Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The parathyroid glands develop from the endoderm of pharyngeal pouches III and IV. These are first identifiable at 5 to 6 weeks of fetal development as thickenings of the anterodorsal branchial pouch epithelium (see Chapter 2 , Applied Embryology of the Thyroid and Parathyroid Glands).
Each pharyngeal pouch III gives rise to the inferior parathyroid gland (parathyroid III) and the corresponding half of the thymus. Both parathyroid III and the thymus (also named “parathymus” ) have complex patterns of migration, and with the descent of the thymus into the thoracic cavity, the associated parathyroid III is also carried until separation from the thymus, being eventually situated near the inferior pole of the thyroid, lower in the neck than the parathyroid IV.
Failure of separation of parathyroid III from the thymus and incomplete migration of this gland may explain instances of aberrant parathyroid III localization found anywhere from the angle of the jaw to the pericardium. As a result of separation failure, inferior parathyroid glands may be found within the thymic tongue or in the anterior mediastinum; however, presence of parathyroid III proximal (i.e., cranial) to parathyroid IV is likely secondary to incomplete migration.
The superior parathyroids (parathyroid IV, also named “thyroid parathyroids”) have a shorter migration distance and are therefore more constant in their final position. These are derived from the fourth branchial pouch together with the ultimobranchial body.
There are usually four parathyroid glands. However 5% to 13% of the normal population have five or more glands, and 2% to 3% have less than four. Of note, several studies have found low glandular weights in individuals with less than four parathyroid glands and concluded that this may be related to the fact that not all glands had been identified.
The glands are distributed symmetrically in the neck in 80% of individuals. The reported combined weight of normal parathyroid glands is between 106 to 166 mg in males and 130 to 168 mg in females, with each gland weighing about 30 to 60 mg. The color of these usually oval-shaped glands varies from yellow to reddish brown, and they measure about 3 to 8 mm in length and 2 to 4 mm in width.
The upper parathyroid glands are present near the crossing of the recurrent laryngeal nerve and the inferior thyroid artery (see Chapter 2 , Applied Embryology of the Thyroid and Parathyroid Glands; and Chapter 55 , Principles in Surgical Management of Primary Hyperparathyroidism). Occasionally the upper glands are present within the carotid sheath or in the retroesophageal or retropharyngeal space. In some instances, the glands may be present in the capsule of the thyroid or, more rarely, within the substance of the thyroid gland. It has been concluded that parathyroid IIIs and IVs and supernumerary glands can all be the source of intrathyroidal parathyroid glands, with a reported incidence of 0.7% to 3.6% in the literature. These glands can be missed by preoperative imaging and be a source of failure during parathyroid surgery for hyperparathyroidism (HPT).
Approximately 60% of the lower parathyroids are located inferior, posterior, or lateral to the lower pole of the thyroid, whereas 17% are present superiorly or on the anterior aspect of the thyroid.
The arterial supply of the upper and lower glands is derived from the superior and inferior thyroid arteries, respectively. The upper gland veins drain into the superior or lateral thyroid veins, whereas the lower glands are drained by the lateral or inferior thyroid veins. Lymphatic drainage derives from a subcapsular plexus into the superior deep cervical, pretracheal, paratracheal, retropharyngeal, and inferior deep cervical lymph nodes.
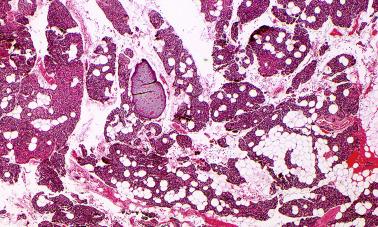
The parathyroid glands have parenchymal and fat-rich stromal components and are enveloped by a thin capsule which extends into the parenchyma, dividing it into multiple lobules. The parenchymal cells are arranged in nests or cords and are separated by richly vascular stromal elements ( Figure 65.1 ). There are four main types of parenchymal cells: chief cells, oxyphil cells, transitional cells, and clear cells. The chief cells (also known as principal cells) are 8 to 10 μm, polyhedral with centrally placed round nuclei, and have dense chromatin and inconspicuous nucleoli. The cytoplasm of the chief cells is faintly eosinophilic and may appear clear due to intracytoplasmic accumulation of glycogen or lipid, particularly in inactive cells. More lipid is found in the cytoplasm of chief cells in adults than children.
