Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Soft tissue is loosely defined as the complex of nonepithelial extraskeletal structures of the body exclusive of the supportive tissue of the various organs and the hematopoietic/lymphoid tissue. It is composed of fibrous (connective) tissue, adipose tissue, skeletal muscle, blood and lymph vessels, and peripheral nervous system.
Fibrous tissue consists primarily of fibroblasts and an extracellular matrix that contains fibrillary structures (collagen and elastin) and nonfibrillary extracellular matrix (“ground substance”). Fibrous tissue is classified according to its texture into loose (most locations) and dense (tendons, aponeuroses, and ligaments). Fibroblasts are responsible for the production of the various extracellular materials, including the many types of collagen. Their shape varies from spindle (especially when stretched along bundles of collagen fibers) to stellate (in myxoid areas). Immunohistochemically, they stain for actin, including smooth muscle actin (SMA), but are negative for h-caldesmon and often desmin as well. Fibrocytes represent the quiescent stage of fibroblasts. Myofibroblasts are modified fibroblasts that show features intermediate between fibroblasts and smooth muscle cells.
Adipose tissue is divided into two major types: white fat , mainly located in the subcutaneous tissue, mediastinum, abdomen, and retroperitoneum; and brown fat , which is concentrated in the interscapular region, neck, mediastinum, axillae, and retroperitoneum (especially perirenal region). Brown fat, whose main function is heat production, is much more conspicuous in infants and children. White fat consists of round or oval cells having most of the cytoplasm occupied by a single large lipid droplet that pushes the crescent-shaped nucleus to the periphery. Brown fat cells are smaller, with an acidophilic multivacuolated cytoplasm and a centrally located nucleus showing fine indentations; mitochondria are numerous at the ultrastructural level.
Skeletal muscle is mainly derived from within myotomes through the formation of myoblasts and eventually of myotubes (muscle fibers). The most distinguishing feature of these fibers is the presence of myofibrils, which are composed of two types of microfilament: thin (made of actin) and thick (made of myosin). The periodic arrangement and interdigitation of thin and thick filaments results in the cross-banding seen at the light microscopic level. The I (isotropic) band is made only of thin filaments, the adjacent A (anisotropic) band is a zone of overlapping thin and thick filaments, and the H band is made up only of thick myofilaments. The I band is divided in its center by the Z line or disc, which is thought to serve as an attachment site for the sarcomere , the repeating individual unit of the muscle fiber.
Vessels are divided into blood vessels and lymph vessels. Blood vessels are further subdivided into arterial and venous compartments joined by a network of capillaries. Blood vessels have two major cell types: endothelial cells (located toward the lumen) and a closely related group of cells including pericytes, smooth muscle cells, and glomus cells (located toward the outside). Ultrastructurally, endothelial cells exhibit numerous pinocytotic vesicles, cytoplasmic microfilaments, specialized cell junctions, microvilli, continuous basal lamina, and—most important—the Weibel–Palade body, a membrane-bound organelle thought to be specific to this cell type and shown to contain the von Willebrand factor (factor VIII-related antigen).
Immunohistochemically, endothelial cells express a number of antigens including factor VIII-related antigen (FVIII-RA), CD34, CD31, FLI-1, and ERG. CD31 is both highly sensitive and specific, although histiocytes and plasma cells are also positive. FVIII-RA is also very specific (the only other positive cell type being the megakaryocyte), but the labile nature of this antigen and its tendency to diffuse out in the tissues limit its utility. FLI-1 (a nuclear transcription factor involved in the pathogenesis of Ewing sarcoma/primitive neuroectodermal tumor [PNET]) is also a sensitive vascular marker. Perhaps most useful is ERG, given its strong nuclear positivity in virtually all benign, intermediate, and malignant vascular tumors, although it too is not completely specific.
The cells of the pericyte–smooth muscle–glomus family are characterized ultrastructurally by cytoplasmic microfilaments exhibiting focal condensations, numerous pinocytotic vesicles, and a thick continuous basal lamina. Immunohistochemically, they show reactivity for actin and myosin; positivity for desmin is largely restricted to smooth muscle cells.
Lymph vessels are lined by endothelial cells exhibiting a much weaker staining for FVIII-RA than endothelial cells from blood vessels. They also stain for Lyve-1 and podoplanin (D2-40).
Peripheral nerves are formed by axons, Schwann cells, perineurial cells, and fibroblasts. Most of the fibroblasts are located in the epineurium , which is the outer sheath of fully developed nerves. Each nerve fascicle is surrounded by the perineurium , a structure continuous with the pia arachnoid of the central nervous system (CNS); unlike Schwann cells, perineurial cells are immunoreactive for epithelial membrane antigen (EMA) and GLUT-1 and negative for S-100 protein. Schwann cells look somewhat similar to fibroblasts at the light microscopic level but are easily distinguished from them immunohistochemically because of their strong immunoreactivity for S-100 protein and ultrastructurally by an intimate relationship to axons (with the formation of mesoaxons) and the presence of a continuous basal lamina that coats the surface of the cell facing the endoneurium.
Soft tissue involvement by infectious processes usually is the result of direct extension from cutaneous, visceral, or osseous foci or the complication of trauma or surgery. Rarely, the process has a hematogenous source.
The severity of the inflammatory reaction and the type of tissue response observed pathologically depend on the type, dose, and virulence of the infecting organism, the resistance of the host tissues, the presence or absence of necrotic tissue, hematoma, or foreign body, and the anatomic features of the infected area.
Clinical types of infectious processes such as hemolytic streptococcal gangrene and necrotizing fasciitis must be diagnosed by clinical appearance and bacteriologic study. N ecrotizing fasciitis is accompanied by severe systemic toxicity and is usually caused by group A streptococci. All of the pyogenic and necrotizing infections result in acute inflammatory tissue reactions that are indistinguishable microscopically. Granulomatous inflammation of soft tissue can be caused by tuberculosis , atypical mycobacteriosis , actinomycosis , blastomycosis , coccidioidomycosis , sporotrichosis , cryptococcosis , and dirofilariasis . A proper search for microorganisms should be made with appropriate special stains and cultures.
Hematoma , if deep and encysted, can simulate a sarcoma both clinically and radiographically. They occur most commonly in and around the tensor fasciae latae and have been variously referred to as ancient hematoma, calcifying myonecrosis, chronic expanding hematoma, and post-traumatic cyst of soft tissues.
Soft tissue tumors constitute a large and heterogeneous group of neoplasms. This chapter deals primarily with tumors located in the somatic soft tissues; it excludes those arising from the soft tissues of the mediastinum, retroperitoneum, and visceral organs and those primarily involving the dermis, such as Kaposi sarcoma and dermatofibrosarcoma protuberans (DFSP).
Traditionally, soft tissue sarcomas have been classified according to a histogenetic concept (e.g., fibrosarcoma as a tumor arising from fibroblasts, osteosarcoma as a tumor arising from osteoblasts). However, it seems clear that most if not all sarcomas arise from primitive multipotential mesenchymal cells, which in the course of neoplastic transformation undergo differentiation along one or more lines. The acceptance of this alternative scheme does not require a change in terminology: a liposarcoma remains as such but is now viewed not as a tumor arising from a lipoblast but as a tumor exhibiting lipoblastic differentiation.
A definite relationship exists between soft tissue tumor type and the age of presentation. For instance, embryonal rhabdomyosarcoma is typically a tumor of infants and children, synovial sarcoma mainly affects adolescents and young adults, and liposarcomas and undifferentiated pleomorphic sarcomas (UPSs) are usually seen in middle-aged and elderly patients. Most soft tissue sarcomas are solitary; synchronous or metachronous multiple sarcomas are exceedingly rare.
For any large soft tissue tumor in which the possibility of malignancy exists, the proper initial diagnostic procedure is to obtain material through excisional or incisional biopsy, core needle biopsy, or percutaneous fine-needle aspiration. The latter technique is being used with increasing frequency in the United States, with rates of accuracy equivalent to those obtainable with frozen section. Excisional or incisional biopsy has the advantage of providing more tissue for diagnosis but, given the risk of bleeding and infection, should be performed only when a definitive diagnosis cannot be made by less invasive techniques. Importantly, site of biopsy or aspiration should be excised in continuity with the tumor at the time of definitive surgery.
