Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Gastroduodenal peptic ulcer disease (PUD) is a common problem with significant geographic variation in prevalence. In Western countries the incidence of PUD has steadily declined. Recent population-based studies have shown a prevalence rate of 4% with 20% of patients having asymptomatic ulcers. In developing countries, the prevalence is much higher. In a recent population-based study from China, 17.2% of the population had endoscopically documented duodenal or gastric ulcers; however, more than 70% of these patients were asymptomatic. Such variations are likely related to the prevalence of Helicobacter pylori , smoking, and use of ulcerogenic drugs, such as nonsteroidal antiinflammatory drugs (NSAIDs). In a systematic review of the literature covering developed countries, the annual incidence of PUD ranged from 0.1% to 0.19% for physician-diagnosed PUD and 0.01% to 0.17% when based upon hospitalized patients. In a 1996 Veterans Affairs study the prevalence of PUD in H. pylori– positive patients was found to be 2%.
In the United States, there has been a decrease in the prevalence and number of hospitalizations for PUD. Between 1993 and 2006 the rate of PUD-related admissions decreased by 30%, with a larger decrease in duodenal ulcer–related admissions than gastric ulcers. Such preferential decreases in duodenal versus gastric ulcer disease likely relate to testing for H. pylori and introduction of more potent and successful therapeutic regimens. The advent of histamine H 2 -receptor antagonists (H 2 blockers) in the 1970s was responsible for an initial 40% decrease in incidence of ulcer operations. The later development of proton pump inhibitors (PPIs) in the late 1980s led to further acid reduction and faster, more efficient healing of active ulcer disease. The development of PPIs has not only influenced elective medical management but also has had an effect in the emergency setting. When combined, PPIs and endoscopic treatment have further decreased the need for emergency operation.
PUD complications include bleeding, perforation, and gastric outlet obstruction. There has been a significant downward trend in the incidence of these complications. Although older studies demonstrated a stable or even an increase in the number of patients admitted with one of these complications, recent studies demonstrated that the rate of perforation and bleeding has been decreasing in the United States. Complications of PUD vary depending on the geographic location, with bleeding being the most common in the United States, whereas obstruction may be more common in other locations in the world.
Risk factors for peptic ulcer complications and their recurrence included NSAID and/or acetylsalicylic acid use, H. pylori infection, and ulcer size greater than or equal to 1 cm. PPI use has reduced the risk of peptic ulcer hemorrhage.
Current indications for surgical intervention are as follows:
Protracted bleeding despite endoscopic therapy. Bleeding is the most common complication of PUD in the United States, with an incidence of approximately 100 per 100,000 population.
Perforation is the second most common with an annual incidence of 11 operations per 100,000 population. Perforations are associated with the highest rate of mortality.
Obstruction occurs as a consequence of scarring following healing of prepyloric and/or duodenal ulcers.
Intractability despite maximum medical therapy is an uncommon indication for operation.
Inability to rule out cancer when an ulcer remains despite treatment and negative endoscopic biopsies. This is of particular importance with gastric ulcers.
The goals of surgical procedures are to
Permit ulcer healing
Prevent or treat ulcer complications
Address the underlying ulcer etiology
Minimize postoperative digestive consequences
No single procedure satisfies all of these objectives. To choose the best operation, the surgeon must consider the characteristics of the ulcer (location, chronicity, type of complication), the likely etiology (acid hypersecretion, drug induced, possible role of H. pylori ), the patient (age, nutrition, comorbid illnesses, condition on presentation), and the operation (mortality rate, side effects). In some respects, all ulcer operations represent a compromise: The morbidity of ulcer disease is replaced by the morbidity of the operation. Finally, surgeon experience must play a role in the choice of operation; nowadays, most surgical residents complete their training with little experience with the more complex procedures. Undoubtedly this influences their choices for both elective and emergent operations.
Initial operations for ulcer disease were based on local control without a good understanding of the physiology involved. As the physiology of digestion and acid production was delineated, the operations changed and were subsequently shifted toward addressing the current understanding of the cause of PUD.
Benjamin Brodie, an English physiologist and surgeon, in 1814, described the vagus nerves and their connection with the production of gastric acid. Then in 1822, William Beaumont, an army surgeon, cared for Alexis St. Martin, a man who sustained a shotgun wound to the abdomen. Beaumont treated his wound but expected him to die; however, the patient survived and was left with a gastrocutaneous fistula. In 1825 Beaumont began to study the patient's digestive process by tying food to a string and inserting it through the fistula into the stomach and observe how it had been digested. He also studied the gastric fluid from the fistula. In 1833 Beaumont published his findings as Experiments and Observations on the Gastric Juice, and the Physiology of Digestion .
