Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Orthotopic liver transplantation has now become the gold standard treatment for end-stage liver disease. However, the ongoing shortage of suitable livers, together with progressively longer waiting lists, prevents many patients from being transplanted; this has led to a significant waiting list mortality rate at most centers. Using livers from living donors is one way to increase the supply of liver grafts but carries with it the disadvantage of donor risk. Splitting the deceased donor liver to use for two recipients represents another method for expanding the donor pool. With split-liver transplantation (SLT), a whole liver from a deceased donor is divided into two functioning grafts, which can then be transplanted into two appropriate-sized recipients. The first SLT was performed by Pichlmayr et al in 1988. Subsequently several centers published their SLT series. The technical aspects of SLT are in many ways similar to the technical aspects of living donor transplants, and these two areas of liver transplantation have developed side by side in many centers, with one program complementing the other. More recently, living donor transplantation has become significantly more common than SLT, especially in areas of the world where deceased donor transplants are not common. The two techniques to expand the donor pool are not mutually exclusive and in fact are usually more successful in programs where both live donor and deceased donor SLT procedures are performed. Ultimately the ability to offer all options for transplant can only benefit the potential recipient and minimize waiting list mortality.
The vast majority of SLT procedures performed to date have been for one adult and one pediatric recipient. Usually the liver is split into a smaller portion consisting of the left lateral segment (which can be transplanted into a pediatric recipient) and the remaining larger extended right lobe (which can be transplanted into a normal-sized adult recipient). The benefits for pediatric recipients have been tremendous, including an expansion of the donor pool and a significant decrease in waiting times and mortality rates. As experience with SLT in this setting has grown, the results have improved. Many single-center series now report equivalent outcomes with split and whole organ deceased donor transplants. Although splitting the liver for an adult and a pediatric recipient has had a significant impact on the expansion of the donor pool for pediatric recipients, it has had no impact on the donor pool for adult recipients, because ultimately this type of split generates only one graft for the adult recipient. Because the majority of individuals on a transplant list are adults, and the majority of waiting list deaths are in adult patients, SLT can have the maximal impact on waiting list mortality if the two grafts generated can be used for two adult recipients. To accomplish this, the liver is generally split into the anatomical right and left lobes, which are then transplanted into two adult-sized recipients. However, splitting the liver for two adult recipients is generally uncommon at this time. There are many reasons why SLT for two adult recipients has remained relatively uncommon. Ideal donors appropriate for splitting have become less common as an increasing number of deceased donors have risk factors that would make them unsuitable for splitting. Similarly, with current allocation rules, appropriate recipients may also be difficult to find because the potential recipients at the top of a waiting list may be too ill to tolerate a partial liver transplant. Other hurdles include the technical complexities and challenges associated with the procedure, the logistics involved with coordinating multiple teams, and the reported results to date. Nonetheless, SLT can play a role in the expansion of the donor pool for adult recipients. Key aspects in trying to optimize results include careful donor and recipient selection, meticulous surgical technique in both the donor and recipient operations, and appropriate methods of allocation to ensure the greatest chance of success. This chapter will cover these areas and present some of the published series with regard to patient outcomes.
Proper donor and recipient selection are crucial to ensuring a good outcome with any SLT. Donors generally have to be ideal; but equally important, suitable recipients that can tolerate partial grafts must be chosen. One obviously wants to prevent the situation in which one deceased donor liver that would have worked in one recipient is split and results in two grafts that do not work in two recipients.
Donors should be medically ideal to minimize the risks of primary nonfunction, especially for recipients of the smaller left lobe graft. Young, hemodynamically stable donors with near-normal liver function test results, short intensive care unit stay (<5 days), and absent or fairly short arrest times should be selected; with such donors, primary nonfunction for the recipients should be uncommon. The upper donor age limit for splitting a liver is unclear, and the criteria likely need to be more stringent when performing a right lobe/left lobe split versus an extended right/left lateral lobe split. Generally donors much beyond an upper age limit of 45 years are likely not to be suitable for splitting. Liver function test results should be close to normal or at most less than three times the upper limit of normal. Donor size plays an important role in determining suitability for right lobe/left lobe splits because the size of the grafts generated in this situation are key predictors of successful outcome in the recipients. The size of the donor correlates to some extent with the size of the liver because we know that the liver constitutes roughly 2% of the total body weight. This obviously holds true only to a certain weight, and obese donors (body mass index > 30) should generally not be considered because of the risk for underlying fatty liver. An intraoperative biopsy can be useful to rule out any significant macrosteatosis, and greater than 10% fat would be a contraindication for splitting the liver. Male donors are generally likely to be more suitable for splitting because their livers tend to be bigger, but this cannot always be reliably predicted. Many donors have a computed tomography scan of the abdomen available (especially if the cause of death was trauma), and it is generally not difficult now to obtain at least crude measures of liver volumes from these scans before donation to help in making an appropriate selection decision.