Performance of frozen sections is useful in determining the type of neoplasm, the degree of malignancy, and the adequacy of surgical margins, but margins are often best assessed grossly at the time the specimen comes to surgical pathology, often done most effectively when the surgeon and pathologist examine the specimen simultaneously.
Light microscopic evaluation of hematoxylin–eosin-stained sections remains the standard technique for the initial diagnostic approach to these tumors and is sufficient in the majority of the cases. However, a significant percentage of cases require ancillary diagnostic techniques including immunohistochemistry, molecular genetic methods, and rarely electron microscopy. Occasionally, histochemical stains may be used (e.g., PAS for assessing for intracytoplasmic crystals in alveolar soft part sarcoma), but many of these stains have been supplanted by other techniques.
The systematic use of cytogenetics has shown the existence of nonrandom chromosomal alterations (mainly translocations) in association with many types of soft tissue tumor. The findings have validated the morphologic approach to classification of soft tissue tumors, helped to refine the boundaries of some entities (such as broadening morphology and location of desmoplastic small cell tumor, merging of spindle cell lipoma with pleomorphic lipoma, and merging of round cell liposarcoma with myxoid liposarcoma), and offered insight into the genesis of the tumors. Furthermore, the molecular alterations (gene fusions) that result from the chromosomal translocations can now be readily demonstrated in routine paraffin-embedded tissues by reverse transcription-polymerase chain reaction (RT-PCR) or fluorescence in situ hybridization (FISH). Such studies can be extremely helpful in the diagnosis of these tumors, especially in small biopsies, tumors with unusual morphology, or tumors in unusual sites. A break-apart FISH probe for EWSR1 is particularly helpful since this gene is implicated in many different soft tissue tumor types, including Ewing sarcoma/PNET, angiomatoid fibrous histiocytoma, extraskeletal myxoid chondrosarcoma, myxoid liposarcoma, clear cell sarcoma, and desmoplastic small cell tumor. Similarly, a break-apart FISH probe for FUS can aid in the diagnosis of low-grade fibromyxoid sarcoma (LGFMS), myxoid liposarcoma, and angiomatoid fibrous histiocytoma. FISH may be a particularly useful technique when evaluating cytologic preparations. As in other fields of oncology, gene expression profiling has significantly contributed to our understanding of the molecular underpinnings of many types of soft tissue tumors. The specific applications of these various methods are described under the respective tumor types.
Some degree of microscopic grading of soft tissue tumors is already built into the conventional microscopic classification of these tumors. For example, DFSP is by definition a low-grade neoplasm, whereas all alveolar rhabdomyosarcomas are high-grade tumors. In addition, several attempts have been made to establish general guidelines for the grading of soft tissue sarcomas independent of their histologic type. The number of grades has varied including two (low grade and high grade), three (I, II, and III, or low grade, intermediate grade, and high grade), and four (I, II, III, and IV) grade systems.
The criteria used to grade sarcomas have included degree of cellularity, pleomorphism, mitotic activity, and necrosis, and these features have been found to be of definite prognostic value for both adult and pediatric soft tissue tumors. However, there are some limitations to grading, including the inherently subjective nature of the evaluation, limited sampling in biopsy material, and the confounding effect of preoperative therapy. Despite these limitations, grading (within the various histotypes and whenever applicable) remains one of the most powerful and inexpensive ways of assessing prognosis in a soft tissue sarcoma. The system devised by the French Federation of Cancer Centers Sarcoma Groups (also known as the FNCLCC grading system) is a three-grade system that is most widely used to grade sarcomas and is based on the evaluation of three separate parameters: tumor differentiation, mitotic rate, and extent of tumor necrosis.
The most widely used staging system is that proposed by the American Joint Committee on Cancer (AJCC), which is largely based on the TNM system, in that it uses the size of the primary tumor (T), the status of lymph nodes (N), the presence of distant metastasis (M), and the tumor's histologic grade (G) ( Tables 41.1–41.4 ).
| HISTOLOGIC TYPE | DIFFERENTIATION SCORE |
|---|---|
| Atypical lipomatous tumor | 1 |
| Well-differentiated leiomyosarcoma | 1 |
| Myxoid liposarcoma | 2 |
| Conventional leiomyosarcoma | 2 |
| Conventional MPNST | 2 |
| Myxofibrosarcoma | 2 |
| Myxoid chondrosarcoma | 2 |
| Conventional angiosarcoma | 2 |
| Round cell liposarcoma | 3 |
| Pleomorphic liposarcoma | 3 |
| Dedifferentiated liposarcoma | 3 |
| Rhabdomyosarcoma | 3 |
| Pleomorphic leiomyosarcoma | 3 |
| Poorly differentiated/epithelioid angiosarcoma | 3 |
| Poorly differentiated MPNST | 3 |
| Synovial sarcoma | 3 |
| Extraskeletal osteosarcoma | 3 |
| Ewing family of tumors | 3 |
| Mesenchymal chondrosarcoma | 3 |
| Clear cell sarcoma | 3 |
| Epithelioid sarcoma | 3 |
| Alveolar soft part sarcoma | 3 |
| Undifferentiated pleomorphic sarcoma | 3 |
| PARAMETER | DEFINITION | |
|---|---|---|
| Tumor differentiation | Score 1: | Closely resembles normal adult mesenchymal tissue; potentially difficult to distinguish from counterpart benign tumor |
| Score 2: | Sarcomas for which histologic type is certain (e.g., myxoid liposarcoma) | |
| Score 3: | Embryonal and undifferentiated sarcoma, synovial sarcoma, sarcomas of uncertain type | |
| Mitotic count | Score 1: | 0–9/10 HPF |
| Score 2: | 10–19/10 HPF | |
| Score 3: | >19/10 HPF | |
| Tumor necrosis | Score 0: | No necrosis |
| Score 1: | <50% tumor necrosis | |
| Score 2: | ≥50% tumor necrosis | |
| Histologic grade | Grade 1: | Total score 2, 3 |
| Grade 2: | Total score 4, 5 | |
| Grade 3: | Total score 6, 7, 8 |
| Definition of Primary Tumor (T) | |
| T Category | T Criteria |
| TX | Primary tumor cannot be assessed |
| T0 | No evidence of primary tumor |
| T1 | Tumor 5 cm or less in greatest dimension |
| T2 | Tumor more than 5 cm and less than or equal to 10 cm in greatest dimension |
| T3 | Tumor more than 10 cm and less than or equal to 15 cm in greatest dimension |
| T4 | Tumor more than 15 cm in greatest dimension |
| Definition of Regional Lymph Node (N) | |
| N Category | N Criteria |
| N0 | No regional lymph node metastasis or unknown lymph node status |
| N1 | Regional lymph node metastasis |
| Definition of Distant Metastasis (M) | |
| M Category | M Criteria |
| M0 | No distant metastasis |
| M1 | Distant metastasis |
| Definition of Grade (G) FNCLCC Histologic Grade—see Histologic Grade (G) |
|
| G | G Definition |
| GX | Grade cannot be assessed |
| G1 | Total differentiation, mitotic count and necrosis score of 2 or 3 |
| G2 | Total differentiation, mitotic count and necrosis score of 4 or 5 |
| G3 | Total differentiation, mitotic count and necrosis score of 6, 7, or 8 |
* For tumors arising in the trunk, extremities, or retroperitoneum.
| WHEN T IS… | AND N IS… | AND M IS… | AND GRADE IS… | THEN THE STAGE GROUP IS… |
|---|---|---|---|---|
| T1 | N0 | M0 | G1, GX | IA |
| T2, T3, T4 | N0 | M0 | G1, GX | IB |
| T1 | N0 | M0 | G2, G3 | II |
| T2 | N0 | M0 | G2,G3 | IIIA |
| T3, T4 | N0 | M0 | G2, G3 | IIIB |
| Any T | N1 | M0 | Any G | IV |
| Any T | Any N | M1 | Any G | IV |
The prognosis of soft tissue tumors depends on a variety of parameters, many of which are interrelated.
Tumor size. There is a definite relationship between tumor size and outcome. This is true for practically all tumor types in which this parameter has been analyzed.
