Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The bicuspid aortic valve (BAV) is the most common congenital heart condition, affecting approximately 1% of the population.
Familial BAV occurs in 9% to 10% of first-degree relatives. Familial aortic aneurysm with or without BAV may occur in certain families; it is inherited as an autosomal dominant condition with incomplete penetrance and inconsistent expression.
BAV may accompany other congenital cardiovascular defects and conditions, including coarctation of the aorta and Turner syndrome.
When transthoracic echocardiography is not diagnostic, transesophageal echocardiography, cardiac magnetic resonance imaging, or cardiac computed tomography may be useful in diagnosing BAV disease.
Aortic dilation, involving the ascending aorta and/or aortic root, occurs frequently in individuals with BAV disease, even in the absence of aortic stenosis or aortic regurgitation, and may occur late after aortic valve replacement.
Abnormal aortic systolic flow patterns through the BAV lead to abnormal regional aortic wall stress and contribute to the aortopathy in this disorder.
The aortopathy of BAV disease is associated with cystic medial degeneration, alterations in signaling pathways and matrix metalloproteinase activity, and apoptosis, all of which place the patient with BAV at increased risk for aortic aneurysm and aortic dissection.
The BAV may be part of certain syndromic and nonsyndromic thoracic aortic aneurysm disorders.
Most patients with BAV will require surgical therapy on the valve and/or aorta during their lifetime.
After BAV replacement, the patient remains at risk for late ascending aortic aneurysm formation and aortic dissection. These complications may be related to the prior valve lesion and aortopathy phenotype, and they are more common with regurgitant than with stenotic valves, especially with the root phenotype. Surveillance of the aorta late after BAV replacement is important.
Bicuspid aortic valve (BAV) is an inherited form of heart disease in which two of the leaflets of the aortic valve fuse during fetal development, resulting in a two-leaflet valve. It is the most common congenital heart disorder and has long been recognized as an important cause of valvular heart disease.
Leonardo da Vinci sketched the bicuspid variant of the aortic valve more than 400 years ago. The clinical and valvular sequelae of the BAV were realized more than 150 years ago. Osler emphasized endocarditis as a complication in 1886. The association of congenital BAV with diseases of the aorta was first recorded by Abbott in 1927. In 1984, the relationship between BAV and aortic dissection risk was emphasized, observing a ninefold greater risk of aortic dissection among BAV patients. Discoveries in genetics and signaling pathways, advances in imaging, and improvements in aortic and valve surgery have improved the understanding and management of BAV and its associated complications.
The prevalence of BAV is approximately 1% of the population, with a 2:1 to 3:1 male-to-female ratio ( Table 11.1 ). In the largest reported autopsy series, involving 21,417 consecutive cases, the prevalence of BAV was 1.37%. In a study of 1075 neonates, the prevalence of BAV was 4.6 per 1000 live births. Among 20,946 military recruits, the prevalence of BAV was 0.8%. Certain groups of patients have a much higher prevalence of BAV than the general population; the rates are approximately 30% to 50% for patients with coarctation of the aorta (CoA) and 30% for Turner syndrome (TS) females.
| Study Authors | Year | Study Population (N) | BAV Prevalence (%) |
|---|---|---|---|
| Olsery | 1886 | 800 | 1.2 |
| Lewis and Grant | 1923 | 215 | 1.39 |
| Wauchope | 1928 | 9,966 | 0.5 |
| Grant et al | 1928 | 1,350 | 0.89 |
| Gross | 1937 | 5,000 | 0.56 |
| Roberts | 1970 | 1,440 | 0.9 |
| Larson and Edwards | 1984 | 21,417 | 1.37 |
| Datta et al | 1988 | 8,800 | 0.59 |
| Pauperio et al | 1999 | 2,000 | 0.65 |
The semilunar valves originate from the mesenchymal outgrowths along the ventricular outflow tract of the primary heart tube. Several molecular signaling pathways have been implicated in its development, including transforming growth factor-β (TGF-β), RAS, WNT/β-catenin, vascular endothelial growth factor (VEGF), and NOTCH signaling. , Anomalous behavior of cells derived from the neural crest has been implicated as a possible cause of BAV because BAV is often associated with congenital malformations of the aortic arch and other neural crest–derived systems. , A primary molecular abnormality of the extracellular matrix may trigger abnormal valvulogenesis. Endothelial nitric oxide (eNOS) signaling may also be important in the pathogenesis of BAV and aortic disease.