Oxyphil cells (also known as oncocytes) are larger than chief cells, measuring 12 to 20 μm, and have abundant granular eosinophilic cytoplasm with abundant mitochondria and nuclei that are larger and more vesicular than those of chief cells. These cells are not found in childhood, but their numbers increase with age. Oxyphil cells may coalesce into nodules or masses in later adulthood and morphologically they may be difficult to distinguish from the Hürthle cells of the thyroid with which they share the finding of prominent cytoplasmic mitochondria ( Figure 65.2 ). Transitional cells, also known as transitional oncocytes, can be considered intermediate between chief cells and oxyphils in both size and morphology. Clear (water clear) cells are 10 to 15 μm in diameter and demonstrate clear cytoplasm often with relatively pyknotic or eccentric nuclei.
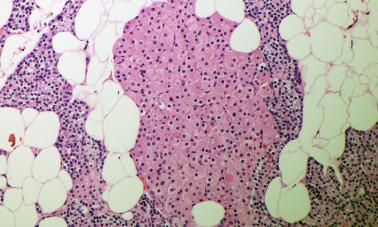
The distribution of fat and parenchymal cells in the parathyroid gland is not uniform, so biopsy specimens from normal glands may predominantly consist of fat, parenchymal cells, or an equal mixture of both. Stromal fat cells tend to be concentrated toward the polar regions of the glands.
The amount of stromal fat is also variable and related to age and nutritional status. Although parathyroid glands in children contain only very small amounts of stromal fat; by early adulthood, stromal fat cells make up approximately 20% of the volume of the glands.
In some circumstances, parathyroid tissue can histologically resemble thyroid tissue, particularly if it is arranged in a follicular-like architecture surrounding secreted parathyroid hormone (PTH), which can resemble thyroid colloid. The observation that parathyroid parenchymal cells have well-defined cytoplasmic membranes compared with the indistinct membranes of thyroid epithelium as well as the absence of oxalate crystals in the parathyroid (which are common in the thyroid) may be useful in this distinction.
HPT is an endocrine disease that is due to excessive secretion of parathyroid hormone (PTH). It is either an intrinsic abnormal change that alters excretion of PTH (primary hyperparathyroidism [PHPT] or tertiary HPT) or an extrinsic abnormal change affecting calcium homeostasis and secondarily stimulating the production of PTH (secondary hyperparathyroidism; S-HPT).
High levels of PTH lead to increased renal tubular resorption of calcium, increased urinary excretion of phosphate, synthesis of 1, 24 (OH) 2 D 3 , and increased bone resorption. Hypercalcemia, in turn, can affect nearly every organ system.
PHPT, the most common cause of hypercalcemia, can be divided into three groups. The majority of patients have elevated serum calcium levels and are categorized in one of the first two groups: symptomatic and asymptomatic.
Asymptomatic PHPT is the most common presentation in countries such as the United States, Canada, and Germany, where biochemical screening is routinely performed. Serum calcium level is usually not more than 1.0 mg/dl above the upper limit of the normal range (8.4-10.2 mg/dl).
Before the introduction of multiphasic biochemical screening programs in the 1970s, most patients with PHPT presented with renal or bone disease (osteitis fibrosa cystica). Osteitis fibrosa cystica is the classical skeletal feature of symptomatic PHPT, which may present with characteristic radiologic findings, such as salt-and-pepper degranulation of the skull, with bone cysts or brown tumors.
Some other effects of hypercalcemia on major target organs include neurocognitive symptoms, such as anxiety and poor concentration, proximal muscle weakness, polyuria, renal stones, and renal insufficiency. The gastrointestinal tract symptoms may range from abdominal pain and constipation to peptic ulcer and pancreatitis.
The third group of patients with PHPT are normocalcemic. These patients persistently have elevated PTH levels, but serum calcium levels are within normal range. The diagnosis of normocalcemic PHPT can be rendered only after exclusion of secondary causes for HPT, such as vitamin D deficiency, malabsorption syndromes, and renal insufficiency (see Chapter 53 , Primary Hyperparathyroidsm: Pathophysiology, Surgical Indications, and Preoperative Workup).
PHPT is most commonly seen in the context of a single benign parathyroid adenoma (PA); that is in about 80% to 85% of patients. Multiglandular disease is seen in approximately 15% to 20% of patients and usually takes the form of four-gland parathyroid hyperplasia, but multiple PAs are well recognized. Parathyroid cancer is a rare cause of PHPT and accounts for less than 0.5% of cases in most series.