Cold ischemic time should be minimized as much as possible in all SLT recipients. This is a crucial aspect because cold ischemic time represents one of the few donor risk factors that can be affected by the transplant team. Ideally cold ischemic times should be less than 10 hours, and less than 8 hours if possible. This requires significant planning with multiple teams and operating rooms so that there can be some degree of overlap between the donor and recipient procedures and at least some degree of overlap between the two recipient procedures.
When selecting appropriate recipients for SLT, important issues are graft size requirement, cause of liver failure, and severity of illness. Critically ill patients with severe portal hypertension and high Model for End-Stage Liver Disease (MELD) scores are generally not good candidates for partial grafts, especially from a deceased donor. Patients with tumors, metabolic diseases, or MELD scores below 30 may be appropriate candidates for these types of splits. Hepatitis C in the recipient does not represent a contraindication for a split graft. A graft weight–to–body weight ratio (GWBWR) of close to 0.8% should likely be the minimum when selecting appropriate recipients. Results with GWBWR ratios of less than 0.8% have been associated with inferior results, though not all series have supported this finding. It is also important to remember that this recommendation is based on data mostly from the living donor literature. A partial graft from a deceased donor likely has more injury than a partial graft from a living donor. Additional stresses on the deceased donor versus the living donor graft include the factors that led initially to the donor’s death, potential hemodynamic instability during the preprocurement management of the deceased donor, and the subsequent cold ischemic time after removal of the organ. Therefore extrapolating the 0.8% value may not be entirely appropriate for SLT from a deceased donor. Nonetheless, our practice has been to try to select recipients in whom we know the GWBWR will exceed 0.8%. This can sometimes be difficult because estimating the size of the partial graft ahead of time can be difficult. Again, there may be an abdominal computed tomography scan available on the donor for review before the procurement. This can be valuable in assessing the anatomy and the size of the two grafts ahead of the procurement. The final decision regarding which recipient is most appropriate can be made intraoperatively during the procurement surgery, once the donor liver has been carefully inspected and the size of the grafts estimated by a trained surgeon.
Another important aspect of the recipient selection process is adequately informing the potential recipient of the splitting procedure and obtaining informed consent. With the current organ allocation system in the United States, the graft is initially assigned to a primary recipient. If the liver is to be split, the second recipient is chosen at the discretion of the center performing the split. This is advantageous for the second recipient, who then bypasses additional waiting time. For primary recipients, however, there is no significant advantage: we are in fact asking them to give a part of “their” new liver to someone else. If the primary recipient is to receive the left lobe (and is therefore likely of smaller size), the issue is not as difficult. A full-size liver graft from a large donor may be difficult to fit into this recipient. Performing the split may allow for a better size match in this situation. But if the primary recipient is to receive the right lobe, the issue is more difficult, because such recipients could easily accommodate the whole graft. In our experience there is usually no hesitation on the part of the recipients to participate in the split, and in some instances some recipients have commented that they tremendously appreciated the opportunity to help another individual by agreeing to the split. Nonetheless, with the present allocation rules, it is crucial to fully inform potential recipients about splitting and to obtain informed consent. This brings up an important question, which is if it is up to the primary recipient to decide if a split should be performed by virtue of their consent. Ideally the center and organ procurement organization should decide ahead of time if this is an appropriate organ for spitting, and the two partial grafts then should be allocated to the two most appropriate recipients near the top of the list, but not necessarily at the very top of the list. These points have been debated for years, and unfortunately there is no good consensus on this at present.