Depth. Superficially located tumors (dermis and subcutaneous tissue) have a much better prognosis than deep-seated ones (intermuscular or intramuscular, retroperitoneal) of similar microscopic type. The difference is largely due to the fact that superficial lesions tend to be considerably smaller at the time of excision and are generally more amenable to complete excision.
Location. Tumors of the retroperitoneum do much worse than microscopically similar lesions located in the extremities. Among the latter, local recurrence has been found to be more frequent with those of the upper extremity than those of the lower extremity.
Microscopic type. Some soft tissue neoplasms (such as atypical lipomatous tumors [ALTs]) are low-grade lesions with no capacity to metastasize, whereas other neoplasms of similar cell type (such as pleomorphic liposarcoma) are highly aggressive and prone to spread distantly.
Vascular invasion. This has been shown to be the strongest predictor of distant metastases in several series.
Surgical margins. Not surprisingly, adequacy of surgical margins is statistically associated with local relapse. The relationship between local recurrence and subsequent risk of metastasis is less clear.
Histologic grade. As already indicated, a relationship has been found between various microscopic grading systems and outcome (see discussion on grading).
Clinical stage. As for most other tumors, this determination—which incorporates several of the previously mentioned parameters, as well as the presence or absence of metastases—is the most powerful prognostic determinator.
Other factors. A number of other prognostic factors have been implicated including DNA ploidy, cell proliferation (MIB-1), and specific genetic alterations such as mutations of TP53 . Along similar lines, claims have been made of a relationship between the type of gene fusion in the sarcomas associated with chromosomal translocations and prognosis (as in alveolar rhabdomyosarcoma and synovial sarcoma; these are discussed in the respective sections).
Soft tissue tumors that are relatively small and/or clearly benign on clinical grounds (such as superficially located lipomas, schwannomas, hemangiomas, and tenosynovial giant cell tumors) can be removed without prior manipulation, but in most others the excision should be preceded by a biopsy or fine-needle aspiration. A few tumors (such as schwannomas) can be safely enucleated, but for most others—even if benign—a rim of uninvolved normal tissue should be excised in continuity with the neoplasm to prevent recurrence. Many soft tissue sarcomas, such as fibrosarcoma, myxoid liposarcoma, and leiomyosarcoma, may appear grossly encapsulated, but microscopic examination will often show tumor cells beyond the apparent capsule; therefore enucleation will usually fail. A wide local excision is particularly important for infiltrative lesions such as desmoid-type fibromatosis and DFSP.
Full-fledged soft tissue sarcomas in children are currently treated, for the most part, by a combination of surgery, radiation therapy, and multidrug chemotherapy, with results that are vastly superior to those obtained in the prechemotherapy era.
The treatment of high-grade soft tissue sarcomas in adults has undergone radical changes. It has been thought for many years that amputation or disarticulation offered the best chances of cure for sarcomas involving an extremity. However, many studies have shown wide local excision with adjuvant therapy to have survival rates similar to those for amputation. Details of therapeutic options go beyond this discussion, but superb reviews on this topic can be found in the recent literature. Similar to other types of neoplasms, increased emphasis has been placed on identifying targeted therapies for patients with recurrent or metastatic soft tissue sarcoma.
The pathogenesis of most soft tissue tumors remains unknown. Environmental, oncogenic, immunologic, and genetic factors have all been implicated and, in fact, several of these factors may be involved in the development of a given tumor.
Trauma is frequently implicated in the development of sarcomas, but trauma often seems to merely call attention to the presence of a tumor which antedated the trauma. Other environmental factors implicated in the pathogenesis of sarcomas include phenoxyacetic acid herbicides, vinyl chloride (which is clearly associated with hepatic angiosarcoma) and radiation exposure. Although dioxin, a contaminant of the defoliant Agent Orange, which was used in Vietnam, has been implicated in sarcomagenesis, case control studies have found no increased risk of sarcomas.
Oncogenic viruses play a role in the development of a small group of sarcomas. The human herpesvirus 8 (HHV8) is clearly the causative agent of Kaposi sarcoma. Epstein-Barr virus (EBV) also seems to play a central role in the development of smooth muscle tumors in patients with immunodeficiencies or immunosuppression.
A number of genetic diseases are associated with soft tissue tumors, including neurofibromatosis type 1 (malignant peripheral nerve sheath tumors [MPNSTs]), familial adenomatous polyposis/Gardner syndrome (Gardner-associated fibroma and fibromatosis), Li-Fraumeni syndrome (germline mutations of TP53 associated with a number of sarcoma types), Carney complex (psammomatous melanotic schwannoma, cutaneous myxoma), and tuberous sclerosis (perivascular epithelioid cell tumor/PEComa).
It should be noted that most soft tissue sarcomas arise de novo rather than from malignant transformation of a preexisting benign tumor, an exception being transformation of neurofibroma into an MPNST. In most cases in which a benign tumor has been thought to become malignant, review of the original material shows the tumor to be malignant from its inception.
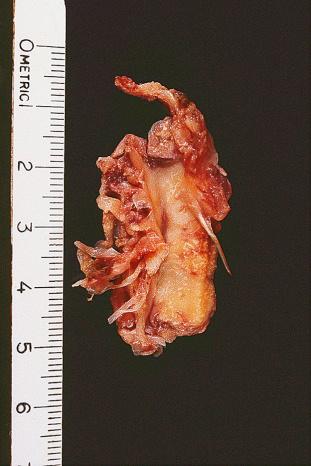
Calcifying aponeurotic fibroma is a distinctive lesion originally described as juvenile aponeurotic fibroma, typically presenting as a soft tissue mass in the hand or wrist of a child or adolescent (peak 8–14 years of age) but sometimes also occurring in the proximal extremities or trunk. At surgery, it may appear as a nodule or as an ill-defined infiltrating mass in the subcutaneous tissue or attached to a tendon ( Fig. 41.1 ). Sometimes, foci of calcification may be detected on gross inspection.
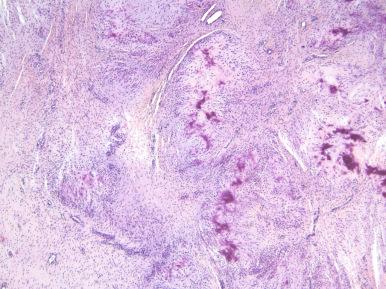
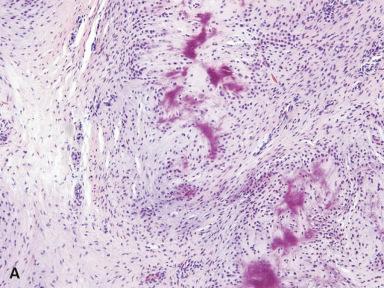
Microscopically, the lesion is characterized by an infiltrative proliferation of cells associated with spotty calcification ( Figs. 41.2 and 41.3 ). Infiltration of fat and skeletal muscle is often seen at the periphery. Mitoses are scarce, and atypical cytologic features are absent. Scattered osteoclast-like giant cells are frequently seen. Cellularity varies from region to region; the cells are plump with ovoid nuclei and indistinct cytoplasm separated by dense fibrous stroma. There may be a cartwheel, linear, or whorled growth pattern. Calcifications vary from fine granules to larger areas with cells radiating out from them in linear columns. Immunohistochemically, the proliferating cells stain variably for actins, CD68, S-100 protein, and even CD99.
Calcifying aponeurotic fibroma can be confused with rheumatoid nodule, schwannoma, soft tissue chondroma, and fibromatosis, including infantile fibromatosis. Local recurrence is common, especially in young children (less than 5 years of age). However, distant metastases do not occur.
