Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The pathologist is often asked to examine liver biopsies obtained to evaluate liver dysfunction in transplant patients, including recipients of liver, renal and bone marrow grafts. For liver transplantation, liver biopsy remains the diagnostic ‘gold standard’ when jaundice and allograft dysfunction develop, because biochemical tests do not adequately discriminate between rejection and other conditions that may develop in the allograft. Moreover, even when serum liver function tests are normal, histological abnormalities (including rejection lesions) may be present. At the time of harvesting or engraftment, the donor liver may also require assessment, sometimes by frozen section, for lesions that may determine whether or not the graft can be used, and that can affect the postoperative course and appearance of subsequent posttransplantation biopsies. This chapter reviews the histopathological features of liver transplant rejection and other conditions affecting the allograft. The concluding sections discuss liver disease in recipients of renal and bone marrow transplants.
The source of the donor allograft is an important consideration when evaluating liver biopsies obtained prior to transplantation, after graft revascularisation or at later times. Biopsies from potential living donors in apparent good health may disclose conditions such as steatohepatitis or primary biliary cholangitis (PBC), which disqualifies their candidacy. Living donor left- or right-lobe grafts, if small in relation to the recipient’s size, may show cholestasis or congestion associated with ascites, prolonged coagulation parameters and impaired metabolic function immediately after transplantation ( small-for-size syndrome ) because of problems in venous drainage and portal hyperperfusion. Postoperative portal tract biliary obstructive changes may also be present due to ischaemia or mechanical obstruction of the large bile ducts associated with this type of procedure. Cadaveric livers are subject to preservation (ischaemia/reperfusion) injury after harvesting and transport to the site of surgery, which in early baseline biopsies is evident as variable degrees of perivenular necrosis, liver-cell ballooning and/or apoptosis. The increased risk of postoperative biliary strictures and bile leaks with ‘donation after cardiac death’ grafts should be kept in mind if cholestasis and features of biliary obstruction are seen in a posttransplantation biopsy. Liver transplantation where ‘extended donor criteria’ are invoked (e.g. advancing age, donation after cardiac death, hepatitis virus infection, others) often requires morphologic assessment prior to the actual transplantation and allograft revascularisation. Donors with known chronic hepatitis B or C benefit from baseline biopsy grading and staging as a source for comparison in the event that later biopsies are obtained. With the advent of direct-acting antiviral agents (DAAs) that effectively treat hepatitis C virus (HCV), there is increased interest in using HCV-seropositive deceased donor livers for transplantation to HCV-seronegative recipients (so-called HCV mismatch transplantation). In the United States, increasing deaths from opioid abuse have increased an available pool of HCV-positive livers (and other organs) for transplantation. The operative principle in ‘HCV mismatch transplantation’ is that antiviral therapy after organ transplantation will effect cure and serologic sustained viral response (SVR). Liver biopsy is likely to remain important in this setting, because antiviral-treated liver transplant recipients with SVR often continue to demonstrate chronic inflammation and fibrosis on liver biopsy and, in certain cases, may also still harbour HCV RNA in liver tissue.
Allograft biopsies obtained soon after transplantation sometimes disclose an unsuspected condition in the donor, such as α 1 -antitrypsin deficiency, iron overload or amyloidosis. Certain known genetic, haematological or immunological disorders in potential donors may constitute contraindications.
Frozen section of potential donor livers may be requested to exclude pre-existing disease. Pathologists providing frozen-section coverage should be aware of three common reasons why frozen sections are requested: (1) to determine whether steatosis is present , and its degree; (2) to exclude changes of chronic hepatitis if the donor is known to be positive for antibodies to hepatitis B core antigen but negative for surface antigen (i.e. possible occult hepatitis B), or is positive for HCV; and (3) to evaluate a mass found in the donor liver. Concern about a substantially fatty liver is based on the increased incidence of primary graft dysfunction or non-function when this is present. The transplant surgeon may be concerned about significant steatosis when the donor liver appears yellow, has rounded edges and shows no surface capsular scratch marks (foci of capsular collagen rupture and disruption, thought to be a procurement phenomenon due to proximity to ice crystals). The degree of macrovesicular (large droplet) fatty change should be categorised according to the percentage of parenchymal involvement as absent, mild (<30%), moderate (30%–60%) or marked (>60%). Transplant surgeons have considered the last category unsuitable for use because of the high risk of primary dysfunction or non-function associated with severe steatosis. Microvesicular (small-droplet) fat is held not to be a contraindication, however, but if it is substantial, it should also be graded and merits discussion with the transplant team because it may delay return of hepatic function and clinical recovery. Diffuse portal mononuclear inflammatory cell infiltrates in donors with markers of hepatitis B or C viral infection support the presence of chronic hepatitis . The significance of this finding needs to be considered by the transplant team. With regard to mass lesions in the donor liver, demonstration of a malignant or metastatic tumour is an obvious contraindication to its use. However, the features of benign lesions such as focal nodular hyperplasia ( Ch. 11 ) are important to recognise because they are often encountered in this setting.
