Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Pancreatic resection and reconstruction remain a therapeutic challenge to surgeons treating neoplastic disease of the pancreas, distal common bile duct, ampulla of Vater, and proximal duodenum. Despite appreciable improvements in perioperative, postoperative, and intraoperative techniques, morbidity for these procedures remains high. The majority of this morbidity is attributed to pancreatic reconstruction and subsequent anastomotic and pancreatic leaks.
The first reported resection of a pancreatic tumor was a distal pancreatectomy for a spindle cell sarcoma performed by Friedrich Trendelenburg in 1882. Despite a postoperative course complicated by wound infection and malnutrition, the patient was discharged home upon his own insistence only to die a few weeks later secondary to acute respiratory failure. Halsted described the first successful resection of a periampullary tumor in 1898 when he performed a local resection along with an anastomosis of the pancreatic duct and bile duct to the duodenum for a patient with obstructive jaundice. Indeed, most early reports of periampullary tumor resection were performed via a transduodenal approach. That same year, Alessandro Codivilla performed the first pancreaticoduodenectomy in a 46-year-old male with a pancreatic head mass; however, the patient died on postoperative day 18. Walther Kausch performed the first successful two-stage pancreaticoduodenectomy in 1909, followed by Hirschel in 1914, who reported the first successful one-stage pancreaticoduodenectomy. By 1910, 20 pancreatic resections were reported in the literature with a 45% in-hospital mortality rate.
General acceptance for the pancreaticoduodenectomy was not seen until Whipple reported his successful two-stage pancreaticoduodenectomies in 1935. Five years later he performed the first anatomic one-stage pancreaticoduodenectomy for a tumor of the head of the pancreas, where he performed an antrectomy and complete removal of the duodenum. The first one-stage pancreaticoduodenectomy in the United States was performed by Trimble in 1941.
Despite numerous technical advances over the next 40 years, morbidity and mortality remained high, 40% to 60% and 20% to 40%, respectively. However, the combination of centralization of pancreatic surgery, as well as the technical advancements led by John Cameron after 1980, resulted in improved outcomes, particularly with mortality, and subsequent widespread acceptance of the procedure.
One of the most important elements in the preoperative evaluation of a patient with pancreatic cancer is determining resectability. This involves evaluation of both clinical and anatomic parameters, ranging from patient performance status to local tumor relationships relative to major vascular structures. Clinical evaluation involves a thorough history and physical examination. History should focus specifically on a personal and family history of pancreatitis or malignancies, such as pancreatic, colon, or breast cancer. Physical examination should look for evidence of advanced disease, such as palpable metastatic disease in the supraclavicular fossa (Virchow node), palpable periumbilical node (Sister Mary Joseph node), palpable perirectal mass (Blumer shelf), ascites, weight loss, and cachexia. Hepatomegaly and a palpable gallbladder (Courvoisier sign) may signify chronic biliary obstruction. Routine laboratory screening (including complete blood count, electrolyte panel, liver function tests, coagulation panel, and albumin) alerts the clinician to anemia, hyperbilirubinemia, and evidence of new-onset diabetes, pancreatitis, and malnutrition. Pertinent serum tumor markers include carbohydrate antigen 19-9 (CA 19-9) and carcinoembryonic antigen (CEA). Of note, CA 19-9 levels are often elevated in setting of biliary inflammation or obstruction, even among patients with benign disease. Accordingly, its low sensitivity (80%) and specificity (60% to 70%) for pancreatic ductal adenocarcinoma make it unsuitable for use as a screening tool but can be useful when trends are assessed, particularly in determining response to therapy.
Appropriate preoperative imaging is essential to assess resectability of pancreatic tumors. Specifically, identification of distant metastases and locally advanced pancreatic tumors involving major vascular structures is paramount. Multiple preoperative imaging modalities are used; however, in most centers, contrast-enhanced pancreas protocol spiral computed tomography (CT) is the preferred modality ( Fig. 100.1 ). Liver lesions greater than 1 cm in size are easily identified. The role of positron emission tomography–CT (PET-CT) in pancreatic cancer remains poorly defined but may be useful in ruling out distant metastatic disease in patients with suspicious lesions. Tumor encasement of the celiac axis or superior mesenteric artery or occlusion/unreconstructible portal vein (PV) and superior mesenteric vein (SMV) tumor involvement is considered locally advanced disease, and nonsurgical options, such as chemotherapy and chemoradiation, are preferred in these instances. Three-dimensional (3D) reconstruction of the mesenteric vessels both in the arterial and venous phases can preoperatively aid in demonstrating critical anatomic relationships. In addition, 3D CT helps identify surgically relevant aberrant anatomy, such as a replaced right hepatic artery.