The discovery of the three separate, yet related, phases of gastric secretion and its involvement in the consumption and digestion of a meal defined the surgical treatment of PUD. The cephalic, gastric, and to a lesser extent the intestinal phases are examples of physiologic discovery molding surgical practice. Ivan Pavlov, a Russian physiologist and physician, described the cephalic phase of gastric secretion. Through his physiologic studies with dogs, Pavlov showed that stimulation of the vagus nerves resulted in the secretion of gastric acid. His discovery earned him the 1904 Nobel Prize in Physiology and Medicine.
The gastric or antral phase of secretion revolves around the work of the physiologist John Edkins, who injected an extract of pyloric mucus membrane “activated by hydrochloric acid or boiling” into the jugular vein of cats. He noted a marked increase in the gastric acid and pepsin secretion. In 1905 he named the active agent “gastrin.” Further study led to the understanding that gastric distention stimulates the release of gastrin from the antrum, resulting in the release of gastric acid. This knowledge was applied directly to the treatment of PUD and became the basis for antrectomy.
The intestinal phase of gastric secretion refers to specific situations when a food bolus, which has not been exposed to gastric acid, comes in contact with the duodenal mucosa. In this situation the stomach is stimulated to secrete acid. Physiologically, food boluses are acidified and upon contact with the duodenal mucosa stimulate an inhibitory response.
In 1881 the Prussian surgeon, Ludwik Rydygier, performed the first successful resection for a gastric ulcer. In the early 1900s the standard operation performed for the treatment of gastric and duodenal ulcers was either a pyloroplasty without vagotomy or a gastroenterostomy. These operations were performed regardless of the presence or absence of obstructive symptoms, and many patients had resolution of their symptoms.
Gastroenterostomy soon surpassed pyloroplasty as the treatment of choice. Charles Mayo presented the Mayo Clinic data on gastroenterostomy for treatment of both gastric ulceration and duodenal ulceration. His data from 647 patients with gastric ulcers showed a mortality rate of 3.2% after gastroenterostomy and less than 2% in the 2734 patients with duodenal ulceration. Eventually, recurrence rates and marginal ulcer formation were recognized, and surgical management began to change. Gastric resection gained favor, and the use of gastroenterostomy alone declined. In 1941 the Mayo Clinic data on the use of subtotal gastrectomy in the treatment of benign gastric ulcer were presented. The data showed a mortality rate of only 2.2% and that other, previously used operations were being performed with much less frequency.
In 1921 Andre Latarjet described the anatomy of the vagus nerves and applied that knowledge clinically by performing an anatomically complete vagotomy for dyspepsia. Subsequently, he observed postoperative issues with inadequate gastric emptying and included a gastrojejunostomy. Despite the improved understanding in vagal anatomy and physiology, therapeutic vagotomy remained an obscure treatment option for PUD. Lester Dragstedt, a physiologist and surgeon at the University of Chicago and later at the University of Florida, was paramount in the development of vagotomy for the treatment of peptic ulcers. Through his animal research, he elucidated the role of acid hypersecretion in the development of ulcers and stated “pure gastric juice as it is secreted by the fundus of the stomach has the capacity to destroy and digest all living tissue, including the wall of the jejunum, duodenum, and even the stomach itself.” Despite his understanding of the physiology, he was reluctant to perform a vagotomy on a human because he was unsure that a person could withstand the operation. However, in 1943 he managed a 35-year-old man with ulcer disease who had failed medical therapy. The patient was offered a subtotal gastrectomy and the patient promptly declined. The patient stated that both his father and his brother had undergone subtotal gastrectomies and that his father had died and his brother was miserable as a result of the operation. Dragstedt performed a bilateral vagotomy through a left thoracotomy. The patient had immediate relief of his symptoms postoperatively. By 1945 Dragstedt had performed vagotomies on 60 patients and, as other surgeons had noted, he began to see postvagotomy “pyloric stenosis.” Although he initially performed a drainage operation only for patients who were symptomatic from the impaired gastric emptying, he later modified his operation and performed abdominal truncal vagotomies (TVs) with a pyloroplasty concomitantly ( Fig. 59.1 ).
