Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Basal cell carcinoma (BCC) and cutaneous squamous cell carcinoma (cSCC) are the most common forms of nonmelanoma skin cancers. Some 70–90% of cSCCs are located in the head and neck region, and 5% of cutaneous skin malignancies metastasize into the parotid or periparotid lymph nodes. Patients with primary lesions >2 cm in diameter, tumor thickness >4 mm, tumor on or around the ear, recurrent lesions, high-grade lesions, microvascular, lymphatic, or perineural invasion (PNI), advanced age, and cSCC in an immunocompromised host, all have higher risk of metastasis.
The incidence of parotid metastasis is on the rise, with the proportion of parotid cancers metastatic to the parotid rising from 31% between 1975 and 1995, to 49% between 1995 and 2015.
Parotid metastases frequency varies between populations in different geographical locations with different levels of sun exposure. In Europe and the United States, the proportion of metastatic tumors in parotid malignancies is around 25%. In Australia and New Zealand, the proportion of metastatic parotid tumors arising from skin malignancies is 60%. Parotid metastasis from cutaneous primary tumors frequently shows delayed presentation after the primary cancer has been treated.
Since the extent of parotid and neck node involvement has an impact on the survival of patients with cSCC, early diagnosis and therapy is essential. However, in the absence of clinical or imaging findings in the parotid gland, the risk of occult metastases is not high enough to warrant elective parotidectomy in many patients with cutaneous SCC.
Parotid surgery for secondary malignant disease of the parotid gland most commonly includes cutaneous malignancies (melanoma and nonmelanoma) and rarely involves metastatic disease from a distant primary site. Metastatic disease to the parotid gland often represents a biologically aggressive tumor that frequently has other pathologically aggressive features, including lymphovascular space (LVI) and PNI, and a propensity for distant spread.
