Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
In 1862 Louis Pasteur's ingenious experiments into the nature of putrefaction were officially endorsed by the Paris Academy of Science. The endorsement signaled an end to the long-held belief that the exposure of organic material to air brought about the spontaneous generation of microorganisms, and the concepts of sepsis and asepsis became firmly established. A mere 3 years later, Joseph Lister demonstrated the incredible implications of antisepsis in his practice of orthopedic surgery. For the first time in recorded history, major surgical procedures could be performed with a reasonable expectation of primary wound healing and recovery. Essential enhancements for preventing and controlling wound sepsis were provided by the antibiotic revolution of the 1940s, ushering in the highly technical, highly invasive, and highly successful era of modern surgery. As noted by McDermott and Rogers, the great achievements of the antibiotic era may be related, in the long run, to its essential role in supporting the advancements of modern surgery. Indeed, surgery as we know it today would be impossible in an environment in which infection was likely or, once established, untreatable.
Despite the fundamental role of antisepsis and antibiotics in the development of modern surgery, implementation of these discoveries in the practice of surgery has not occurred without opposition. As late as 1880, for example, William Halstead was ordered from the operating theater when he challenged a senior surgeon's disregard for antiseptic techniques. The early use of antibiotics for prophylaxis in surgical procedures was also questioned as respected academicians freely voiced their disapproval of antimicrobial prophylaxis in clean surgical procedures. For a number of years the value of prophylactic antibiotics in preventing infections of the surgical wound remained in doubt. A consensus in favor of their use did not emerge until two concepts of perioperative prophylaxis and infection were established. First, investigators in Cincinnati and Boston demonstrated that, despite the use of standard aseptic techniques, Staphylococcus aureus could be regularly isolated from the operative field. It became apparent that aseptic technique could decrease but not eliminate bacterial contamination of the surgical field; therefore it appeared plausible that perioperative antibiotics could supplement aseptic techniques in containing the inevitable contamination of the operative wound.
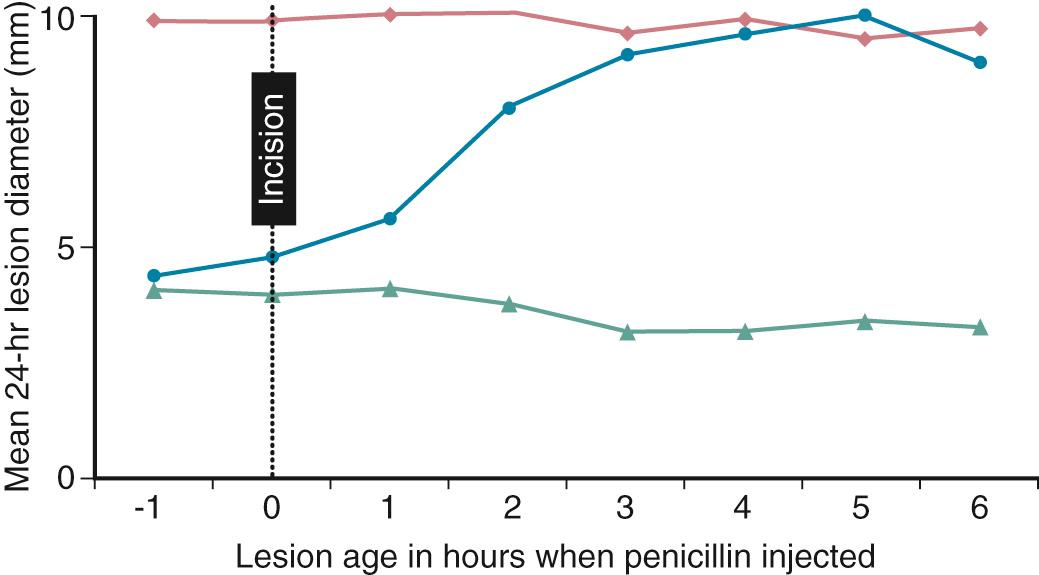
The second major finding involved the timing of the administration of the prophylactic antibiotic. As early as 1946, Howes had noted a correlation between the amelioration of infection and the interval between the contamination of the wounds and the administration of antibiotics. Several years later, Miles and colleagues and Burke, working with a guinea pig model of wound infection, demonstrated the remarkable brevity of the “window” of prophylactic efficacy. They noted that antibiotics given shortly before or at the time of bacterial inoculation of the subcutaneous tissue of the guinea pig produced a notable diminution in the size of the subsequent wound induration compared with lesions in animals not receiving antimicrobial prophylaxis ( Fig. 313.1 ). By delaying the administration of antibiotics by only 3 or 4 hours, resulting lesions were identical in size to those of animals receiving no antimicrobial prophylaxis whatsoever. Thus “failures” of antimicrobial prophylaxis