Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Q11.1 What is the spectrum of dermatologic conditions that human herpes virus (HHV) infections can cause? (Pg. 115, Table 11.1 )
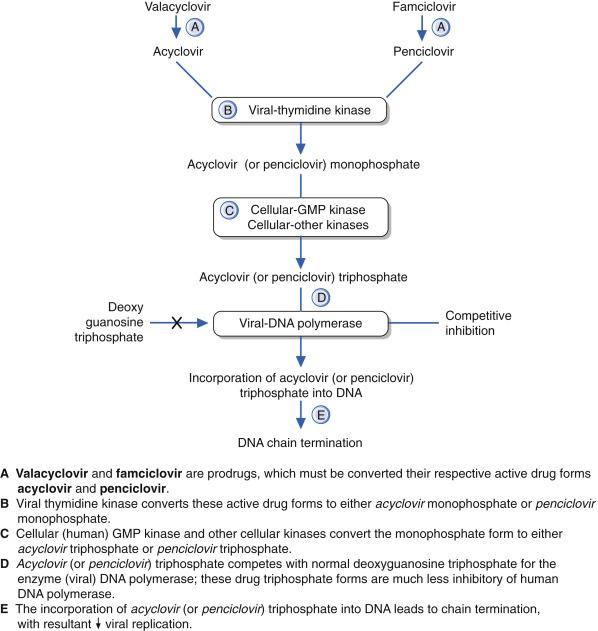
Q11.2 What are the two primary steps (one step with two parts) by which acyclovir reaches the form that inhibits viral replication (similar steps for valacyclovir and famciclovir) (Pg. 115, Fig. 11.2 )
Q11.3 How common is acyclovir resistance, and what are the clinical implications of this resistance? (Pgs. 118, 122)
Q11.4 What is the rationale for the use of acyclovir or valacyclovir in patients with recurrent erythema multiforme, and which regimens are most effective? (Pgs. 118, 120)
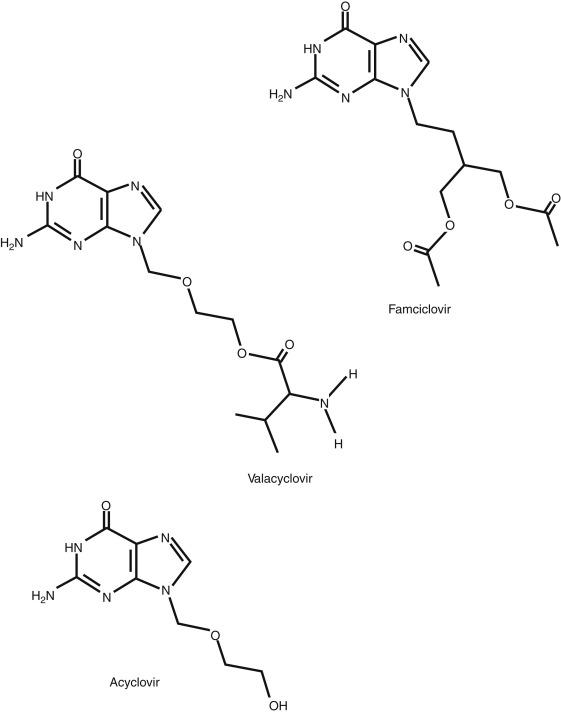
Q11.5 Of the three drugs for HHV infections discussed in this chapter, which two are defined as ‘prodrugs’ for another active drug? (Pgs. 119, 121)
Q11.6 What advantages does valacyclovir have over acyclovir in treating herpes zoster? ( Pgs. 119, 120)
Q11.7 How does the bioavailability differ between acyclovir, valacyclovir, and famciclovir? How might this relate to treating varicella-zoster virus (VZV) infections and herpes simplex virus (HSV) infections?) (Pg. 121)
Q11.8 What are the most important clinical circumstances that may justify long-term antiviral suppressive therapy for recurrent HSV infections? (Pg. 122)
Q11.9 What are the key points concerning the development of the VZV vaccine and the priorities for clinical use of this vaccine? (Pg. 122)
Q11.10 What are the two available vaccines to prevent shingles and how are they different? (Pg. 123)
Q11.11 Which human immunodeficiency virus (HIV) medications function as pharmacokinetic enhancers to boost the concentrations of other antiretroviral medications? (Pg. 124)
Q11.12 Concerning vaccine development for HIV prevention, (1) what are several of the methods of development used, and (2) what is the only vaccine that has demonstrated any efficacy in clinical trials? (Pg. 124x2)
Advisory Committee on Immunization Practices
Acyclovir
Acquired immunodeficiency syndrome
Azidothymidine ( same as zidovudine)
Centers for Disease Control and Prevention
Cytochrome P-450
Gastrointestinal
Famciclovir
US Food and Drug Administration
Highly active antiretroviral therapy
Human herpes virus
Human immunodeficiency virus
Herpes simplex virus
Herpes zoster
Integrase strand transfer inhibitor
Measles/mumps/rubella
Nucleoside reverse transcriptase inhibitor
Nonnucleoside reverse transcriptase inhibitor
Postherpetic neuralgia
Protease inhibitor
Pregnancy prescribing category
Prescription or medication
Stevens–Johnson syndrome
Toxic epidermal necrolysis
Thymidine kinase
Valacyclovir
Varicella-zoster virus
Thanks to Patricia C. Lee, George D. Magel, Kassie A. Haitz, Whitney J. Lapolla, Catherine M. DiGiorgio and Natalia Mendoza for their input into this chapter and/or prior editions of this chapter.