Needle liver biopsies are obtained as part of a liver transplantation protocol or because of clinical deterioration. Discussion with the clinical team and careful review of pertinent radiographic, biochemical and microbiological findings are critical to biopsy interpretation and institution of appropriate therapy. Serial biopsies may be necessary to resolve difficult diagnostic problems.
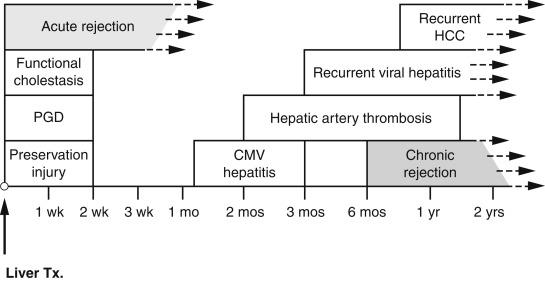
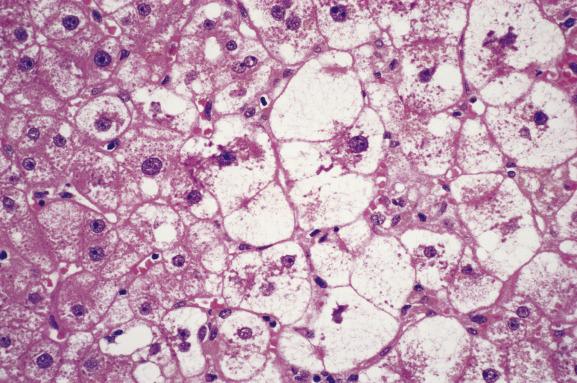
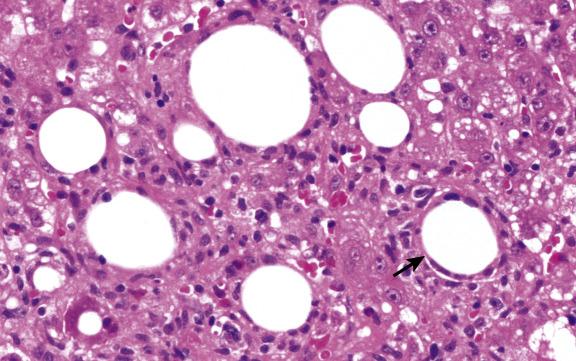
There are many causes of allograft injury in addition to rejection ( Box 16.1 ), and these should be considered in the context of the time elapsed since transplantation ( Fig. 16.1 ). For several weeks following transplantation, functional cholestasis may be present and must, if possible, be distinguished from the cholestasis of acute rejection, bile-duct obstruction, hepatitis, drug toxicity and sepsis. Bile is present within hepatocytes and canaliculi. This impairment of bile flow can be explained by exposure of the donor liver to cold ischaemia and reperfusion injury (preservation injury) with resultant damage to liver-cell organelles. Liver-cell death due to preservation injury actually shows features of both necrosis and apoptosis (necrapoptosis). Early postoperative cholestasis may also be due to a ‘small-for-size’ graft. Cholestasis may be accompanied by hepatocellular ballooning in perivenular regions ( Fig. 16.2 ) or in a diffuse distribution. In the absence of frank perivenular necrosis, ballooning does not confer an unfavourable prognosis. Hypoperfusion liver damage in the perioperative period may result in necrosis in periportal or perivenular regions and sometimes an irregular subcapsular band of infarction. If the donor liver is fatty, rupture of hepatocytes affected by preservation injury may rarely cause sinusoidal engorgement by lipid vacuoles ( lipopeliosis ) ( Fig. 16.3 ).
