare, very severe disorders of keratinization inherited as an autosomal recessive trait in most families owing to mutations in the ABCA12 gene. The skin of affected infants is markedly thickened, hard (armor-like), and hyperkeratotic, with deep crevices running transversely and vertically. The fissures are most prominent over areas of flexion. Rigidity of the skin around the eyes results in marked ectropion, although the globe is usually normal. The ears and nose are underdeveloped, flattened, and distorted, and the lips are everted and gaping, producing a “fish-mouth” deformity. The nails and hair may be hypoplastic or absent. Extreme inelasticity of the skin is associated with flexion deformity of all joints. The hands and feet are ischemic, hard, and waxy in appearance, with poorly developed distal digits.
Most harlequin fetuses are born prematurely, usually between 32 and 36 weeks’ gestation, adding to their morbidity and mortality. Complications relate to an impaired skin barrier with associated hypernatremic dehydration, altered thermoregulation, increased metabolic demands, and increased risk of respiratory failure and sepsis. Although the disease still has a high mortality, therapeutic trials of oral retinoids combined with multidisciplinary intensive care have resulted in dramatic survival of several of these infants. All surviving infants develop chronic, severe ichthyosis.
The collodion baby is less severely affected than the infant with harlequin ichthyosis and may represent a phenotypic expression of several genotypes. The infant is covered with a cellophane-like membrane, which by its tautness may distort the facial features and the digits. The membrane is shiny and brownish yellow, resembling an envelope of collodion or oiled parchment, and may be perforated by hair. This condition eventuates in lamellar ichthyosis or congenital ichthyosiform erythroderma in approximately 60% of patients. Sjögren-Larsson syndrome, Conradi-Hünermann syndrome, trichothiodystrophy, dominant lamellar ichthyosis, and neonatal Gaucher disease account for the remainder of the infants born with a collodion membrane. Rarely, shedding of the collodion membrane results in a normal underlying integument (i.e., lamellar exfoliation of the newborn). Collodion babies often are born prematurely. The presence of ectropion, eclabium, and crumpled ears causes these infants to resemble one another for the first few days of life. Fissuring and peeling begin shortly after birth, and large sheets may desquamate, revealing erythema of variable intensity. These infants have an abnormal epidermal barrier, which leads to complications such as dehydration, electrolyte imbalance, temperature instability, and cutaneous and systemic infection. Complete shedding of the collodion membrane may take several weeks. Pedigree information and histopathologic examination of a skin biopsy are additional aids in the delineation of the specific type of ichthyosis.
Management of ichthyotic patients in the newborn period varies from managing mild cosmetic problems with excessive dryness and scaling to treatment of potentially life-threatening illness because of deficits in the epidermal barrier and subsequent infection. Table 94.4 lists the primary complications that form the primary targets of therapy for ichthyosis. Severely affected infants require aggressive topical care, oral systemic retinoids (Acitretin), liberal use of bland emollients, and careful monitoring of electrolyte needs. The infant should be placed in an environment with increased humidity and every effort taken to prevent infections. Keratolytic agents if necessary can be used with caution in the neonatal period. The long-term goal of ichthyotic therapy is to eliminate scaling owing to excessive cornification and reduce dryness (xerosis) of the skin without inducing irritation. Although the stratum corneum is often thick and scaly, the barrier is compromised, and there are significant risks of systemic absorption of potentially toxic substances applied topically. Avoidance of corneal desiccation and ulceration is critical if ectropion is present.
| Complications with Ichthyosis | Therapeutic Management Strategies |
|---|---|
| Restricted chest movement, contractures of limbs and soft tissues, sucking and feeding problems, compartment syndromes | Respiratory support, nasogastric feeding, liberal emollients with daily bathing and/or retinoids to thin and remove scale, monitor tissue perfusion and compartment pressure, orthopedic and plastic surgery evaluation of contractures, ophthalmology consult for ectropion and avoidance of corneal desiccation |
| Risk for bacterial infections and sepsis caused by impaired epidermal barrier | Watch for clinical signs of localized infection and/or sepsis with institution of aggressive culturing and treatment, topical antibiotics to fissures, reverse isolation |
| Risk of dehydration and electrolyte imbalance | Monitor fluid balance and electrolytes, humidified incubator to decrease evaporative water loss, intravenous hydration and/or alimentation |
| Fragile, easily traumatized skin; risk of absorption of topical preparations | Minimal handling; avoid aggressive adhesives; use self-adherent wraps or gauze; use bland emollients; avoid silver sulfadiazine, lactic acid, and salicylic acid preparations; antibacterial soaps |
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here