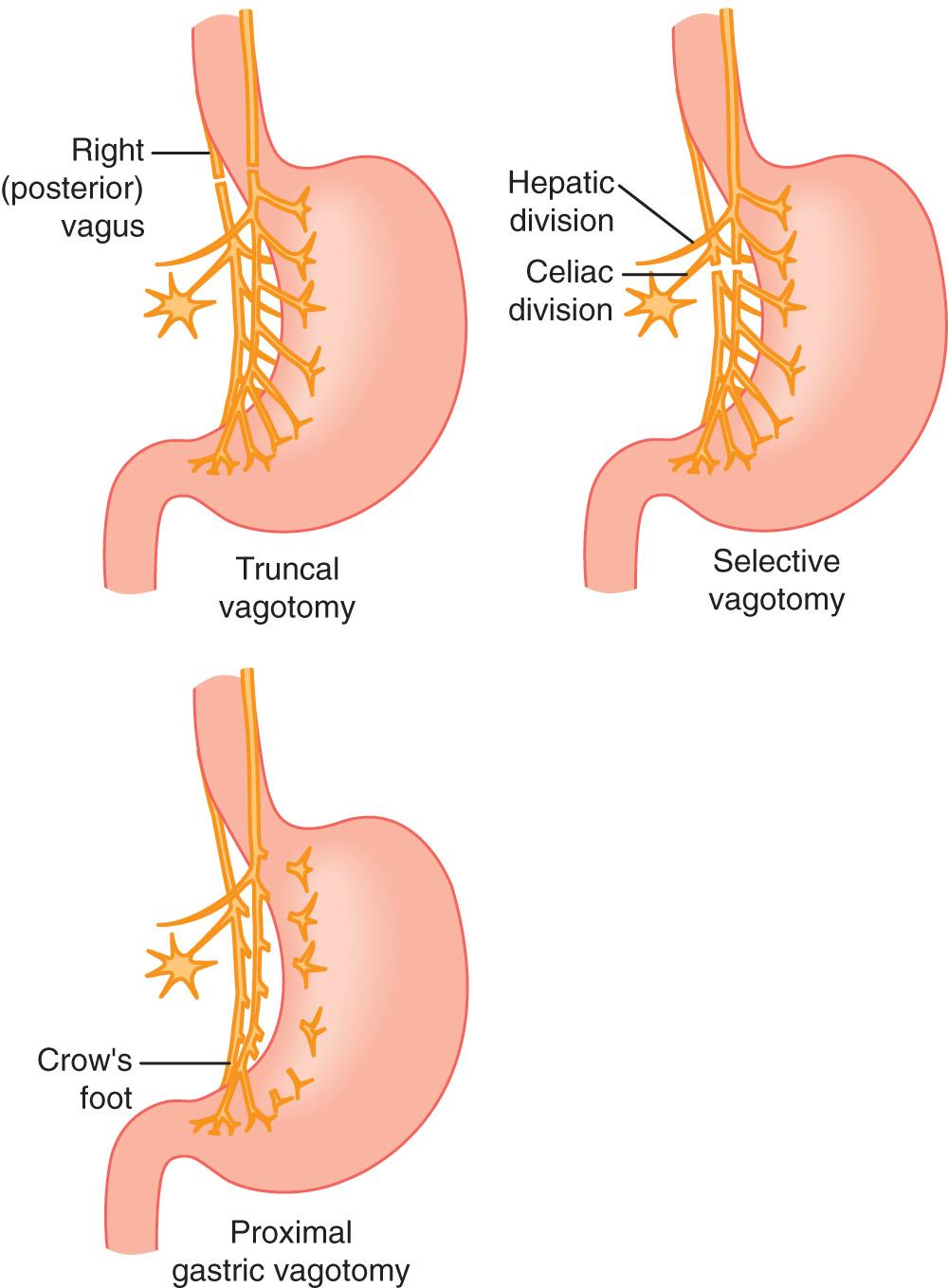
Vagotomy and drainage was gradually accepted because of its relatively equal results in regard to resolution of symptoms and a lower mortality when compared with resective procedures. Dragstedt considered his vagotomy surgical technique “the most important contribution of his career.” George Crile reported the Cleveland Clinic data: gastrectomy was associated with an ulcer recurrence rate and a mortality rate approximately three times higher than that in the vagotomy group. Goligher et al. compared vagotomy and pyloroplasty with other operations for duodenal ulceration and found that vagotomy and pyloroplasty had a recurrence rate at 2 years of 6.3%, vagotomy and enterostomy of 3.6%, whereas gastric resection had 0% recurrence.
The surgical dictum of the time was that the treatment of ulcer disease was directed at reducing acid secretion. Vagotomy was used to eliminate the cephalic phase of acid secretion, which was considered to be the major contributor in duodenal ulceration. Antrectomy was the solution to eliminating the gastric phase of acid secretion, considered to be a major cause of gastric ulceration. Incorporating both vagotomy and antrectomy would effectively reduce, if not eradicate, acid production, thus continuing the “no acid, no ulcer” dogma of the time, and eradicated the need for a drainage procedure.
