Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
This chapter focuses on clinical gastroenterology issues of interest to pathologists, including endoscopic diagnosis and management of Barrett’s esophagus, management of intestinal metaplasia in the setting of chronic gastritis, and surveillance in patients with inflammatory bowel disease, colon polyps, and colon cancer.
Most authorities recommend that men with chronic reflux symptoms lasting 5 or more years and at least two known risk factors undergo an upper endoscopy to screen for Barrett’s esophagus (BE) ( Box 2.1 ). The benefits of screening programs for BE are controversial because of a lack of sufficient evidence that such programs improve survival rates or are cost-effective. Furthermore, data regarding the overall survival benefits of surveillance endoscopy for BE are inconsistent. For example, a recent meta-analysis demonstrated that surveillance endoscopy for BE was associated with detection of earlier stage esophageal adenocarcinoma and may therefore provide a small survival benefit; however, the impact of lead- and length-time bias on survival remains unclear. It should be stated, however, that the prospect of performing a prospective, randomized control trial to properly assess the potential benefits of screening and surveillance for BE are greatly limited by the low rate of cancer progression, with an annual incidence of only 0.1% to 0.3%. The current standard of care dictates that if BE is diagnosed, the patient should be entered into an endoscopic surveillance program for early detection of dysplasia and adenocarcinoma.
Screening for Barrett’s esophagus should be considered in men with chronic (>5 years) and/or frequent (weekly or more) symptoms of gastroesophageal reflux (heartburn or acid regurgitation) and two or more risk factors for Barrett’s esophagus or esophageal adenocarcinoma.
Risk factors include:
Age 50 years or older
White race
Presence of central obesity (waist circumference >102 cm or waist-to-hip ratio >0.9)
Currently smoking or history of smoking
Confirmed family history of Barrett’s esophagus or esophageal adenocarcinoma (in a first-degree relative)
In the past, endoscopic surveillance was undertaken only in patients who were medically fit to undergo esophagectomy. However, with the advent of nonsurgical ablative endoscopic technique and endoscopic mucosal resection (EMR), the number of patients eligible for surveillance has increased. Current guidelines recommend that EMR or endoscopic submucosal dissection be performed when mucosal irregularity, such as nodularity, is identified in the setting of BE on endoscopy, and the adjunct use of narrow band imaging (NBI) technology may aid the endoscopist in identifying such abnormalities. EMR provides therapeutic benefit, but also allows for proper histologic assessment and staging.
Aggressive treatment of reflux with proton pump inhibitors (PPIs) is warranted before surveillance endoscopy because active inflammation with repair can mimic dysplasia. Current guidelines recommend against obtaining biopsies in mucosal areas where there is endoscopic evidence of erosive esophagitis. Long-term once-daily treatment with a PPI is recommended for patients with BE because of evidence that once-daily PPI may decrease the risk of esophageal adenocarcinoma.
Currently, endoscopic surveillance is performed by obtaining four-quadrant biopsies at 2-cm intervals with the use of cold biopsy forceps. In addition, specific attention is paid to mucosal abnormalities such as ulcers, irregular lesions, nodules, and polyps, and these lesions should be submitted separately. In patients with known or suspected dysplasia, biopsies should be obtained at 1-cm intervals instead of the standard 2 cm. Studies have shown that endoscopy with magnification and NBI allows for better localization of high-grade dysplasia (HGD) and may allow more targeted and fewer biopsies. ,
The recommended interval of surveillance for dysplasia in patients with BE is determined by the presence and degree of dysplasia found ( Table 2.1 ). In the absence of dysplasia, the surveillance interval is every 3 to 5 years with four-quadrant biopsies every 2 cm. If the pathology is indefinite for dysplasia, a repeat endoscopy after optimization of acid suppressive medications for 3 to 6 months should be performed. If pathology remains indefinite for dysplasia on subsequent endoscopy, a surveillance endoscopy is recommended in 1 year.
| Nondysplastic Barrett’s esophagus |
|
| Indefinite for dysplasia |
|
| Low-grade dysplasia |
|
| High-grade dysplasia |
|
If dysplasia of any grade is identified on pathology, review by two pathologists, at least one of whom has specialized expertise in gastrointestinal (GI) pathology, is recommended, given the interobserver variability in the interpretation of dysplasia. If low-grade dysplasia (LGD) is confirmed, endoscopic ablative therapy such as radiofrequency ablation (RFA) should be considered. However, endoscopic surveillance every 12 months is also considered a reasonable alternative.