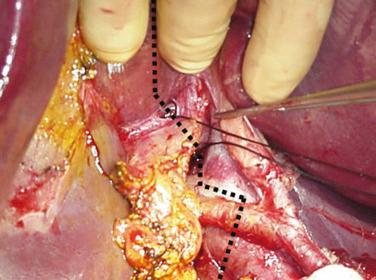
The SLT procedure can be divided into two parts: the donor operation and the recipient surgery (which in turn consists of the surgery for the right lobe and left lobe recipients). Both parts of the procedure are technically demanding, and the success of this procedure rests on performing a meticulous division of the donor liver and then subsequent implantation in the two recipients. When the liver is to be split for two adult recipients, it is usually transected in its midplane, generating two similar-sized grafts—the larger anatomical right lobe and the smaller anatomical left lobe. The transection plane should stay to the right of the middle hepatic vein so that this structure is retained with the left lobe ( Fig. 53-1 ). Segment IV makes up a crucial part of the left lobe, and hence the middle hepatic vein should be preserved with the left lobe to ensure no congestion. Loss of the middle hepatic vein may affect drainage of segments V and VIII in the right lobe graft. If significant draining vessels are identified, they can easily be reconstructed on the back table using vascular conduits from the deceased donor. Regarding the dissection in the hilum ( Fig. 53-2 ), our preference has been to leave the full length of the hilar vascular structures intact with the left lobe. The right-sided hilar structures are usually larger than the left-sided structures. Therefore leaving the main vessels intact with the left lobe makes that transplant easier. The common bile duct is left maintained with the right lobe graft, however, so that there is a greater chance of having a single bile duct orifice to reconstruct on both the right and left lobe grafts. One crucial technical point for the recipient operation is ensuring adequate venous outflow of the grafts to prevent congestion. Preserving the cava with the right lobe graft helps to maximize outflow by preserving all inferior hepatic veins. This also allows for back-table reconstruction of any segment V and VIII veins draining from the right lobe to the middle hepatic vein.
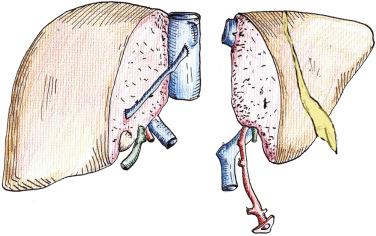
No standard operative technique yet exists for such splitting of livers; each center has developed its own technique, with subtle variations. The majority of these techniques involve dividing the liver in its midplane, thereby generating two grafts consisting of the anatomical right lobe (segments V, VI, VII, VIII) and the anatomical left lobe (segments I, II, III, IV). The middle hepatic vein and left hepatic vein are preserved with the left lobe graft, as are the main trunks of the hepatic arterial and portal venous systems.
The donor operation begins with a careful examination of the liver to assess quality, size, and anatomy. An intraoperative cholangiogram is an easy test to obtain that can give useful information about the anatomy of the biliary tree. The right lobe is not mobilized, and all short hepatic veins draining the posterior aspect of the right lobe into the inferior vena cava (IVC) are thus preserved. The left lobe (including the caudate lobe) is completely mobilized away from the underlying IVC. The confluence of the left and middle hepatic veins is encircled with an umbilical tape. The plan is to preserve the IVC with the right lobe graft and the middle hepatic vein with the left lobe graft. By preserving the donor IVC with the right lobe, all short hepatic veins (small and large) draining the right lobe are kept intact. Also, major hepatic vein tributaries to the middle hepatic vein tributaries can be reconstructed on the back table in cold preservative solution. Doing so maximizes outflow from the right lobe, minimizes warm ischemic time, and simplifies implantation of the right lobe.
The porta hepatis is then carefully examined to evaluate the hepatic arterial anatomy. Regarding the dissection in the porta, our preference has been to leave the full length of the main vascular structures intact with the left lobe (i.e., the common hepatic artery with the celiac trunk and the main portal vein, see Fig. 53-2 ). The right lobe then retains just the right-sided vascular structures: the right hepatic artery and the right portal vein. The right-sided hilar structures are usually larger than the left-sided vascular structures. Therefore leaving the main vessels intact with the left lobe makes that transplant easier. The inflow blood supply to the right lobe (arterial and portal) is isolated. The intraoperative cholangiogram can help guide the biliary dissection by giving valuable information regarding the biliary anatomy. Given the long extrahepatic course of the left hepatic duct, the common bile duct is maintained with the right lobe graft; the point of transection of the bile duct is at the junction of the left hepatic duct with the common hepatic duct so that there is a greater chance of having a single bile duct orifice to reconstruct on both the right and left lobe grafts. It is useful to divide the common bile duct just above the duodenum and pass a biliary probe through the cut end proximally to help decide on the exact site for transection of the biliary system between the two lobes.