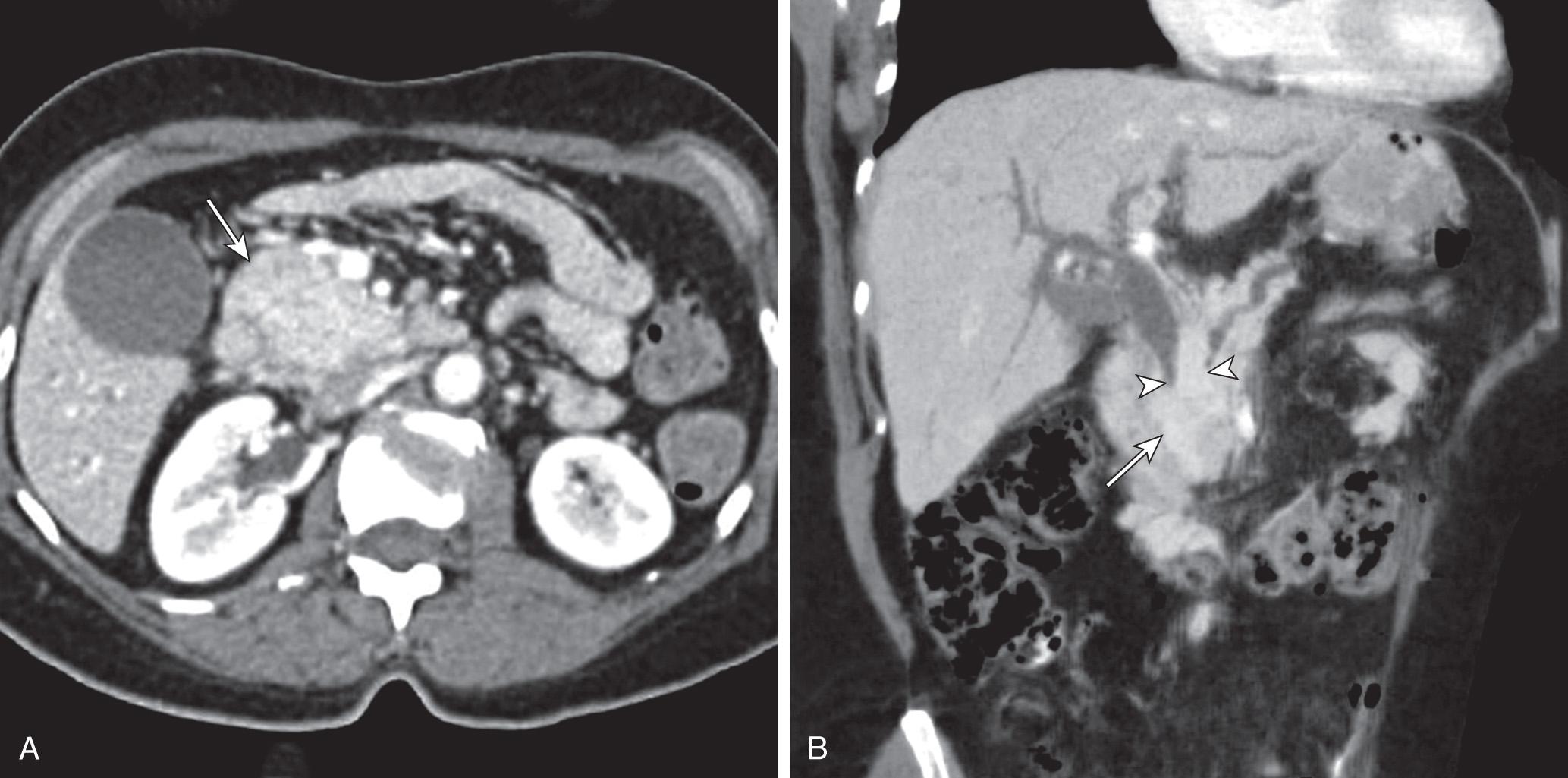
The use of endoscopic ultrasound (EUS) for preoperative evaluation is center dependent. The ability of EUS to predict vascular involvement remains controversial. Moreover, the procedure is operator dependent and not particularly useful in the determination of distant metastatic disease. Its main utility is in obtaining a tissue sample via fine-needle aspiration, particularly in patients with locally advanced/unresectable disease prior to beginning chemotherapy. Biopsy is associated with a low incidence of complications, and it is generally reserved for unresectable lesions or patients who may benefit from neoadjuvant therapy prior to resection. Preoperative biliary decompression is associated with increased wound infections and prolonged hospital stay but may be beneficial for malnourished patients or those presenting with jaundice/cholangitis in whom surgery may be delayed.
Endotracheal intubation is necessary. Central venous catheters and invasive arterial monitoring lines are inserted as needed, with peripheral venous access typically being sufficient. A nasogastric tube is placed and preoperative antibiotic prophylaxis, as well as deep venous thrombosis prophylaxis, is administered. The patient is positioned in the supine position.