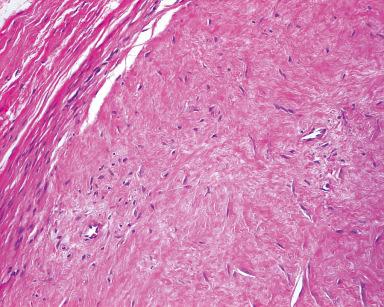
Fibroma of tendon sheath is a well-circumscribed, often lobulated tumor found attached to tendon or tendon sheath. Microscopically, it is composed of dense fibrous tissue containing spindle and sometimes stellate mesenchymal cells deposited in a collagenous stroma ( Fig. 41.4 ), although myxoid foci may be seen. Frequently, there are dilated or slit-like channels, particularly at the periphery of lobules. Occasionally, a component of bizarre tumor cells unaccompanied by mitoses is seen (so-called pleomorphic fibroma of tendon sheath). Ultrastructurally, most of the cells have features of myofibroblasts and, by immunohistochemistry, the cells express actins but not desmin. The translocation t(2;11)(q31–32;q12) has been identified in rare cases. More recently, some cases of fibroma of tendon sheath, particularly those that are highly cellular, have been shown to harbor rearrangements of USP6 , suggesting a relationship to nodular fasciitis in these cases. Interestingly, however, these alterations were not identified in any classic cases of fibroma of tendon sheath. This suggests that lesions previously identified as cellular fibromas of tendon sheath are probably underrecognized examples of tenosynovial fasciitis. The lesion is benign and only very rarely recurs.
Collagenous fibroma (desmoplastic fibroblastoma) is a benign lesion usually centered in the subcutaneous tissue and with a wide anatomic distribution, usually arising in adults. A subset of these lesions may occur in deeper locations including intramuscular examples. The most distinctive microscopic feature is the presence of stellate fibroblasts (together with ordinary spindle-shaped fibroblasts) separated by a collagenous matrix with or without myxoid features ( Fig. 41.5 ). By immunohistochemistry, most cases show staining for SMA but are negative for desmin, consistent with myofibroblastic differentiation. Some have suggested this lesion could represent the end stage of a reactive process. However, aberrations of 11q12 suggest a neoplastic process, as well as a possible link to fibroma of tendon sheath. Interestingly, a recent study found consistent expression of FOSL1 in collagenous fibroma, while none of the cases of fibroma of tendon sheath showed expression of this antigen, suggesting these are distinct lesions.
Nuchal-type fibroma is a benign lesion typically found in the posterior aspect of the neck and characterized microscopically by hypocellular bundles of thick collagen fibers, with entrapped adipose tissue and traumatic neuroma-like structures. A high percentage of the patients are diabetic. However, up to 30% occur in other soft tissue sites (buttocks, extremities, among others), and hence the reason the term nuchal-type fibroma is preferred over nuchal fibroma.
Gardner-associated fibroma has overlapping features with nuchal-type fibroma but seems distinct given its close association with Gardner syndrome/familial adenomatous polyposis. These patients tend to be younger than those with nuchal-type fibroma, with most patients presenting in the first decade of life. Most arise in the back and paraspinal region, and histologically there is close resemblance to nuchal-type fibroma, although entrapped nerves are not conspicuous. The majority of cells express CD34, CD99, and nuclear β-catenin staining. This lesion is benign, but it may recur if incompletely excised, and rare cases transform into a full-blown fibromatosis. This process is probably distinct from nuchal fibrocartilaginous pseudotumor , which is characteristically located in the deep soft tissue overlying the posterior aspect of the lower cervical vertebrae and thought to be due to fibrocartilaginous metaplasia of the nuchal ligament, probably trauma induced.
Superficial acral fibromyxoma (also referred to as digital fibromyxoma ) is a soft tissue tumor with a predilection for the fingers and toes and composed of spindle- to stellate-shaped usually CD34-positive tumor cells arranged in a nondescript, storiform, or fascicular pattern of growth, embedded in a myxoid, fibromyxoid, or collagenous stroma ( Fig. 41.6 ). A cellular variant has also been described. Some believe this lesion may be related to cellular digital fibroma , but a recent study detected frequent RB-1 loss or deletions, suggesting a relationship to other members of the RB-1 deleted tumor family (spindle cell lipoma, mammary-type myofibroblastoma, and cellular angiofibroma). The tumor arises in patients of all ages, with a 2 : 1 male to female ratio. Although most are cured by local excision, up to 25% recur.
Angiofibroma of soft tissue is a recently described fibrovascular tumor which most commonly arises in the soft tissues of the extremities. The tumor shows a proliferation of bland spindled cells in a myxocollagenous matrix with a distinct network of thin, finely branched blood vessels. The cells variably express EMA, desmin, SMA, and CD34, but they characteristically show aberrations of the NCOA2 gene on 8q13, which can be detected by FISH. The lesion is benign and only rarely recurs following local excision.
Fibrous hamartoma of infancy is a tumorlike condition seen almost exclusively during the first 2 years of life and sometimes present at birth. It predominates in boys, and the most common locations are the region of the axilla, shoulder, and upper arm. It is almost always solitary. Grossly, it is poorly circumscribed and composed of white fibrous tissue intermixed with islands of fat.
Microscopically, the distinctive feature of this lesion is an organoid pattern with three distinct types of tissue being present: (1) well-differentiated spindle cells of fibroblastic/myofibroblastic appearance accompanied by deposition of collagen; (2) mature adipose tissue; and (3) immature cellular areas arranged in a whorling pattern and resembling primitive mesenchyme ( Fig. 41.7 ). Very rare cases with overt sarcomatous-appearing foci have been reported, characterized by high cellularity, high nuclear grade, and brisk mitotic activity. Immunoreactivity for actin (and sometimes desmin) is found mainly in the spindle cell areas, suggesting myofibroblastic differentiation. Recently a case of fibrous hamartoma of infancy with an EGFR exon 20 insertion/duplication mutation was reported. Although there may be local recurrence, the clinical course is benign.
Nodular fasciitis is a distinctive pseudosarcomatous myofibroblastic proliferation that is important because of its ability to simulate a malignant process. It can affect patients of all ages but is most prevalent in young adults, with a peak between 20 and 40 years of age.
The most common locations are the upper extremities (particularly the flexor aspect of the forearms), trunk, and neck, but this lesion has been described in virtually every anatomic location. It presents as a rapidly growing mass of short duration, sometimes following known trauma. It usually extends above the fascia into the subcutis, but it may grow beneath it into skeletal muscle, remain within the fascia as a fusiform expansion, or even be centered in the dermis.
Microscopically, the lesion is characterized by a cellular spindle cell growth set in a loosely textured myxoid matrix. Microcystic change, red blood cell extravasation, and lymphocytic infiltration are also often present ( Figs. 41.8 to 41.11 ). A feature of diagnostic significance is the presence of undulating wide bands of collagen lined on the sides by spindle cells. Some areas have a storiform growth pattern. Focal metaplastic bone formation may be present, establishing a link between nodular fasciitis and myositis ossificans. The high cellularity of the lesion and the presence of numerous mitotic figures are responsible for the frequent confusion of this lesion with a sarcoma. However, the constituent cells lack nuclear hyperchromasia, a critical feature in distinguishing a reactive process from a sarcoma.
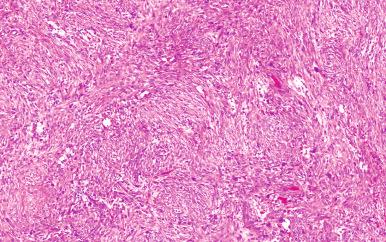
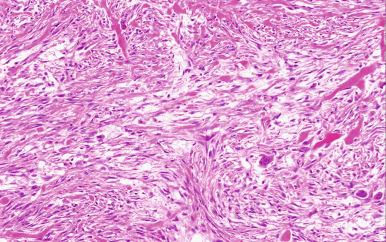
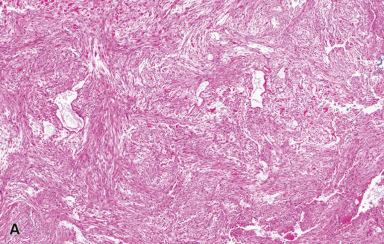
Ultrastructurally and immunohistochemically, many of the proliferating spindle cells have features of myofibroblasts with strong expression of SMA. This lesion is completely benign and often resolves spontaneously. If a lesion originally diagnosed as nodular fasciitis recurs, it is wise to re-review the original pathology since recurrence is so uncommon. Interestingly, there are a number of reports of clonal chromosomal rearrangements involving USP6 on 17p13 and MYH9 on 22q13, which has led some authors to favor the interpretation that it is a neoplastic process.
