Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
In search of the ideal meniscal substitute, natural and synthetic materials for meniscal replacement have been reported with variable outcomes. Although meniscus allograft transplantation is the only clinically available option for total meniscus replacement, disadvantages to its use include limited donor supply, the need for size matching, the risk of disease transmission and the possibility of an immune response to affect healing and incorporation. Therefore there is an acute need to develop alternative strategies for meniscus replacement and regeneration. This chapter summarises progress in the field and the translational potential of cell-free scaffolds and biomaterials developed for meniscus replacement and regeneration.
Tissue engineering is a rapidly evolving field in biomedical research aimed at developing biological substitutes that restore, maintain or improve tissue function. In general, tissue engineering is based on a combination of cells, scaffolds and biological or biomechanical signals to stimulate neo-tissue formation. The classic tissue engineering paradigm involves the stimulation of cells, in vitro or in vivo, to migrate and proliferate through a three-dimensional (3D) scaffold, leading to production of functional replacement tissue. The scaffolds should ideally promote uniform cell adhesion, migration, differentiation and proliferation. The microstructure and composition of the scaffold should also promote proper phenotypic expression by infiltrating cells and functional extracellular matrix production.
Although a number of approaches have been reported for tissue engineering of the meniscus, an optimal meniscus replacement has yet to be discovered. Importantly, many of the published tissue engineering studies do not report the mechanical properties of the meniscal constructs or repair tissue, which is a critical outcome measure given the tissue’s intrinsic functions. There is also a lack of standardised and objective outcome measures across studies to compare the effectiveness of different meniscal scaffolds and no consensus on the definitions or benchmarks for success.
An ideal meniscus replacement should have biological and biomechanical properties that are similar to the native meniscus ( Fig. 20.1 ). Importantly, a functional meniscus replacement should ultimately restore the meniscus structure, function and anisotropic properties. The scaffold should also maintain a synergistic relationship with the articular cartilage, provide joint protection and reduce the onset and progression of osteoarthritis. Further, the ideal meniscus replacement should resorb or degrade slowly enough to allow for ingrowth and regeneration of the tissue’s vascular, structural, cellular and extracellular matrix components. The meniscal scaffold should also be nontoxic to the joint and not evoke a foreign body reaction or immune response on degradation. Lastly the optimal scaffold should be readily available for point-of-care clinical use, minimally invasive, implantable with technical ease and reimbursable by federal and private insurance providers.
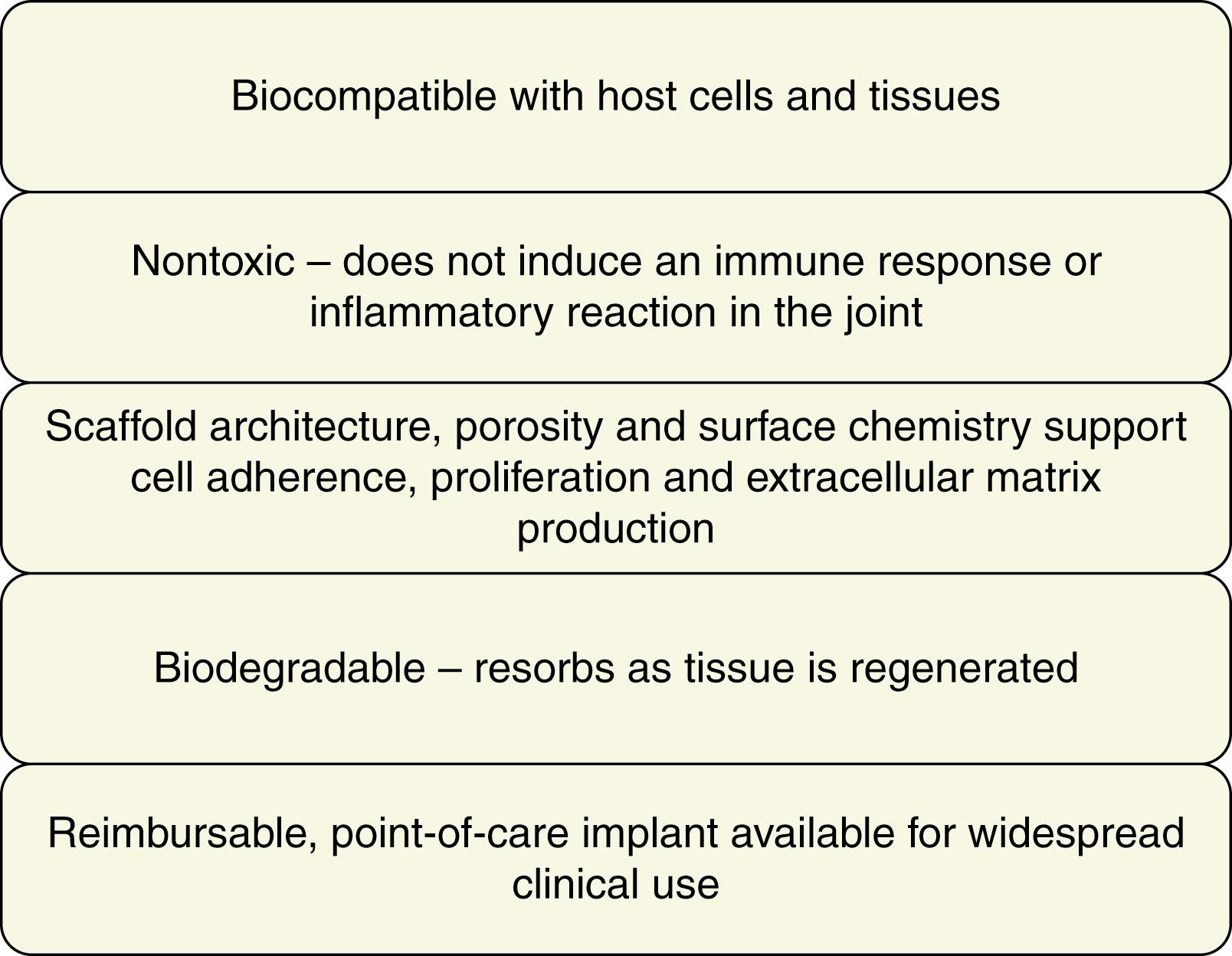