The first step of the procedure is to determine resectability and rule out distant disease. Routine use of staging laparoscopy remains controversial. Some surgeons use it routinely, whereas others prefer to use staging laparoscopy only in select cases when there is a higher probability of metastatic disease, such as adenocarcinoma of body and tail of the pancreas or in the setting of high CA 19-9 levels. With the increased sensitivity of modern imaging techniques, many surgeons argue that staging laparoscopy has a low yield, making it less necessary.
Many surgeons prefer an upper abdominal midline incision; however, a bilateral subcostal incision may also be used. The abdomen is explored for evidence of metastatic disease. This exploration includes the liver, peritoneum, omentum, transverse mesocolon, and all serosal surfaces. Involvement of periportal and celiac axis lymph nodes or involvement of the base of the transverse mesocolon is not necessarily a contraindication to resection. The base of the transverse mesocolon may be taken with the specimen, including a segment of the middle colic artery when necessary, given that the marginal artery generally maintains blood supply to the transverse colon.
A wide Kocher maneuver is performed to evaluate local major vascular relationships. Of note, preoperative cross-sectional imaging offers the most accurate assessment of the relationship of the tumor to the vessels, especially the superior mesenteric artery. With mobilization of the duodenum, one can assess whether there is a component of uncinate process involvement with the superior mesenteric artery. In addition, this maneuver allows the surgeon to evaluate for tumor extension into the inferior vena cava and aorta. Next, the common hepatic artery and proper hepatic artery are assessed to confirm resectability. The PV SMV confluence is also assessed for tumor involvement. The hepatoduodenal ligament is incised. The common hepatic duct is identified and divided close to the level of the cystic duct. A more proximal margin on the hepatic duct is sometimes needed for superior pancreatic tumors arising from the head of pancreas. Assessment for a replaced right hepatic artery, typically located lateral and posterior to the common bile duct, is imperative, and the artery should be protected when identified. A replaced hepatic artery should also be readily visible with state-of-the-art preoperative imaging. When an accessory right hepatic artery is present (i.e., a right hepatic artery traveling lateral and posterior to the common bile duct in the presence of a native right hepatic artery), the accessory replaced right hepatic artery can often be ligated with impunity. Rarely, a completely replaced common hepatic artery may be seen originating from the superior mesenteric artery (type IX anatomy); recognition, preservation, or reconstruction of this vessel is critical.
After defining the arterial anatomy, the gastroduodenal artery (GDA) is identified and test clamped to ensure adequate flow in the proper hepatic artery. In instances in which the celiac axis is partially or completely occluded, the hepatic artery flow may be dependent on the GDA. If a weak pulse is noted in the hepatic artery at test clamping, division of the median arcuate ligament is a feasible option to diminish stenosis or occlusion of the celiac axis. After confirming an adequate pulse in the proper hepatic artery, the GDA is ligated and divided. The anterior PV can now be dissected free from the posterior surface of the neck of the pancreas. For a classic Whipple, a distal gastrectomy is performed, which includes ligation of the right gastric artery. For pylorus-preserving pancreaticoduodenectomies (PPPDs) the first portion of the duodenum is divided 3 cm distal to the pylorus. Numerous studies comparing classic Whipple and PPPD have reported no major differences in morbidity or survival. However, other studies have noted decreased delayed gastric emptying (DGE) with classic Whipple compared with less operative time, operative blood loss, and red blood cell transfusion for patients undergoing PPPD.
Attention is then turned to the third and fourth portions of the duodenum. This portion of the duodenum is adequately mobilized, allowing for the anterior surface of the SMV to be dissected free. The right gastroepiploic vein and artery are identified and ligated. Cephalad dissection off the anterior surface of the SMV avoids venous branches and ultimately clears a plane anterior to the PV and SMV, posterior to the neck of the pancreas ( Fig. 100.2 ). The transverse pancreatic arterial arcade is controlled with four 3-0 silk stay sutures placed superiorly and inferiorly. The pancreatic neck is slowly transected using electrocautery. The duct is identified and transected with a scalpel when possible. The jejunum is divided distally, approximately 15 to 20 cm distal to the ligament of Treitz. The mesentery is carefully divided, and the proximal jejunum and fourth portion of the duodenum are passed posterior to the mesenteric vessels to the right of the operative field.
