Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The first skin autograft was described by Reverdin in 1871, and the use of allograft skin as a clinical method for wound coverage soon followed. In 1874, Thiersch published a report about a small series of patients on whom he had used partial-thickness skin grafts. This led to extensive trials of harvesting extremely thin grafts, leaving some of the surface epithelium behind to aid in donor site healing. Results from the use of these small, thin grafts, known as “Thiersch grafts,” “pinch grafts,” “epidermis grafts,” or “razor grafts” were generally so unsatisfactory for resurfacing large areas that they were typically limited to the treatment of small ulcerated wounds. The first successful use of allogeneic skin for burn wound coverage was reported by Girdner in 1881. Five years later, Thiersch described the histologic anatomy of skin engraftment which popularized the clinical use of split-thickness skin grafts.
Storage of human skin did not begin until the early 1900s, when Wentscher reported his experience with the transplantation of human skin that had been refrigerated for 3–14 days; however it was not until the 1930s that blood and tissue banking took their place in the clinical practice of medicine. The clinical utility of allograft skin in burn wound coverage was first described in 1938, when Bettman reported his success in the treatment of children with extensive full-thickness burn injuries. Webster and Matthews later described the successful healing of skin autografts stored for 3 weeks at 4–7°C; however it was not until 1949, following the establishment of the United States Navy tissue bank, that modern-day skin banking began.
The establishment of skin banking signaled the beginning of significant research related to the processing, preservation, and storage of human tissues. Baxter explored the histologic effects of freezing on human skin and discovered that the formation of ice crystals caused the destruction of skin architecture. This was followed in 1952 by the pioneering research of Billingham and Medawar who demonstrated that skin could be effectively cryopreserved using glycerol. Soon afterward, Taylor was able to demonstrate that the addition of glycerol to storage solutions decreased ice crystal formation in frozen tissues. These advancements permitted Brown and Jackson to popularize the use of allogeneic human skin grafts as biologic dressings for extensive burns and denuded tissue. By 1966, Zaroff had reported the 10-year experience using allograft skin in the treatment of thermally injured patients at the Brooke Army Medical Center. In this report, he described the mechanical and physiologic advantages of allograft skin as a biologic dressing. In 1966, Cochrane reported the first successful use of frozen autologous skin grafts following controlled-rate freezing in 15% glycerol and rapid rewarming prior to implantation. This was followed by Morris's report demonstrating the beneficial effects of using allogeneic skin in the treatment of infected ulcers and other contaminated wounds and Shuck et al.'s report suggesting the potential use of allogeneic skin in the treatment of traumatic wounds based on their Vietnam War experience. These increased uses of allograft skin led to further research into the beneficial effects of allograft skin on wound healing, including its association with a reduced incidence of bacterial infections and the stimulation of wound bed neovascularization.
Bondoc and Burke are credited with the establishment of the first functional skin bank in 1971. Their experience with allograft skin led to a report of successful burn wound excision and allografting with temporary immunosuppression in children with extensive injuries. Today allograft skin remains an ideal temporary cover for extensive or excised cutaneous or soft tissue wounds, particularly when sufficient autograft skin is not available or when temporary wound coverage is desired.
Another biologic dressing with a long history in the treatment of burned patients is amniotic membrane, with the first use for the purpose taking place in 1912 by Sabella a few short years after it was originally used in skin transplantation by Davis in 1910. During the immediate period following these reports, the use of amniotic membranes was attempted as permanent skin replacements, although the grafts were rejected. In 1952, Douglas was the first to report the use of amniotic membrane as a temporary burn wound covering. Since that time, amnion has been one of the tools available in the treatment of partial-thickness burns. Amnion has been shown to alleviate pain, prevent infection, reduce pruritus, and accelerate wound healing. Our institution has used amniotic membrane for temporary total ocular surface coverage in the treatment of toxic epidermal necrolysis, which has shown improvements in corneal clarity, conjunctival scaring, and dry eye symptoms at 3-month follow-up as compared to a historical group that received maximal medical treatment. The preparation and storage of amnion has been a role in many skin banks around the world.
The widespread use of allograft skin in the management of patients with extensive burn, traumatic, and soft tissue injuries has had a major impact on the number of skin banking facilities over the past two decades or so. Consequently the majority of skin banks have been founded in close proximity to regional burn centers or within the burn center hospitals themselves. Skin banks must therefore maintain a close working relationship with regional burn centers not only to meet the specific needs of the burn surgeon but also to help generate community support for skin donation through combined educational outreach programs.