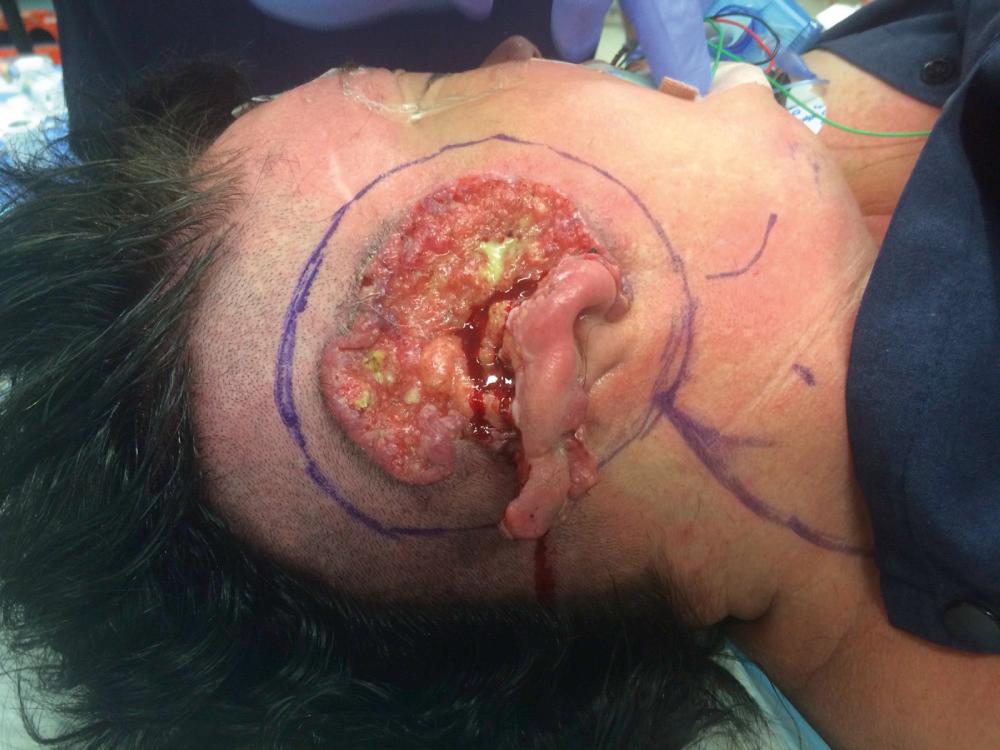
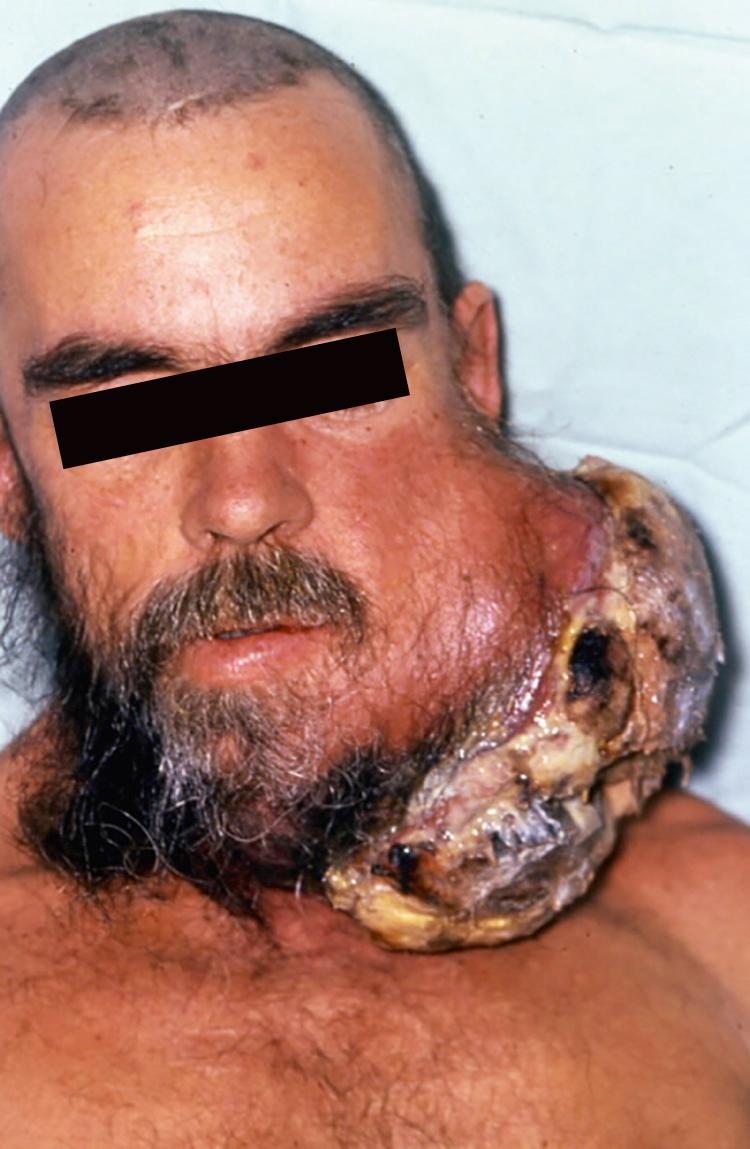
Direct infiltration of malignant disease into the parotid gland may be seen in cutaneous malignancies, including BCC ( Fig. 49.1 ) or cSCC. Parotidectomy for metastases from primary tumors in non-head and neck sites, may be indicated for symptom control ( Fig. 49.2 ). Primary palliative radio-chemotherapy and immunotherapy should also be considered in selected cases.
Intraparotid nodes are commonly first-echelon drainage basins from melanoma of the anterior scalp, temple, forehead, and pinna. Melanoma <1 mm has a low risk of metastasis. There is no role for elective node dissection because of the complexity of drainage patterns in the head and neck. The evidence relating to sentinel node biopsy (SNB) for 1B or higher cutaneous melanoma in the parotid region can improve staging, selection of patients for adjuvant treatment, and assurance to patients with negative biopsy. However, the overall survival benefit has not been proven with parotidectomy and neck dissection versus observation, despite large randomized trials.
Previous history of skin cancers in the head and neck region, or the presence of a suspected skin lesion, fixed parotid mass pain, rapid growth in an existing parotid mass, facial nerve (FN) paralysis, skin involvement, or cervical lymph node metastasis, suggests parotid malignancy. FN paralysis is present in 12–19% of patients with a malignant parotid mass independent of tumor size. If FN sacrifice is likely, then a multidisciplinary team with experts on facial reanimation should be involved in management.
Cutaneous cancers of the head and neck metastasizing into the parotid gland may be associated with increased expression of epidermal growth factor and are particularly aggressive and associated with poor outcome, requiring a different diagnostic regimen and surgical and adjuvant management.