Viral diseases in dermatology can be very frustrating to treat. Prevention strategies, such as vaccines, proper sanitation, vector control, blood testing, condom use/abstinence, and education remain essential to managing viral spread. Once viruses, such as human herpes viruses (HHV) and human immunodeficiency virus (HIV) are acquired, antiviral agents are essentially the sole method of treatment. A large number of antiviral medications have been approved by the US Food and Drug Administration (FDA) during the past 3 decades. New antiviral agents and vaccines are continuously being researched for more effective control of these viral diseases.
To date, there are over 30 FDA-approved systemic antiviral drugs for treatment of infections caused by HHV and HIV, as well as for hepatitis viruses, influenza, and so on. This chapter primarily addresses the current use of systemic antiviral agents (against HHV) in dermatology, as well as new agents currently under investigation. Also provided is a brief overview of antiviral therapy for HIV infections.
HHV are double-stranded, linear deoxyribonucleic acid (DNA) viruses that cause a variety of illnesses. Q11.1 The HHV family includes herpes simplex virus type 1 and type 2 (HSV-1 and HSV-2), which most frequently produce herpes labialis (cold sores) and genital herpes, respectively; however, both types of lesion can be caused by either virus. HSV-1 and -2 have also been shown to cause gingivostomatitis, ocular disease, herpes gladiatorum, eczema herpeticum, herpetic whitlow, neonatal herpes, lumbosacral herpes, herpetic keratoconjunctivitis, herpes encephalitis, cervicitis, and erythema multiforme. HHV type 3 is also known as varicella-zoster virus (VZV). It is more commonly called chickenpox in its primary form and herpes zoster (HZ) or shingles in its recurrent form. The remaining members of the HHV family and resulting conditions are listed in Table 11.1 . The three primary drugs that have efficacy against HSV-1, HSV-2, and VZV are acyclovir, valacyclovir, and famciclovir ( Table 11.2 ).
| Hhv Number | Older Nomenclature | Resultant Diseases |
|---|---|---|
| HHV 1 | Herpes simplex virus type 1 (HSV-1) | Herpes labialis, etc. |
| HHV 2 | Herpes simplex virus type 2 (HSV-2) | Genital herpes, etc. |
| HHV 3 | Varicella-zoster virus (VZV) | Chicken pox, HZ |
| HHV 4 | Epstein-Barr virus (EBV) | Mononucleosis, Burkitt lymphoma |
| HHV 5 | Cytomegalovirus (CMV) | CMV retinitis |
| HHV 6 | No specific name | Roseola infantum, etc. |
| HHV 7 | No specific name | Roseola infantum, Pityriasis rosea a , etc. |
| HHV 8 | Kaposi sarcoma herpes virus | Kaposi sarcoma (classic and epidemic) |
a The causal role of HHV 7 in pityriasis rosea has not been fully established.
| Generic Name | Trade Name | Generic Available | Tablet Size | Oral Suspensions | Topical Preparation |
|---|---|---|---|---|---|
| Acyclovir | Zovirax | Yes | 200, 400, 800 mg | Yes | Yes |
| Valacyclovir | Valtrex | Yes | 500, 1000 mg | No | No |
| Famciclovir | Famvir | Yes | 125, 250, 500 mg | No | No |
Acyclovir (9-2-hydroxyethoxymethyl guanine or acycloguanosine) (ACV), a guanosine analog, is the most well-known and widely used antiviral drug in the world ( Fig. 11.1 ). ACV is available in intravenous, oral, and topical formulations, but its oral bioavailability is low (∼20%). Q11.2 Activation of ACV requires phosphorylation by herpes-specific thymidine kinase (TK) before bi- and triphosphorylation by host cellular enzymes. The active triphosphorylated ACV inhibits viral DNA polymerase by serving as an obligate chain terminator (i.e., complete and irreversible inhibition of further viral DNA synthesis) ( Fig. 11.2 ). Furman and co-authors suggested that activated triphosphate of ACV is substantially more effective in inactivating the viral polymerase than the cellular DNA polymerase. Table 11.3 contains the key pharmacologic concepts for ACV.