that had been noted in earlier clinical studies were related to the fact that administration of preoperative antibiotics had been inappropriately timed. Although these observations have been challenging to reproduce in vitro, the experience of current practice has evolved to mandate that whenever possible surgical antimicrobial prophylaxis should be administered so as to ensure adequate tissue levels of antimicrobials from the time of the initial surgical incision until closure. The efficacy of prophylactic antibiotics has now been verified for most major surgical procedures, with a wide variety of antimicrobials when care has been given to provide adequate serum and tissue levels of antibiotics during the surgical procedure. Perioperative antibiotics and aseptic techniques have become routine aspects of care in most major surgical procedures.
Despite efforts to prevent surgical site infections (SSIs), these outcomes are not uncommon. The 2014 Healthcare-Associated Infection Prevalence study from the Centers for Disease Control and Prevention (CDC) estimated an annual national burden of more than 157,000 SSIs among hospitalized adult patients, a figure that does not include those patients with an SSI that did not require hospitalization. Often neglected in these estimates, SSIs associated with ambulatory surgical procedures result in a substantial rate of postsurgical acute care visits for clinically significant SSIs in 4.8 per 1000 procedures across the eight states studied. Another analysis estimated that SSIs led to an added $20,785 and 11.2 days length of stay per event and a total of $3.2 billion in attributable costs in the United States in 2008. A patient who develops an SSI while hospitalized has a greater than 60% greater risk of being admitted to the intensive care unit, is 15 times more likely to be readmitted to the hospital within 30 days after discharge, and incurs an attributable extra hospital stay of 6.5 days, leading to a direct cost of an additional $3000 per infection. SSIs due to methicillin-resistant S. aureus (MRSA), in particular, have also been shown to have a higher mortality than those due to methicillin-sensitive strains of the organism.
Technologic advances (e.g., the introduction of minimally invasive procedures and robotic surgery) and the emergence of antibiotic-resistant organisms have led to additional challenges in the prevention of SSIs. In addition, the prevention of SSI has moved to the forefront of surgical quality improvement programs, highlighting important issues regarding antimicrobial prophylaxis (e.g., drug dosing in obese patients, the specific timing of antibiotic administration, and the role of anti-MRSA prophylaxis). Reflecting the increasing desire for transparency of infection prevention program effectiveness, public reporting of facility-specific colon and abdominal hysterectomy SSI performance is now tied directly to reimbursement. Despite these advances over 150 years since the discoveries of Pasteur and Lister, much remains to be learned about the pathophysiology, prevention, and surveillance of SSIs.
Whether a wound infection occurs after surgery depends on a complex interaction between the following: (1) patient-related factors (e.g., host immunity, nutritional status, comorbid conditions); (2) procedure-related factors (e.g., implantation of foreign bodies, degree of trauma to the host tissues); (3) microbial factors (e.g., tissue adherence and invasion); and (4) use of preventive measures (e.g., perioperative antimicrobial prophylaxis).
