Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The renal pelves, ureters, urinary bladder, and urethra (except the terminal portion) are lined by a special form of transitional epithelium called urothelium . Urothelium is composed of five to six layers of cells with oval nuclei, often with linear nuclear grooves, and a surface layer consisting of large, flattened “umbrella cells” with abundant cytoplasm. This epithelium rests on a well-developed basement membrane, beneath which is a lamina propria. The lamina propria in the bladder contains wisps of smooth muscle that form a discontinuous muscularis mucosa. It is important to differentiate the muscularis mucosa from the deeper well-defined larger muscle bundles of the detrusor muscle (muscularis propria), since bladder cancers are staged on the basis of invasion of the latter. If urine flow is obstructed and intravesical pressures rise, the bladder musculature undergoes hypertrophy.
The ureters lie throughout their course in a retroperitoneal position. Retroperitoneal tumors or fibrosis may entrap and obstruct the ureters. In middle-aged and older women, relaxation of pelvic support leads to prolapse (descent) of the uterus, pulling with it the floor of the bladder. In this fashion the bladder is protruded into the vagina, creating a pouch (cystocele) that fails to empty readily with micturition. In males, the seminal vesicles and prostate have similar close relationships, being situated just posterior and inferior to the neck of the bladder. Thus, enlargement of the prostate, so common in middle to later life, constitutes an important cause of urinary tract obstruction. Subsequent sections discuss the major pathologic lesions of the ureters, urinary bladder, and urethra.
Congenital anomalies of the ureters are found in about 2% to 3% of all autopsies ( Table 21.1 ). Although most have little clinical significance, certain anomalies may lead to obstruction of the flow of urine and thus cause clinical disease.
|
Ureteropelvic junction (UPJ) obstruction is the most common cause of hydronephrosis in infants and children. Cases that present early in life preferentially affect males, are bilateral in 20% of cases, and are often associated with other anomalies. There is agenesis of the contralateral kidney in a minority of cases. In adults, UPJ obstruction is more common in women and is most often unilateral. The condition is most commonly ascribed to abnormal organization of smooth muscle bundles or excess stromal deposition of collagen between smooth muscle bundles at the UPJ, or in rare cases to extrinsic compression of the UPJ by abnormal renal vessels.
Primary tumors of the ureter are rare. Benign tumors are generally of mesenchymal origin. Fibroepithelial polyp, a tumor-like lesion often occurring in children, is composed of loose, vascularized connective tissue overlaid by urothelium. Primary malignant tumors of the ureter resemble those arising in the renal pelvis, calyces, and bladder. The majority are urothelial carcinomas ( Fig. 21.1 ). They occur most frequently during the sixth and seventh decades of life and cause obstruction of the ureteral lumen. They commonly occur concurrently with urothelial carcinomas of bladder or renal pelvis.
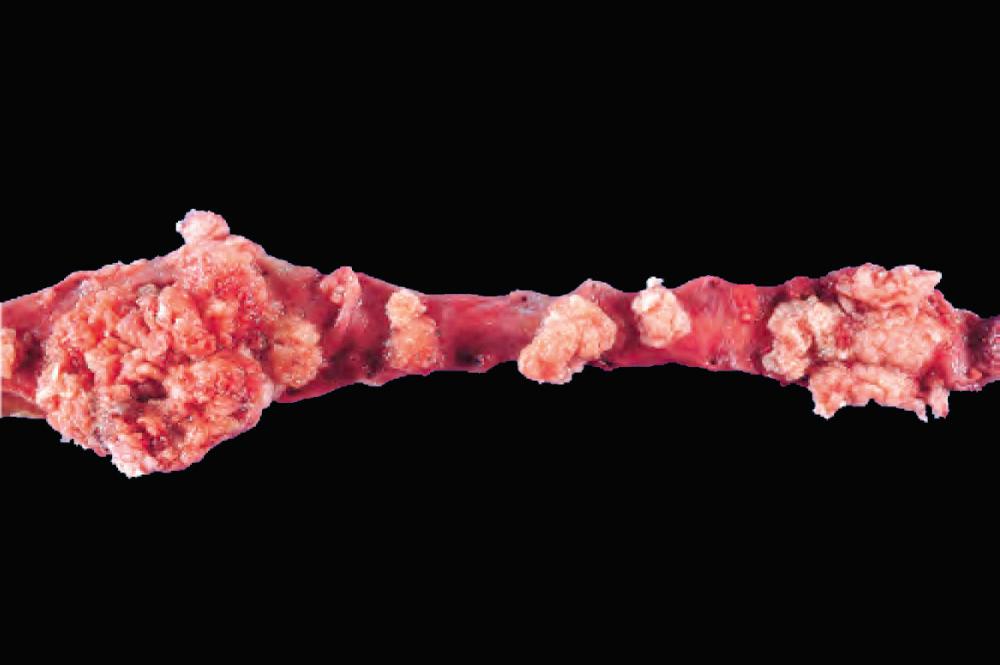
Intrinsic and extrinsic lesions may obstruct the ureters and may give rise to hydroureter, hydronephrosis, and pyelonephritis ( Chapter 20 ). Unilateral obstruction typically results from proximal intrinsic or extrinsic causes (e.g., stones, neoplasms etc.), whereas bilateral obstruction arises from distal causes, such as nodular hyperplasia of the prostate ( Table 21.2 ).
| Cause of Obstruction | Characteristics/Mechanisms |
|---|---|
| Intrinsic | |
| Calculi | Typically of renal origin |
| Usually small (5 mm in diameter or less) | |
| Impact at loci of ureteral narrowing—ureteropelvic junction, where ureters cross iliac vessels and where they enter bladder—and cause excruciating “renal colic” | |
| Strictures | May be congenital or acquired |
| Tumors | Urothelial carcinomas arising in ureters |
| Rarely, benign tumors or fibroepithelial polyps | |
| Blood clots | Massive hematuria from renal calculi, tumors, or papillary necrosis |
| Neurogenic | Interruption of the neural pathways to the bladder |
| Extrinsic | |
| Pregnancy | Physiologic relaxation of smooth muscle or pressure on ureters at pelvic brim from enlarging fundus |
| Periureteral inflammation | Salpingitis, diverticulitis, peritonitis, sclerosing retroperitoneal fibrosis |
| Endometriosis | With pelvic lesions associated with scarring |
| Tumors | Cancers of the rectum, bladder, prostate, ovaries, uterus, cervix; lymphomas, sarcomas |
This rare lesion is characterized by a fibrotic proliferative inflammatory process that encases retroperitoneal structures and causes hydronephrosis. The disorder occurs in middle to late age and is more common in males than females. At least a subset of these cases is related to IgG4-related disease, a recently described entity associated with elevated levels of serum immunoglobulin G4 (IgG4) and fibroinflammatory lesions rich in IgG4-secreting plasma cells, that also affects other organs ( Chapter 6 ). Other etiologies for retroperitoneal fibrosis include drug exposures (ergot derivatives, β-adrenergic blockers), inflammatory conditions (vasculitis, diverticulitis, Crohn disease), or malignancies (lymphomas, urinary tract carcinomas). Most, however, have no obvious cause and are considered primary or idiopathic (Ormond disease).
Microscopic examination typically reveals fibrous tissue containing a prominent infiltrate of lymphocytes, often with germinal centers, plasma cells (frequently IgG4-positive), and eosinophils. Treatment initially involves corticosteroids, although many patients eventually become resistant and require ureteral stents or surgical extrication of the ureters from the surrounding fibrous tissue (ureterolysis).
Ureteral obstruction is clinically significant because it can lead to hydroureter, hydronephrosis, or pyelonephritis, thus compromising renal function.
In children, congenital UPJ obstruction is the most common obstructive lesion.
In adults, ureteral obstruction may be acute (e.g., due to obstructing calculi) or chronic (e.g., due to intrinsic or extrinsic tumors or rarely idiopathic conditions such as sclerosing retroperitoneal fibrosis).