Rare cases of apparent PHPT may result from the paraneoplastic production of PTH by nonparathyroid tumors, but most cases of malignancy-associated hypercalcemia result from the paraneoplastic production of PTH-related protein.
In most patients with PHPT, the disease is sporadic, without a personal or family history of PHPT or other endocrinopathies. These patients are predominantly female and present typically in the sixth decade with uniglandular involvement. The heritable forms account for 10% to 15% of cases of PHPT and represent a clinically and genetically heterogenous group of disorders (see Chapter 60 , Surgical Management of Multiglandular Parathyroid Disease; and Chapter 62 , Parathyroid Management in the MEN Syndromes). Common hereditary forms of disease include familial cancer predisposition syndromes (multiple endocrine neoplasia type 1 [MEN 1], MEN 2A, MEN 4, and hyperparathyroidism-jaw tumor [HPT-JT] syndrome) and hereditary “hypercalcemic” syndromes (familial hypocalciuric hypercalcemia [FHH-1, FHH-2, FHH-3], and neonatal severe HPT [NSHPT]). Familial isolated hyperparathyroidism (FIHPT) is a rare and nonsyndromic cause of heritable PHPT.
Heritable forms of the disease typically have multiglandular parathyroid involvement and equivalent rates of occurrence in females and males.
A single parathyroid adenoma (PA) accounts for the overwhelming majority of cases of primary hyperparathyroidism (PHPT); 80% to 85% of cases. Adenomas can occur at any age, although most become evident clinically in the fourth decade. They occur more commonly in females, with a female-to-male ratio of 3:1 to 4:1.
Although most PAs occur sporadically, germline mutations associated with syndromic and nonsyndromic diseases are found in about 5% of cases. There is a growing body of evidence supporting the presence of various molecular genetic drivers in sporadic cases. For instance, somatic alterations in MEN1 and CCND1 / PRAD1 genes have emerged as two important drivers in the development of 25% to 40% of sporadic PAs.
It is suggested that nonprotein-coding genes, regulatory elements, or epigenetic derangements may also have roles in the majority of PAs.
A previous history of exposure to ionizing radiation is suggested as a predisposing factor to the formation of sporadic parathyroid tumors.
The multiple endocrine neoplasia (MEN1) gene on chromosome 11q13 is a tumor suppressor gene that encodes menin, a nuclear protein with a high degree of functional versatility. Germline inactivating mutations of this gene are responsible for MEN 1 syndrome. Somatic-only mutations of the MEN1 gene occur in 40% to 50% of sporadic parathyroid tumors in most series.
The tumor suppressor role is thought to be exerted by regulation of Wnt/β-catenin signaling.
CCND1 / PRAD1 (11q13) is a proto-oncogene that encodes cyclin D1 protein. Somatic activating alterations of the CCND1 / PRAD1 gene, causing excess cyclin D1 signaling that is thought to inactivate the retinoblastoma protein (Rb) and to induce cellular proliferation, have been reported in 20% to 40% of sporadic PAs. Rearrangement of the CCND1 / PRAD1 gene can occur with the PTH promoter gene in about 8% of cases, and/or other enhancers/promoters, leading to amplification or transcriptional activation of cyclin D1.
Somatic mutations in the CaSR gene, although well-documented in hereditary forms of HPT, have not been identified in sporadic disease. Nonetheless, despite identification of aberrant CaSR signaling in a considerable subset of sporadic PAs, characterized by reduced expression of vitamin D receptors (VDRs) and CaSRs, the exact underlying mechanisms have not been elucidated.
Given the fact that the pathogenesis of > 50% of sporadic PAs remains unknown, it is postulated that undiscovered somatic alterations causing deregulation of CaSR signaling may play an important role in parathyroid tumorigenesis.
Whole-exome sequencing, DNA methylation studies, and microRNA profiling studies have revealed additional somatic mutations involving ZFX , CTNNB1 , EZH2 , and POT1 genes as well as important epigenetic alterations in sporadic parathyroid tumors.
Most adenomas involve a single gland and occur with equal frequency in the upper and lower parathyroids. These lesions occur in a variety of ectopic sites, including the thymic tongue, thymus gland proper, retroesophageal space, or within the thyroid gland. Rare examples of adenoma have been recorded in the pericardium, vagus nerve, and soft tissues adjacent to the angle of the jaw. Adenomas may rarely involve two glands, although the distinction from hyperplasia in these cases is often problematic.