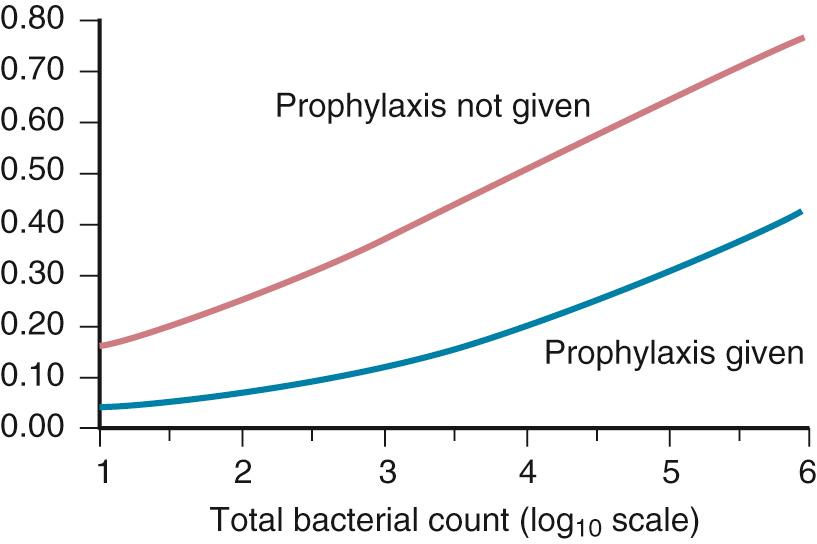
Bacterial contamination of the surgical wound is inevitable. State-of-the-art aseptic technique has been associated with a dramatic drop in, but not the elimination of, this phenomenon. Even under laminar flow operating room (OR) environments, bacteria can be isolated from wound surfaces at the close of the surgical procedure. The importance of the microbial load in determining whether or not a wound becomes infected has been long appreciated and is relevant even in the era of the routine administration of antimicrobial prophylaxis for most major surgical procedures ( Fig. 313.2 ). Historically, operations have been stratified on the basis of the expected quantity of bacteria introduced into the operative site during surgery ( Table 313.1 ). Although the magnitude of bacterial inoculation into the wound still has some predictive value regarding the risk of developing a wound infection, patient- and procedure-related risk factors also contribute greatly to this risk ( Table 313.2 ).
| Class I: Clean Wound |
| An uninfected operative wound in which no inflammation is encountered, and the respiratory, alimentary, genital, or uninfected urinary tract is not entered. In addition, clean wounds are primarily closed and, if necessary, drained with closed drainage. Operative incisional wounds that follow nonpenetrating (blunt) trauma should be included in this category if they meet the criteria. |
| Class II: Clean-Contaminated Wound |
| An operative wound in which the respiratory, alimentary, genital, or urinary tracts are entered under controlled conditions and without unusual contamination. Specifically, operations involving the biliary tract, appendix, vagina, and oropharynx are included in this category, provided no evidence of infection or major break in technique is encountered. |
| Class III: Contaminated Wound |
| Open, fresh, accidental wounds. In addition, operations with major breaks in sterile technique (e.g., open cardiac massage) or gross spillage from the gastrointestinal tract, and incisions in which acute, nonpurulent inflammation is encountered are included in this category. |
| Class IV: Dirty-Infected Wound |
| Old traumatic wounds with retained devitalized tissue and those that involve existing clinical infection or perforated viscera. This definition suggests that the organisms causing postoperative infection were present in the operative field before the operation. |
| Patient Factors |
|
| Procedural Factors |
|
| Proceduralist Factors |
|
a Likely a surrogate marker for severity of underlying illness and comorbidities.
Numerous organisms have been described as wound pathogens, and the origin of the inoculum is not established with certainty for most infections. The patient's endogenous skin flora, with gram-positive organisms in general, and staphylococcal species in particular, are the predominant cause of incisional infections of clean surgical procedures. Over the past decade, the microbiology of SSIs has evolved due to the emergence of various multidrug-resistant pathogens, with a concerning proportion of reported SSIs due to multidrug-resistant gram-negative pathogens ( Table 313.3 ). S. aureus colonization of the patient's nares is a major risk factor for developing a S. aureus SSI, a particular concern in selected populations, such as diabetic individuals and recipients of hemodialysis, who have S. aureus colonization rates in excess of 50%. S. aureus and Streptococcus epidermidis and streptococcal wound infections can also occur in clusters, sometimes with a particular surgeon or nurse implicated in their spread. Unusual and hard-to-culture species, including nontuberculous mycobacteria, Nocardia spp., Legionella spp., Mycoplasma hominis , and Cutibacterium (Propionibacterium) acnes (an organism associated with shoulder arthroplasty SSI), have caused surgical site infections as well.