Broad categories of scaffolds investigated for meniscal replacement include synthetic polymers, hydrogels, extracellular matrix components and tissue-derived materials. Natural polymeric scaffolds offer a distinct advantage for scaffold fabrication in that they are easily modified and the biomechanical and biological properties of the construct can be manipulated during the fabrication process, therefore allowing the engineered construct to resemble tissues for a specific application. They are also less likely than synthetic polymers to incite a strong inflammatory response in vivo . The hydrophobic composition of synthetic polymers may also hinder cell growth in a 3D architecture. Thus a natural polymeric scaffold can be designed in such a way that it is biocompatible and biodegradable, allows for diffusion of nutrients and metabolites, provides mechanical support, has appropriate porosity and provides an aqueous environment for cellular encapsulation, proliferation and tissue production.
Hydrogel scaffolds are fabricated from natural or synthetic polymers, consisting of hydrophilic building blocks that are insoluble because of the presence of cross-links. Examples include alginate, collagen, agarose, chitosan, gelatine and hyaluronic acid. The latter polymers are formed from soluble monomers, multifunctional polymers (macromers) and nano- or microparticles. In addition, a variety of crosslinking processes may be used to affect the chemical and physical properties for a specific application. Potential chemical crosslinking agents include glutaraldehyde, genipin, adipic dihydrazide, and bis(sulfosuccinimidyl) suberate. Thermal, ionic and free radical crosslinking methods may also be employed, depending on the type of polymer to be investigated. Controlling hydrogel density and crosslinking chemistry of the scaffolds can result in subsequent control over mechanical properties and tissue in-growth rates via scaffold resorption. Because hydrogels have functional and structural similarities to native extracellular matrix, they also have the potential to provide cells with a biologically relevant microenvironment that fosters cell proliferation, migration and extracellular matrix production. , Other advantages include the chemical diversity of hydrogels under an array of different environmental factors, including temperature, pH, electric field, ultrasound and salinity.
Collagen is a naturally occurring matrix polymer and the most abundant extracellular matrix protein. It contains three polypeptide α chains forming a right-handed collagen triple helix. The meniscus is composed predominantly of type I collagen, but there is variation in the type and structure of collagens between the vascular and avascular zones, as well as between the superficial and deep zones. Type I collagen significantly contributes to normal matrix composition and the biomechanical properties of the meniscus. Collagen fibres are primarily responsible for the tensile strength of the meniscus, and the longitudinal fibres are critical for normal meniscal function and chondroprotection in that they transfer vertical compressive loads into circumferential hoop stress. Consequently a meniscal replacement intended for clinical translation should be able to recapitulate the force transmission properties of the meniscus in vivo .
High-density collagen gels have been investigated for fabrication of cell-seeded meniscal constructs with improved mechanical and biochemical properties compared with alginate gels with or without insulin-like growth factor supplementation. , The Collagen Meniscal Implant (CMI) is a porous meniscus implant derived from purified bovine collagen and will be discussed in another section (see Meniscal Scaffolds in Clinical Use).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here