Several different types of congenital abnormalities of the bladder are recognized; these are variously associated with increased risk of infection or neoplasia.
Vesicoureteral reflux is the most common and serious congenital anomaly. As discussed in Chapter 20 , vesicoureteral reflux predisposes to ascending pyelonephritis and loss of renal function. Abnormal connections between the bladder and the vagina, rectum, or uterus may create congenital vesicouterine fistulae.
Diverticula are pouch-like invaginations of the bladder wall that vary from less than 1 cm to 10 cm in diameter and may be congenital or acquired. Congenital diverticula may be due to a focal failure of development of the normal musculature or to a urinary tract obstruction during fetal development. Acquired diverticula are most often associated with prostatic hyperplasia, producing urinary outflow obstruction. The resulting increase in intravesical pressure causes outpouching of the bladder wall and the formation of diverticula. Although most diverticula are small and asymptomatic, they may come to clinical attention since they constitute sites of urinary stasis and predispose to infection and the formation of bladder calculi. Rarely, carcinoma may arise in bladder diverticula; such tumors are on average more advanced in stage due to the thin or absent muscularis propria layer of diverticula.
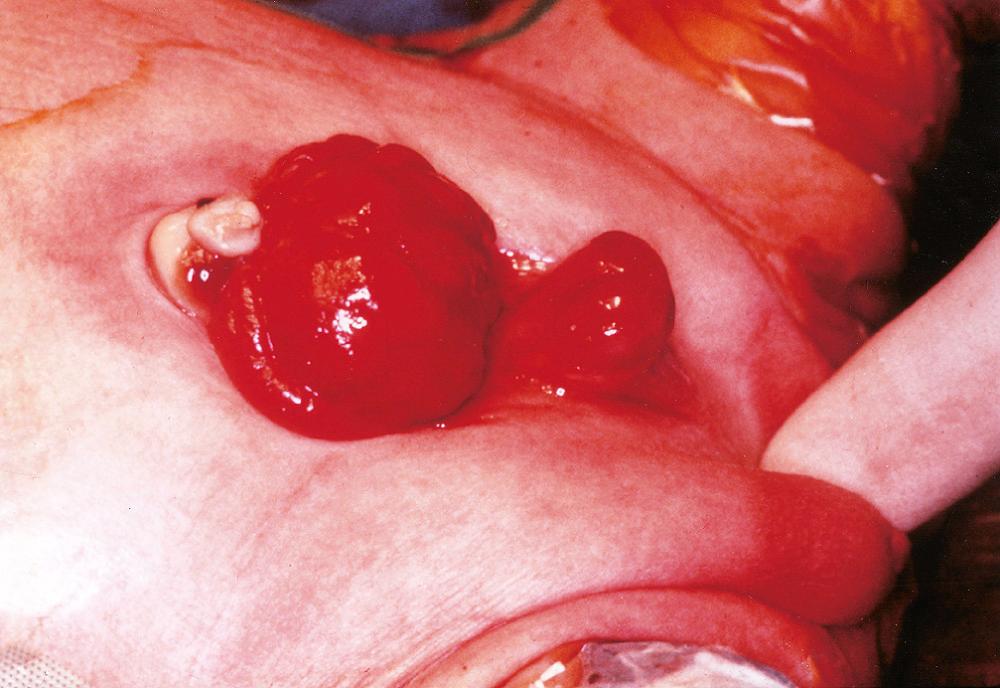
Exstrophy of the bladder is a developmental failure in the anterior wall of the abdomen and the bladder. As a result, the bladder communicates directly with the abdominal surface ( Fig. 21.2 ). The exposed bladder mucosa may undergo colonic glandular metaplasia and is subject to chronic infection that often spreads to the upper urinary tract. Exstrophy is associated with an increased risk of adenocarcinoma in the bladder remnant.
Urachal anomalies. The urachal canal connects the fetal bladder with the allantois and normally is obliterated at birth. Rarely, it remains fully or partially patent. The former creates a fistulous urinary tract connection between the bladder and umbilicus. When only a central region of patent urachus persists, a urachal cyst lined by urothelial or metaplastic glandular epithelium is formed. Urachal cysts are at increased risk for neoplastic transformation, mostly presenting as adenocarcinomas (see Neoplasms). These account for only a small minority of bladder cancers overall (0.1% to 0.3%), but constitute 20% to 40% of bladder adenocarcinomas.
Bacterial pyelonephritis is frequently preceded by infection of the urinary bladder, with retrograde spread of microorganisms into the kidneys and their collecting systems (discussed in Chapter 20 ). Predisposing factors for cystitis include bladder calculi, urinary obstruction, diabetes mellitus, instrumentation, and immune deficiency. The most common etiologic agents of cystitis are the coliforms: Escherichia coli, followed by Proteus, Klebsiella, and Enterobacter. Women are more likely to develop cystitis as a result of their shorter urethras. Tuberculous cystitis is almost always a sequel to renal tuberculosis. Candida albicans and, much less often, cryptococcal agents may cause cystitis, particularly in immunosuppressed patients or those receiving long-term antibiotics. Schistosomiasis (Schistosoma haematobium) remains an important cause of cystitis in certain African and Middle Eastern countries. Viruses (e.g., adenovirus), Chlamydia, and Mycoplasma may also cause cystitis. Adenovirus and BK virus infections may result in hemorrhagic cystitis. Gas-forming bacteria (such as Clostridium perfringens ) result in emphysematous cystitis (gas-filled vesicles in the bladder wall).
Most cases of cystitis produce nonspecific acute or chronic inflammation of the bladder. In acute cystitis, there is hyperemia of the mucosa and neutrophilic infiltrate, sometimes associated with exudate. Persistence of the bacterial infection leads to chronic cystitis associated with mononuclear inflammatory infiltrates.
Other types of non-infectious cystitis include iatrogenic, follicular, and eosinophilic cystitis. Patients receiving systemic chemotherapy or pelvic irradiation may develop iatrogenic cystitis. Cytotoxic agents, such as cyclophosphamide, may cause hemorrhagic cystitis. Acute and chronic radiation cystitis may occur following the irradiation of the bladder region. Follicular cystitis is characterized by the presence of lymphoid follicles within the bladder mucosa and underlying wall. Eosinophilic cystitis, manifested by infiltration of the submucosa by eosinophils, typically is a nonspecific subacute inflammation but may also be a manifestation of a systemic allergic disorder.
All forms of cystitis are characterized by a triad of symptoms: (1) frequency, which in acute cases may necessitate urination every 15 to 20 minutes; (2) lower abdominal pain localized over the bladder region or in the suprapubic region; and (3) dysuria (pain or burning on urination). The local symptoms of cystitis may be merely disturbing, but these infections may be antecedents to pyelonephritis, a more serious disorder ( Chapter 20 ). Cystitis is sometimes a secondary complication of an underlying disorder associated with urinary stasis, such as prostatic hypertrophy, cystocele, calculi, or bladder neoplasms. These primary lesions must be corrected before the cystitis can be relieved.
Several variants of cystitis have distinctive causes or morphologic appearances.
Interstitial cystitis is a disorder of unknown etiology that occurs most frequently in women. The associated pain syndrome is defined by the American Urological Association as “an unpleasant sensation (pain, pressure, discomfort) perceived to be related to the urinary bladder, associated with urinary tract symptoms of more than six weeks duration, in the absence of infection or other identifiable causes.” It is characterized by intermittent, often severe, suprapubic pain; urinary frequency; urgency; hematuria; and dysuria. Typical cystoscopic findings include mucosal fissures and punctate hemorrhages (glomerulations). Microscopically, the pathologic findings are nonspecific; mast cells are often increased in the submucosa, but the pathogenic significance of this finding is uncertain. The main role of biopsy is to rule out carcinoma in situ (CIS), which clinically mimics interstitial cystitis. Treatment is largely empiric. Some cases are associated with chronic mucosal ulcers (Hunner ulcers); this is termed the late (classic, ulcerative) phase. Late in the course, transmural fibrosis may lead to a contracted bladder.