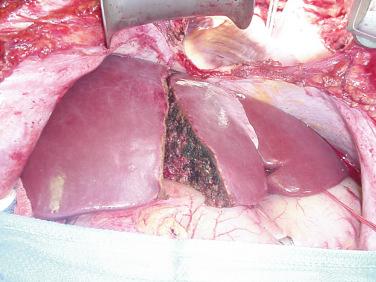
The final step is transection of the hepatic parenchyma itself. The transection plane should stay to the right of the middle hepatic vein, so that this vein is retained with the left lobe ( Fig. 53-3 ). The transection can be performed in situ, using a device such as a Cavitron Ultrasonic Surgical Aspirator (CUSA), or ex situ after the liver is removed. Our preference is in situ splitting, which has several advantages over the ex situ technique. First, it decreases the total cold ischemic time. Performing the split on the back table could add up to 2 to 3 hours of cold ischemia. Also, there is likely some warming of the liver on the back table, even if the split is being performed in a cold ice bath of preservative solution. Even a warming of the liver by a few degrees may have a negative impact on the outcome. Performing the split in situ also has other advantages. Significantly less bleeding occurs when the organs are reperfused. The two liver grafts can be assessed in the donor immediately after parenchymal transection and before vascular interruption, to ensure no significant ischemia at the borders of the cut edge of the liver ( Fig. 53-4 ). Previous SLT series have shown superior results with in situ versus ex situ liver splits. For all these reasons we feel that the actual splitting of the donor liver should be performed in situ. Disadvantages of the in situ procedure include the time added to the procurement during the parenchymal transection and the potential for hemodynamic instability in the donor if there is any significant bleeding. This can affect not only the liver itself but also the other organs that are being removed during the procurement. Once the transection is complete, the liver and other abdominal organs are flushed with cold preservative solution as usual and the liver is removed. On the back table the previously isolated vasculature to the right lobe is divided to completely separate the two grafts. Any important hepatic venous tributaries from the right lobe draining into the middle hepatic vein (segment V or VIII) that had been divided during the parenchymal transection can now be reconstructed. This can be done by using vascular conduit from the deceased donor and anastomosing the cut surface veins to the preserved donor IVC using vascular conduit (such as iliac vein) from the deceased donor ( Fig. 53-5 ).
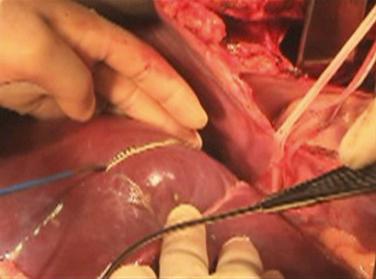
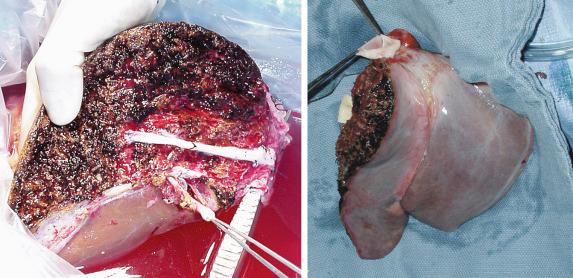
The preceding description is our preferred technique, but several variations have been described. Two important ones include the cava splitting method and the middle hepatic vein splitting technique. In the cava splitting method the left lobe is not mobilized from the IVC. Rather the cava is split down the middle on the back table, thus preserving a portion of it as a patch with both the right and left lobes. This technique has the advantage of preserving all of the outflow of the left lobe, including the short hepatic veins. The middle hepatic vein splitting technique involves ex situ transection of the hepatic parenchyma with division of the middle hepatic vein longitudinally along its length. This can then be reconstructed in both the right and left lobe grafts using a vein patch, hence preserving the middle hepatic vein with both lobes and maximizing outflow for both grafts.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here