Hubert et al. from the Mayo Clinic presented their results for vagotomy with antrectomy with a mean 17-year follow-up. They showed an operative mortality rate of 1.1% and an ulcer recurrence rate of 0.7%; the incidence of major postoperative complications was less than 1%. Extensive controversy revolved around the decision to perform a vagotomy and pyloroplasty versus a vagotomy with antrectomy. The idea that the two procedures both had a place in the treatment of ulcer disease was gradually accepted. At the time it was well established that for recurrent disease the optimal operation was vagotomy with resection. It was believed that, although both operations had equivalent results as to resolution of symptoms, vagotomy with antrectomy was associated with a lower recurrence rate, whereas vagotomy with drainage was associated with a lower mortality rate.
The selective vagotomies (SVs) were thought to be the answer to the question of how to decrease acid secretion and at the same time limit the known and occasionally debilitating postoperative morbidity of the previously discussed operations. The basis behind the more SVs was the work done by Griffith and Harkins in 1957. They showed that fibers originating from the vagal trunks along the lesser curvature of the stomach innervate small groupings of parietal cells and as they approach the pylorus, they have a primary motor function.
The goal of selective gastric vagotomy (SGV), first performed in the 1960s, was to limit the side effects encountered with other operations. However, gastric stasis was still a factor. In response to the motor denervation, a pyloroplasty or other drainage procedure was needed. Siim et al. followed 105 patients for up to 13 years and showed satisfactory resolution of symptoms in only 77% of those with low-grade ulcerations. In the individuals with high-grade/severe ulcer disease, only 19% had good results. In addition to the poor clinical results, there was a 15% recurrence rate. Ultimately, they concluded that “selective gastric vagotomy has no place in elective treatment.”
Proximal gastric vagotomy (PGV), also referred to as a parietal cell vagotomy or highly SV, does not affect the distal motor function and thus does not impede gastric emptying. In the 1970s PGV became the most popular operation for elective treatment of PUD because of its lower mortality and morbidity rates and the omission of a drainage procedure. van Heerden et al. reported the data on 223 patients from 1973 to 1977 at the Mayo Clinic. With a 39-month mean follow-up, the incidence of postoperative adverse effects was less than 3%, with 0% deaths and a 4.9% recurrence rate. At the end of their study, they concluded that “proximal gastric vagotomy is an effective, safe, and satisfactory option.”
Hoffmann et al. compared TV to SGV and PGV in a randomized controlled study of 248 patients with an 11- to 15-year follow-up. They found recurrence rates of 28.5% (TV), 37.4% (SGV), and 39.3% (PGV). Although there was a trend with lower recurrence in the TV group, the difference between the three groups failed to show statistical significance. Overall, the patient satisfaction for the three groups was similar, with approximately two-thirds of the patients being satisfied. The findings led to the conclusion that “none of the three forms of vagotomy can be recommended as the standard operative treatment.”
As the discovery of H 2 blockers and PPIs emerged, surgical indications decreased. The improved understanding of the pathogenesis of ulcer disease with the discovery of H. pylori and understanding the impact of NSAIDs further decreased the need for operation. Management became primarily medical in nature, with operation reserved for the emergency treatment of bleeding and perforation. This chapter discusses elective operations for intractable peptic ulcers and emergency procedures for complications.
The decades of surgical therapy dominating the treatment of PUD have been followed by a period of potent acid-reducing medication use that has now been replaced with a short-term regimen targeting the elimination of H. pylori infection. Discussions of the best operation have been replaced with a discussion of the best drug combination to treat the various manifestations of both gastric and duodenal peptic ulceration. A 10-day to 2-week course of drug therapy directed against H. pylori has an ulcer recurrence rate equivalent to TV with pyloroplasty. Although emergency operations for both peptic ulcer bleeding and perforation are still occasionally required, even their incidences are on the wane.
The introduction of histamine H 2 -receptor antagonists in 1977 radically changed the need for elective surgical therapy of PUD. Yet, it was the discovery of the association of Campylobacter pyloridis (renamed H. pylori in 1989) with peptic ulceration by Warren and Marshall in 1982 that truly revolutionized our understanding of ulcer pathogenesis and its treatment. They received the Nobel Prize for this work in 2005. Epidemiology studies revealed a strong association between H. pylori infection and both gastric and duodenal ulcer disease. Treatment of the infection resulted in long-term cure of peptic ulcers. Despite the development of potent antisecretory drugs and treatment for H. pylori infection, PUD remains an important clinical problem because of the widespread use of NSAIDs. The cause of peptic ulcers is complex and multifactorial, as they result from the interplay of the effects of gastric acid and pepsin and the gastric mucosal barrier. Any entity that either increases acid and pepsin secretion or weakens the mucosal barrier can result in ulcers ( Box 59.1 ).
