Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Modern stereotactic functional surgery includes lesioning and stimulation procedures. Both have their advantages and disadvantages, and their choice should be tailored to the therapeutic aims and circumstances in the individual patient.
Evidence for the efficacy of stereotactic surgery in mental health disorders has been variable. Randomized controlled trials have provided level 1 evidence of efficacy of functional neurosurgery for obsessive-compulsive disorder and Tourette syndrome.
Deep brain stimulation for cluster headache is a therapy that has been directly informed by the results of functional neuroimaging and can decrease headache load and improve quality of life.
Deep brain stimulation for epilepsy is still being explored, and its place in the armamentarium of epileptologists remains to be determined.
Mental health disorders, pain, and epilepsy have been recognized in the medical literature since antiquity, but two millennia later they continue to represent a clinical challenge and many patients have medically intractable disease. As with movement disorders, these conditions have been treated with stereotactic lesioning procedures since the mid-20th century, and interest has focused on deep brain stimulation (DBS) since its resurgence in the 1990s. The appeal of selectively and reversibly modulating discrete brain areas has led to a large focus of research by the international neurosciences community into DBS in particular for epilepsy and mental health disorders such as depression, obsessive-compulsive disorder (OCD), and Tourette syndrome. Surgery remains one aspect of these patients' continuing multimodal care rather than a cure. This chapter focuses on the indications with most utilization in current practice and also addresses those still under experimental investigation.
Surgical intervention for psychiatric disorders should be undertaken as part of a management schedule driven by a multidisciplinary team and led by psychiatry expertise. Stereotactic surgery has been undertaken since the 1950s for these disorders and includes both lesioning and electrical stimulation (for a review, see Hariz, 2016).
Gilles de la Tourette described this syndrome, which manifests as sudden brief tics, both vocal and motor, often preceded by urges. The underlying pathophysiology is poorly understood. It is proposed that abnormal activity of striatal neurons suppress the substantia nigra pars reticulate and globus pallidus internus (GPi). The latter two suppress thalamic activity and unwanted, inappropriate actions. However, thalamic inhibition is lost in Tourette syndrome. Tics, as well as being socially limiting, can also be violent, resulting in accidental harm to sufferers or others. In severe forms, medical therapy such as alpha-2 agonists, antipsychotics, benzodiazepines, and botulinum toxin injections are insufficient to control the symptoms, and therefore surgery has been explored in these patients.
Trials in small numbers of patients have shown amelioration in tics and helped inform the understanding of the condition. Targets have included the anterior cingulate cortex (ACC) (cingulotomy), limbic leucotomy (which includes cingulotomy and subcaudate tractotomy), the thalamus (ventrolateral nuclei; Fig. 58.1 ), and zona incerta (for a review, see Houeto, 2009). Significant adverse effects such as hemiplegia or dystonia were reported.
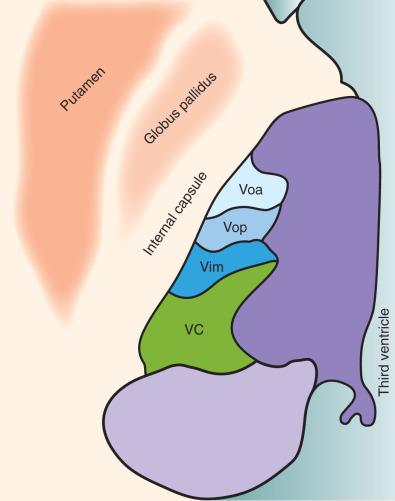
DBS entails implantation of 1.3-mm diameter electrodes within these critical structures and then provides modulation neural of activity rather than injury. Furthermore, the stimulation is modifiable and can be balanced between efficacy and adverse stimulation effects. It is also reversible. DBS has therefore been a desirable alternative to explore. The thalamic lesion effects observed by Hassler formed the foundation for modern DBS trials in Tourette syndrome. Vanderwalle and colleagues targeted DBS electrodes to the centromedian and ventral oralis internus nuclei and the substantia periventricularis. Significant improvements were found at the follow-up of 8 months to 5 years in three patients. A 72% to 90% improvement in frequency of tics per 10 minutes was observed using high-frequency, wide pulse width stimulation. Six hardware revision procedures were required, which is a high demand.
A randomized, double-blinded, crossover trial at Queen Square enrolled 15 severe, medically refractory Tourette syndrome patients with a mean age of 34.7 years, and in 13 data were complete. All patients received bilateral GPi electrode implantation ( Fig. 58.2 ) and were randomized to stimulation and no stimulation in a 1 : 1 ratio for the first 3 months, followed by the reverse condition for the next 3 months. Patients and scorers were blinded. The mean score on the Yale Global Tic Severity Scale improved by 7.2 points, from 87.9 baseline to 80.7, in the control no-stimulation group. In the stimulation group, the mean Yale Global Tic Severity Scale results improved significantly by 19.6 points (from 87.9 to 68.3).