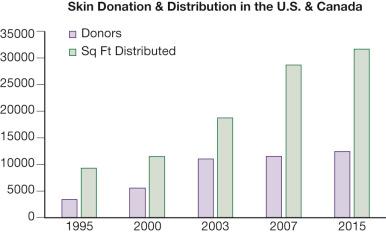
From 1969 to 1988, there was a steady growth in the number of skin banks; however this number declined, reaching its nadir in 2002. Since that time, however, there has been a steady increase in the number of skin banking facilities to its current total of 81 American Association of Tissue Banks (AATB)-accredited tissue banks that recover, process, store, and/or distribute skin for transplantation. In 1983, DeClement and May estimated that as much as 32,000 square feet of skin might be needed in burn and wound care centers. Fig. 14.1 shows the total number of skin donors as well as total number of square feet of skin distributed by skin banks throughout the United States and Canada from 1995 through 2015.
As skin banking facilities grew in number, it became apparent that policies and procedures required standardization. This was quite difficult initially because there was insufficient data to develop a consensus regarding standards of practice. As early as 1976, the AATB had begun to address this issue by the formation of a Skin Council. This provided a forum for the discussion of skin banking practices and was complemented by the activities of the American Burn Association's Skin Banking Special Interest Group. The Standards and Procedures Committees were created in 1977 and produced the first guidelines for tissue banking in 1979. The first Standards for Tissue Banking were published in 1984, and tissue-specific technical manuals (including skin) were developed in 1987. Since that time, the Standards have been modified and refined based on consensus and, where available, supportive scientific research, with the latest edition, the 14th, released in 2016. In addition, shortly after the development and promulgation of its Standards for Tissue Banking, the AATB created an inspection and accreditation committee in 1982 and began conducting voluntary inspections in 1986. This program continues today and is important in ensuring that tissue banks adhere not only to AATB Standards but also the U.S. Food and Drug Administration (FDA) regulations governing all aspects of human tissue banking. In 1992, the AATB presented its Year 2000 Plan, outlining the institution's goals of supporting tissue and cell banks and ensuring the safety and quality of tissues, as well as advancing and strengthening the AATB. The AATB has worked with many organizations, such as the FDA, the Centers for Disease Control (CDC), and the American Burn Association, to update standards and quickly respond when necessary, such as through the timely creation of instructions and methods to screen potential donors for emerging infectious diseases, including West Nile, Ebola, and Zika viruses in order to reduce risk of infection.
The increasing use of allograft skin in specialized burn care centers has been one of the driving forces behind the growth and development of skin banks in the United States. The general indications for its use in wound management are listed in Box 14.1 . Allograft skin possesses many of the ideal properties of biologic dressings and plays a major role in the surgical management of extensive wounds when autologous tissue may not be immediately available ( Box 14.2 ). It reduces evaporative water loss and the exudation of protein-rich fluids, prevents wound desiccation, and suppresses microbial proliferation. Wound pain is lessened, and this is associated with better patient compliance with occupational and physical therapy. By restoring the physiologic barrier at the wound surface, the allografts reduce heat loss through the wound and mitigate the hypermetabolic response to burn injury. The frequent and unpredictable demand for allograft skin in specialized burn care centers has prompted the growth and development of local and regional skin banks throughout the world.
Coverage of extensive wounds where autologous tissue is not available
Coverage of widely meshed skin autografts
Extensive partial-thickness burns
Extensive epidermal slough
Stevens-Johnson syndrome
Toxic epidermal necrolysis
Staphylococcal scalded skin syndrome
Testing the wound bed's ability to accept autograft
Template for the delayed application of keratinocytes.
Reduce water, electrolyte, and protein loss
Prevent desiccation of tissue
Suppress bacterial proliferation
Reduce wound pain
Reduce energy requirements
Promote epithelialization
Prepare wounds for definitive closure
Provide dermal template for epidermal grafts.
Fresh allograft skin represents the gold standard for all biologic dressings employed for temporary wound closure based on a number of its distinctive properties compared to cryopreserved skin ( Box 14.3 ). Its availability is critically important for the surgeon faced with the need to provide immediate coverage of large excised burn wounds. Fresh allografts become well-vascularized, stimulate neovascularization in the underlying wound bed, and prepare the recipient sites for permanent coverage with autologous skin. In addition, fresh allografts tolerate modest wound contamination and adhere better to the freshly excised subcutaneous fat than do cryopreserved grafts. The allogeneic skin is usually removed once the patient's donor sites have healed sufficiently for reharvesting or once autologous cultured skin is available for permanent wound closure.