Helicobacter pylori infection
Drug induced
Nonsteroidal antiinflammatory drugs
Low-dose aspirin
Acid hypersecretory state
Zollinger-Ellison syndrome
Retained gastric antrum
Anastomotic ulcer following gastric surgery
Severe physiologic stress
Tumors
H. pylori is the most common chronic bacterial infection in humans. Once acquired, infection persists and may or may not produce gastroduodenal disease. A number of factors determine whether H. pylori infection causes disease: the pattern of histologic gastritis induced; changes in homeostasis of gastrin and acid secretion; gastric metaplasia in the duodenum; interaction of H. pylori with the mucosal barrier; and the strain of H. pylori present. There is a great deal of variation in the virulence of different strains of H. pylori . Some genotypes of H. pylori appear to be particularly toxic and are more common in patients with peptic ulcers. These are vacA and cagA positive. There is also a genetic predisposition to acquire H. pylori infection.
H. pylori colonizes the entire gastric epithelium. However, the severity of the chronic mucosal inflammation is variable and the resultant clinical scenario is dependent on the distribution of the inflammation. The incidence of H. pylori in the setting of gastric ulcers is between 80% and 90% and up to 100% in the setting of duodenal ulcers. In patients with duodenal ulcer, density of infection and severity of inflammation are greatest in the distal antral region with sparing of the acid-secreting body mucosa. After H. pylori eradication, the gastric mucosal changes revert to normal.
In gastric ulcers, the body and antrum are affected to a similar degree. In this case, gastric acid secretion can be decreased because of the more severe involvement of the parietal cell region. In response to the same stimulation with gastrin, duodenal ulcer patients with H. pylori produce more acid than infected patients without ulcers. This may result from an impaired acid-secreting ability of the nonulcer H. pylori -infected patient's more diseased acid-secreting fundus mucosa. Increased gastric acid can lead to the development of gastric metaplasia in the duodenal bulb. This is a necessary forerunner to colonization of the duodenal epithelium with H. pylori , because H. pylori exclusively binds to the gastric epithelium. The metaplastic, H. pylori -colonized, duodenal epithelium then becomes more susceptible to acid and pepsin effects and ulceration. After the eradication of H. pylori infection, gastric metaplasia in the duodenum does not revert to normal, but with the elimination of the infection, the risk of ulcer recurrence is eliminated.
H. pylori infection impairs the negative feedback of gastrin release by somatostatin secreted by antral D cells. Somatostatin causes inhibition of gastrin release through a paracrine effect. Production of alkaline ammonia by the bacteria on both the surface epithelium and in the antral glands prevents the D cells from properly interpreting the level of acid present. This leads to improperly low levels of somatostatin, and thus loss of gastrin inhibition. Chronic hypergastrinemia caused by H. pylori exerts a trophic effect and hyperplasia of the acid-secreting parietal cells.
Infection with H. pylori also interferes with the neural connections between the antrum and fundus that downregulate acid production. This impaired neural control, coupled with hypergastrinemia, leads to further increases in acid production. With H. pylori eradication, the hypergastrinemia rapidly resolves. Resolution of acid hypersecretion occurs much more slowly.
The inflammatory response caused by H. pylori infection of the gastric mucosa leads to cytokine production, mainly, interleukin (IL)-8. IL-8 acts as a potent chemotactic and attracts neutrophils and acute inflammatory cells into the submucosa. Other cytokines include IL-17 and IL-18. In a recent animal model, increased serum level of IL-17 was found to correlate with severity of gastritis; this correlation was not observed with changes in serum level of IL-8 and IL-18.
Complex interactions occur between H. pylori and host defense mechanisms that affect the occurrence of peptic ulceration. Duodenal ulcers appear to be predominantly related to increased acid production, whereas in gastric ulceration, defense mechanism breaches appear to prevail. Despite these differences in mechanisms, H. pylori eradication effectively cures PUD and prevents relapses. In addition, the rate of ulcer healing is accelerated if antibiotics effective against H. pylori are given in addition to drugs that suppress acid.
NSAIDs increase the risk of peptic ulcers. NSAIDs are the most commonly identified risk factor for peptic ulcer bleeding, especially in older adults; the risk is drug specific and dose dependent. NSAIDs decrease the mucosal defense by suppression of prostaglandin synthesis in gastric and duodenal mucosa. Controlled trials with cyclooxygenase-2 (COX-2)-selective inhibitors have demonstrated a reduction in the risk of gastroduodenal ulcers and their associated complications.
