Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Myopathic changes are seen in disorders that primarily affect muscle
Neurogenic changes in skeletal muscle result from disorders of innervation: peripheral nerve, motor neuron disease, or neuromuscular junction
Depends on the specific disease process
Myopathies usually present with the following:
Proximal muscle weakness, usually symmetric
Malaise and fatigue
No sensory complaints or paresthesias
Atrophy and hyporeflexia are late findings
Serum creatine kinase (CK) levels are usually elevated
Electromyography (EMG) shows “myopathic” changes
Neurogenic muscle disorders present with the following:
Usually distal muscle weakness
Sensory complaints and paresthesias
Atrophy and hyporeflexia early in the disease course
Serum CK levels are usually normal
Electromyography shows “neurogenic” changes
Depends on the specific disease process
There are no specific imaging findings that help differentiate between myopathic and neurogenic muscle changes
Myopathic features include the following:
Variability in fiber size and shape with rounded myofibers
Large and small myofibers are randomly and diffusely distributed
Type 1 fiber predominance
Increased internal nuclei (more than 3%) and nuclear chains
Myofiber necrosis (pale staining or hyaline fiber) and phagocytosis
Regenerating (basophilic) myofibers
Fiber splitting
Increased endomysial connective tissue and deposition of adipose tissue
Endomysial, perimysial, or perivascular infiltrates of T lymphocytes and macrophages are prominent in some myopathies
Myofibrillar architectural changes such as moth-eaten fibers, ring fibers, lobulated and whorled fibers
Sarcoplasmic inclusions and vacuoles are characteristic of some myopathies
Neurogenic features include the following:
Variability in myofiber size and shape with angulated fibers
Two populations of myofibers are seen in denervated muscle: atrophic fibers (denervated) and normal or hypertrophied fibers
Atrophic fibers are generally clustered in groups (small group atrophy)
Large group atrophy occurs when an entire fascicle becomes atrophic
Fiber type grouping (groups of both types have to be identified in the biopsy)
Hypertrophic myofibers are usually type 1
Atrophic angulated myofibers are of both types
Pyknotic nuclear clumps
Atrophic angulated myofibers stain intensely with oxidative stains
Myofiber architectural changes including target fibers and targetoid fibers
Oxidative enzymes such as NADH-TR highlight myofiber architectural changes in both myopathic and neurogenic muscle
Specific immunostudies (inflammatory stains, sarcolemmal proteins) depending on the histologic features and clinical impression
Myopathic changes are seen in a wide range of acquired and inherited disorders including inflammatory myopathies, metabolic disorders, muscular dystrophies, and toxic exposure, among others
Neurogenic changes are seen in numerous inherited and acquired clinical disorders caused by defects in upper or lower motor neurons or the peripheral nerve including, but not limited to, amyotrophic lateral sclerosis (ALS), spinal muscular atrophy (SMA), hereditary motor and sensory neuropathies (HMSN), and inflammatory peripheral neuropathies