Cranial fasciitis is a distinct variant of nodular fasciitis generally seen in infants and characterized by involvement of the skull with erosion of the outer table of the cranium. It shows histologic and immunohistochemical features identical to nodular fasciitis, including strong expression of SMA. Birth trauma has been implicated as causative in at least some cases.
Intravascular fasciitis is another morphologic variant of fasciitis in which involvement of the wall and lumen of medium-sized veins and arteries occurs. This is an exaggerated expression of a phenomenon seen frequently in ordinary nodular fasciitis and which constitutes a useful diagnostic sign, that is, the fact that, at the periphery of the lesion, the walls of small to medium-sized vessels are involved by the reactive mesenchymal process.
In proliferative fasciitis the location of the lesion, rapidity of growth, and self-limited nature are the same as those of nodular fasciitis, but the presence of large basophilic cells resembling ganglion cells indicates a link with proliferative myositis (see subsequent discussion) ( Fig. 41.12 ). It usually affects adults, although it can also be seen in children. As in the other conditions described in this section, myofibroblasts are the cells that predominate in this proliferation. It should be mentioned here that the presence of ganglion-like cells in a soft tissue lesion does not guarantee that the condition is of a reactive nature. Indeed, we have seen several cases of soft tissue sarcoma containing similar-appearing cells.
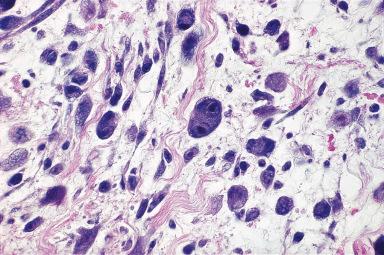
Nodular fasciitis and the variants described previously are characteristically located in the somatic soft tissues, but reactive fasciitis-like lesions with a somewhat different morphologic appearance (even more sarcomatoid) can develop from the stromal tissue of a variety of organs, such as the bladder, prostate, vulva, vagina, and cervix (see respective chapters).
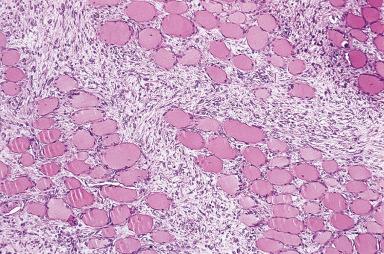
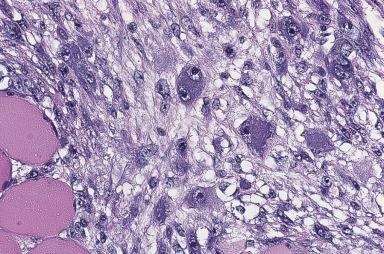
Proliferative myositis can be confused with sarcoma not only clinically but also microscopically. The skeletal muscles of the shoulder, thorax, and thigh are most commonly affected. Most patients are over the age of 45 years, but it can also present in children. Grossly, there is typically induration of muscle with an ill-defined scar-like lesion ( Fig. 41.13 ). Microscopically, a cellular proliferation rich in fibroblasts and myofibroblasts is seen surrounding individual muscle fibers, creating a checkerboard pattern ( Fig. 41.14 ). The hallmark of the lesion is the presence of large basophilic cells with vesicular nuclei and prominent nucleoli, resembling ganglion cells or rhabdomyoblasts ( Fig. 41.15 ). Conservative surgery is curative.

Other pseudoneoplastic myofibroblastic proliferations pathogenetically related to nodular fasciitis and representing an exaggerated reaction to injury include proliferative funiculitis (involving the spermatic cord and probably secondary to ischemia or torsion) and atypical decubital fibroplasia , otherwise known as ischemic fasciitis . This is a myofibroblastic proliferation that predominantly involves soft tissue overlying bony prominences, often occurring in debilitated or immobilized patients. Most of those affected are elderly (peak incidence in eighth or ninth decade), but this lesion rarely affects young patients as well. The soft tissues of the shoulder, chest wall, and hip or sacrococcygeal region are most commonly affected. Microscopically, a zonal pattern best appreciated at low magnification is seen with a central liquefactive zone surrounded by proliferating vessels and myofibroblasts, some of which have smudgy chromatin or resemble the ganglion-like cells in proliferative fasciitis/myositis ( Fig. 41.16 ). The lesion is benign but can recur if the patient remains debilitated.
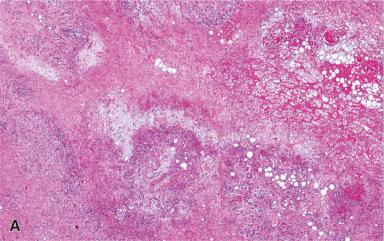
Myositis ossificans and related conditions such as fibro-osseous pseudotumor of the digits are located in the soft tissue and are pathogenetically and histologically linked to the previous entities ( Fig. 41.17 ). They are more fully discussed in Chapter 40 .
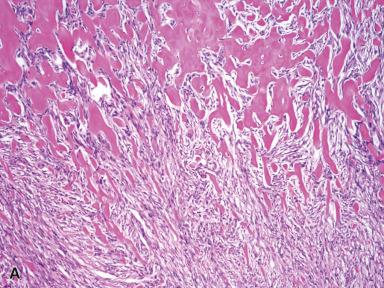
This is a distinctive benign neoplasm that arises in lymph nodes, most commonly in the groin, usually in middle-aged adults. Microscopically, the tumor is well circumscribed with residual nodal tissue at the periphery and is composed of bland spindle cells arranged in short fascicles with vague nuclear palisading. The most distinctive feature is the presence of thick densely eosinophilic collagen (so-called amianthoid fibers) that is variably found throughout the tumor ( Fig. 41.18 ). The cells have myofibroblastic features, as they express actins but not desmin. Recently, mutations of CTNNB1 (β-catenin gene) have been described. The tumor does not recur or metastasize.
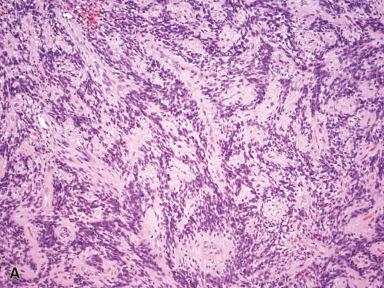
Mammary-type myofibroblastoma arises in both males and females with a slight male predominance. Although initially described to occur in the breast, the inguinal region is actually the most common location (45% of cases), followed by the lower extremities, trunk, breast, and intraabdominal/retroperitoneum, although many extra-mammary sites have been described. Patients present with a slowly growing, well-circumscribed mass. The tumor is composed of a haphazard arrangement of slender spindle cells separated by collagen bundles ( Fig. 41.19 ). The cells may be arranged in short fascicles or packets, and they have bland chromatin, inconspicuous nucleoli, and occasional intracytoplasmic inclusions. Some cases have extensive amounts of mature adipose tissue, and others have focal thick-walled blood vessels. Extensive hyalinization and myxoid stroma may also be seen. Tumors composed predominantly of epithelioid cells have also been described. By immunohistochemistry, the cells typically coexpress CD34 and desmin, with variable expression of SMA. The tumor consistently shows rearrangements or deletions of 13q14, resulting in loss of Rb-1 expression by immunohistochemistry. Given overlapping morphology with spindle cell lipoma and cellular angiofibroma, and the fact that these all show loss of Rb-1, it is now believed these lesions are a family of closely related tumors. Mammary-type myofibroblastoma is benign and is generally cured by local excision, with very rare local recurrences.
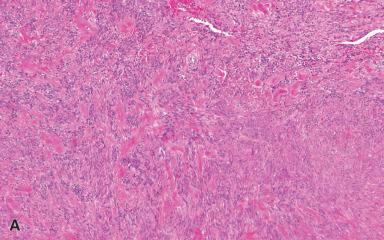
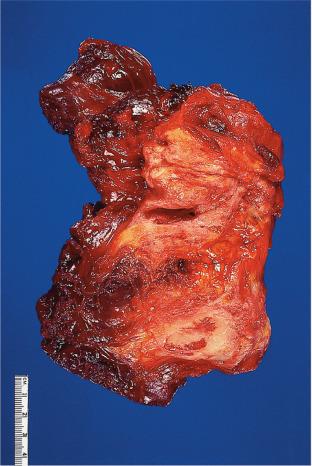
Elastofibroma is a benign, poorly circumscribed process involving almost exclusively the subscapular region of elderly individuals, although isolated cases have been seen in a number of other locations including the gastrointestinal tract. Multicentric and familial cases have also been described. There is often a history of strenuous, repetitive manual labor. At surgery, the lesion is usually found at the apex of the scapula, beneath the rhomboid and latissimus dorsi muscles.