Malakoplakia is a distinctive chronic inflammatory reaction that appears to stem from acquired defects in phagocyte function. It arises in the setting of chronic bacterial infection, mostly by E. coli or occasionally Proteus species, and occurs with increased frequency in the setting of immunosuppression, such as in renal transplant recipients.
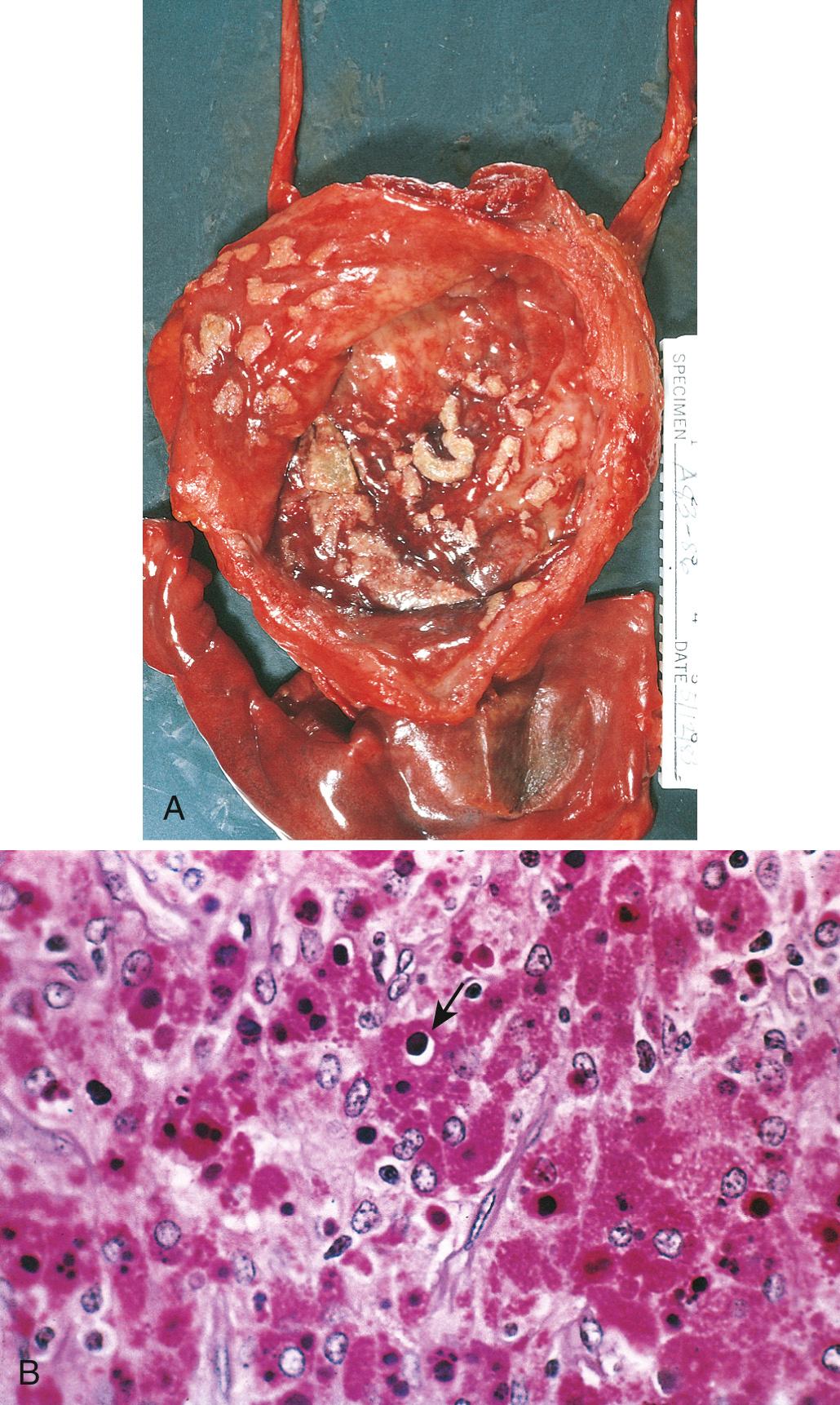
Cystoscopically, malakoplakia takes the form of soft yellow, slightly raised mucosal plaques, 3 to 4 cm in diameter ( Fig. 21.3A ). Histologically, the lesion is composed of aggregates of large foamy macrophages with occasional multinucleate giant cells and lymphocytes. The macrophages have abundant granular cytoplasm due to phagosomes stuffed with particulate and membranous debris of bacterial origin, which are believed to accumulate due to defective phagolysosome function. In addition, laminated mineralized concretions resulting from deposition of calcium in enlarged lysosomes, known as Michaelis-Gutmann bodies, are typically present within the macrophages ( Fig. 21.3B ). Similar lesions have been described in other organs including the colon and other parts of the gastrointestinal tract, brain, lungs, bones, endometrium, kidneys, prostate, and epididymis.
Polypoid cystitis is an inflammatory lesion resulting from irritation of the bladder mucosa, most commonly as a result of instrumentation, including indwelling catheters. The urothelium is thrown into broad bulbous polypoid projections as a result of marked submucosal edema. Polypoid cystitis may be mistaken for papillary urothelial carcinoma, both clinically and histologically.
Several lesions that stem from true metaplasia or conditions that mimic metaplasia affect the bladder.
Cystitis glandularis and cystitis cystica. These are common lesions of the urinary bladder in which nests of urothelium (von Brunn nests) grow downward into the lamina propria. Here, epithelial cells in the center of the nest undergo metaplasia and take on a cuboidal or columnar appearance (cystitis glandularis) or retract to produce cystic spaces lined by flattened urothelium (cystitis cystica) . Because the two processes often coexist, the condition is typically referred to as cystitis cystica et glandularis . In a variant of cystitis, glandularis goblet cells are present, and the epithelium resembles intestinal mucosa (intestinal or colonic metaplasia). Both variants can arise in the setting of inflammation and metaplasia. Extensive and multifocal intestinal metaplasia is a precursor to adenocarcinoma.
Squamous metaplasia. As a response to chronic injury, the urothelium is often replaced by nonkeratinizing or keratinizing squamous epithelium, which is a more durable lining. This should be distinguished from glycogenated squamous epithelium that is normally found in women at the trigone region of bladder. Extensive multifocal keratinizing squamous metaplasia is a precursor to dysplastic lesions and in situ and invasive squamous cell carcinoma. A classic example of this sequence is seen with bladder schistosomiasis, which commonly produces squamous metaplasia and is associated with squamous cell carcinoma in areas in which it is endemic.
Nephrogenic adenoma. Nephrogenic adenoma is an unusual lesion that may not be a form of true metaplasia. There is convincing evidence from renal transplant recipients showing that at least some of these lesions are caused by implantation and growth of renal tubular cells at sites of bladder mucosa erosion. The overlying urothelium is focally replaced by cuboidal epithelium, which can assume a papillary growth pattern. Although the lesions are typically less than 1 cm in size, larger lesions have been reported that can produce signs and symptoms that raise a suspicion of cancer. In addition, the tubular proliferation can infiltrate the underlying lamina propria and superficial detrusor muscle, microscopically mimicking a malignant process.
The bladder can be involved by a number of inflammatory lesions, many of which manifest with frequency and dysuria.
Acute or chronic bacterial cystitis is extremely common, particularly in women, and results from retrograde spread of colonic bacteria in most cases.
Other forms of cystitis have iatrogenic causes, such as radiation cystitis and hemorrhagic cystitis due to chemotherapy.
Some inflammatory or metaplastic bladder lesions are significant in that they may clinically and/or histologically mimic bladder cancer, including malakoplakia, polypoid cystitis, cystitis cystica et glandularis, and nephrogenic adenoma.