Different pathways were shown to be responsible for BAV leaflet orientation in animal models: one that relies on a nitric oxide–dependent epithelial-to-mesenchymal transformation and another that results from the distorted behavior of neural crest cells. GATA5 gene expression is restricted to endocardial cushions, and targeted inactivation of GATA5 in mice leads to development of BAV, involving several signaling pathways, including that of NOTCH . The ubiquitin fusion degradation 1–like gene (UFD1L) , which is expressed in the embryonic outflow tract, is downregulated in BAV tissue. NOTCH1 encodes for a transmembrane protein that is important in cardiac embryogenesis, including the aortic and pulmonary valves and aorta. NOTCH1 mutations (9q34.3) are found in a small number of families with BAV and ascending aortic aneurysms. , Animal models propose deficient signal exchanges among the endocardial cushions, second heart field, and neural crest cells (with different embryologic origins of the outflow tract and aortic segmental anatomy) as a link between BAV and aortic wall abnormalities , ( Fig. 11.1 ).
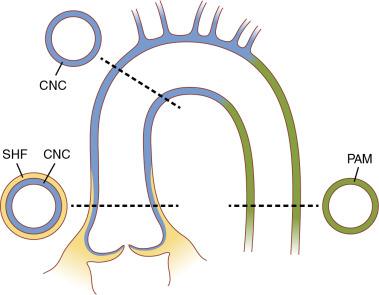
Family studies report the prevalence of BAV among first-degree relatives of an individual with a BAV is 9% to 10%. , The inheritance of BAV is consistent with an autosomal dominant pattern with reduced penetrance. Monozygotic twins do not necessarily both have BAV, highlighting the incomplete penetrance of this disorder.
Although BAV is a heritable trait, the genetic causes remain elusive. In most patients, no specific genetic variant has been discovered. Mutations in NOTCH1 are associated with familial and nonfamilial BAV and may result in aortic aneurysms and early aortic calcification. Mutations in other single genes such as GATA5, GATA6, and NKX2-5 have been associated with BAV; ACTA2 , TGFB2, LOX, SMAD6 , and other genes have been associated with BAV and thoracic aortic aneurysm ( Table 11.2 ). Polymorphisms in other genes may be associated with BAV risk. BAV is also associated with genetic conditions such as DiGeorge syndrome, Loeys-Dietz syndrome, Anderson syndrome (i.e., KCNJ2 mutation), and complex congenital heart conditions such as Shone complex and hypoplastic left heart syndrome. One cross-sectional exome-wide association study found no association signals for BAV with or without thoracic aortic aneurysm, but whole-genome sequencing may provide more comprehensive understanding of the genetics of BAV. ,
| Mutated Gene | Genetic Defect and Phenotype |
|---|---|
| ACTA2 | FTAA with premature CAD, CVD, livedo reticularis; BAV in 3% |
| ELN | BAV may occur with cutis laxa |
| FBN1 | Marfan syndrome or nonsyndromic FTAA; reported increased frequency of BAV |
| FLNA | BAV may occur with X-linked cardiac valve dysplasia |
| GATA5 | Rarely associated with familial BAV |
| GATA6 | Loss-of-function mutation associated with BAV |
| KCNJ2 | Anderson syndrome; associated with BAV |
| LOX | FTAA; associated with BAV |
| MAT2A | FTAA and BAV |
| MYH11 | FTAA; BAV may occur |
| NKX2-5 | Rarely associated with familial BAV |
| NOTCH1 | Isolated BAV with AS, familial BAV or FTAA |
| SMAD3 | Syndromic FTAA (LDS type 3, aneurysm-osteoarthritis syndrome); may have BAV |
| SMAD6 | Rarely associated with BAV, AS, and CoA |
| TGFB2 | Syndromic FTAA (LDS type 4); may have BAV |
| TGFB3 | Syndromic FTAA (LDS type 5); may have BAV |
| TGFBR1 TGFBR2 | Syndromic (LDS) or nonsyndromic FTAA; BAV in 2%–17% |
BAV anatomy typically includes unequal cusp size due to fusion of two cusps, leading to one larger cusp ( Fig. 11.2 ). Fusion most commonly occurs between the right and left coronary leaflets (70%–86%). There may also be fusion between the right and noncoronary leaflets (12%) or between the left and noncoronary leaflets (3%). , Several classification systems have been used to describe BAVs based on the fused cusp orientation (i.e., right-left, right-noncoronary, or left-noncoronary) and the presence or absence of a raphe ( Fig. 11.3 ). The raphe (89% of cases) is the site of fusion of the conjoined cusps. The presence of a raphe is associated with a higher prevalence of valve dysfunction.