Most adenomas are ovoid or reniform and have a smooth surface, but occasional tumors may be multilobate and therefore at higher risk for recurrence because they may be removed incompletely during initial surgery. On cross-section, adenomas are encapsulated and soft in consistency, with a tan to orange-brown color. Foci of cystic change are common, particularly in large adenomas. Cystic parathyroid tumors may be associated with prominent pericapsular fibrosis attributable to previous episodes of rupture and repair. Adherence of fibrotic tissue from a previously ruptured cystic adenoma to the adjacent thyroid or soft tissues may lead to a clinical suspicion of carcinoma at surgical exploration.
Fibrosis is also seen in some patients with multiple endocrine neoplasia (MEN) syndromes and also after procedures on parathyroid glands, such as fine-needle aspiration (FNA; for PTH measurement and/or cytologic examination) and ethanol injection.
PAs are composed of variable proportions of chief cells, clear cells, oxyphilic cells, or transitional cells; however, chief cell adenomas are the most common type ( Figures 65.3 and 65.4 ). The tumor cells can be arranged in cords, nests, sheets, papillary, pseudopapillary, and follicles with palisaded appearances around blood vessels. The follicle-like structures often contain a densely eosinophilic colloid-like material morphologically similar to colloid found in the thyroid, and the distinction between parathyroid and thyroid tissue in such instances may be particularly difficult, if not impossible, at frozen section. However, colloid in thyroid tissue frequently contains birefringent oxalate crystals, which are absent from the colloid-like material in parathyroid tumors. The colloid-like material in parathyroid often stains with Congo red with green birefringence under polarized light. Focal calcification of the luminal parathyroid colloid may also occur, which is similar to that seen in oncocytic thyroid neoplasms.

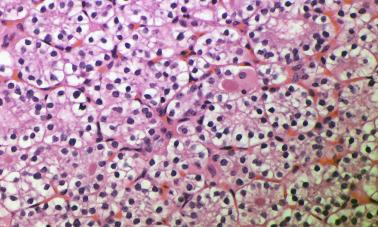
The nuclei of neoplastic cells often appear round and central and are generally larger than those in the adjacent normal gland. These contain dense chromatin and occasionally small nucleoli. The cytoplasm of neoplastic chief cells is faintly eosinophilic but may appear clear because of the accumulation of glycogen deposits. Scattered pleomorphic and hyperchromatic nuclei as well as multinucleate cells are relatively common and occasionally may form nodular aggregates.
Although scattered mitotic figures may be present in most cases, the proliferative index of most adenomas as assessed by Ki-67 (MIB-1) is generally < 5%.
Adenomas also may contain other cell types, including groups of oncocytes and transitional oncocytes and can even display a multinodular appearance, with individual nodules of chief cells or oncocytes. The nodules often have a higher proliferative index than the non-nodular portions of the adenoma.
A rim of residual normal or even atrophic gland is present in approximately 50% of adenomas (see Figure 65.3 ). The rim is often separated from the adenomas by a capsule, but in some cases, the cells of the rim appear to merge imperceptibly into the adenoma. The probability of finding a rim of normal gland is greater in smaller adenomas than in larger tumors. Typically, the cells within the rim are smaller than the adenoma cells and often contain a single large lipid droplet. If lipid is present within the adenoma cells, it tends to be more finely distributed within the tumor cells. The parathyroid chief cells within the rim generally contain more abundant immunoreactive PTH and messenger RNA than do the adenoma cells.
The absence of a rim of normal gland does not exclude the diagnosis of adenoma because the preexisting normal gland may have been overgrown or the rim may have been lost during sectioning. Although adenomas have traditionally been described as having no stromal fat, some tumors may contain stromal fat cells distributed diffusely throughout the tumor. In some instances, a small biopsy of an adenoma may be misinterpreted as a normal gland because of the abundant fat. Knowledge of the size of the gland would circumvent this potential pitfall. Occasional adenomas may contain abundant fibrous tissue, chronic inflammation, and hemosiderin deposits.
Several adenoma variants have been described.