Bladder cancer is the ninth most common cancer type worldwide and is responsible for significant morbidity and mortality. It is the fourth most common cancer in American men (7% of all new cases). The overwhelming majority (>95%) of bladder tumors are of epithelial origin ( Table 21.3 ), with urothelial neoplasms being by far the most common type followed by squamous and glandular neoplasms.
|
Urothelial neoplasms represent about 90% of all bladder tumors and run the gamut from small benign lesions that do not recur to aggressive cancers that are often fatal. Many of these tumors are multifocal at presentation. Though most common in the bladder, all of the urothelial lesions described here may be seen at any site where there is urothelium, from the renal pelvis to the distal urethra.
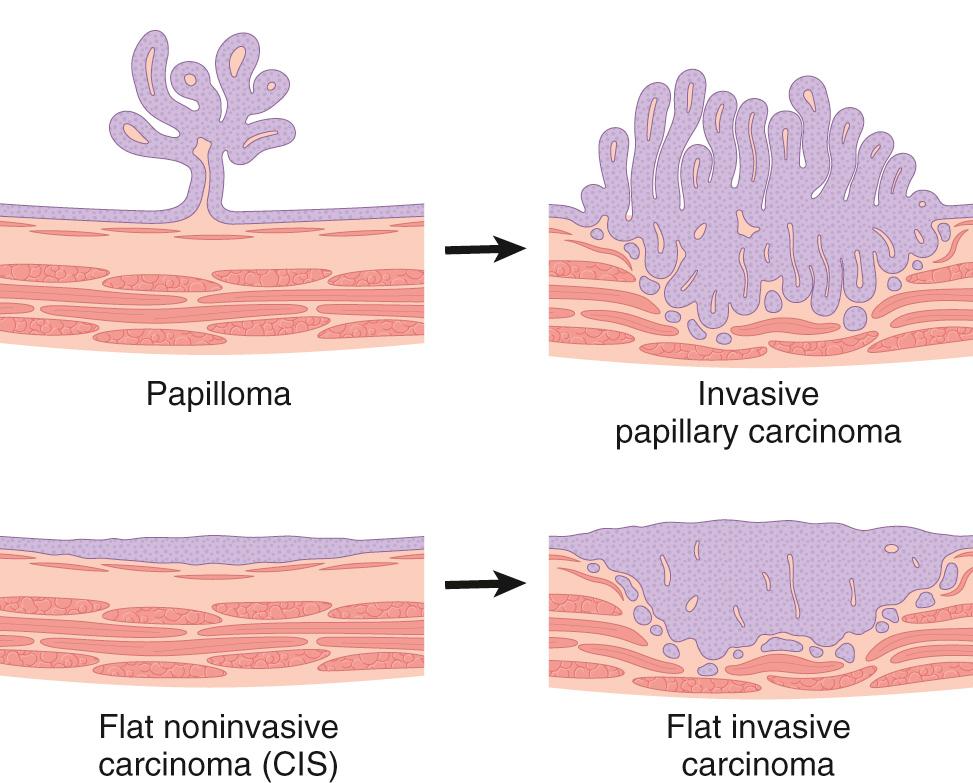
There are two distinct precursor lesions to invasive urothelial carcinoma: noninvasive papillary tumors and flat noninvasive urothelial carcinoma in situ (CIS) ( Fig. 21.4 ). The most common precursor lesions are the noninvasive papillary tumors, which originate from papillary urothelial hyperplasia. These tumors have a range of atypical changes and are graded according to their biologic behavior. The other precursor lesion to invasive carcinoma, flat noninvasive urothelial carcinoma, is referred to as CIS. As discussed in Chapter 7 , CIS is a term used to describe epithelial lesions that have the cytologic features of malignancy but are confined to the epithelium, showing no evidence of basement membrane invasion. Such lesions are considered to be high grade. In about one-half of individuals with invasive bladder cancer, the tumor has already invaded the bladder wall at the time of presentation, and precursor lesions are not detected. It is presumed in such cases that the precursor lesion was destroyed by the high-grade invasive component, which typically appears as a large, ulcerated mass. Although invasion into the lamina propria worsens the prognosis, the major decrease in survival is associated with invasion of the muscularis propria (detrusor muscle). Once muscularis propria invasion occurs, there is a 30% 5-year mortality rate.
The incidence of bladder cancer is higher in men (male-to-female ratio of 3 : 1), in higher income nations, and in urban dwellers. About 80% of patients are between 50 and 80 years of age. Bladder cancer, with rare exceptions, is not familial.
Several factors have been implicated in the causation of urothelial carcinoma, including the following:
Cigarette smoking is clearly the most important influence, increasing the risk threefold to sevenfold, depending on the duration and type of tobacco use. Between 50% and 80% of all bladder cancers among men are associated with the use of cigarettes. Cigars, pipes, and smokeless tobacco are associated with a smaller risk.
Industrial exposure to aryl amines, particularly 2-naphthylamine and related compounds, as pointed out in our earlier discussion of chemical carcinogenesis ( Chapter 7 ). Cancer appears 15 to 40 years after the first exposure.
Schistosoma haematobium infections in endemic areas (Egypt, Sudan) are an established risk. The ova are deposited in the bladder wall and incite a brisk chronic inflammatory response that induces progressive mucosal squamous metaplasia, dysplasia, and, in some instances, neoplasia. Seventy percent of the cancers are squamous, the remainder being urothelial or glandular (the least common type).
Long-term use of analgesics is implicated, as it is in analgesic nephropathy ( Chapter 20 ).
Heavy long-term exposure to cyclophosphamide, an immunosuppressive agent, induces, as noted, hemorrhagic cystitis and increases the risk of bladder cancer.
Irradiation, often administered for other pelvic malignancies, increases the risk of urothelial carcinoma occurring several years following exposure.
Analysis of the genomes of precursor lesions and invasive bladder carcinomas has identified two relatively distinct molecular pathways of tumor progression ( Fig. 21.5 ). Non–muscle-invasive papillary cancers often have gain-of-function alterations that increase signaling through growth factor receptor pathways, such as amplifications of the FGFR3 tyrosine kinase receptor gene and activating mutations in the genes encoding RAS and PI 3-kinase. These tumors frequently recur but progress to muscle-invasive bladder cancer in only about 20% of cases. The majority of muscle-invasive bladder cancers develop by progression from “flat” CIS. Mutations that disrupt the function of p53 and RB are prevalent in all muscle-invasive cancers, but occur early in the development of CIS and later in the progression of papillary cancers.

Additional work is ongoing to further characterize the genetic and molecular landscape of bladder cancer. Consistent with epidemiologic data suggesting that environmental carcinogens have an important role, bladder cancers have a high burden of somatic mutations, comparable to other carcinogen-induced cancers, such as lung cancer and melanoma . As in other cancers, recurrently mutated oncogenes and tumor suppressor genes include genes involved in cell cycle regulation, chromatin regulation, DNA repair, and growth factor signaling pathways . Analyses of RNA expression suggest the existence of several relatively distinct molecular subtypes; further work is needed to fully understand their biologic and clinical significance.
The appearance of urothelial tumors varies from purely papillary to nodular or flat. Papillary lesions are red, elevated excrescences ranging in size from less than 1 cm in diameter to large masses up to 5 cm in diameter ( Fig. 21.6 ). Multiple discrete tumors are often present.