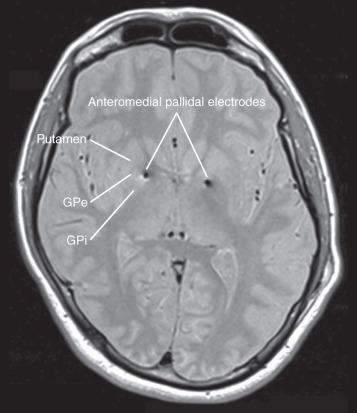
Serious adverse effects occurred in three patients. Two patients encountered hardware infection at the battery site requiring explantation. Due to the efficacy they had enjoyed, they reentered the trial and underwent DBS reimplantation. Hypomania with tic deterioration occurred in one patient who required admission, benzodiazepines, and stimulation adjustment. All three adverse events were resolved by active treatment. Less severe events occurred in 67% of patients, including mild dysarthria, insomnia, and irritability but resolved in 60%. Two patients received posteroventral GPi stimulation on the basis of concurrent dystonic features, whereas all others underwent anteromedial GPi stimulation. The study was not designed to evaluate the superiority of one target over the other.
Overall, trials of DBS for Tourette syndrome have been positive albeit variable in terms of brain targets, selection criteria, hardware, and stimulation strategies. This has hindered regulatory body approval. The Tourette Association of America launched a DBS registry and database to address this matter.
The prevalence of OCD in the general population is 2% to 3%. In severe cases, it can significantly limit daily activities, social interaction, and ability to work. Treatment includes cognitive behavioral therapy and medication. Selected intractable cases have undergone surgical treatment for the last half-century.
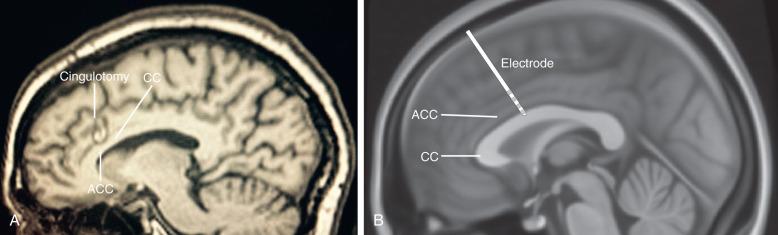
The neural circuits underlying OCD are still being defined, but a body of evidence implicates the frontal lobes and basal ganglia with the fronto-striatal-pallidal-thalamic-frontal loop, which passes through the anterior limb of the internal capsule (ALIC). The two most common lesioning procedures for OCD are anterior cingulotomy ( Fig. 58.3A ) and anterior capsulotomy. These are generally safe and well-tolerated procedures. A series of 1000 radiofrequency cingulotomies at Massachusetts General Hospital found no deaths or infections. Seizure was present in only 1% of patients and other adverse effects were mild or short lived including headache, temperature, and transient urinary dysfunction. Careful patient selection is necessary and severity objectively scored, for example, using the Yale-Brown Obsessive Compulsive Scale (YBOCS) score, accepting those with a score worse than 20, and disability severity assessed by the Global Assessment Function (GAF) of less than 50. In a review of 20 studies encompassing 108 capsulotomy patients, there was a 51% improvement in YBOCS at 61 months. About 25% to 30% of patients receiving cingulotomy for OCD in Ballantine's series were significant responders with a further 10% to 15% partial responders.
More recently, Gamma Knife capsulotomy using radiosurgery has been shown to improve median YBOCS by 28.6% compared to a sham of 5.8%, ( p = .04988). Two out of eight patients who received treatment responded. The strength of this study of 16 patients was the convincing use of a surgical sham whereby the patients lay either in the Gamma Knife chamber or in a specially built sham chamber attached to the Gamma Knife. As the radiosurgery procedure does not produce any sensations or external features, patient blinding was of high standard.
The two most promising anatomic targets for DBS are the ALIC and the anteromedial subthalamic nucleus (amSTN). Nuttin and colleagues demonstrated rapid improvement in OCD in one patient by stimulating the ALIC bilaterally. They further implanted six patients with bilateral ALIC DBS. The mean YBOCS was 19.8 during stimulation compared to 32.2 off stimulation. Over the years, this group has progressively moved its target more posteriorly toward the bed nucleus of the stria terminalis and has published its results of 24 patients with significant benefit of stimulation over sham in 17 of these. The Amsterdam group reported similar improvement with DBS of the ALIC, again with significant improvement in the YBOCS comparing active to sham stimulation.
A multicenter French study demonstrated the efficacy of amSTN DBS in severe refractory OCD in a sham crossover design study with 16 patients. However, long-term clinical results have not yet been published.
Pepper and associates reviewed 20 studies evaluating outcomes in 62 patients undergoing DBS to the ventral caudate/ventral striatum and nucleus accumbens. They found an overall 40% improvement in YBOCS. However, this was significantly less than the anterior capsulotomy group's 51% improvement. Moreover, significant side effects were more common with DBS. Therefore lesioning still offers an important treatment option for these patients, even in the era of DBS. Whether DBS or lesioning is employed, responders appear to experience progressive improvement in symptoms over time. Moreover, there appears to be a synergistic effect with cognitive behavioral therapy in patients who did not improve or could not engage with this therapy prior to surgery.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here