Rapidity and strength of adherence to the wound
Control of microbial growth
Rapidity of revascularization
Reproducible clinical results
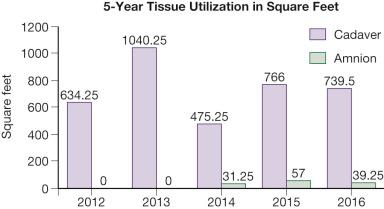
The use of fresh allografts has become extremely limited in recent years, however, due to increased FDA regulations put in place to reduce risk of disease transmission. When fresh allograft is not available, cryopreserved skin is an excellent alternative for temporary wound coverage. Although frozen cryopreserved skin generally has less measurable viability than fresh skin, it is currently difficult to maintain continuous and ample stores of fresh skin beyond 14 days. It has therefore been standard skin banking practice to cryopreserve allograft skin within 7–10 days of recovery if it is not going to be utilized within the time period that viability can be maintained. Fig. 14.2 depicts the quantitative use of allograft skin and amnion in thermally injured children treated at the Shriners Hospital for Children in Galveston, Texas, since 2012.
Alternative wound coverings such as Integra dermal regeneration template have provided alternatives to allograft skin for the treatment of excised burn wounds in patients with extensive full-thickness burn injuries. When compared with artificial dermis, fresh, refrigerated allograft has been shown to have a better rate of engraftment than the dermal regeneration template. A study performed at our institution in 2007 indicated that Integra can safely be used for wound coverage in children with burns of more than 50% total body surface area (TBSA) burned with an associated attenuation of post-burn hypermetabolism with no increased risk of infection.
Another use of allograft skin has been its application as an overlay on top of widely expanded, meshed autologous skin grafts ( Fig. 14.3 ). This technique was originally described utilizing meshed allograft and provides immediate, as well as both temporary and permanent, wound closure. Our institution utilizes 2 : 1 meshed cadaver skin for the coverage of widely expanded autografts (with a ratio greater than 2 : 1). Because it is usually less viable than fresh skin, it functions more as a biologic dressing than as a temporary skin replacement. Its adherence to the underlying wound bed results in the relief of pain and the limitation of exudative and water losses, and it reduces the need for frequent dressing changes. As the underlying wound bed reepithelializes, the allograft slowly separates without disturbing the delicate underlying epithelium. These properties of frozen allograft are also utilized in the coverage of partial-thickness wounds. Studies by Rose and Naoum demonstrated more rapid healing times and shorter hospital stays for children with extensive partial-thickness burns when treated with early wound débridement and allografting compared to conventional topical antimicrobial therapy. However due to lower costs and ease of use, Shriners Hospitals for Children – Galveston primarily uses xenograft for partial thickness burns.

There has been some concern that allografts may induce an inflammatory rejection response resulting in delayed reepithelialization of underlying autografts; therefore the use of lyophilized tissue has been suggested because the lyophilization process destroys cellular components and results in a diminished immunologic response from the graft recipient. A 2013 study of 11 patients treated with lyophilized allograft showed no evidence of immunologic reactions due to the administration of allograft.
The clinical use of cultured epidermal autografts (CEA) in the care of burn patients was first described by O'Connor et al. in 1981. Since that time, there have been numerous reports supporting its use as a permanent skin replacement for patients with extensive full-thickness burn injuries. This methodology has not been without problems, however, with many authors describing variable take rates and instability of the grafts. Cuono first advocated the use of allogeneic skin with CEA, allowing the allograft skin to vascularize before removing the antigenic epidermal layer by dermabrasion. Hickerson et al. reported results on five burn patients demonstrating more than 90% CEA take on the allogeneic dermis and supple, durable grafts up to 4 years postoperatively. A recent multicenter randomized trial compared patients with full-thickness burns who were treated with split-thickness skin autografts to those who were additionally treated with CEA. The application of CEA resulted in faster wound epithelialization, improved perception of scarring, and improved pigmentation and elasticity of the skin at follow-up.
The past decade or so has also witnessed the development of an acellular allogeneic dermal matrix (AlloDerm) as a template for the simultaneous application of thin split-thickness autografts. The potential advantage of the dermal template is reasoned to be the use of a thinner autologous skin graft resulting in more rapid donor site healing and reduced morbidity. A multicenter clinical trial demonstrated equivalence of this technique with a standard split-thickness meshed autograft; however autograft take rates were somewhat lower than that for controls and varied from center to center. In addition, the allogeneic dermal grafts measured only 36–116 cm 2 and were only evaluated up to 180 days post-grafting. AlloDerm has also been used as a template for CEA; however there have been only a few anecdotal reports related to this potential application.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here