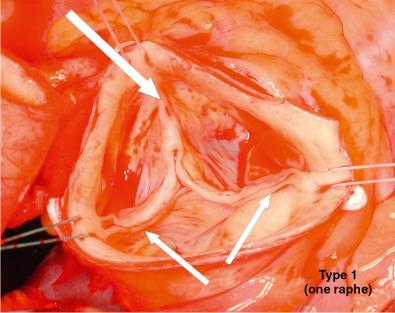
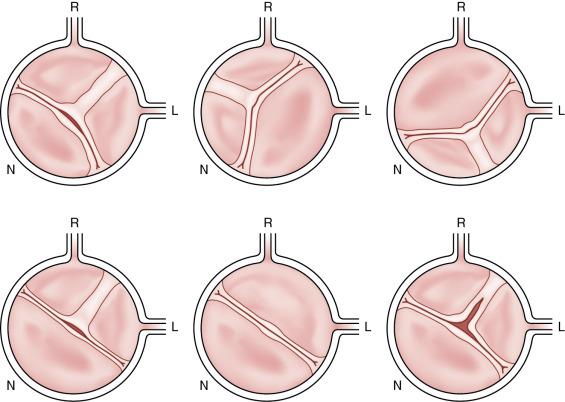
In most instances, BAV is an isolated cardiovascular finding. However, BAV may coexist with other congenital cardiovascular defects or syndromes.
BAV is associated with CoA in 30% to 50% of patients with CoA, whereas CoA has been found in 6% of patients with BAV. , Interrupted aortic arch is also frequently associated with BAV. Coexistence of BAV and CoA is predominantly associated with right-left coronary cusp fusion (85%). During infancy and adolescence, valvular dysfunction in patients with BAV and CoA tends to occur at the same rate as in those with isolated BAV. However, during adulthood, valvular complications from BAV, such as aortic stenosis (AS) or aortic regurgitation (AR), are more prevalent among people with both CoA and BAV.
Pediatric patients with CoA and BAV may have smaller aortic dimensions than those with isolated BAV. However, individuals with CoA and BAV have larger aortas than those with a CoA alone, and even those with CoA alone have been found to have aortic dimensions similar to those of the normal tricuspid aortic valve (TAV) population. BAV is an important predictor of subsequent aortic wall complications (i.e., aortic aneurysm, aortic dissection, and aortic rupture) in patients with CoA. ,
Among patients with BAV and CoA, the aortic abnormalities are not confined to the ascending aorta, suggesting a more diffuse process. Hypertension and intrinsic aortic wall disease may be important in the pathogenesis of aortic dissection related to CoA. Among CoA patients, BAV is a major risk factor for death or future cardiovascular events. Individuals with BAV require long-term follow-up of the coarctation repair, the BAV, and the ascending aorta.
TS, which is caused by complete or partial absence of one X chromosome, is associated with cardiovascular defects in up to 75% of cases. BAV is the most common defect, occurring in up to 30% of cases; other associated defects include ascending aortic dilation, CoA, pseudocoarctation, elongated aortic arch, and hypoplastic left heart syndrome. Identification of a left-sided outflow tract lesion in a female infant should prompt evaluation for TS.
The finding of BAV in TS is associated with larger aortic dimensions at the annulus, sinuses, sinotubular junction, and ascending aorta. Because TS patients have short stature, ascending aortic dimensions may be significantly dilated relative to body surface area (BSA), and absolute measurement of the aortic root or ascending aortic diameter may not predict the risk of aortic dissection. Prophylactic aortic surgery is recommended for these patients at smaller aortic dimensions, and the aortic size should be indexed to BSA. ,
Congenital coronary anomalies have been described in patients with BAV, including a short left main coronary artery and increased frequency of left-dominant circulation (24%–57%). Isolated congenital coronary artery anomalies, particularly with a separate ostia for the left anterior descending and circumflex arteries, are associated with BAV and are related to fusion of the left and right coronary cusps. High take-off coronary arteries are more common in BAV with associated congenital heart disease. Coronary artery ectasia is also reported in BAV patients.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here