Graft rejection
Humoral (hyperacute)
Acute (cellular)
Chronic (ductopenic)
Functional cholestasis
Preservation injury
Bile-duct obstruction
Thrombosis of hepatic artery or portal vein
Infections and sepsis
Drug toxicity
Recurrence of original disease
Neoplastic disease
Posttransplant lymphoproliferative disease
Hepatocellular carcinoma
In evaluating posttransplant biopsies, special attention should be paid to the portal tracts, the major sites of rejection lesions. The type of cellular infiltrate, the bile ducts, portal-vein branches and hepatic arterioles are examined to distinguish rejection from other conditions with portal tract pathology, particularly bile-duct obstruction , recurrent viral hepatitis , drug toxicity and immunosuppression-related lymphoproliferative disease (see ‘Differential diagnosis in transplant biopsies’ section, later). The perivenular region also requires inspection for possible preservation injury, cholestasis or inflammation, and for necroinflammation which may accompany portal tract lesions in more severe cases of acute rejection. The lobular parenchyma shows few alterations in rejection apart from cholestasis and the occasional apoptotic bodies and scattered liver-cell mitoses which develop as the allograft equilibrates to the appropriate size for the recipient. As a result, in cases where confusion arises in the interpretation of portal changes, it is important to evaluate the lobular parenchyma carefully for evidence of intercurrent diseases such as viral or drug hepatitis. The pathologist should always bear in mind that a given biopsy may show superimposed features attributable to several different posttransplantation complications.
The histopathological lesions of liver allograft rejection have been well characterised and are classified as humoral rejection , acute ( cellular ) rejection and chronic ( ductopenic ) rejection , as recommended by an international working party which met in 1994. Acute and chronic rejection are the most common forms seen in clinical practice.
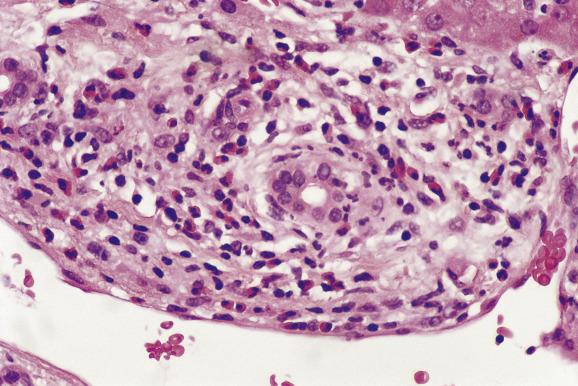
Antibody-mediated rejection (AMR) is rare after liver transplantation and has been best studied in recipients of ABO-incompatible allografts. Microvascular damage evolves over the first few hours after transplantation, consisting of sinusoidal infiltrates of neutrophilic leukocytes, fibrin and red blood cells associated with focal haemorrhages. This progresses to portal and periportal oedema with coagulative and haemorrhagic necrosis and a ductular reaction over the next few days ( Fig. 16.4 ). Immunofluorescent studies show linear deposits of immunoglobulin G (IgG) or IgM, complement fractions C1q, C3 and C4 and fibrinogen in arterial walls. The graft may remain stable in some patients for the first few days, however, possibly because of Kupffer-cell protection against the effects of circulating antibodies. Graft failure within 2–4 weeks is associated with a progressive and marked rise in serum aminotransferase activity. The liver appears mottled and cyanotic at gross examination. Recipients of ABO-unmatched livers may also develop graft-versus-host haemolysis , associated with erythrophagocytosis and Kupffer-cell siderosis .