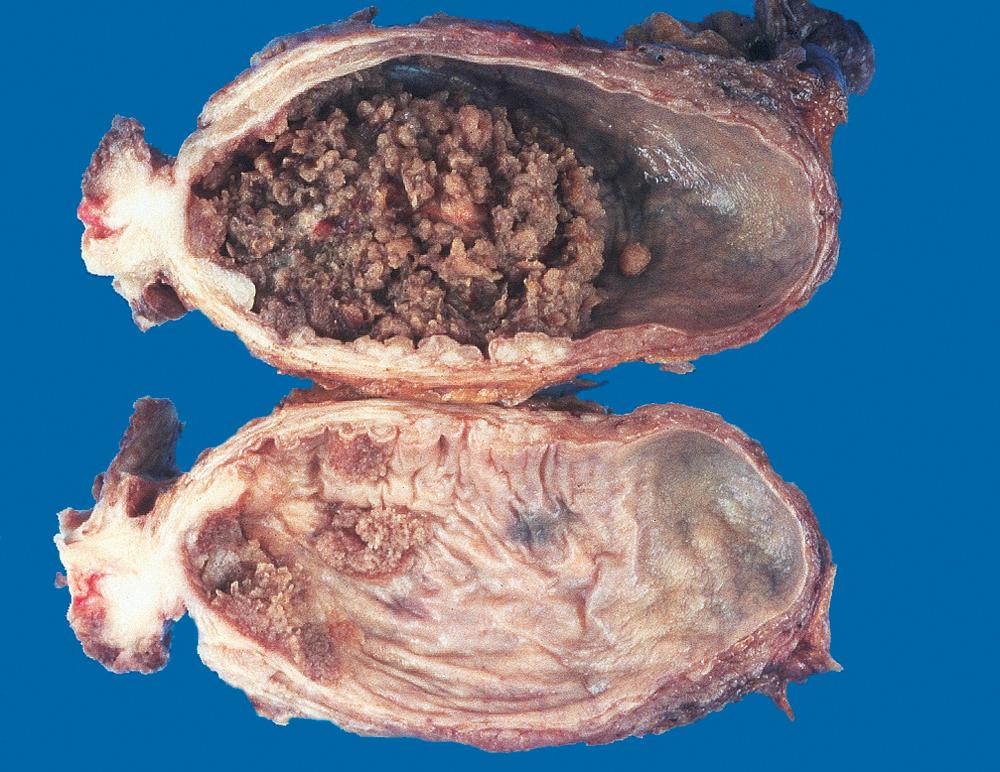
Table 21.3 lists the grading system of urothelial tumors. Papillomas represent 1% or less of bladder tumors and are often seen in younger patients. These tumors typically arise singly as small (0.5 to 2 cm), delicate structures superficially attached to the mucosa by a stalk and are referred to as exophytic papillomas. The individual finger-like papillae have a central core of loose fibrovascular tissue covered by epithelium that is histologically identical to normal urothelium ( Fig. 21.7A ). Recurrences and progression are rare but may occur. In contrast to exophytic papillomas, inverted papillomas are completely benign lesions consisting of inter-anastomosing cords of cytologically bland urothelium that extend down into the lamina propria; they simulate an invasive process.
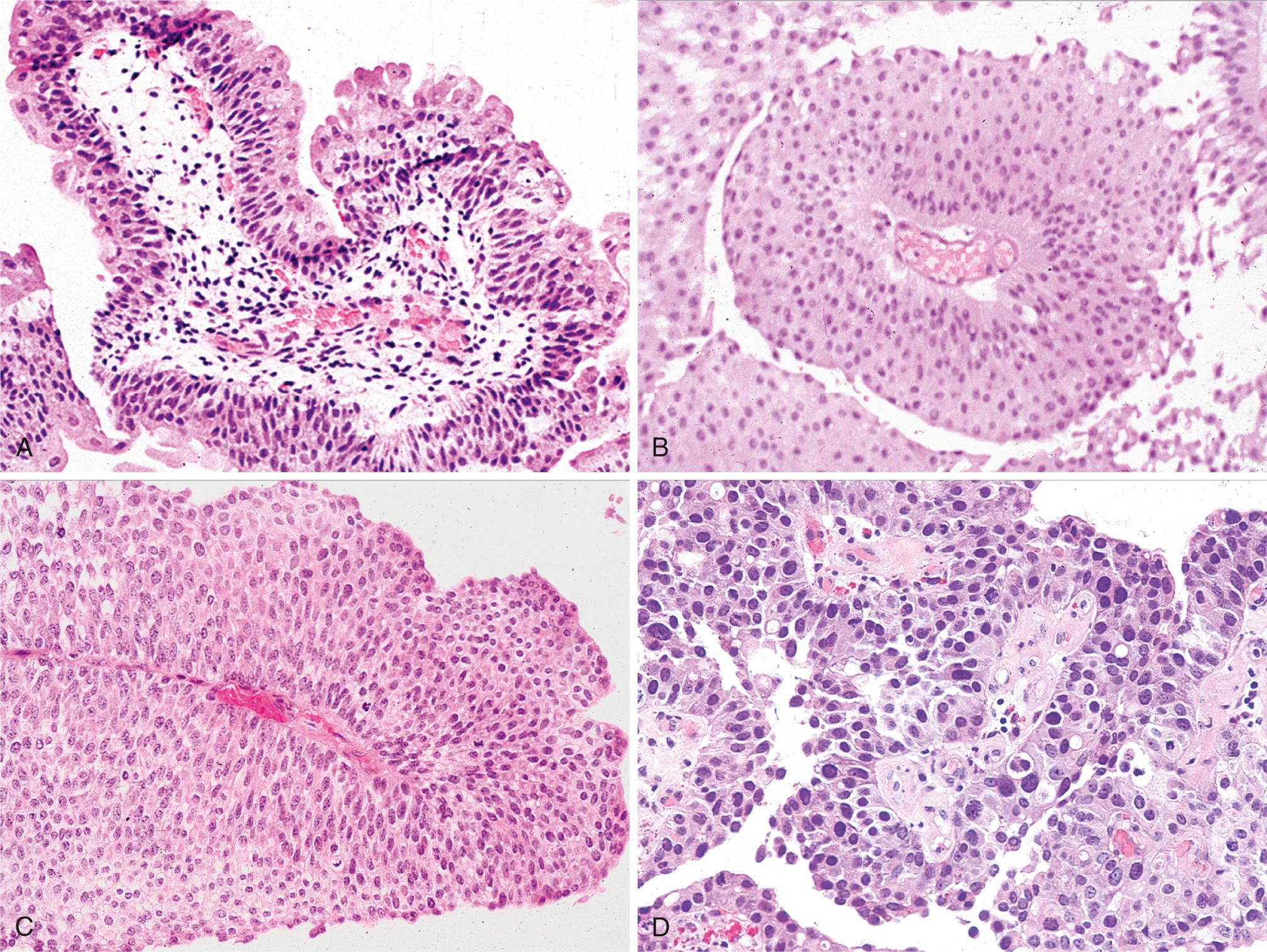
Papillary urothelial neoplasms of low malignant potential (PUNLMP) share many histologic features with papillomas, differing only in having thicker urothelium with greater density of cells ( Fig. 21.7B ) At cystoscopy, these tumors tend to be larger than papillomas and may be indistinguishable from papillary cancers. Recurrent tumors usually show the same morphology; progression to tumors of higher grade may occur but is rare.
Low-grade papillary urothelial carcinomas have an orderly architectural appearance and low-grade cytologic atypia. The cells are evenly spaced (i.e., maintain polarity) and cohesive. There are scattered hyperchromatic nuclei, infrequent mitotic figures predominantly toward the base, and slight variation in nuclear size and shape ( Fig. 21.7C ). These low-grade cancers may recur and, infrequently, may also invade. Only rarely do these tumors pose a threat to life.
High-grade papillary urothelial carcinomas contain dyscohesive cells with large hyperchromatic nuclei, irregular nuclear chromatin, and prominent nucleoli. Some of the tumor cells are highly anaplastic ( Fig. 21.7D ). Mitotic figures, including atypical ones, are frequent. Architecturally, there is disarray and loss of polarity. As compared to low-grade lesions, these tumors have a much higher incidence of progression to muscle-invasive bladder cancer and have a significant potential for metastasis to regional lymph nodes and systemic spread (e.g., to liver and lung).