In patients with flat HGD, guidelines now recommend endoscopic ablative therapy over esophagectomy or intensive endoscopic surveillance, given the proven efficacy of techniques such as RFA and the favorable adverse effect profile of ablative therapy compared to that with surgery.
The most common causes of chronic gastritis are Helicobacter pylori infection, environmental exposures including smoking, and autoimmune processes. Biopsies obtained during endoscopy from patients with chronic gastritis may reveal gastric intestinal metaplasia (GIM). Currently, the overall pooled prevalence of GIM in the United States has been estimated to be 4.8%. However, the prevalence of GIM can be significantly higher in certain subgroups. For example, a study from the United States demonstrated that 13% of patients at low risk for gastric cancer and 50% of patients at high risk had GIM on biopsies from normal-appearing gastric mucosa.
In the United States, where the incidence of gastric cancer is low, endoscopic screening for gastric cancer has not been endorsed. However, because GIM has been recognized as a precursor lesion to gastric cancer, there has been considerable debate as to whether identification of GIM warrants future surveillance endoscopic evaluations. The first evidence-based guideline was put forth by the American Gastroenterological Association (AGA) in 2020. In this guideline, the AGA does not recommend routine endoscopic surveillance in patients with GIM. This recommendation is primarily based on data that suggest that findings of GIM portend a 0.16% annual and 1.6% 10-year cumulative risk for incident gastric cancer. However, the AGA remarks that patients at higher risk for gastric cancer who “put a high value on potential but uncertain reduction in gastric cancer mortality, and who put a low value of potential risks of surveillance endoscopies,” may elect for surveillance. Risk factors for gastric cancer include incomplete (at least partial colonic type) versus complete (small intestinal type metaplasia), extensive (involving the gastric body plus either antrum and/or incisura) versus limited (involving the gastric antrum and/or incisura only), and a family history of gastric cancer. In contrast, guidelines by the American Society of Gastrointestinal Endoscopy (ASGE) in 2015 recommend surveillance endoscopy for patients with GIM who are at increased risk of gastric cancer because of ethnic background (e.g., Asian heritage) or family history. Surveillance intervals are not delineated in these guidelines, but rather, it is suggested that surveillance intervals be individualized.
With regard to surveillance, the AGA recommends against routine short-interval repeat endoscopy for the purpose of risk stratification. However, it is suggested that patients may choose to repeat endoscopy to establish the anatomic extent of GIM—to undergo “gastric mapping” with biopsies throughout the stomach. Specifically, the Sydney protocol, which requires five biopsies (two from the antrum at the lesser and greater curvature, two from the body at the lesser and greater curvature, and one from the incisura), is recommended for the purposes of assessing the extent of GIM. Additionally, the guidelines suggest that patients who are at increased overall risk for gastric cancer (racial/ethnic minorities, immigrants from regions with high gastric cancer incidence, or individuals with a family history of a first-degree relative with gastric cancer) may elect to undergo repeat endoscopy in 1 year to undergo targeted biopsies of any visible abnormalities within the gastric lumen and untargeted biopsies (of at least the gastric antrum and body). No statement is provided with respect to the frequency of surveillance intervals thereafter.
Pernicious anemia, which can occur as a result of autoimmune chronic atrophic gastritis, is another potential risk factor for gastric cancer and gastric neuroendocrine tumors. A twofold to threefold increased risk for gastric cancer has been noted, depending on the location and duration of disease. Clear guidelines for surveillance in atrophic gastritis have not yet been established. Current guidelines from the ASGE advocate performance of a screening upper endoscopy at the time of diagnosis of pernicious anemia to identify lesions such as neuroendocrine tumor or gastric cancer and/or to perform endoscopy on patients with pernicious anemia in whom upper GI symptoms develop.
Inflammatory bowel disease (IBD), which comprises ulcerative colitis (UC) and Crohn’s disease, is the second most common inflammatory disorder after rheumatoid arthritis. There are approximately 1.6 million patients with IBD in the United States, and as many as 700,000 new cases of IBD are diagnosed every year. The Centers for Disease Control and Prevention has estimated that there are 3.1 million patients with IBD in the United States. Each year, between 20,000 and 25,000 new cases are diagnosed. The peak incidence occurs between the ages of 15 and 35, with males and females equally affected.