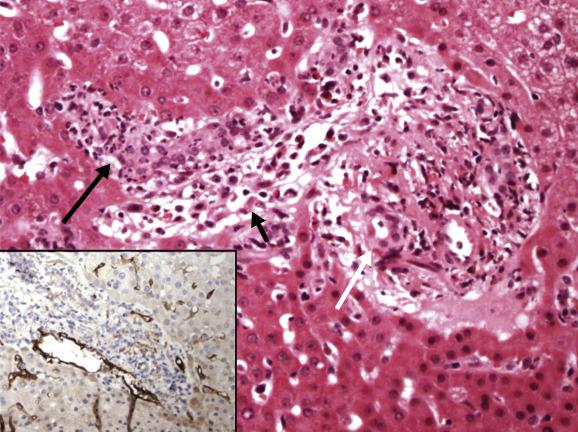
A potential role for antibody-mediated rejection is sometimes considered in the ABO-compatible recipient with early or late allograft dysfunction/failure who has high-titre donor-specific antibodies. Recent histopathological data on AMR were incorporated at the most recent (2016) meeting of the Banff Working Group to provide guidelines for histopathological criteria for AMR, which include (1) endothelial cell hypertrophy (‘hobnail’ endothelial cells) involving portal veins, capillaries and inlet venules (periportal sinusoids), accompanied by (2) marginated and/or intraluminal monocytes, neutrophils or eosinophils adherent to and embedded into the endothelium. C4d immunohistochemical staining of paraffin-embedded biopsies should be scored as minimal (<10% portal tracts), focal (10%–50% portal tracts) or diffuse (>50% portal tracts) positivity of portal vein and portal capillaries, with inlet-venule staining usually a feature seen in the highest score (diffuse) category ( Fig. 16.4 , inset ). While AMR is uncommon relative to T-cell-mediated acute cellular rejection (discussed in detail in the next section), when AMR is suspected as a cause of chronic allograft dysfunction and/or fibrosis, the recommended workup should include assessment of donor-specific antibodies, histopathological evaluation and C4d staining.
Acute (cellular) rejection , the most common form of rejection, is a cell-mediated immune injury directed at bile-duct epithelium and the endothelium of portal-vein branches and terminal hepatic venules. This usually occurs within the first month to 6 weeks after transplantation, but may be seen later if immunosuppression is lowered or discontinued. The characteristic histological triad of cellular rejection includes portal inflammation , bile-duct damage and endotheliitis ( endothelialitis ). Endotheliitis is not present in all cases. The portal inflammatory lesion is typically heterogeneous, with lymphocytes predominating among plasma cells, neutrophils and, occasionally, large lymphoid cells, some in mitosis ( Fig. 16.5 ). Eosinophils are often abundant ( Fig. 16.6 ), which is a very helpful diagnostic sign that acute rejection is present.
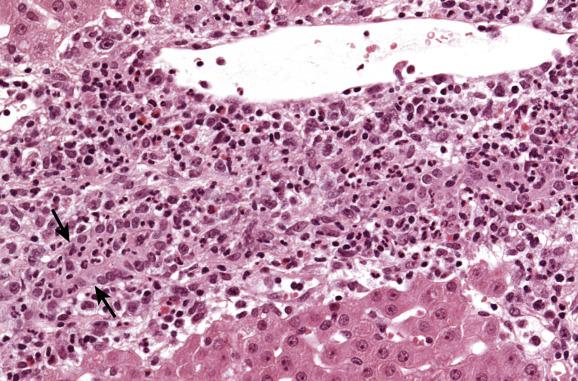
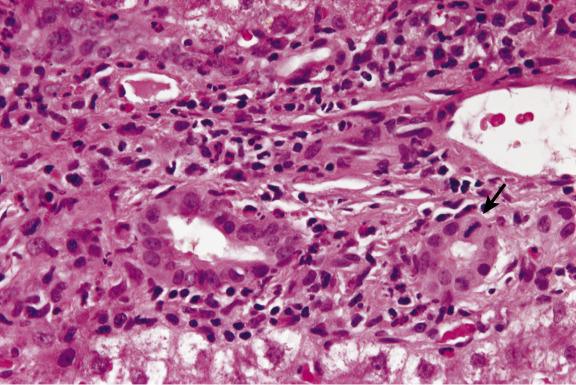
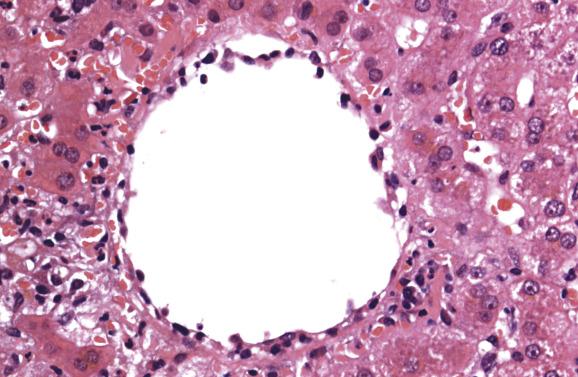
Bile ducts are surrounded and infiltrated by immune cells, and damage to their epithelium takes the form of variation in nuclear size, vacuolation of cytoplasm, regions of cell stratification or cell loss and irregularity of duct outlines ( Figs 16.5–16.7 ). Endotheliitis comprises attachment of lymphoid cells to the endothelium of portal-vein branches or terminal hepatic venules, variable degrees of endothelial damage, subendothelial inflammation ( Figs 16.5 and 16.6 ) and lifting off of endothelial cells from the underlying vein wall ( Fig. 16.8 ). Sinusoidal endotheliitis is occasionally also present, but can be a prominent pattern in certain cases. Mild focal endotheliitis is sometimes found in association with hypoperfusion damage in baseline biopsies, but extensive endotheliitis in the postoperative period is very characteristic of rejection. Necrosis of perivenular hepatocytes, accompanied by endotheliitis of terminal hepatic venules and expansive portal inflammatory lesions involving the periportal parenchyma, is indicative of severe acute rejection. Central perivenulitis (central venulitis) —characterised by endotheliitis of terminal venules, drop-out and apoptosis of nearby hepatocytes (sometimes with focal sinusoidal congestion and dilatation)—often presages later episodes of acute rejection and chronic ductopenic rejection. Central perivenulitis is a common expression of rejection in paediatric allografts. It is sometimes the chief manifestation of rejection, with few or no portal tract changes, as isolated central perivenulitis in both paediatric and adult allografts, many months or longer after transplantation (see ‘Late liver allograft dysfunction’ section, later).