The presence of gastric acid contributes to NSAID injury by converting superficial mucosal lesions to deeper ulcers. In addition, acid interferes with platelet aggregation and impairs ulcer healing. Acid suppression is the mainstay in the therapy of NSAID-associated ulcer disease. Risk factors that influence PUD in NSAID users include history of ulcer; advancing age; high-dose NSAIDs; steroids; aspirin; anticoagulants; and H. pylori infection. The use of COX-2 inhibitor and PPI can significantly reduce complications associated with NSAID intake.
Even at very low doses (75 mg daily), aspirin decreases gastric mucosal prostaglandin levels and can cause significant gastric lesions. The effect of aspirin is dose dependent, and ulcer complications are twofold to fourfold higher in patients taking 75 to 300 mg daily compared with controls. PPIs, given with low-dose aspirin, can significantly decrease the risk of developing peptic ulceration.
Both Zollinger-Ellison (ZE) syndrome as a consequence of gastrinoma, and retained gastric antrum after antrectomy with gastrojejunal anastomosis (so-called retained excluded antrum) result in peptic ulceration secondary to high levels of gastrin secretion. In cases of retained excluded gastric antrum, the residual gastric antral tissue is constantly bathed in a fluid with a high pH (nonacid), resulting in continuous secretion of gastrin. Fortunately, because of the infrequency of antrectomy in current surgical practice, this clinical situation is rarely encountered. In both disease states, high levels of serum gastrin result in gastric acid hypersecretion and resultant peptic ulceration. Serum gastrin elevations are also seen in chronic atrophic gastritis as a consequence of the lack of gastric acid secretion (typically achlorhydria) causing chronic G-cell stimulation.
The pathophysiology of stress ulceration is multifactorial and undefined. A breakdown of the gastroduodenal mucosal barrier, often a result of severe physiologic stress and splanchnic hypoperfusion, combined with gastric acid may lead to ulceration and bleeding. After splanchnic perfusion is restored, a reperfusion injury can further exacerbate the condition. It can develop within hours in critically ill patients, typically starting in the fundus and spreading distally. Prior to the development of effective medical therapy to reduce or eliminate gastric acid, this was a feared and highly lethal condition, often requiring total or near-total gastrectomy for control in extremely ill patients. Even with such heroic measures, mortality was extremely high. With the advent of histamine H 2 -receptor antagonists and PPI therapy, the primary goal of stress ulcer therapy has been to prevent clinically important bleeding by identifying those patients at risk for the development of stress ulceration ( Box 59.2 ) and administering appropriate prophylactic measures. Fortunately, acid-reducing medication effectively prevents significant bleeding in nearly all patients at risk for stress ulceration. Esophagogastroduodenoscopy (EGD) is the first line of intervention. It aids with the diagnosis. However, treatment is usually unsuccessful secondary to the diffuse nature of the bleeding. Angiography should be considered in patients who fail endoscopic intervention. Angiography can facilitate embolization of the bleeding vessel(s), which is usually the left gastric artery, or can help reduce the rate of bleeding by selective vasopressin infusion. Operative intervention is considered as the last resort in patients.
Respiratory failure requiring mechanical ventilation >48 h
Coagulopathy or anticoagulation
Acute renal insufficiency
Acute hepatic failure
Sepsis
Hypotension
Brain or spinal cord injury
History of gastrointestinal bleeding
Low intragastric pH
Burn involving >35% of body surface area
Major operation (>4 h)
High-dose corticosteroids (>250 mg/day hydrocortisone or equivalent)
The treatment of PUD has undergone a significant change. The previous use of elective surgical means to treat PUD has faded into history, and medical therapy has moved to the forefront of current treatment. Elective surgical procedures for PUD are limited these days to patients with gastric outlet obstruction because of long-standing, untreated or poorly treated ulcer disease. These patients are rare, and many are treatable with endoscopic dilation with or without stenting.
In a meta-analysis including 2102 patients with PUD, the 12-month ulcer remission rates for gastric and duodenal ulcers were significantly higher in patients who were successfully eradicated of H. pylori infection when compared with those with a persistent infection (97% and 98% vs. 61% and 65%, respectively). H. pylori eradication even without concurrent acid suppression therapy heals greater than 85% of duodenal ulcers. Confirmation of H. pylori eradication should be strongly considered for all patients receiving treatment because of the availability of accurate, relatively inexpensive, and noninvasive tests. All patients with duodenal ulcer(s) should receive antisecretory therapy to facilitate ulcer healing; however, the duration of therapy will vary depending upon ulcer characteristics, risk factors for recurrent PUD, and the presence of ulcer complications. In patients with uncomplicated duodenal ulcer who test positive for H. pylori , PPI, given for 10 to 14 days, along with the antibiotic regimen to eradicate H. pylori , is usually adequate to induce healing, and additional PPI therapy is not needed as long as they are asymptomatic following therapy. Thus medical therapy for duodenal ulcer has shifted away from an antisecretory/antacid or surgical approaches to an antimicrobial strategy.