CIS (or flat urothelial carcinoma) is defined by the presence of cytologically malignant cells within a flat urothelium ( Fig. 21.8 ). CIS may range from full-thickness cytologic atypia to scattered malignant cells in an otherwise normal urothelium, the latter termed pagetoid spread. A common feature shared with high-grade papillary urothelial carcinoma is a lack of cohesiveness, which leads to shedding of malignant cells into the urine. When shedding is extensive, only a few CIS cells may be left clinging to a largely denuded basement membrane. On cystoscopy CIS usually appears as an area of mucosal reddening, granularity, or thickening without an evident intraluminal mass. It is commonly multifocal and may involve most of the bladder surface and extend into the ureters and urethra. If untreated, 50% to 75% of CIS progresses to invasive cancer.
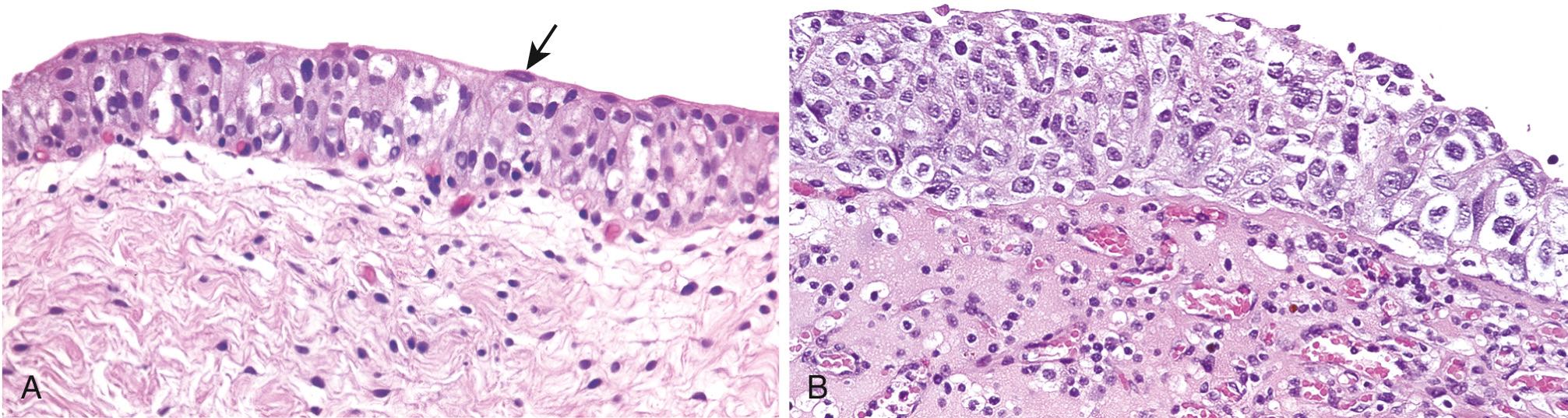
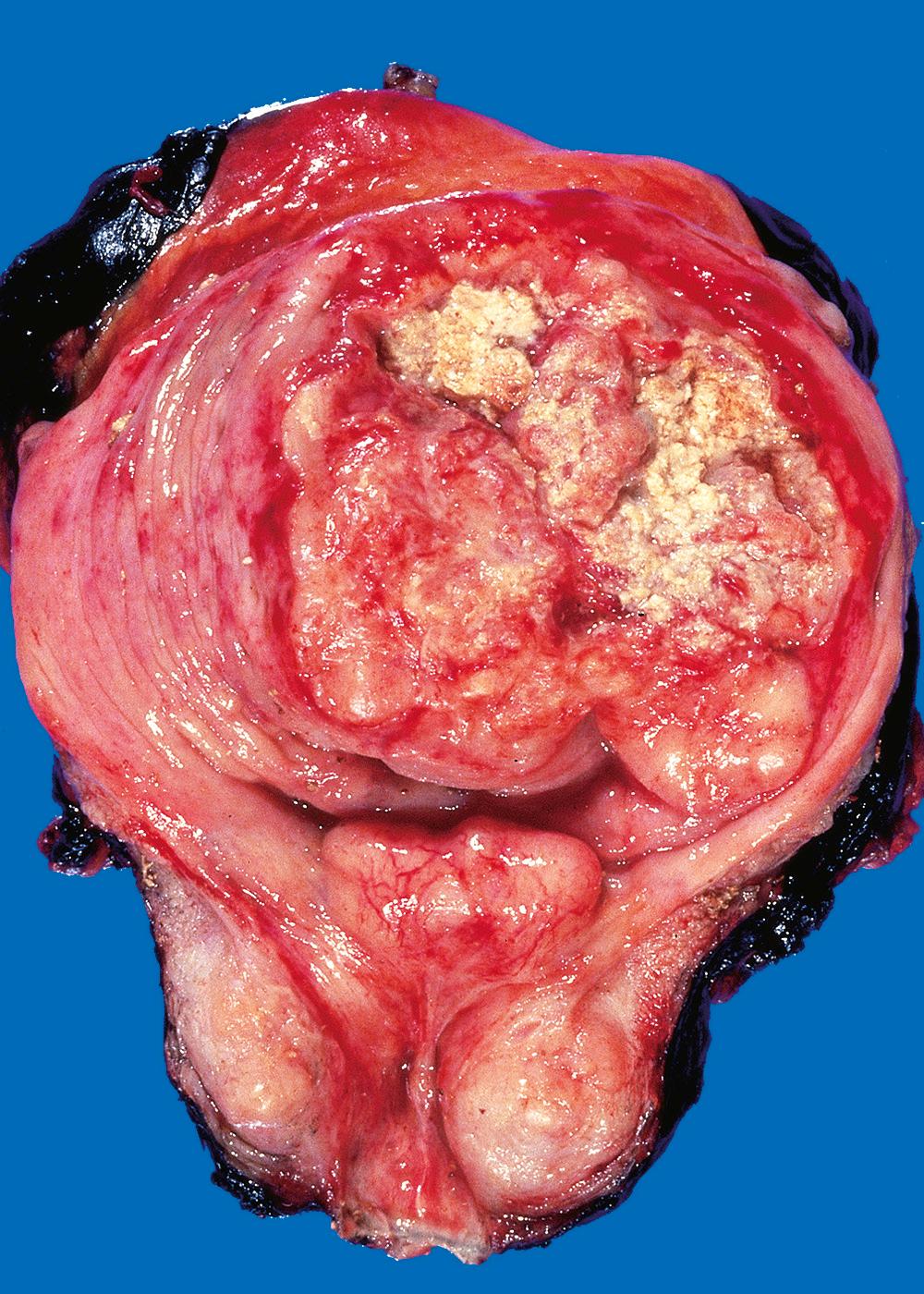
Invasive urothelial carcinoma ( Fig. 21.9 ) may be associated with papillary urothelial cancer, usually high grade, or adjacent CIS. The extent of spread (stage), based primarily on depth of invasion in the bladder wall, at the time of initial diagnosis is the most important prognostic factor. Stage also determines treatment modality, with invasion of the muscularis propria layer being an indication for radical cystectomy or radiation therapy with neoadjuvant or adjuvant chemotherapy ( Table 21.4 ).
| WHO/ISUP Grades (2016) |
| Flat Lesions |
|
| Exophytic Papillary Lesions |
|
Unusual variants of urothelial cancer include a nested variant with deceptively bland cytology, lymphoepithelioma-like carcinoma, micropapillary carcinoma, and sarcomatoid carcinoma (see Table 21.3 ).
Painless hematuria is the most common symptom of bladder cancer. Frequency, urgency, and dysuria may accompany hematuria. Occasionally, obstruction of the ureteral orifice may lead to pyelonephritis or hydronephrosis.