The goal of colonoscopic surveillance is to minimize the morbidity and mortality from colorectal cancer (CRC) with appropriate and timely referral for colectomy. Close interaction between the endoscopist and the pathologist is crucial in the management of IBD. Although no prospective randomized studies have been performed to evaluate the efficacy of surveillance colonoscopy to detect dysplasia or CRC in UC patients, case-control studies suggest a reduction in mortality in those undergoing surveillance. Surveillance colonoscopy should optimally be performed when the patient is in clinical remission, because active inflammation may hinder the endoscopic identification of mucosal abnormalities and histologic diagnosis of dysplasia.
Biopsies taken during surveillance colonoscopy are graded as positive for dysplasia, negative for dysplasia, or indefinite for dysplasia. Dysplasia is further classified as LGD or HGD. The greatest interobserver variability among pathologists lies in the interpretation of the LGD and indefinite-for-dysplasia categories. Therefore, once dysplasia is detected, a second pathologist with expertise in interpretation of GI biopsies should confirm the diagnosis. Dysplastic mucosa may be characterized as either flat or raised. Flat dysplasia is generally considered to be endoscopically undetectable, is identified by random biopsies, and can be further classified as either multifocal or unifocal. Raised or polypoid dysplasia was historically referred to by the acronym DALM : dysplasia-associated lesion or mass. However, an international consensus statement on the management of dysplasia in IBD in 2015 recommended that the term DALM be abandoned in favor of a modified Paris classification for describing lesions on colonoscopy.
Current guidelines from the American College of Gastroenterology (ACG), the American Gastroenterology Association (AGA), and the British Society of Gastroenterology (BSG) recommend that colonoscopic surveillance begin 8 to 10 years after the onset of symptoms of colitis in patients with UC or with Crohn’s colitis involving at least one third of the colon.
The results of the colonoscopy determine the extent of disease, and appropriate patients are then entered into a surveillance program. Repeated surveillance colonoscopy is performed every 1 to 3 years (AGA) or 1 to 5 years (BSG). Patients with coexisting primary sclerosing cholangitis should begin surveillance colonoscopy at the time of diagnosis of liver disease and continue annually thereafter regardless of the extent of disease. Other risk factors associated with an increased risk of developing colorectal neoplasia include CRC in a first-degree relative, male sex, ongoing active endoscopic or histologic inflammation, and anatomic abnormalities such as a foreshortened colon, stricture, or multiple inflammatory pseudopolyps. Several studies have correlated increased severity of colonoscopic, macroscopic, and histologic inflammation with a higher risk of CRC. , Patients with proctitis or distal proctosigmoiditis are not at increased risk for the development of CRC and do not need to undergo surveillance.
There is wide variability in the practice of surveillance by gastroenterologists and inconsistency in the management of patients with dysplasia. , Current guidelines recommend the use of chromoendoscopy and directed biopsies or four-quadrant random biopsies every 10 cm for a minimum of 33 total colonic biopsies, or 6 biopsies in 6 pathology specimen bottles. The recommendation for 33 biopsies is based on a retrospective analysis that revealed a 90% positive predictive value for dysplasia with 33 biopsy specimens and a 95% positive predictive value with more than 56 specimens. It is also recommended that in patients with UC, four-quadrant biopsies should be taken every 5 cm in the distal sigmoid and rectum, given the increased risk of carcinoma in those areas. Other endoscopists obtain six specimens from each of the following sections: cecum and ascending colon, transverse colon, descending colon, sigmoid, rectosigmoid, and rectum. Additional biopsies should be obtained from any suspicious mucosal lesions. The BSG guidelines recommend pancolonic dye spraying and targeted biopsies of abnormal mucosa. If chromoendoscopy is not used, two to four random biopsies every 10 cm should be taken.
As stated previously, the use of the term DALM has been discouraged because it relates to description of dysplastic lesions in the setting of IBD. Rather, polypoid and nonpolypoid dysplastic lesions are considered “visible” if identified on endoscopic examination and lend consideration of resection. Alternatively, dysplastic lesions are termed “invisible” if detected by random biopsies. Criteria for what constitutes a lesion as endoscopically resectable are not clearly specified in published guidelines to date and largely depend on the comfort level of polypectomy by the performing endoscopist and/or availability of techniques such as EMR and endoscopic submucosal dissection (ESD). The goal of endoscopic resection is to obtain an en bloc resection with negative margins; if this cannot be achieved, surgery is indicated. SCENIC guidelines recommend that patients with larger lesions removed in a piecemeal fashion, or by EMR or ESD, should undergo repeat colonoscopy in 3 to 6 months, whereas patients with smaller lesions resected en bloc may return for colonoscopy in 1 year. Figure 2.1 describes the algorithm proposed by the ASGE in 2015 for management of an endoscopically visible lesion in the setting of colitis.