| Drug Name | Peak Levels | Bioavailability (%) | Protein Binding (%) | Half-Life | Metabolism | Excretion |
|---|---|---|---|---|---|---|
| Acyclovir | 1.5–2.0 hours | 15–30 | 9–33 | 1.3–1.5 hours | No hepatic microsomal metabolism | Roughly equal urine and fecal |
| Valacyclovir | Uncertain | 54.50 | 13.5–17.9 | 2.5–3.3 hours | No hepatic microsomal metabolism; conversion to acyclovir | Roughly equal urine and fecal |
| Famciclovir | 0.9 hours | 77 | <20 | 2.3–3.0 hours | No hepatic microsomal metabolism; conversion to penciclovir | 73% urine, 27% fecal; mostly penciclovir, also 6-deoxypenciclovir |
Indications for ACV are found in Box 11.1 . Box 11.2 details various risks of ACV.
Primary episode
Recurrent episodes
Suppressive therapy
Chicken pox
HZ
Immunocompromised patients (such as HIV infections)
Recurrent erythema multiforme (presumed/proven caused by HSV)
Other subsets of herpes simplex infections (see text)
Primary episode
Recurrent episodes
Suppressive therapy
HZ
Immunocompromised patients (such as HIV infections)
Recurrent erythema multiforme (presumed/proven caused by HSV)
Other subsets of herpes simplex infections (see text)
Primary episode
Recurrent episodes
Suppressive therapy
Immunocompromised patients (such as HIV infections)
HZ
Immunocompromised patients (such as HIV infections)
Other subsets of herpes simplex infections (see text)
Primary varicella
| Contraindications | |
|
|
| Boxed Warnings | |
|
|
| Warnings & Precautions a | |
| General Concerns | Major Organ Toxicities
|
| Pregnancy Prescribing Status | |
|
|
a Under “Warnings & Precautions” these adverse effects can be considered relatively high risk or important clinical scenarios to avoid.
b See Chapter 65 , Dermatologic Drugs During Pregnancy and Lactation, for detailed explanations of terms for “Newer rating” based on 2015 US Food and Drug Administration rulings.
ACV can be administered topically, orally, and intravenously. The oral form is the most widely used for HSV infections. In the therapy of genital HSV, oral ACV is indicated for treatment of the initial episode and recurrent disease, as well as for suppressive therapy. For first-episode genital HSV, the original recommended dose was 200 mg 5 times daily for 10 days. Although ACV has greater efficacy when used for first-episode genital HSV, it also shows significant benefit in recurrent disease if therapy is initiated during the prodromal phase. For recurrent genital HSV, originally ACV was dosed as 200 mg 5 times daily for 5 days. Far more commonly, clinicians use ACV dosed as 400 mg 3 times daily (TID) for 10 days (first-episode HSV) or 5 days (HSV recurrences). The decreased frequency leads to greater convenience and increased patient compliance. Suppressive therapy is recommended for frequent recurrences. Continuous suppressive therapy with ACV 400 mg twice daily reduces recurrence of genital HSV by 80% to 90%, and reduces asymptomatic viral shedding of HSV-2 by 95%. Suppressive ACV in pregnant females beginning before 36 weeks’ gestation reduces the risk of perinatal transmission, viral shedding, and the number of caesarean deliveries.
ACV is also beneficial in recurrent orofacial HSV (herpes labialis). ACV 200 mg 5 times daily for 5 days expedites crusting, but does not appear to reduce healing time significantly. Maximal therapeutic benefits are seen when therapy is initiated during the prodromal stages before vesicle formation. Suppressive therapy should be considered for patients with severe or frequent recurrences defined as six or more episodes per year or those with a history of ocular HSV disease. This guideline no doubt has flexibility based on individual patient circumstances. ACV suppressive therapy following initial ocular herpes has been shown to reduce the recurrence by 45% in the first year. Topical ACV is also promoted for the treatment of orofacial HSV. Although it has been suggested that topical ACV has limited efficacy because of inadequate penetration of the drug through the stratum corneum, recent data demonstrate transdermal penetration of ACV can be enhanced by using different vehicles, percutaneous absorption enhancers (such as collagen microparticles, oleic acid, nonionic surfactants to name a few), and iontophoresis.