| PATHOGEN | NO. OF ISOLATES TESTED | PERCENT OF ISOLATES RESISTANT |
|---|---|---|
| Staphylococcus aureus | ||
| Resistant to oxacillin/methicillin (MRSA) | 8738 | 42.6% |
| Enterococcus faecium | ||
| Resistant to vancomycin (VRE) | 1342 | 58.4% |
| Escherichia coli | ||
| Extended-spectrum cephalosporin resistant | 6816 | 15.3% |
| Fluoroquinolone resistant | 6816 | 30.9% |
| Carbapenem resistant | 6816 | 0.7% |
| Multidrug resistant | 6816 | 6.5% |
| Pseudomonas aeruginosa | ||
| Extended-spectrum cephalosporin resistant | 2617 | 9.9% |
| Fluoroquinolone resistant | 2617 | 11.5% |
| Carbapenem resistant | 2617 | 7.7% |
| Multidrug resistant | 2617 | 4.3% |
| Enterobacter spp. | ||
| Extended-spectrum cephalosporin resistant | 2056 | 27.5% |
| Carbapenem resistant | 2056 | 3.4% |
| Multidrug resistant | 2056 | 2.4% |
| Klebsiella spp. | ||
| Extended-spectrum cephalosporin resistant | 2319 | 11.3% |
| Carbapenem resistant | 2319 | 3.3% |
| Multidrug resistant | 2319 | 4.6% |
| Acinetobacter spp. | ||
| Carbapenem resistant | 174 | 33.3% |
| Multidrug resistant | 174 | 32.9% |
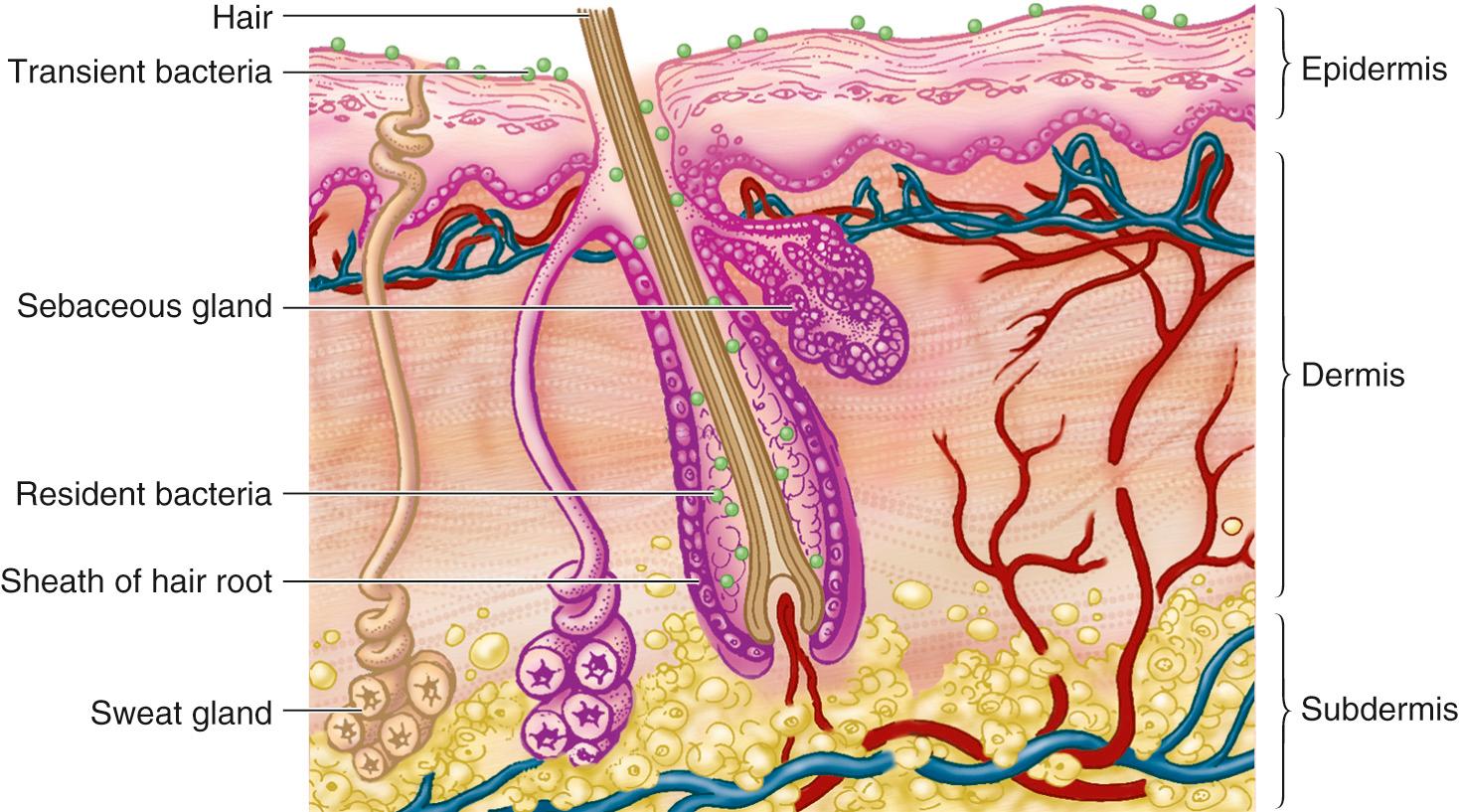
Modern methods of antisepsis can reduce but not eliminate the skin-associated bacteria of surgical patients. This limitation derives, in part, from the localization of up to 20% of skin-associated bacteria in skin appendages, such as hair follicles and sebaceous glands ( Fig. 313.3 ). Because these sites are beneath the skin's surface, bacteria residing there are not eliminated by topical antisepsis. Transection of these skin structures by surgical incision may carry the patient's resident bacteria deep into the wound and set the stage for subsequent infection.