Oxyphilic adenomas (also known as oncocytic adenomas ) make up approximately 5% of all PAs. A cutoff of 75% of neoplastic cells demonstrating oxyphil differentiation has been proposed to define this entity ( Figure 65.5 ). Experience is limited, but oxyphilic adenomas have been reported to be more frequently associated with symptomatic patients, and with higher preoperative serum calcium and PTH levels. These adenomas are also more difficult to localize on preoperative ultrasound examination.
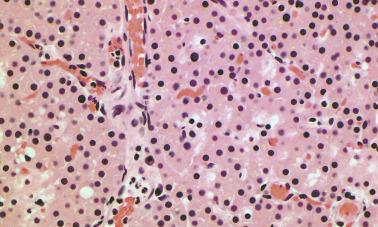
Water-clear cell adenomas are rare and characterized by the presence of cells with multiple cytoplasmic vacuoles, similar to those seen in clear cell hyperplasia. Their primary significance is that they may mimic metastatic clear cell renal carcinoma on histologic examination ( Figure 65.6 ).
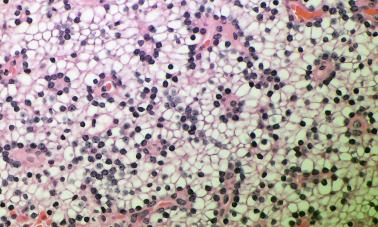
Lipoadenomas (hamartomas) are rare tumors and are characterized by the proliferation of both parenchymal and stromal elements. The stromal elements include abundant fibroadipose tissue with varying degrees of myxoid change and variable numbers of lymphocytes ( Figure 65.7 ). The parenchymal components are usually present in the form of compressed cell cords. Lipoadenomas should be distinguished from so-called large normal or fat rich parathyroid glands by the presence of increased acinar tissue as well as adipose tissue.
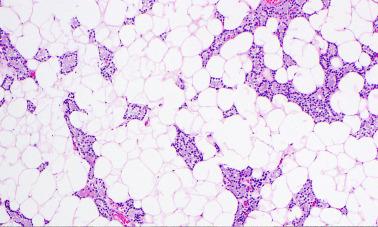
Microadenomas can be defined as tumors measuring less than 0.6 cm in diameter and/or weighing less than 200 mg. They may be overlooked on surgical exploration and frozen section. In several reported cases, microadenomas became apparent only after serial sectioning of paraffin-embedded samples. Some studies have shown that when the first resected abnormal parathyroid gland is small (< 200 mg), the likelihood of multiglandular disease and operative failure is higher.
Double adenomas are rare and have been reported in less than 2% of patients with PHPT. Patients who have been diagnosed with double adenomas are more likely to relapse after surgery either with a third or fourth adenoma or multiglandular hyperplasia. Differentiation between adenomas and asymmetric nodular hyperplasia, based on morphology alone, may be somewhat arbitrary at times. In addition, emerging molecular data suggest that the nodules in parathyroid nodular hyperplasia commonly represent clonal proliferation and as such, are more closely related to adenomas. Therefore, distinction between multiple adenomas and nodular hyperplasia has become more controversial.
Atypical adenomas represent a controversial pathologic entity. These tumors show some histopathological features of parathyroid carcinoma, such as band forming fibrosis, increased mitotic activity, cytologic atypia, and the presence of tumor cells within a thickened capsule, but they lack the definite diagnostic features of malignancy ( Figure 65.8 ).
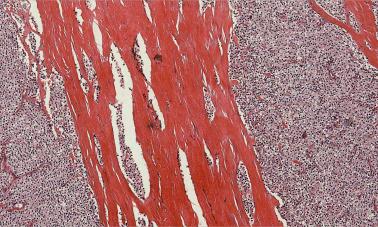
It should be noted that fibrosis can occur in many situations, including previous manipulation (post-FNA biopsy), MEN syndromes, lithium intake, long-standing chronic renal failure-related parathyroid hyperplasia, in the setting of parathyroiditis, or in large adenomas undergoing episodes of rupture/infarction and repair. Fibrotic bands at the periphery of a parathyroid that does not have a true anatomic capsule may be a pitfall and mimic a tumor cell within a thickened capsule.