The subsequent treatment and course of bladder carcinoma depend on tumor grade and stage ( Table 21.5 ), with the key variable being whether the tumor is muscle-invasive or not. Noninvasive papillary urothelial tumors and those that solely invade the lamina propria (together referred to as non–muscle-invasive) constitute the majority (70% to 80%) of urothelial neoplasms. Twenty percent to 30% of bladder cancers are muscle-invasive with only a minority of these originating in patients with a prior history of non–muscle-invasive disease.
| pTNM, AJCC 8th Edition | |
| Tumor | |
| pTX | Primary tumor cannot be assessed |
| pT0 | No evidence of primary tumor |
| Ta | Noninvasive papillary carcinoma |
| Tis | Urothelial carcinoma in situ: “flat tumor” |
| T1 | Tumor invades lamina propria (subepithelial connective tissue) |
| T2 | Tumor invades muscularis propria |
| T2a | Tumor invades superficial muscularis propria (inner half) |
| T2b | Tumor invades deep muscularis propria (outer half) |
| T3 | Tumor invades perivesical soft tissue |
| T3a | Tumor invades perivesical soft tissue microscopically |
| T3b | Tumor invades perivesical soft tissue macroscopically (extravesical mass) |
| T4 | Extravesical tumor directly invades any of the following: prostatic stroma, seminal vesicles, uterus, vagina, pelvic wall, abdominal wall |
| T4a | Extravesical tumor invades directly into prostatic stroma, seminal vesicles, uterus, or vagina |
| T4b | Extravesical tumor invades pelvic wall or abdominal wall |
| Regional Lymph Nodes (pN) | |
| NX | Lymph nodes cannot be assessed |
| N0 | No lymph node metastasis |
| N1 | Single regional lymph node metastasis in the true pelvis (perivesical, obturator, internal and external iliac, or sacral lymph node) |
| N2 | Multiple regional lymph node metastasis in the true pelvis (perivesical, obturator, internal and external iliac, or sacral lymph node metastasis) |
| N3 | Lymph node metastasis to the common iliac lymph nodes |
| Metastasis | |
| M1 | Distant metastasis |
| M1a | Distant metastasis limited to lymph nodes beyond the common iliac arteries |
| M1b | Non–lymph node distant metastases |
Initial treatment of non–muscle-invasive tumors is guided by the pathologic findings. For small, localized low-grade papillary tumors, diagnostic transurethral resection is the only procedure needed. CIS and papillary tumors that are large, high grade, multifocal, have a history of recurrence, or are associated with lamina propria invasion are treated with intravesical instillation of an attenuated strain of Mycobacterium bovis called bacillus Calmette-Guérin (BCG), which elicits a local inflammatory reaction that destroys the tumor.
Unfortunately, non–muscle-invasive bladder cancers have a high tendency to recur (up to 70%) and are at risk for progression to a higher grade or stage. The risk of recurrence and progression is related to several variables, including tumor size, stage, grade, multifocality, prior recurrence, and the presence of CIS in the surrounding mucosa. Most recurrences appear at sites other than that of the original lesion, yet are clonally related, apparently arising from shedding and implantation of cells from the original tumor at a distant site. Other recurrences are clonally distinct and represent a second primary tumor. Papillomas, PUNLMP, and low-grade papillary urothelial cancer have a 98% 10-year survival rate regardless of the number of recurrences, and only a few tumors (<10%) progress to higher-grade lesions. In contrast, high-grade papillary urothelial carcinomas invade and lead to death in about 25% of cases. Patients with CIS only, as opposed to CIS associated with infiltrating urothelial carcinoma, are less likely to progress to muscle-invasive cancer (28% versus 59%) or die of disease (7% versus 45%).
Because of the risk of recurrence and progression, patients with non–muscle-invasive urothelial neoplasms need lifelong surveillance and follow-up. Surveillance cystoscopy, biopsies, and urine cytologic examinations contribute mightily to the huge burden on health care resources (over 3 billion dollars annually in the United States alone) imposed by bladder cancer. Several noninvasive assays to screen for recurrence have been developed, but none have been proven to have sufficient sensitivity and specificity to replace cystoscopy.
Muscle-invasive bladder carcinoma is treated with radical cystectomy or cystoprostatectomy, or radiation therapy with neoadjuvant and/or adjuvant chemotherapy. In addition to muscle-invasive cancers, radical cystectomy is also indicated in cases of (1) CIS or high-grade papillary cancer refractory to BCG and other intravesical therapies; (2) CIS extending into the prostatic urethra and ducts, sites where instilled BCG does not come into contact with the neoplastic cells; and (3) occasional cases of non–muscle-invasive papillary urothelial high-grade carcinoma, such as multifocal lesions that are too large and extenisive to completely eradicate by transurethral resection.
Once bladder carcinoma metastasizes, treatment options are limited. Most metastatic tumors respond poorly to chemotherapy, which produces 5-year survival rates of only 15%. As with other cancers linked to environmental carcinogens with high mutational loads, a subset (roughly 30%) of metastatic bladder carcinomas respond to immune checkpoint inhibitors, sometimes dramatically, providing some hope for this group of patients.
Squamous cell carcinomas resembling squamous cell cancers occurring at other sites make up 3% to 7% of bladder cancers in the United States but are much more frequent in countries where urinary schistosomiasis is endemic. Pure squamous cell carcinomas arise from atypical keratinizing mucosa (squamous dysplasia and CIS) and are nearly always associated with chronic bladder irritation and infection. Mixed urothelial carcinoma with areas of squamous carcinoma is more frequent than pure squamous cell carcinoma. Most are invasive, fungating tumors or are infiltrative and ulcerative. The level of cellular differentiation varies widely, from well-differentiated lesions producing abundant keratin to anaplastic tumors with only focal evidence of squamous differentiation. Adenocarcinoma of the bladder is rare and histologically identical to adenocarcinomas seen in the gastrointestinal tract. Some arise from urachal remnants or in association with extensive intestinal metaplasia (discussed earlier).
Small-cell carcinoma, indistinguishable from small-cell carcinomas of the lung, occasionally arise in the bladder, often in association with urothelial, squamous, or adenocarcinoma (see Table 21.3 ). As with small-cell carcinomas in other anatomic sites, these very aggressive tumors are strongly associated with loss-of-function mutations in the TP53 and RB tumor suppressor genes.
A great variety of benign mesenchymal tumors may arise in the bladder, having the histologic features of their counterparts elsewhere. Even collectively, they are rare. The most common is leiomyoma. They tend to grow as isolated, intramural (submucosal), encapsulated, oval-to-spherical masses up to several centimeters in diameter.
True sarcomas are distinctly uncommon in the bladder. Inflammatory myofibroblastic tumors and various carcinomas that assume sarcomatoid growth patterns are more common than true sarcomas. As a group, sarcomas tend to produce large masses (up to 15 cm in diameter) that protrude into the vesicle lumen. Their soft, fleshy, gray-white gross appearance suggests their mesenchymal nature. The most common bladder sarcoma in infancy or childhood is embryonal rhabdomyosarcoma. In some of these cases they manifest as a polypoid grape-like mass (sarcoma botryoides). The most common bladder sarcoma in adults is leiomyosarcoma ( Chapter 26 ).
Secondary malignant involvement of the bladder is most often due to direct extension of cancers arising in adjacent organs, mainly the cervix, uterus, prostate, and rectum. Lymphoma may involve the bladder as a component of systemic disease. Metastatic spread of solid tumors to the bladder may occur but is very rare.
Bladder cancer is most common in older males, and cigarette smoking constitutes one of the most important risk factors.
Painless hematuria is a common presenting symptom of bladder cancer and requires clinical investigation by cystoscopy and/or urine cytology analysis to rule out urothelial neoplasia.
Non–muscle-invasive bladder cancer is associated with gain-of-function mutations in growth factor signaling pathways and frequently recur but rarely progress in stage or grade.
Muscle-invasive bladder cancers are associated with inactivation of TP53 and RB tumor suppressor genes and often develop from “flat” carcinoma in situ, with or without a high-grade papillary component.
Molecular subtyping of bladder cancer has identified several molecular subtypes that are being evaluated for prognostic and potential therapeutic significance.
Other epithelial bladder tumor variants may occur, either alone or mixed with urothelial carcinoma, including squamous cell carcinoma, adenocarcinoma, and small cell carcinoma.