Descriptive and semi-quantitative grading of acute rejection can effectively be accomplished using the scoring system presented in the Banff international consensus document ( Table 16.1 ). Using the semi-quantitative approach of assigning a numerical score to each component of the acute rejection triad, a total Rejection Activity Index ( RAI ) can be conveyed in the biopsy report. Alternatively, a simpler global assessment of the biopsy as showing indeterminate, mild, moderate or severe changes of acute rejection can be used ( Table 16.2 ). The choice of grading system, as with grading and staging for chronic hepatitis, should be made after discussion with clinicians.
| Category | Criteria | Score |
|---|---|---|
| Portal inflammation | Mostly lymphocytic inflammation involving, but not noticeably expanding, a minority of the triads | 1 |
| Expansion of most or all of the triads by a mixed infiltrate containing lymphocytes with occasional blasts, neutrophils and eosinophils | 2 | |
| Marked expansion of most or all of the triads by a mixed infiltrate containing numerous blasts and eosinophils with inflammatory spillover into the periportal parenchyma | 3 | |
| Bile-duct inflammation damage | A minority of the ducts are cuffed and infiltrated by inflammatory cells and show only mild reactive changes, such as increased nucleus-to-cytoplasm ratio of the epithelial cells | 1 |
| Most or all of the ducts are infiltrated by inflammatory cells. More than an occasional duct shows degenerative changes such as nuclear pleomorphism, disordered polarity and cytoplasmic vacuolisation of the epithelium | 2 | |
| As above for 2, with most or all of the ducts showing degenerative changes or focal luminal disruption | 3 | |
| Venous endothelial inflammation | Subendothelial lymphocytic infiltration involving some, but not most, of the portal and/or hepatic venules | 1 |
| Subendothelial infiltration involving most or all of the portal and/or hepatic venules | 2 | |
| As above for 2, with moderate or severe perivenular inflammation that extends into the perivenular parenchyma and is associated with perivenular hepatocyte necrosis | 3 |
∗ The Rejection Activity Index (RAI) is the sum of the scores for each of the three components of acute rejection. RAI ≥ 4 (mild), RAI ≥ 6 (moderate or severe).
| Global assessment ∗ | Criteria |
|---|---|
| Indeterminate | Portal inflammatory infiltrate that fails to meet the criteria for the diagnosis of acute rejection (see text) |
| Mild | Rejection infiltrate in a minority of the triads that is generally mild and confined within the portal spaces |
| Moderate | Rejection infiltrate, expanding most or all of the triads |
| Severe | As above for moderate, with spillover into periportal areas and moderate-to-severe perivenular inflammation that extends into the hepatic parenchyma and is associated with perivenular hepatocyte necrosis |
∗ Verbal description of mild, moderate or severe acute rejection could also be labelled as grades I, II and III, respectively.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here