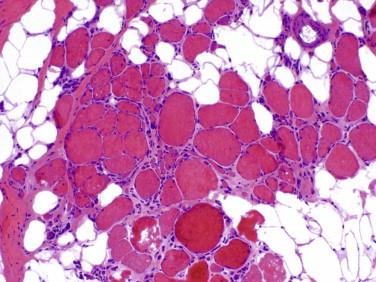
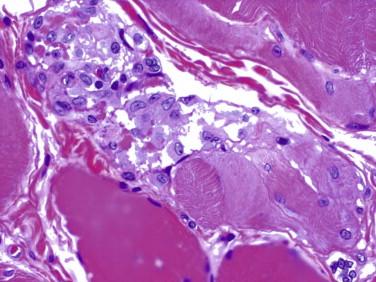
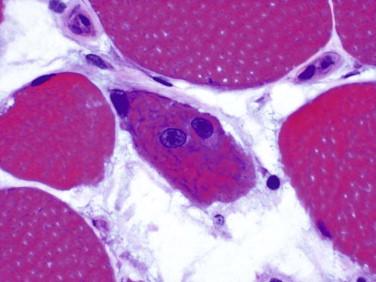
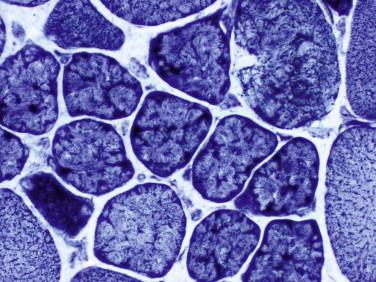
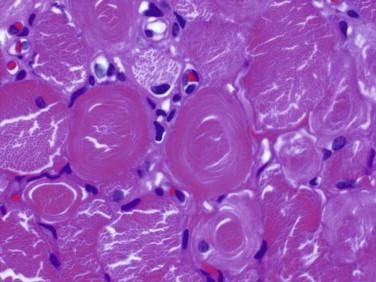

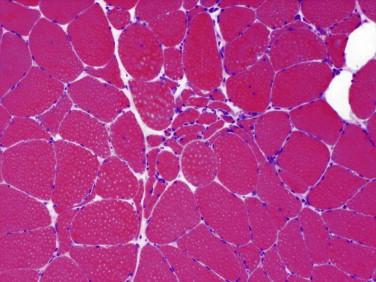
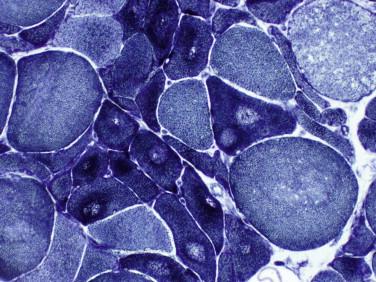
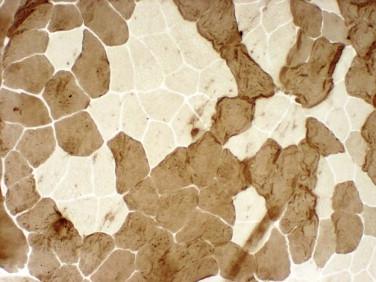
Idiopathic inflammatory myopathy with associated characteristic cutaneous findings
Rare disease with an estimated incidence of 9.63 cases per million people
More common in women (women-to-men ratio of 2 : 1)
Adult-onset dermatomyositis has peak onset age of 50 years
Juvenile dermatomyositis affects children, usually between 5 and 10 years
Heliotrope (violaceous) skin rash over the upper eyelids
Skin findings usually antedate muscle manifestations
Groton papules or scaly erythematous eruptions over the extensor surfaces of the knuckles, elbows, or knees
Progressive symmetric proximal limb muscle weakness with insidious onset, myalgia, tenderness, and dysphagia
Neuromuscular symptoms similar to polymyositis
Involvement of organ systems such as the lung, heart, and kidneys may occur
Strong association with the development of malignancies
Juvenile dermatomyositis
Variable muscle weakness
Associated with general symptoms of malaise, fever, lethargy
Calcinosis in chronic cases
Laboratory abnormalities include increased serum creatine kinase (CK) usually 5 to 50 times the reference range, increased serum aldolase, increased erythrocyte sedimentation rate (ESR), and myoglobulinuria
Antibodies may be detected in the serum, including antinuclear antibodies (ANA), myositis-specific antibodies, and anti-RNA antibodies such as Jo-1 and Mi-2 antibodies
Electromyography (EMG) reveals a combination of spontaneous fibrillation potentials, positive sharp waves, and polyphasic, short duration potentials on voluntary contractions indicative of irritable myopathy
Muscle disease responds to corticosteroid therapy
Other immunosuppressive agents (azathioprine, cyclophosphamide, cyclosporine) may be used in patients unresponsive to corticosteroids and to treat skin disease
Prognosis is generally favorable; however, recovery may be slow and incomplete
MRI findings are nonspecific and reveal increased signals in areas of edema and inflammation
May be used to select the site of biopsy
Perifascicular myofiber atrophy
Inflammation is focal and predominantly perivascular and perimysial
Inflammatory infiltrates are composed predominantly of macrophages, B cells, and CD4+ T lymphocytes
Decreased capillary density especially in the perifascicular region, which may lead to myofiber infarction
Electron microscopy reveals tubuloreticular inclusions in endothelial cells of intramuscular arterioles and capillaries
Humoral mediated immune reaction against capillaries with activation of complement, deposition of C5b-9, lysis of endothelial cells, and loss of capillaries with resulting ischemia