For contaminated procedures, wound pathogens frequently are among the bacterial species that comprise the normal flora of the viscus entered during the surgical procedure. Enteric gram-negative pathogens and anaerobic bacteria (e.g., Bacteroides fragilis ) are common pathogens of wounds after colonic procedures, and polymicrobial infections are common in this setting. Infection by a particular species, however, does not correlate directly with its quantitative presence among the normal flora, but by the particular virulence attributes of bacteria.
Although numerous sources of bacterial contamination of surgical wounds have been described, it is virtually impossible to identify with certainty the source(s) and route(s) of contamination. The direct inoculation of a patient's endogenous flora at the time of surgery is believed to be the most common mechanism; however, others undoubtedly occur. Transmission from contaminated surgical instruments or surgical material; hematogenous seeding from preexisting infection of a nonwound site; and contamination from either the skin, mucous membranes, or clothing of operating room staff have been implicated as potential sources of microbial contamination. Outbreaks of group A streptococcal wound infection have been traced to the anal or vaginal carriage of this organism by operating room personnel, and epidemiologic investigation of these outbreaks indicated that airborne contamination of the operative field had occurred. Infections, including Candida albicans osteomyelitis and diskitis after lumbar laminectomy, have been traced to the use of artificial fingernails worn by surgical staff, leading many hospitals to ban the use of artificial or long natural fingernails in health care workers with direct patient care responsibilities. A worldwide outbreak of SSI in cardiac surgery patients associated with aerosol dissemination of and surgical field contamination with Mycobacterium chimaera by heater-cooler units used intraoperatively illustrates the potential for airborne spread and development of SSI due to pathogens with low pathogenic potential, incubation periods of months to years, and the unintended consequences of new technologies used in the operating suite.