Microscopically, collagen bundles alternate with numerous acidophilic, refractive cylinders often containing a central dense core, both of which stain strongly with elastic stains ( Figs. 41.20 and 41.22 ). Occasionally, adipose tissue is found intermingled with the collagen and elastic fibers, a variation in the theme that has been dignified with the name elastofibrolipoma .
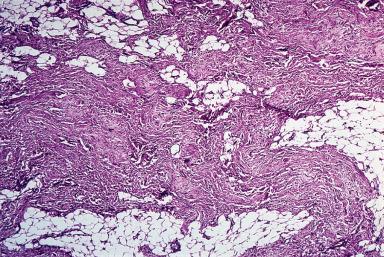
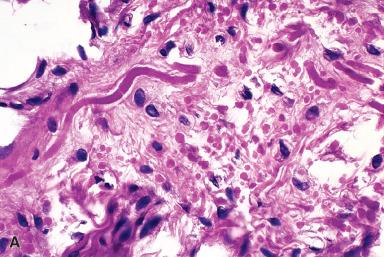
The biochemical composition of the fibers is that of elastin but with an amino acid composition slightly different from that of normal elastic tissue. The collagen deposited in the lesion is a mixture of types I, II, and III; the presence of type II collagen is perplexing because this is normally present only in articular cartilage and some ocular structures. Traditionally, this lesion has not been regarded as a true neoplasm but rather a reactive hyperplasia involving abnormal elastogenesis ; however, several recent reports have found cytogenetic alterations of various types.
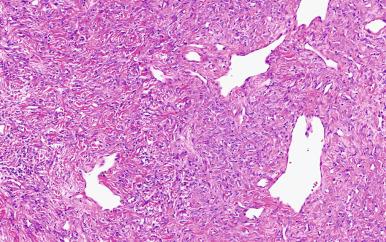
This neoplasm is ubiquitous in soft tissues and has been described in virtually every anatomic site, although it is most common in the pleura. The tumor is composed of fibroblastic spindle cells arranged in a patternless pattern and associated with wire-like collagen and often a staghorn or hemangiopericytoma-like vascular pattern ( Figs. 41.23 and 41.24 ). Other features may include myxoid change, mature adipose tissue, floret-like giant cells (formerly referred to as giant cell angiofibroma), and even dedifferentiation. Although CD34 is virtually always expressed, this marker lacks specificity. More recently, STAT6 has been found to be a more sensitive and specific marker, which is not unexpected given the identification of NAB2-STAT6 gene fusions in a significant subset of these tumors. Although most cases are clinically benign, some are both histologically and clinically malignant, particularly those with areas of high cellularity, necrosis, and greater than four mitotic figures per 10 high power field (HPF).
The generic term desmoid-type fibromatosis was originally proposed by Stout for a group of related conditions having in common the following features:
Proliferation of well-differentiated fibroblasts and myofibroblasts
Infiltrative pattern of growth
Presence of a variable (but usually abundant) amount of collagen between the proliferating cells
Lack of cytologic features of malignancy
Low or absent mitotic activity
Aggressive clinical behavior characterized by repeated local recurrences but lack of capacity to metastasize distantly
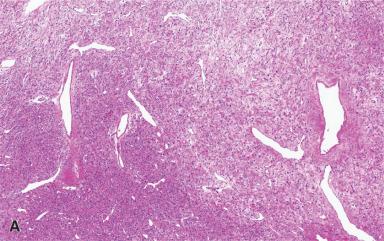
Grossly, these lesions are often large, firm, and white, with ill-defined outlines and an irregularly whorled cut surface ( Fig. 41.25 ). Microscopically, most of the proliferating cells have features of fibroblasts or myofibroblasts ( Figs. 41.26 and 41.27 ). Other light microscopic features commonly encountered in fibromatosis are a perivascular lymphocytic infiltrate located at the advancing edge of the lesion, and thick-walled often ectatic vessels sharply outlined from the surrounding tissue.
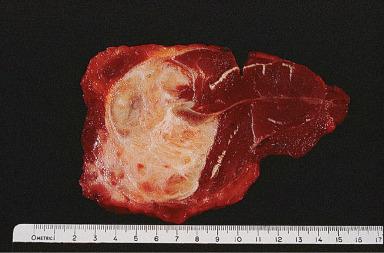
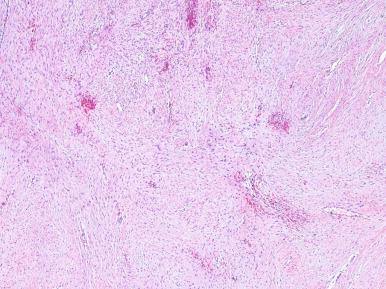
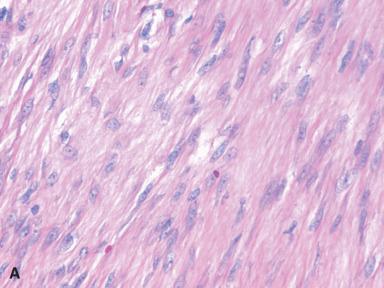
Clonal chromosomal aberrations are present in approximately half of the deep-seated fibromatoses but only in 10% of those located superficially. Trisomies 8 and 20 and loss of 5q material represent the recurring cytogenetic changes. At the molecular level, there are activating mutations of CTNNB1 (encoding β-catenin), leading to frequent nuclear expression of β-catenin. For cases associated with Gardner syndrome, there is germline mutation of the APC gene (5q22), together with loss of heterozygosity at the locus. The end result is similar, since both APC and β-catenin are components of the Wnt signaling pathway. The cells of fibromatosis are also immunoreactive for SMA and desmin, with variable staining for calponin, and estrogen receptor-beta (but not estrogen receptor-alpha or CD34).
Some pathologists add the adjective aggressive to some forms of fibromatosis to emphasize its potential behavior. We do not use the term, since we regard it as redundant, being that all deep-seated fibromatoses are potentially aggressive. Other authors have gone even further and have used well-differentiated fibrosarcoma as a synonym for the histologically more cellular or clinically more aggressive types of fibromatosis. We are opposed to this terminology because the designation of sarcoma endows this lesion in the mind of many surgeons with a metastasizing potential that it simply does not possess. Although some cases are challenging, we always attempt to distinguish between fibromatosis and well-differentiated fibrosarcoma, reserving the latter term for tumors showing atypical cytologic features.
Most soft tissue fibromatoses are in intimate contact with skeletal muscles and their aponeuroses—hence their designation as musculoaponeurotic fibromatosis , although the WHO refers to these lesions as desmoid-type fibromatosis. Although such lesions often arise in the abdominal wall in women during or following pregnancy, in our experience, this lesion is almost as common in men and in other locations, such as the shoulder girdle, head and neck area, and thigh. It can also occur in the mediastinum, pleural cavity (with lung invasion), retroperitoneum, abdominal cavity (see subsequent discussion), and breast, among many other sites.
The treatment of deep fibromatoses should be individualized, and, in fact, a number of modalities can be utilized, including surgery alone or in conjunction with radiation and cytotoxic and noncytotoxic drugs (tamoxifen, Gleevec, and nonsteroidal anti-inflammatory drugs including sulindac). The tumor does have a strong propensity for local recurrence, but some authors have advised against the re-excision of a recurrent lesion that does not appear to be growing. In the study by Gronchi et al., 76% of tumors locally recurred by 10 years. Tumor size was associated with recurrence risk, but surprisingly, microscopic deposits of surgical margins were not predictive of recurrence. Recently, Colombo and colleagues found that sporadic tumors with mutations in codon 45 in the β-catenin gene (CTNNB1) have a greater tendency to locally recur.