Obstruction of the bladder outlet is of major clinical importance because of its eventual effect on the kidney. In males, the most common cause is enlargement of the prostate gland due to benign prostatic hyperplasia (BPH). Bladder obstruction is less common in females and is most often caused by cystocele of the bladder. Infrequent causes are (1) congenital urethral strictures; (2) inflammatory urethral strictures; (3) inflammatory fibrosis and contraction of the bladder; (4) bladder tumors, either benign or malignant; (5) invasion of the bladder neck by tumors arising in contiguous organs; (6) mechanical obstructions caused by foreign bodies and calculi; and (7) injury of nerves controlling bladder contraction (neurogenic bladder).
In the early stages there is only thickening of the bladder wall due to smooth muscle hypertrophy. With progressive hypertrophy the individual muscle bundles greatly enlarge and produce trabeculation of the bladder wall (see Fig. 21.10 ). In the course of time, crypts form and may be converted into diverticula.
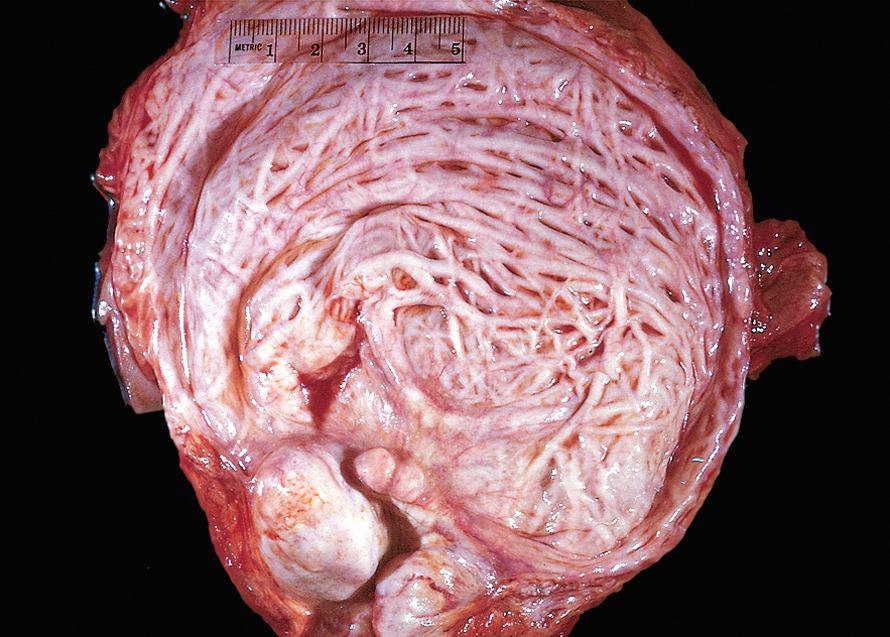
In some cases of acute obstruction or in terminal disease when the patient's normal reflex mechanisms are depressed, the bladder may become extremely dilated. The enlarged bladder may reach the brim of the pelvis or even the level of the umbilicus. In such cases, the bladder wall is markedly thinned and lacks trabeculations.
Urethritis is classically divided into gonococcal and nongonococcal causes. Gonococcal urethritis is one of the earliest manifestations of this venereal infection. Nongonococcal urethritis is common and can be caused by several different organisms. Various strains of Chlamydia (e.g., Chlamydia trachomatis ) are the cause of 25% to 60% of nongonococcal urethritis in men and about 20% in women. Mycoplasma (Ureaplasma urealyticum) also accounts for symptoms of urethritis in many cases. Urethritis is often accompanied by cystitis in women and by prostatitis in men. In many instances of suspected bacterial urethritis, no organism can be isolated. Some urethritis is truly noninfectious in origin. An example of such an inflammatory urethritis is a disorder known as reactive arthritis (formerly Reiter syndrome), which is associated with the clinical triad of arthritis, conjunctivitis, and urethritis ( Chapter 26 ).
The morphologic changes are entirely typical of inflammation in other sites within the urinary tract. The urethral involvement is not itself a serious clinical problem but may cause considerable local pain, itching, and frequency and may warn of more serious disease at higher levels of the urogenital tract.
Urethral caruncle is an inflammatory lesion that presents as a small, red, painful mass about the external urethral meatus, typically in older females. It consists of inflamed granulation tissue covered by an intact but friable mucosa, which may ulcerate and bleed with the slightest trauma. Surgical excision affords prompt relief and cure.
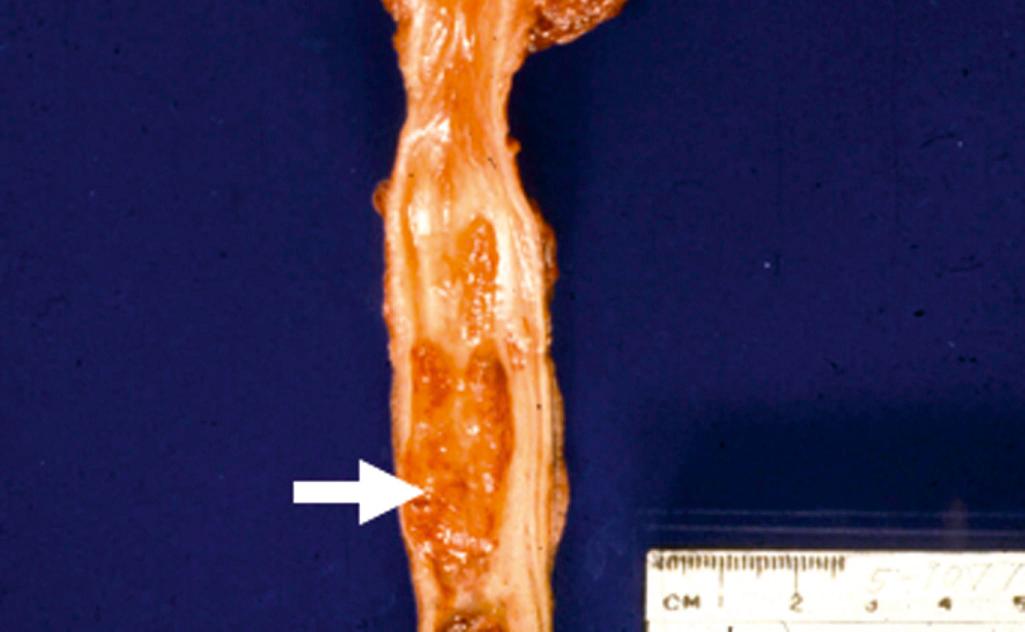
Benign epithelial tumors of the urethra include squamous and urothelial papillomas, inverted urothelial papillomas, and condylomas. Primary carcinoma of the urethra is an uncommon lesion ( Fig. 21.11 ). Cancers arising within the proximal urethra tend to show urothelial differentiation and are analogous to those occurring within the bladder, whereas lesions originating within the distal urethra are more often squamous cell carcinomas and HPV-related. Adenocarcinomas are infrequent in the urethra and generally occur in women. Neoplasms of the prostatic urethra are addressed later in the section on the prostate.
The penis is involved by many congenital anomalies; only those that are clinically significant are discussed here.
Malformation of the urethral groove and canal may create an abnormal opening either on the ventral surface of the penis (hypospadias) or on the dorsal surface (epispadias) . Either of these anomalies can be associated with failure of normal descent of the testes and with malformations of the urinary tract. Hypospadias, the more common of the two, occurs in approximately 1 in 300 live male births. Even when isolated, these urethral defects may have clinical significance because the abnormal opening is often constricted, resulting in urinary tract obstruction and an increased risk of ascending infections. When the orifices are situated near the base of the penis, normal ejaculation and insemination are hampered and may be a cause of sterility.
When the orifice of the prepuce is too small to permit its normal retraction, the condition is designated phimosis. An abnormally small orifice may result from anomalous development but is more frequently the result of repeated bouts of infection that cause scarring of the preputial ring. Phimosis is important because it interferes with cleanliness and permits the accumulation of secretions and detritus under the prepuce, favoring the development of secondary infections and increasing the risk for penile carcinoma.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here