The relative importance of hematogenous seeding (i.e., inside-to-out ) of the surgical wound is somewhat unclear. Evidence of this route of seeding of the surgical wound site has been mainly noted with procedures involving implantation of prosthetic devices. In addition, although it is generally accepted that prosthetic valves and hips are at risk for an indefinite period of time for hematogenous seeding and infection postoperatively, it is difficult to ascertain with certainty whether a late postoperative infection results from intraoperative bacterial seeding of the prosthetic device, followed by prolonged dormancy, or from a true postoperative hematogenous event. In a randomized, prospective, controlled study of antimicrobial prophylaxis in total hip replacement, Carlsson and coworkers demonstrated that deep wound infections that developed over 2.5 years after surgery were more likely to have occurred among placebo versus cloxacillin recipients (13.7% vs. 3.3%, respectively; P < .05). These data suggest that bacteria inoculated into wounds at the time of surgery may lie dormant for years, rendering differentiation of the precise source of late wound infection virtually impossible. If late hematogenous seeding of a surgical wound with or without prosthetic material can occur, it is reasonable to assume that wounds are even more vulnerable to seeding and secondary infection during the immediate postoperative period. During this time, surgical incisions are hyperemic from the trauma of the surgery and endothelialization of intravascular prosthetic materials has not yet had time to occur. Moreover, the regular use of indwelling intravascular catheters probably increases the risk of bacteremia; however, information with which to judge the relative contribution of intraoperative versus postoperative hematogenous seeding of the surgical incision is unavailable.
The requirement for large inocula in the early models of S. aureus soft tissue infection gave the misleading impression that cooperative interaction between bacteria may be required to establish a wound infection. Later models involving foreign bodies demonstrated median infective dose (ID 50 ) values of fewer than 100 colony-forming units (CFU) with polytetrafluoroethylene (PTFE) tissue cages, 10 CFU with PTFE vascular grafts, and as low as 1 CFU with dextran microbeads. These data demonstrate the pathogenic potential of a single bacterium to produce wound infection, provided that it is inoculated into a suitable niche.
The role of coliforms and anaerobes in abdominal sepsis has been elucidated in a model that involves inserting a gelatin capsule containing a standardized inoculum of pooled cecal contents into the peritoneal cavities of rats. Acute peritonitis and septicemia from coliforms caused rapid death in 37% of the animals, and all of the survivors developed abscesses with anaerobes as the predominant organisms. The capsular polysaccharide of Bacteroides fragilis promotes abscess formation and may reduce phagocytosis. In experimental models, immunization against capsular polysaccharide can protect against abscess formation after inoculation with B. fragilis by a T-cell–dependent mechanism, except in the presence of foreign material. Also, B. fragilis produces a variety of tissue-damaging enzymes, including hemolysin, chondroitin sulfatase, neuraminidase, and hyaluronidase.
Much of our understanding of the pathophysiology of wound infection and the nature of the surgical wound derives from investigational models. Early investigations suggesting that the efficacy of antibiotics in preventing wound infection is limited to only a few hours after the moment of bacterial inoculation suggested that the wound microenvironment is not static. It is likely that rapid changes are occurring among microbial factors, such as a shift from exponential to stationary-phase growth with an accompanying decrease in bacterial susceptibility to antibiotics and possibly the expression of different microbial virulence factors. Wound-related changes must also occur, such as gradually diminishing tissue perfusion and antibiotic delivery related to increased tissue oncotic pressure brought about by the effect of inflammatory mediators on vascular permeability.
Investigations of S. aureus infection in the skin of human volunteers by Elek and Conen conclusively established the role of foreign material in potentiating wound infection. By including suture material with the intradermal staphylococcal inoculum, the number of organisms required to establish a skin pustule could be reduced 10,000-fold relative to lesions without sutures (i.e., a fall from 5 × 10 6 organisms to 3 × 10 2 organisms in the inoculum). These investigators further suggested that “other circumstances may lead to the unhindered growth of small inocula, including heavily traumatized tissues, burns, or devitalized tissues distal to the ligated vessels. This may be the explanation of the traditional surgical view that untidy operative techniques predispose to infection.” Because in clean and clean-contaminated surgical procedures, quantitative bacterial inoculation into the wound is small, tissue devitalization at a gross or microscopic level provides a niche wherein a small bacterial inoculum may grow in relative isolation from the host's defenses, which plays a major role in the pathogenesis of infection.