Given the significant interobserver variability and lack of clinicopathological correlations in most cases, these borderline tumors cannot be classified with certainty. However there is good evidence that the majority of atypical adenomas do not recur. Advances in molecular pathology and the use of new ancillary tests may help better refine these tumors and identify those with an increased risk of recurrence. Features reported to be associated with an increased risk of recurrence include an elevated Ki-67 proliferative index, loss of parafibromin expression by IHC, galectin-3 positivity, PGP9.5 positivity, retinoblastoma protein (Rb) loss, bcl2 loss, decrease in p27, hTERT expression, decrease in mdm2, and Adenomatous polyposis coli (APC) loss of function.
Hyperparathyroidism-jaw tumor (HPT-JT) syndrome is a rare complex autosomal-dominant disorder characterized by the presence of parathyroid tumors, including PAs, parathyroid carcinomas, and fibro-osseous lesions of the maxilla or mandible. These patients harbor pathogenic germline mutations of the cell division cycle protein 73 ( CDC73 gene, previously known as HRPT2 ) that maps to 1q31.2 and encodes the protein parafibromin. CDC73 functions as a genuine tumor suppressor gene that is involved in the regulation of p53 and also as a component of the human PAF1 complex, which controls RNA polymersase II-mediated transcription.
In HPT-JT syndrome, hyperparathyroidism (HPT) commonly occurs at a young age with median first onset between the third and fourth decade. These patients also have an increased risk of developing parathyroid carcinoma, ranging from 15% to 37.5% in different case series.
Somatic-only biallelic CDC73 mutation/inactivation is also strongly associated with parathyroid carcinoma occurring outside the setting of HPT-JT syndrome.
The combined result of multiple studies in parathyroid carcinomas with unequivocal histologic features of malignancy and biological evidence of malignant behavior (i.e., recurrence or metastasis) show a CDC73 mutation frequency of up to 77%. However, the relationship between CDC73 mutation and malignancy is much less strong when malignancy is defined by histologic criteria alone, perhaps reflecting the difficulty in making a correct histologic diagnosis of malignancy prospectively. In any case, the incidence of CDC73 mutation in unselected parathyroid tumors is much less than 1%.
Loss of immunohistochemical expression of parafibromin has been used as a marker of biallelic CDC73 mutation inactivation in parathyroid tumors. However, the use of parafibromin IHC has been associated with difficulties and controversies. Although some groups have found parafibromin IHC to be useful both in differential diagnosis of atypical parathyroid tumors and as a marker of underlying CDC73 mutation, others have found it technically difficult to perform and interpret.
The parafibromin-deficient parathyroid neoplasms have been recently suggested to represent a distinct subgroup of parathyroid tumors that display a distinct morphology. These tumors are characterized by a sheet-like growth of neoplastic cells, frequently interrupted by an arborizing vasculature. These cells, at least focally, have eosinophilic cytoplasm with a quality distinct from typical granular cytoplasm of the oxyphil cells. There is nuclear enlargement and distinctive perinuclear cytoplasmic clearing, speckled chromatin, and sometimes prominent nucleoli ( Figure 65.9, A and B ). Clinically, these patients are usually younger, have larger tumors, and have more marked hypercalcemia. These neoplasms show complete loss of nuclear immunostaining with parafibromin. The reported incidence of large-scale germline deletions of CDC73 in these tumors is up to 35%, and often both sequencing and multiplex ligation-dependent probe amplification assay (MLPA) studies may be required to identify these mutations.
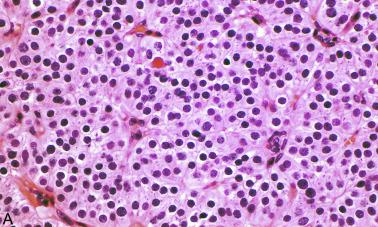
Although there is extensive literature linking CDC73 mutations with parathyroid carcinoma, the role of CDC73 mutation as a marker of malignancy in atypical parathyroid tumors remains controversial. In one study, 40% of parafibromin IHC-negative parathyroid tumors also fulfilled histologic criteria for parathyroid carcinoma, and it was recommended that a lower threshold for malignancy may be appropriate in parafibromin-deficient tumors. Although isolated case reports of morphologically benign tumors with parafibromin loss and CDC73 mutation that have metastasized have been presented, metastasis in the absence of invasive disease is rare. The primary significance of identifying parafibromin-deficient parathyroid tumors is therefore the association with malignancy, to identify potential hereditary disease, and to initiate surveillance in view of the risk of metachronous disease.