Immunostudies demonstrate a predominance of B cells and CD4+ cells (both T lymphocytes and dendritic cells) in the inflammatory infiltrate
Deposition of the membrane attack complex of complement (C5b-9) in capillaries is an early event
Aberrant sarcolemmal expression of major histocompatibility complex I (MHC-I) may be seen, especially in the perifascicular region
Connective tissue disorders with muscle and skin involvement such as systemic lupus erythematosus
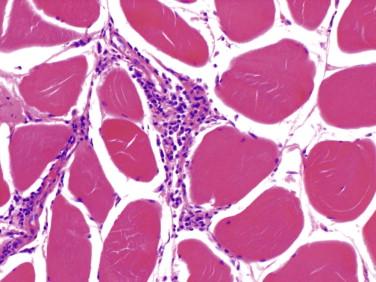
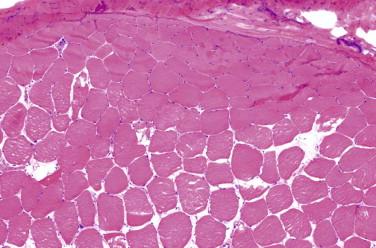

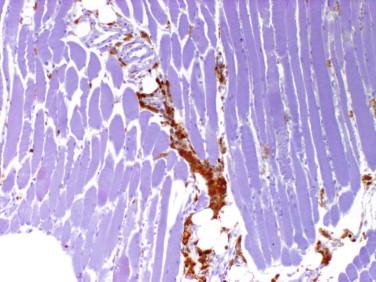
Idiopathic inflammatory disease that affects predominantly the skeletal muscle; may occur in setting of systemic inflammatory/autoimmune disease
Rare disease with an incidence of 0.5 to 8.4 cases per million people
More common in women (women-to-men ratio of 2 : 1)
Peak incidence between 45 and 60 years; it rarely affects children
Progressive symmetric proximal limb muscle weakness with insidious onset
Myalgia and tenderness may be present
Dysphagia is present in one third of patients
Extraocular muscles are usually spared
Involvement of organ systems such as the lung, heart, and kidneys is uncommon
Laboratory abnormalities include increased serum creatine kinase (CK) usually 5 to 50 times the reference range, increased serum aldolase, increased erythrocyte sedimentation rate (ESR), and myoglobulinuria
In some cases, antibodies may be detected in the serum, including antinuclear antibodies (ANA), myositis-specific antibodies, and anti-RNA antibodies such as Jo-1 and signal recognition particle (SNP) antibodies
Electromyography (EMG) reveals a combination of spontaneous fibrillation potentials, positive sharp waves, and polyphasic, short duration potentials on voluntary contractions indicative of irritable myopathy
Polymyositis may be associated with other connective tissue diseases
Majority of cases respond to corticosteroid therapy
Other immunosuppressive agents (azathioprine, cyclophosphamide, cyclosporine) may be used in patients unresponsive to corticosteroids
Prognosis is generally favorable; however, recovery may be slow and incomplete
MRI findings are nonspecific and reveal increased signals in areas of edema and inflammation
May be used to select the site of biopsy
Myopathic features
Evidence of myofiber necrosis, phagocytosis, and regeneration
Prominent endomysial inflammatory infiltrates composed predominantly of T lymphocytes and macrophages
T lymphocytes also surround and invade non-necrotic myofibers
Inflammatory infiltrates can be small and multifocal
Evidence suggests an antigen-directed and MHC-I restricted cytotoxicity mediated by CD8+ T cells
Immunohistochemical studies for CD3 (T lymphocytes) and CD68 (macrophages) are useful for identification and localization of inflammatory cells
CD8+ T lymphocytes predominate
Major histocompatibility complex I (MHC-I) is abnormally up-regulated on the sarcoplasm of myofibers even in areas devoid of inflammation
Inclusion body myositis