Most peptic ulcers respond to medical treatment. However, in some individuals the ulcer is either refractory to conventional therapy or recurs following successful initial treatment. A refractory peptic ulcer is defined as an endoscopically proven ulcer greater than 5 mm in diameter that does not heal after 12 weeks of treatment with a PPI. On the other hand, a recurrent peptic ulcer is defined as an endoscopically proven ulcer greater than 5 mm in diameter that develops within 12 months following complete ulcer healing documented by repeat endoscopy. Prior to labeling the ulcer disease as intractable, it is important to rule out the following:
Cancer by performing endoscopy with adequate biopsy of the ulcer edge and base.
Gastrinoma by measuring fasting serum gastrin.
Total serum calcium should be measured to screen for hyperparathyroidism.
Ulcerogenic medication (e.g., NSAIDs, aspirin).
Persistent H. pylori infection by undergoing additional tests to confirm eradication. Ideally patients should be off of the PPI for at least 2 weeks to reduce false-negative results.
Chronic smoking, although smoking does not appear to be a risk factor for ulcer relapse after H. pylori has been eradicated.
After these have been ruled out and when operative intervention is being considered, the strategy continues to be based on reduction of acid secretion. Gastric distention is an important stimulant of gastrin release by G cells, which are mainly located in the antrum; thus decompressing the stomach in patients with bleeding ulcers and gastric outlet obstruction secondary to ulcer is important to reduce gastrin and hence acid release. Acid release can surgically be reduced by dividing the vagus (cephalic phase), and eliminating hormonal stimulation from the antrum (gastric phase). Each of these maneuvers has consequences in terms of the normal physiology of the upper gastrointestinal tract that tend to be amplified when the procedures are combined, such as with vagotomy and antrectomy. In the past, the choice of operation involved weighing the risk of recurrent ulceration with the possibility of postoperative complications and long-term sequelae (postgastrectomy syndromes). This decision dilemma prompted a large number of trials comparing these procedures in the surgical literature. Improvements in medical therapy, particularly treatment of H. pylori , have markedly reduced the risk of ulcer recurrence, rendering much of these data obsolete. Thus surgical decision-making has become confusing with little quality data available from the post– H. pylori era. The choices for surgical intervention for intractable duodenal ulcer disease include either a vagotomy with or without a drainage procedure or with a gastric resection.
The rationale for vagotomy is the elimination of direct cholinergic stimulation of gastric acid secretion. The released acetylcholine stimulates acid secretion via a specific receptor on the parietal cell. Vagotomy also renders the acid-producing parietal cells less responsive to histamine and gastrin. The distal portion of the anterior and posterior trunks send branches to the antrum and pylorus that serve a primarily motor function. Gastric motility is affected by the antral and pyloric branches of the vagus that stimulate peristaltic activity of the antrum and relaxation of the pylorus. The celiac branch of the posterior vagus mediates small intestine motility, whereas the hepatic branch mediates bile flow and gallbladder motility.
TV results in a variety of physiologic alterations in the stomach. Acid secretion is drastically reduced because of diminished cholinergic stimulation of parietal cells, and the cephalic phase of gastric secretion is essentially eliminated. There is a 75% decrease in basal acid secretion and a 50% decrease in maximum acid output. The increased intraluminal stomach pH leads to elimination of the negative feedback on gastrin secretion; therefore, this results in increased serum gastrin levels and gastrin cell hyperplasia. As a result of loss of reflex relaxation of the gastric fundus, there is rapid emptying of liquids. Similarly, TV affects distal gastric motility, resulting in difficulty in emptying solids. Because of the latter alterations, approximately 20% to 30% of patients develop gastric atony, which leads to stasis and chronic abdominal pain and distention. For that reason, it is recommended that after a TV patients should undergo a drainage procedure to counteract the nonrelaxing pylorus, which acts as an obstruction. The various drainage procedures available are discussed later. There are four types of vagotomy to consider: truncal, selective, proximal gastric, and supradiaphragmatic. Truncal and proximal gastric are commonly used to treat PUD, whereas selective and supradiaphragmatic vagotomies are used infrequently.