Adenomas must be distinguished from hyperplasias, and knowledge of the results of ultrasonography, 99m Tc-sestamibi scintigraphy, operative findings, and intraoperative PTH assays is essential for this distinction. Most adenomas involve a single gland, whereas enlargement of at least two glands may be apparent on 99m Tc-sestamibi scintigraphy in cases of double adenomas and hyperplasias. Multiple studies have confirmed the ability of intraoperative PTH assays to guide surgical decision-making by accurately predicting whether or not additional hypersecreting glands remain in the neck after the removal of glands identified by preoperative localization studies (see Chapter 59 , Intraoperative PTH Monitoring During Parathyroid Surgery). The presence of a single enlarged and hypersecreting gland with three normal glands is virtually diagnostic of adenoma. In current practice, the largest gland, as identified by 99m Tc-sestamibi scintigraphy or ultrasonography, is removed, and if the level of PTH as determined by intraoperative PTH drops by more than 50% within 10 minutes after excision, the surgical procedure is concluded without having to identify the remaining glands (see Chapter 59 , Intraoperative PTH Monitoring During Parathyroid Surgery).
Accurate diagnosis of parathyroid proliferations at the time of the intraoperative assessment can be difficult or even impossible. The primary role of the intraoperative pathologic examination is to confirm the presence of parathyroid tissue in the resected specimen and also to confirm the glandular abnormality (enlarged cellular gland) in patients with HPT. The evaluation of a second parathyroid gland or performing fat stains on frozen tissue was a common approach in the past, but the recent development and adoption of rapid intraoperative PTH assay appears to be a more precise tool to distinguish adenoma from hyperplasia. (see Chapter 59 Intraoperative PTH Monitoring During Parathyroid Surgery).
With the exception of rare double adenomas, PAs and carcinomas are almost always solitary lesions, whereas hyperplasia represents multiglandular proliferation that can be asymmetric and asynchronous. The presence of an atrophic rim of hypocellular nontumorous parathyroid tissue adjacent to a cellular proliferation often helps in accurate diagnosis of PA in the appropriate clinical and biochemical context, but an enlarged parathyroid gland, lacking a normal/atrophic rim, should be classified as an “enlarged cellular parathyroid gland,” as hyperplasia cannot be excluded. A diagnosis of PA can be rendered after surgery if biochemical cure is achieved and carcinoma is excluded.
It should be noted that hyperplastic glands can also present small areas of normocellular tissue, which is indistinguishable from those surrounding PAs.
In one study of more than 600 cases, a small subgroup of patients with enlarged glands (double adenoma or hyperplasia) who were not diagnosed using 99m Tc-sestamibi scintigraphy, ultrasound, or intraoperative PTH could only have been discovered with the traditional four-gland exploration approach.
This raises the question as to whether enlarged glands that are not hypersecreting at initial surgery will cause recurrent HPT in the future. Follow-up studies have demonstrated a cure rate of 98% with the focused approach; however, some studies have questioned the validity of this approach (see Chapter 59 , Intraoperative PTH Monitoring During Parathyroid Surgery).
The distinction of PA from other tumors occurring in the head and neck region is usually straightforward. On occasion, however, it can be difficult to distinguish PAs from thyroid follicular cell tumors, C-cell neoplasms, and paragangliomas. Because a small number of PAs may occur within the thyroid and may have a follicular architecture, the correct interpretation of fine-needle aspiration (FNA) biopsies, small tissue samples, or frozen section may be particularly challenging. As noted in a prior section, thyroid follicular colloid often contains birefringent oxalate crystals, whereas parathyroid tissue does not. Immunohistochemistry is of particular value in the distinction of follicular and parathyroid tumors, but in practice can only be performed on fixed tissue. Follicular cells are typically positive for thyroglobulin and thyroid transcription factor (TTF)-1, whereas parathyroid chief cells are negative for these markers but are positive for chromogranin and PTH. Glial cell missing 2 (GCM2) transcription factor is involved in embryonic parathyroid development. Although this transcription factor appears sensitive and specific for parathyroid lesions, its staining intensity and extent is variable in parathyroid neoplasia and may be negative in cystic parathyroid lesions. Some PAs may be difficult to distinguish from medullary carcinomas. The latter tumors are, however, positive for calcitonin, chromogranin, and carcinoembryonic antigen. Paragangliomas are negative for TTF-1 but are reactive for chromogranin and S-100–positive sustentacular cells that are typically present around the cell nests.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here