Juvenile fibromatosis is an antiquated term that has been applied to fibromatoses occurring in children and adolescents. There is very little either on clinical or microscopic grounds that differentiates fibromatosis in children from that occurring in other age groups. There are, however, three variants of fibromatosis that predominantly occur in childhood that present a distinctive clinicopathologic picture: fibromatosis colli (congenital torticollis), inclusion body fibromatosis (infantile digital fibromatosis), and infantile myofibroma/ myofibromatosis.
Fibromatosis colli (congenital torticollis) is a type of fibromatosis affecting the lower third of the sternocleidomastoid muscle and appearing at birth or shortly thereafter, sometimes bilaterally. It is frequently associated with various congenital anomalies including rib cage anomalies and congenital hip dysplasia. An association between complicated deliveries (particularly breech deliveries) and fibromatosis colli has been established. Although some instances of spontaneous regression have been recorded, this condition usually necessitates resection of the muscle. Microscopically, there is partial replacement of the sternocleidomastoid muscle by a fibroblastic proliferation of varying cellularity admixed with atrophic skeletal muscle fibers.
Inclusion body fibromatosis (infantile digital fibromatosis) is a form of fibromatosis usually restricted to childhood. The typical location is on the exterior surface of the end phalanges of the fingers and toes, but it may also occur outside the digits and at sites such as the oral cavity and breast. The lesions can be solitary or multiple and either present at birth or appear during the first 2 years of life. However, morphologically identical lesions in adults are on record. A distinctive microscopic feature, generally not observed in other forms of fibromatosis, is the presence of peculiar eosinophilic cytoplasmic inclusions, often situated close to the nucleus and separated by a narrow clear zone ( Fig. 41.28 ). A trichrome stain can highlight these inclusions. Ultrastructural findings suggest they represent bundles of microfilaments. The spindle cells stain for SMA, desmin, calponin, and CD99. Inclusion body fibromatosis has a high tendency for local recurrence.
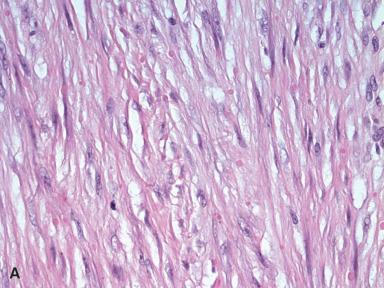
Very rare cases of inclusion body fibromatosis lacking inclusion bodies may be a component (and sometimes the first clinical manifestation) of the syndrome known as terminal osseous dysplasia with pigmentary defects .
Infantile myofibroma/myofibromatosis presents as solitary (myofibroma) or multiple (myofibromatosis) nodules in the skin, soft tissues, or bone, either limited to these sites or associated with internal organ involvement. A large majority of the cases occur before the age of 2 years, and approximately 60% are congenital. However, this lesion can also occur in adults. Solitary forms are more common in males, and multicentric forms are more common in females. A familial incidence has been suggested, and evidence for both an autosomal dominant and recessive pattern of transmission has been proposed. Microscopically, peripheral areas that resemble smooth muscle alternate with hemangiopericytoma-like areas and foci having a more typical fibroblastic appearance ( Fig. 41.29 ). Central necrosis and intravascular growth may be present. Both immunohistochemical and ultrastructural studies suggest the lesion is largely composed of myofibroblasts. However, a whole range of differentiation exists between fibroblasts and fully developed, desmin-positive smooth muscle cells. Indeed, myofibromatosis seems to be a member of a family of tumors showing vessel-related myoid differentiation, which also includes glomangiopericytoma and myopericytoma given that tumors with overlapping features exist. Solitary and multicentric lesions confined to the bone and soft tissues carry an excellent prognosis, with frequent spontaneous regression. The prognosis is far less favorable in patients with multiple visceral lesions.
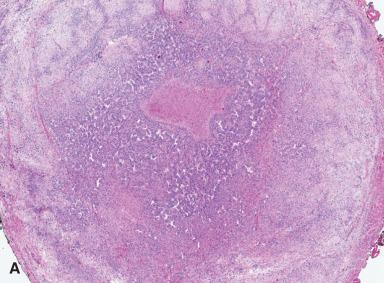
Interestingly, recurrent somatic PDGFRB mutations have been identified in sporadic infantile and solitary adult myofibromas, but these mutations were not identified in cases of angioleiomyoma and myopericytoma. However, recurrent PDGFRB alterations were identified in myopericytomatosis, suggesting a relationship between myopericytomatosis and infantile myofibroma/myofibromatosis. Alterations in PDGFRB also suggest the possible utility of tyrosine kinase inhibitors as a potential therapy for these lesions. A recurrent SRF-RELA fusion was recently found in highly cellular variants of myofibroma and myopericytoma, but cases of typical myofibroma and myopericytoma lack this fusion.
Lipofibromatosis is a minor variation on the theme of infantile fibromatosis, in which a spindle cell component of fibroblastic appearance (mainly located in septa and skeletal muscle) is admixed with mature adipose tissue. This lesion affects children from birth to up to 10 years of age, usually arising in the muscles of the extremities, trunk, or head and neck. Like other forms of fibromatosis, this lesion frequently recurs but does not metastasize.
Recently, a series of locally aggressive lipofibromatosis-like neural tumors with recurrent NTRK1 gene fusions were identified. These lesions occurred in children and young adults with an infiltrative growth pattern similar to that seen in lipofibromatosis, but with variable cytologic atypia and immunoreactivity for S-100 protein and CD34 suggesting neural differentiation.
Juvenile hyaline fibromatosis/infantile systemic hyalinosis is a distinctive group of lesions affecting children and characterized microscopically by cords of spindle cells embedded in an eosinophilic hyalinized matrix within the connective tissue of the skin, oral cavity, articular capsule, and bone. Several studies have found mutations of the capillary morphogenesis genes 2 (CMG2) on 4q21 in both diseases, confirming these are part of a disease spectrum. Inheritance is in an autosomal recessive manner, and there is a distinct predilection for patients of Middle Eastern descent.
The superficial fibromatoses derive their names from their particular location. Penile fibromatosis (Peyronie disease) will be discussed in greater detail elsewhere. Palmar fibromatosis ( Fig. 41.30 ) is also known as Dupuytren contracture and plantar fibromatosis as Ledderhose disease. These conditions occur predominantly in adults but can also be seen in children and adolescents. Contracture of the fingers or toes is the leading clinical manifestation. The lesions can be multiple and bilateral and may coexist in the upper and lower extremities. The plantar form tends to be more localized than its palmar counterpart. Microscopically, both often show a cellular proliferative phase (especially for the plantar lesions), which may lead to a mistaken diagnosis of fibrosarcoma. It is well to remember that fibrosarcoma of the palmar and plantar areas is exceptional and that the differential diagnosis of a cellular spindle cell tumor of the sole is usually between fibromatosis, synovial sarcoma, malignant melanoma, and Kaposi sarcoma. The cells are spindled with open chromatin and small nucleoli and are separated by variable amounts of collagen. Older lesions are often less cellular and more collagenous.

The association of soft tissue tumors, usually Gardner-type fibroma and desmoid-type fibromatosis, with colonic polyposis and occasionally multiple osteomas is known as Gardner syndrome . In this condition, the fibromatosis has a particular tendency to involve intra-abdominal structures, especially the mesentery, and to manifest itself following a surgical procedure in the area. It is important not to misdiagnose intra-abdominal fibromatosis involving the intestinal wall (a not uncommon occurrence) as a gastrointestinal stromal tumor (GIST). Along these lines, it should be mentioned that—despite early statements to the contrary—fibromatosis is a CD117-negative tumor, as opposed to the vast majority of GISTs. As previously discussed, patients with Gardner syndrome can also develop Gardner-associated fibroma , a soft tissue tumor most often located in the back or paraspinal region and characterized microscopically by a bland hypocellular proliferation of spindle cells embedded in a background of haphazardly oriented coarse collagen fibers, similar to nuchal-type fibroma.
Adult-type fibrosarcoma can arise from superficial and deep connective tissues such as fascia, tendon, and periosteum; they often grow slowly and appear well circumscribed. They usually are soft and may contain area of necrosis and hemorrhage ( Fig. 41.31 ).