Intravenous ACV is reserved for severe illness and in the immunocompromised because of its markedly greater bioavailability (∼100%). Indications include disseminated HSV disease, complicated primary infection, neonatal infection, eczema herpeticum, herpes encephalitis, and other HSV subsets that fails oral therapy. Several randomized, placebo-controlled trials have found ACV suppressive therapy among people dually infected with HSV-2 and HIV-1 to reduce risk of HIV-1 progression and slowed CD4 cell count falling to less than 350 cells per μL. The effect of ACV on the clinical course of HIV is mediated through decreases in HIV viral load, the primary determinant of HIV disease progression, and via suppression of HSV-2-mediated inflammation.
Q11.3 Although ACV resistance is low in immunocompetent patients, HSV-2-resistant isolates are more common in immunocompromised patients. In ACV-resistant strains, antiviral susceptibilities should be determined and foscarnet or cidofovir used as primary therapy. See Tables 11.4 and 11.5 regarding the use of ACV for HHV infections in immunocompetent and immunocompromised patients.
| Clinical Scenario | Acyclovir | Valacyclovir | Famciclovir |
|---|---|---|---|
| Herpes simplex—primary | 200 mg 5×/day for 10 days a | 1000 mg BID for 10 days | 250 mg TID for 10 days |
| Herpes simplex—recurrences | 200 mg 5×/day for 5 days a | 500 mg BID for 3 days b | 125 mg BID for 5 days |
| Herpes simplex—suppression | 400 mg BID | 500 mg qD c | 250 mg BID |
| HZ—acute treatment | 800 mg 5×/day for 7–10 days | 1000 mg TID for 7 days | 500 mg TID for 7 days |
| Primary varicella—children | 20 mg/kg QID up to 800 mg/dose for 5–7 days | 20 mg/kg TID up to 1000 mg/dose for 5 days | Not well evaluated |
a Less frequent dosing regimens include ACV 400 mg TID 10 days (primary); ACV 400 mg TID 5 days (recurrences).
b Shorter dosing regimen VACV 1000 mg 2 BID x1 day.
c Recommended VACV 1000 mg daily suppressive dose (single dose) if 10 or more HSV recurrences each year.
| Clinical Scenario | Acyclovir | Valacyclovir | Famciclovir |
|---|---|---|---|
| Herpes simplex—primary | 200–400 mg 5×/day for 10 days or 5 mg/kg IV q8h for 7–10 days | 500–1000 mg BID for 7–10 days | 250 mg TID for 7–10 days |
| Herpes simplex—recurrent | At least 400 mg TID for 5 days | 500 mg BID for 3 days or 1000 mg daily for 5 days | 250–500 mg BID for 5 days |
| Herpes simplex—suppression | At least 400 mg BID | 500 mg or 1000 mg daily | 250–500 mg BID |
| HZ—adults | 800 mg 5×/day for 7–10 days | 1000 mg TID for 7–10 days | 500 mg TID for 7–10 days |
| Primary varicella—children | 10–20 mg/kg IV q8h for 5 days or 1000 mg TID for 5–7 days | 1000 mg TID for 5–7 days | No studies reported |
When used to treat first-episode VZV, the recommended adult dose of oral ACV is 800 mg 5 times daily for 7 days and 20 mg/kg 4 times daily (up to a maximum of 800 mg per dose) for children. For efficacious treatment, ACV must be initiated within the first 24 to 72 hours after appearance of the characteristic skin eruption. A meta-analysis revealed that ACV can shorten the duration of fever and reduce the number of lesions; however, its effects on pruritus and onset of new lesions were not consistent. ACV treatment of chickenpox, even in otherwise healthy children, is considered cost effective because it allows the child to return to school at least 2 days earlier, thus enabling the parent to return to work sooner. Intravenous ACV is recommended for pregnant women with any evidence of pneumonia because of the high fetal morbidity and mortality. No ACV directly-related fetal risk has been documented.
ACV is used for recurrences of VZV known as HZ (or shingles ). Acute HZ requires 800 mg of ACV to be taken 5 times daily for 7 to 10 days. Placebo-controlled trials indicate accelerated healing time when therapy is initiated within 1 to 2 days of the initial symptoms and signs of infection. Although there is much controversy regarding the effects of ACV on postherpetic neuralgia (PHN), recent studies have demonstrated reduction in zoster-related pain following antiviral treatments. Intravenous ACV (10 mg/kg TID for 7–10 days) is used in the immunocompromised and those with severe trigeminal nerve distribution.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here