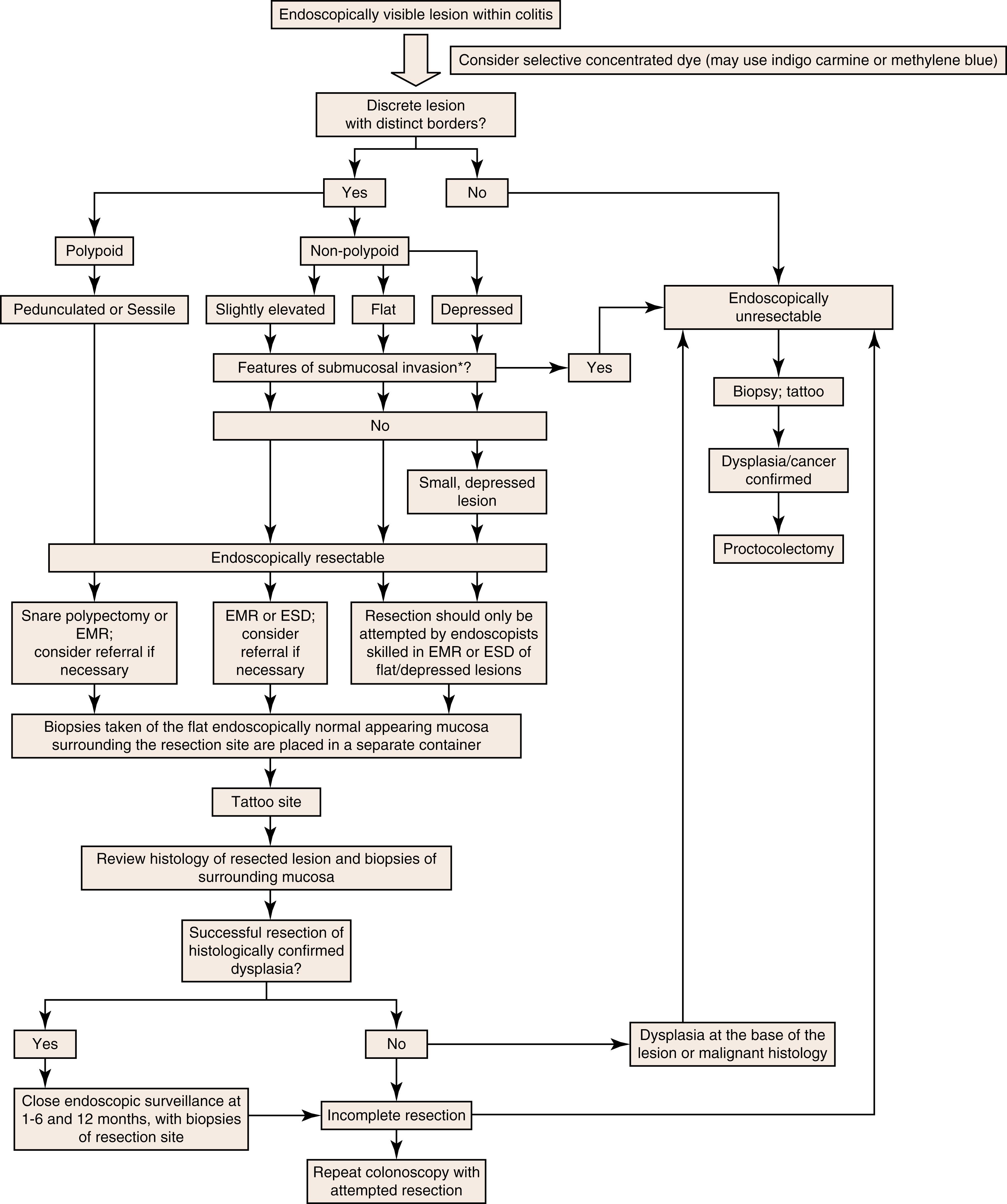
The AGA and BSG guidelines previously recommended proctocolectomy in cases of “flat” HGD. More recently, it has been suggested that if LGD or HGD is confirmed on random biopsy (so-called “invisible dysplasia”) by an expert GI pathologist, a repeat colonoscopy should be performed with enhanced imaging techniques, such as chromoendoscopy, to assess for the presence of a visible lesion, which may be resected; if a visible lesion is not identified, colectomy should be considered. Recommendations from the ASGE and SCENIC international consensus on the detection and management of dysplasia in patients with IBD are listed in Box 2.2 and Box 2.3 , respectively. ,
We recommend that all patients with ulcerative colitis (UC) or Crohn’s disease colitis undergo a screening colonoscopy 8 years after disease onset to (1) reevaluate extent of disease and (2) initiate surveillance for colorectal neoplasia.