Microscopically, the well-differentiated tumors (low grade) are easily recognized as fibroblastic; the cells are arranged in long fascicles that intersect at acute angles, resulting in a “herringbone” appearance ( Fig. 41.32 ). Mitotic activity varies. High-grade tumors have closely packed, less well-oriented, smaller, rounder tumor cells with a less distinct fascicular arrangement, greater mitotic activity, and often extensive necrosis. It should be remembered that many other soft tissue tumors, particularly synovial sarcoma, MPNST, and UPS may harbor areas closely resembling fibrosarcoma. Careful examination of different blocks of the tumor coupled with select adjunctive studies will provide the correct diagnosis in almost all instances. Although a pleomorphic type of fibrosarcoma probably exists, one should question the diagnosis of fibrosarcoma in the presence of a soft tissue sarcoma with numerous tumor giant cells. As a matter of fact, the diagnosis of fibrosarcoma (especially the low-grade type) should be viewed as a diagnosis of exclusion. Both immunohistochemical and ultrastructural studies have shown most of the cells show fibroblastic features, although it is common to also find cells with myofibroblastic differentiation, including the expression of SMA. The distinction of fibrosarcoma from fibromatosis is critical, given that the former has a significant risk of metastasis and the latter only has a risk of local recurrence. Generally, the more superficial and lower grade the tumor, the better the prognosis. In the study by Scott et al., the overall rate of recurrence was 42% at 5 years. The status of the surgical margins was strongly predictive of local recurrence, but neither tumor grade nor stage were predictive factors. The treatment of choice is radical excision, and postoperative radiation therapy should be considered if microscopic residual or positive margins are encountered. Since subclinical microscopic metastases are presumed to exist in many patients at the time of surgery, adjuvant chemotherapy has been recommended following surgical excision of high-grade tumors.

Congenital fibrosarcoma (infantile fibrosarcoma) is an extremely cellular tumor characterized by very rapid growth and the capability for extensive local invasion, but its metastatic rate is negligible. Microscopically, this tumor is composed of a cellular proliferation of spindle cells that are fairly uniform and arranged in fascicles or bundles, often admixed with lymphocytes and a hemangiopericytoma-like vascular pattern. At the molecular level, it is characterized by the ETV6–NTRK3 gene fusion, which results from the chromosomal translocation t(12;15)(p13;q25). This gene fusion is not present in adult-type fibrosarcoma or other pediatric fibroblastic tumors in the differential diagnosis. In addition, trisomies of chromosomes 8, 11, 17, and 20 are common. Of interest, cellular congenital mesoblastic nephroma of the kidney shows identical genetic alterations, suggesting that these two entities are closely related.
Low-grade fibromyxoid sarcoma (LGFMS) , also known as Evans tumor, is a low-grade sarcoma with deceptively bland histologic features. Most patients are young to middle-aged adults and present with a slowly enlarging mass in the deep soft tissue of the extremities, although this tumor has been reported in virtually every anatomic site. The tumor is of low to moderate cellularity with bland spindle-shaped cells deposited in a variably myxoid and collagenous matrix. There is often an elaborate network of capillaries in the myxoid zones, which are usually longer and less organized in comparison to the delicate vasculature of myxoid liposarcoma ( Figs. 41.33 to 41.35 ). Some tumors show collagen rosettes (formerly known as hyalinizing spindle cell tumor with giant rosettes) ( Fig. 41.36 ), and others show cords of epithelioid cells with features overlapping that seen in sclerosing epithelioid fibrosarcoma (see later). Rare cases show higher-grade areas with increased cellularity, mitotic activity, and nuclear pleomorphism. By immunohistochemistry, many express EMA and virtually all show strong staining for MUC4. The vast majority harbor translocations involving either CREB3L2 or CREB3L1 with the FUS gene, resulting from either a t(7;16) or t(11;16), respectively. Rare cases show involvement of EWSR1 rather than FUS .




Despite its bland histologic appearance, LGFMS can locally recur and metastasize. In the study by Folpe et al. in 2000, of 54 patients with follow-up, only 5 (9%) developed local recurrence, and 3 (6%) had metastatic disease; only 1 patient died of their tumor. In a more recent study by Evans in 2011 with long-term follow-up, the rates of local recurrence, metastasis, and death were 64%, 45%, and 42%, respectively.
Sclerosing epithelioid fibrosarcoma is a variant of fibrosarcoma that simulates the appearance of infiltrating carcinoma. It is composed of small, round to ovoid tumor cells embedded in a dense hyalinized stroma. The scanty cytoplasm often has a clear appearance, and there may be a single file pattern of growth ( Fig. 41.37 ). Necrosis and bone invasion may be found. Up to 50% express EMA, and, like LGFMS, most express MUC4, particularly those with FUS translocations. Some cases exhibit the same distinctive chromosomal translocation [t(7;16)] of LGFMS, suggesting a close relationship between these two tumor types. Other examples of sclerosing epithelioid fibrosarcoma (like LGFMS) show involvement of EWSR1 rather than FUS . The tumor is associated with a high incidence of local recurrence and distant metastasis.

Myxoinflammatory fibroblastic sarcoma (MIFS) , also known as inflammatory myxohyaline tumor, is a low-grade sarcoma most commonly found in the distal extremities of adults. This lesion can simulate an inflammatory condition, Hodgkin lymphoma, and various other types of soft tissue sarcomas, including myxofibrosarcoma. Microscopically, it has an infiltrative multinodular quality and a polymorphic cellular composition in a hyaline or myxoid background ( Fig. 41.38 ). There is a dense mononuclear inflammatory infiltrate containing scattered stromal cells of either epithelioid or spindle shape, including some with large bizarre nuclei and prominent nucleoli resulting in a resemblance to Reed–Sternberg cells or virus-infected cells. The immunohistochemical profile is nonspecific, but at least a subset of tumors have a recurrent t(1;10)(p22;q24) involving TGFBR3 on chromosome 1 and MGEA5 on chromosome 10. Interestingly, this alteration has been identified in hemosiderotic fibrolipomatous tumor (also known as hemosiderotic fibrohistiocytic lipomatous tumor), and some cases of pleomorphic hyalinizing angiectatic tumor (PHAT) suggesting a relationship among these tumors. Local recurrence is common, but distant metastases are exceptionally uncommon.

A particularly difficult issue in the classification of soft tissue tumors is the role played by myofibroblasts. Some authors have taken a very restrictive view of the definition of a myofibroblast (including the requirement of ultrastructural confirmation), whereas others do not believe ultrastructural confirmation should be required. Part of the problem is that there are no specific immunohistochemical markers for myofibroblasts, although the most common immunophenotype is strong staining for SMA with an absence of staining for desmin and h-caldesmon.
The other problem concerns the fact that cells with myofibroblastic features can be found in a large number of benign and malignant soft tissue lesions, including lesions in which they constitute only a small percentage of the neoplastic cells. Therefore, if there is to be a category of myofibroblastic tumors, it would be wise to reserve it for lesions that are composed almost entirely of cells having myofibroblastic features.
Inflammatory myofibroblastic tumor is a distinctive entity that has gone by a number of different names, including inflammatory pseudotumor and inflammatory fibrosarcoma, among many others. Although they have been described in virtually every anatomic site, many of the cases arise in the mesentery or retroperitoneum of children or adults, often accompanied by anemia and fever.
Histologically, a variety of patterns may be seen including a fasciitis-like pattern, cellular spindle cell areas, and hypocellular hyalinized zones. Lymphocytes and plasma cells are often intermixed within any of these patterns ( Fig. 41.39 ). The constituent cells show the immunohistochemical features of myofibroblasts, as previously discussed, and up to 50% also stain for ALK. There is imperfect correlation between ALK staining and ALK rearrangements approximately 50% of these tumors harbor clonal rearrangements of the ALK gene at 2p23. Recently, gene fusions involving other kinases have been identified, including ROS1 and PDGFRB . A number of other fusion partners have been identified including TPM3, TPM4, CARS, ATIC, SEC31L1, RANBP2, and CLTC . Tumors in the abdomen and retroperitoneum have a higher propensity for aggressive clinical behavior than those arising elsewhere, including frequent local recurrences and definite risk of metastasis. Although surgery is the mainstay of therapy, ALK inhibitors such as crizotinib have been found to be efficacious in treating aggressive lesions.

Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here