TV (see Fig. 59.1 ) involves division of the anterior and posterior vagal trunks after they emerge below the diaphragm. The first step is to incise the peritoneal covering of the gastroesophageal junction. The peritoneum is opened horizontally, from the lesser curvature to the cardiac notch at the greater curvature. The surgeon uses thumb and right index finger for blunt dissection to encircle the esophagus. A Penrose drain is placed around the lower esophagus to place more effective downward traction on the gastroesophageal junction. When encircling the esophagus, the surgeon stays wide of the esophagus to prevent inadvertent entry into the lumen and to include the vagal trunks. In the course of this maneuver, the posterior vagal trunk usually will be palpated as a taut cord anterior to the aorta. A single anterior vagal trunk is usually identified in the anterior midportion of the esophagus, 2 to 4 cm above the gastroesophageal junction. It is not uncommon for vagal fibers to be distributed among two or three smaller cords at this level. These trunks are individually lifted up, and 2- to 4-cm segments of each are separated from surrounding tissues. A 1- to 2-cm length of nerve is resected and a clip is applied to the cut ends of the nerve. The “criminal nerve” of Grassi also may be identified wrapping around the cardiac notch from its origin in the posterior trunk and is a common cause of incomplete vagotomy.
The posterior vagal trunk is usually identified along the right edge of the esophagus. If the anterior vagus has already been divided, the esophagus is more mobile. This mobility allows downward traction on the gastroesophageal junction, causing the posterior vagus to “bowstring” and making it easier to identify. A 2- to 4-cm segment is separated from surrounding tissues, its margins marked with clips, and resected. The resected portions of the anterior and posterior vagal trunks should be sent to pathology for frozen section. This procedure completely denervates the stomach and eliminates vagal innervation to the pancreas, small intestine, proximal colon, and hepatobiliary tree. Although this procedure significantly reduces acid secretion, it also markedly alters gastric motility. As discussed earlier, some form of gastric emptying procedure should be performed.
The SV procedure (see Fig. 59.1 ) was developed in an attempt to decrease the incidence of postvagotomy diarrhea and ameliorate the increased incidence of gallbladder stasis, which may lead to increased gallstone formation. The vagal fibers are divided distal to the takeoff of the hepatic branch(es) from the anterior vagus and the celiac branch(es) from the posterior vagus. This procedure is technically more demanding than TV and requires a more careful and meticulous dissection. This technique spares vagal innervation to the gallbladder and intestine while completely denervating the stomach. Because the vagal pyloric innervation is also eliminated, a drainage procedure is still required. The primary reason for the development of this technique was its presumed lower side-effect profile. However, a prospective randomized study failed to show substantial benefit for SV over TV. The incidence of diarrhea following an SV was no different when compared with TV. The introduction of PGV with its lower side-effect profile and the elimination of the need for a drainage procedure resulted in a limited use of SV as a therapeutic option.
PGV is also known as parietal cell vagotomy and highly SV (see Fig. 59.1 ). The rationale for PGV is to eliminate the vagal stimulation to the acid-secreting portion of the stomach without interrupting motor innervation to the antrum and pylorus. The operation involves severing all branches of the vagus nerve along the lesser curvature that innervate the corpus and fundus of the stomach, while preserving the hepatic and celiac branches, as well as the distal vagal branches extending to the antrum and pylorus. The end result of this procedure is the same reduction in acid secretion that occurs after TV (basal and stimulated acid secretion are reduced by more than 75% and 50%, respectively) but without the troublesome stasis and gastric atony. Because the distal motor nerves are preserved, emptying of solids is normal; however, the nerves affecting receptive relaxation are divided, and some rapid emptying of liquids may occur. The alteration in liquid emptying is usually minimal. This procedure is associated with the lowest morbidity rate of all vagotomy procedures and became the operation of choice in many centers despite a reported ulcer recurrence rate of between 5% and 20%. A meta-analysis of 12 trials confirmed that PGV has the highest recurrence rate when compared with TV with pyloroplasty, but fewer long-term side effects. PGV also has been compared with TV in a randomized trial, in which it was shown to have a lower incidence of dumping syndrome and weight loss. Although the ulcer recurrence rates were higher with PGV, this was not significant when prepyloric ulcers (for which PGV is not an adequate operation) were excluded.
PGV is a complex and lengthy procedure, and, to help to simplify the procedure, several variations have been described. They usually consist of a posterior TV and a more selective ablation of the anterior vagal fibers to the gastric fundus and body. Hill and Baker performed a posterior TV with an anterior PGV (Hill-Baker procedure). Taylor combined the posterior TV with anterior lesser-curve seromyotomy (Taylor procedure). Randomized studies confirm the superiority of the Taylor procedure to TV and document equal outcomes to PGV with a shorter operative time. With the decreased incidence of elective ulcer surgery, such operations are not commonly used. However, such approaches are popular for laparoscopic treatment of ulcer disease.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here