We recommend surveillance colonoscopy be performed every 1 to 3 years beginning after 8 years of disease in patients with UC with macroscopic or histologic evidence of inflammation proximal to and including the sigmoid colon and for patients with Crohn’s colitis with more than one-third of colon involvement.
We recommend chromoendoscopy with targeted biopsies as the preferred surveillance technique to maximize dysplasia detection.
We suggest that chromoendoscopy-targeted biopsies are sufficient for dysplasia surveillance in patients with inflammatory bowel disease (IBD) and that consideration should be given to taking two biopsies from each colon segment for histologic staging to assess extent and severity of inflammation.
We suggest that random biopsies with targeted biopsies of any suspicious-appearing lesions remain a reasonable alternative for dysplasia surveillance if the yield of chromoendoscopy is reduced by significant underlying inflammation, significant pseudopolyposis, or poor preparation or if chromoendoscopy is not available.
We recommend that patients with IBD whose polypoid dysplastic lesions have been removed completely receive endoscopic surveillance at 1 to 6 months and at 12 months, with yearly surveillance examinations thereafter.
We suggest that patients with IBD whose nonpolypoid dysplastic lesions have been removed completely receive endoscopic surveillance at 1 to 6 months and at 12 months, with yearly surveillance thereafter.
We recommend proctocolectomy in patients with IBD if a detected lesion is not endoscopically resectable, if there is evidence of dysplasia at the base of the lesion, or if endoscopically invisible high-grade dysplasia or multifocal low-grade dysplasia is found in the colon during a high-quality chromoendoscopy examination.
When performing surveillance with white-light colonoscopy, high definition is recommended rather than standard definition.
When performing surveillance with standard-definition colonoscopy, chromoendoscopy is recommended rather than white-light colonoscopy.
When performing surveillance with high-definition colonoscopy, chromoendoscopy is suggested rather than white-light colonoscopy.
When performing surveillance with standard-definition colonoscopy, narrow-band imaging is not suggested in place of white-light colonoscopy.
When performing surveillance with high-definition colonoscopy, narrow-band imaging is not suggested in place of white-light colonoscopy.
When performing surveillance with image-enhanced high-definition colonoscopy, narrow-band imaging is not suggested in place of chromoendoscopy.
After complete removal of endoscopically resectable polypoid dysplastic lesions, surveillance colonoscopy is recommended rather than colectomy.
After complete removal of endoscopically resectable nonpolypoid dysplastic lesions, surveillance colonoscopy is suggested rather than colectomy.
For patients with endoscopically invisible dysplasia (confirmed by a gastrointestinal pathologist) referral is suggested to an endoscopist with expertise in inflammatory bowel disease surveillance using chromoendoscopy with high-definition colonoscopy
Studies have demonstrated that the use of chromoendoscopy can greatly increase the detection rate of dysplasia in patients with UC who have been enrolled in a surveillance program. Chromoendoscopy with targeted biopsies revealed significantly more dysplastic lesions than conventional colonoscopy with random biopsies. The overall sensitivity of chromoendoscopy for predicting neoplasia was 93% to 97%. Given these findings, consensus guidelines from several organizations have endorsed the use of chromoendoscopy in surveillance colonoscopy by trained endoscopists. , As more data regarding chromoendoscopy become available and new imaging techniques are developed, guidelines for surveillance endoscopy in patients with IBD will be refined to reflect these advances. It is also likely that molecular biology techniques will play a more important role in the future as an adjunct to endoscopic biopsy.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here