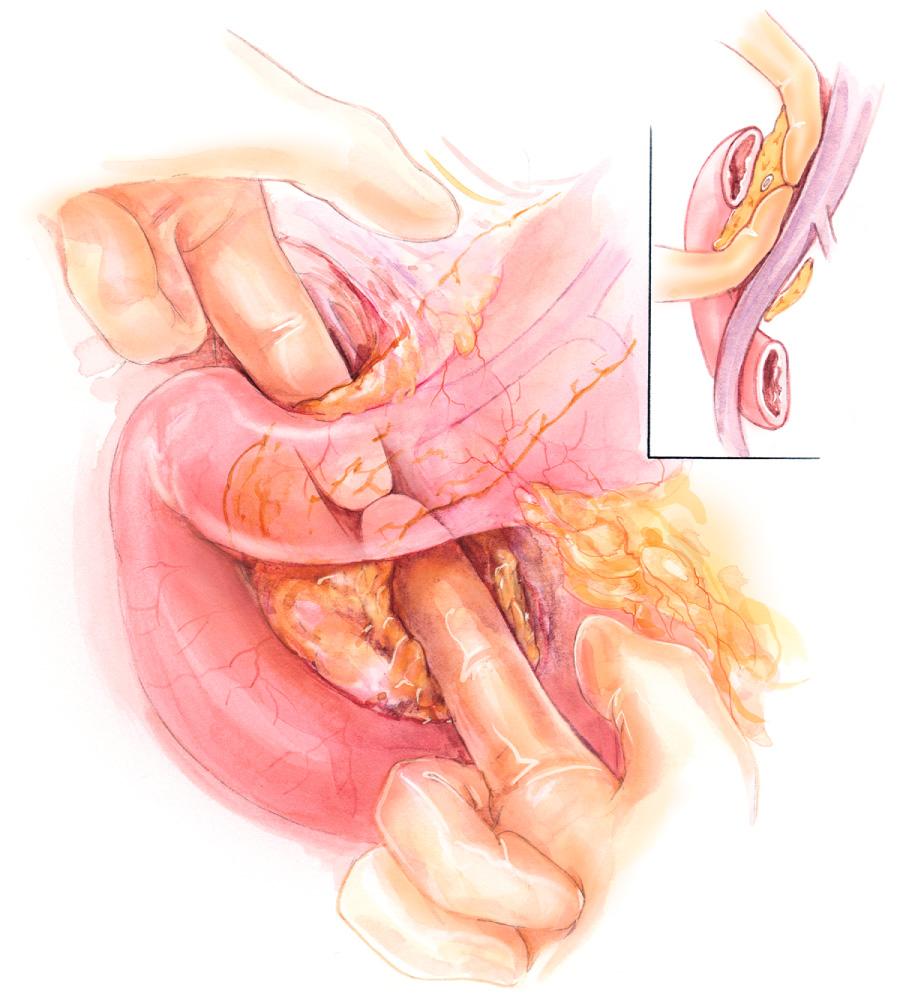
The specimen now remains attached by the remaining attachments of the head and uncinate process, which are subsequently separated from the SMV and SMA through sharp dissection, and serial division of retroperitoneal tissue along the superior mesenteric artery with an electronic dissector (e.g., harmonic scalpel, LigaSure device). The dissection should remain flush with the mesenteric vessels to ensure pancreatic tissue and lymph nodes are excised in their entirety. The specimen consists of the distal stomach or first portion of the duodenum, neck, head, and uncinate process of the pancreas, gallbladder, distal biliary tree, and approximately 10 cm of the jejunum. The pancreatic neck, bile duct, and retroperitoneal margins are marked for intraoperative frozen section. Alternatively, the common hepatic duct and pancreatic neck margins can be sent for examination earlier in the procedure while the main specimen is removed.
En bloc resection to include the PV or SMV in pancreatic cancer was first proposed by Fortner in 1973. With the increasing use of neoadjuvant therapies, PV and SMV resection in borderline resectable pancreatic tumors is increasingly being performed. State-of-the-art cross-sectional imaging is very accurate in identifying vascular involvement; as such, the surgeon should rarely be caught off guard and should be prepared for the possibility of vascular reconstruction. Adequate proximal and distal control is obtained by mobilization of the PV and SMV. Primary anastomosis is generally possible for short segment resections (<3 to 4 cm) after adequate mobilization of the portal and SMV (and sometimes the liver itself). For long segment reconstruction, interposition grafts are used, including cadaveric vein or autologous vein, such as the internal jugular or left renal vein.
Arterial encasement of the superior mesenteric artery, hepatic arteries, or celiac axis is generally accepted as a contraindication to resection. However, visceral arterial resections with or without vein resections have been reported. Unlike the outcomes among patients undergoing PV, which have been demonstrated to be comparable to Whipple without vascular resection, visceral artery resection may be associated with worse short- and long-term outcomes. Therefore vascular resection in pancreatic cancer should be used very selectively, being largely restricted to patients who have received neoadjuvant chemotherapy/chemoradiation. In addition, such complex operations should be performed only at experienced centers. In general, outcomes after arterial resection remain disappointing and should therefore not be considered a standard approach, although arterial resection may be beneficial in select cases.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here