Investigational models have demonstrated how technical variables of the surgical procedure influence the risk of infection. Some suture materials appear to have a stronger adjuvant effect on infection than others. Whether the use of the electrosurgical knife, which can damage host tissues via the transfer of heat, is an adjuvant for infection is controversial. Clinical studies examining the use of electrocautery have not revealed an increased risk for SSI with the use of this technique, with a recent Cochrane analysis noting a nonsignificant difference (7.7% vs. 7.4%) in wound infection rates between electrosurgery vs. scalpel.
Operative procedures produce systemic and local changes in the immune defense mechanisms of the host. Neutrophil function and serum opsonizing capacity become impaired. The microbicidal activity of neutrophils obtained postoperatively from patients undergoing abdominal hysterectomy is 25% less than that of neutrophils harvested from the same patients preoperatively, and it takes 9 days to return to normal. The depletion of opsonizing factors within the abscess milieu also may contribute to decreased neutrophilic bactericidal function.
Major surgical procedures compromise the host defenses in other ways. Surface levels of human leukocyte antigen–DR antigens on the circulating monocytes of patients are reduced after major surgery. However, it has been shown that defects in T-cell proliferation and cytokine secretion after major surgery involve an inability of T cells to respond to T-cell receptor- and CD28 coreceptor-mediated signals rather than problems with antigen presentation by monocytes-macrophages. Many factors can reduce core body temperature during a surgical procedure, including ambient OR temperatures, lack of body covering, and intravenous (IV) fluids administration. Anesthetic agents also impair thermoregulatory control and may impact vasoconstriction and shivering thresholds. In the setting of perioperative hypothermia, neutrophils have reduced chemotaxis, impaired ingestion of staphylococci, and diminished superoxide production. Hypothermia can also trigger vasoconstriction and lead to low tissue oxygen tension, which is itself a risk factor for SSI. Because of these risks, interventions aimed at reducing perioperative normothermia and hypoxia have been examined to prevent SSIs (see “ Prevention of Surgical Site Infections ”).
Perioperative blood transfusion has been associated with an increased rate of postoperative infections, including wound infection, with donated white blood cell (WBC)-induced immunosuppression implicated as the culprit. Decreases in cell-mediated immunity and increases in cytokine levels (interleukin [IL]-2 receptor, IL-6) have been demonstrated in mice and humans after transfusion. However, clinical trials comparing the receipt of standard whole or buffy-coat–depleted blood products (which, despite the moniker, still retain some donor WBCs) with WBC-depleted products, or the administration of allogenic versus autologous blood, have failed to provide a consensus answer to transfusion's role in the development of postoperative infections. A restrictive transfusion approach (transfusion once hemoglobin falls below 7–8 g/dL) was associated with a reduced risk for the development of health care–associated infections when compared with more liberal strategies, but only six studies included in this meta-analysis examined surgical wound infections as a study outcome. Studies describing this potential transfusion effect have been criticized for using nonstandardized outcome definitions, failing to account for known risk factors for postoperative infection, and using only univariate analyses. Blood transfusion may simply serve as a marker for unidentified patient comorbidities, and more rigorously designed studies are needed before general conclusions regarding the role of blood transfusion in wound infection can be determined.
In cardiac surgery the patient may be exposed to hypothermia, cardiopulmonary bypass (CPB), and relative arterial hypotension throughout much of the procedure. Exposure of blood to CPB depletes serum complement, causes systemic release of proinflammatory cytokines, and adversely affects neutrophilic function. Furthermore, protein denaturation and chylomicron aggregation may contribute to small vessel occlusion and tissue hypoxia, as well as overwhelm the capacity of the reticuloendothelial system to clear infectious agents from the blood. This raises the possibility that postbypass patients may be predisposed to develop infections via hematogenous bacterial seeding as a result of reduced reticuloendothelial clearance.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here