Options to consider for tissue diagnosis are fine needle aspiration (FNA) cytology, core needle biopsy (image guided or direct), or open biopsy. Preoperative tissue diagnosis is helpful, particularly in metastatic disease, to stage direct adjuvant therapy or to consider other means of management. Open biopsy may be warranted in a select group of cases, including patients with a high suspicion of malignancy where FN sacrifice is likely, or in patients with suspected lymphoma and in those patients where diagnosis cannot be made on FNA or core biopsy.
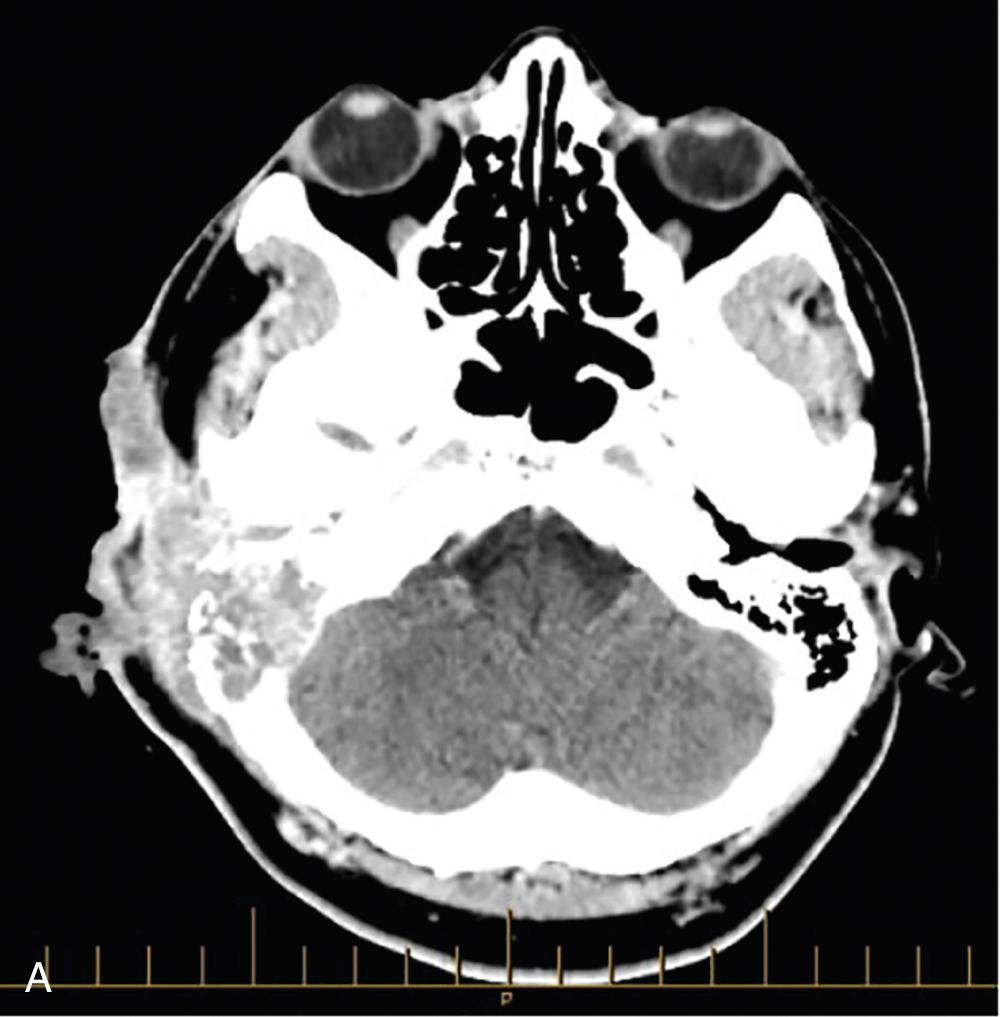
Imaging helps define the extent of the disease ( Fig. 49.3A ). This includes local invasion into surrounding structures, PNI, cervical metastasis, and evaluation of distant metastasis. CT is utilized for tumors extending into bony structures when an extended lateral temporal bone resection (LTBR) is planned. Magnetic resonance imaging (MRI) is useful in evaluating deep lobe, parapharyngeal space, base of skull, and cranial nerve involvement. Identifying these characteristics may alter surgical approach and classify the patient as unresectable. Positron emission tomography (PET) scan is useful in staging and follow-up.
The surgical management of parotid metastases from cSCC has focused on parotidectomy, neck dissection ( Fig. 49.3B ), and soft tissue resection. LTBR is indicated in selected patients. The extent of surgery is tailored to the extent of disease.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here