Limb-girdle muscular dystrophy
Toxic or drug-induced myopathies (alcohol, statins)
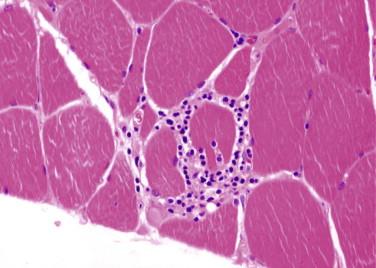
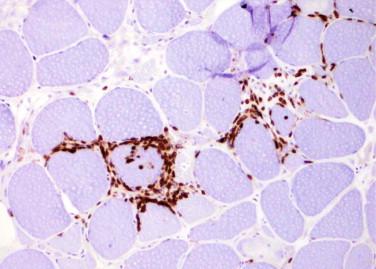
Most common myopathy in adults over 50 years old, characterized by inflammatory myopathy with rimmed vacuoles
Accounts for 16% to 28% of inflammatory myopathies in North America
Affects individuals older than 50 years (mean age of onset is 56 to 60 years)
Men are more commonly affected than women (ratio of 3 : 1)
Hereditary inclusion body myopathies:
Hereditary group of disorders characterized by progressive muscle weakness
Most are of late onset, but congenital and childhood forms have been described
Muscle biopsy reveals rimmed vacuoles with accumulation of the same proteins as in sporadic inclusion body myositis but no inflammatory infiltrate
Several genetic defects have been identified
Insidious onset of proximal leg and distal arm weakness with marked asymmetry
Dysphagia is present in a majority of patients
May be associated with autoimmune disorders (systemic lupus erythematosus [SLE], Sjögren syndrome, and sarcoidosis, among others)
Strong association to MHC antigens HLA-DR3, DR52, and B8
Serum creatine kinase (CK) may be normal or elevated (up to 10 times the upper normal limit)
Myositis-associated autoantibodies are usually not found
Electromyography (EMG) reveals increased insertional activity and short duration polyphasic motor unit potentials
Nerve conduction studies are consistent with superimposed peripheral neuropathy
Refractory to immunosuppressive therapy
Prognosis is that of slow progression to disability
MRI is of limited diagnostic value
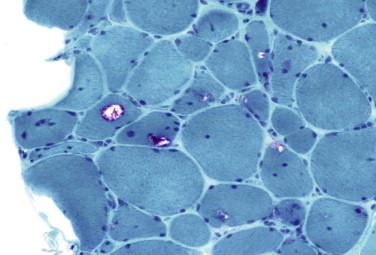
Myopathic features
Small angulated myofibers may be scattered through the biopsy or in clusters
Endomysial mononuclear cell inflammatory infiltrate with invasion of non-necrotic myofibers
Gomori trichrome stain reveals rimmed vacuoles and ragged-red areas
Intracellular amyloid deposits (Congo red positive)
Eosinophilic intracytoplasmic inclusions
Occasional cytochrome oxidase (COX) negative fibers
Electron microscopy reveals 15- to 21-nm tubulofilaments in the sarcoplasm and structurally abnormal mitochondria
Inflammatory infiltrate is predominantly composed of CD8+ T lymphocytes
MHC-I expression in the sarcolemma
The vacuoles may be immunoreactive for β-amyloid, β-amyloid precursor protein, ubiquitin, α-synuclein, phosphorylated tau, TDP-43, and prion protein, among others
Unknown
Immune-mediated causes and degenerative processes have been proposed
Polymyositis
Amyotrophic lateral sclerosis (ALS)


Inherited neuromuscular disorders (muscular dystrophy) caused by mutations in the Dystrophin gene
Duchenne muscular dystrophy (DMD) is the most common muscular dystrophy
Prevalence of 1 in 3500 live male births worldwide (63 cases per million)
Becker muscular dystrophy (BMD) has a prevalence of approximately 24 cases per million
Almost exclusively affects males because of X-linked inheritance pattern
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here