Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Annually, 12,390 new cases occur in the United States—fewer than 1%–2% of cancer diagnoses.
No specific etiologic agent is identifiable in the majority of cases.
Occasional cases are related to previous radiation, chemical exposure, alkylating chemotherapeutic agents, or chronic lymphedema.
Genetic conditions related to soft tissue sarcoma include Li-Fraumeni syndrome, neurofibromatosis, tuberous sclerosis, basal cell nevus syndrome, Gardner syndrome, and retinoblastoma.
Core needle biopsy (large lesions) or excisional biopsy (small lesions) is performed.
Pathologic review of histologic subtype, grade, and assessment of margins (excisional biopsies) is undertaken.
Imaging includes magnetic resonance imaging (MRI) or computed tomography (CT) of the primary site.
Chest radiography or chest CT (preferred) is also performed.
High-grade histologic type, deep location, and T2 tumor size are independent adverse prognostic factors for distant metastasis and survival.
Presentation with recurrent disease and positive surgical margins (gross or microscopic) are independent adverse prognostic factors for local recurrence.
Individual patient prognosis may be estimated with use of a nomogram or newer techniques.
The American Joint Committee on Cancer (Union for International Cancer Control) system uses criteria that include grade, size, and location relative to the investing muscular fascia, nodal status, and distant metastases. Other means to characterize risk of recurrence of soft tissue sarcomas include nomograms and bayesian belief networks.
Primary therapy includes surgical resection with an adequate margin of normal tissue. For extremity lesions, a limb-sparing approach is possible in more than 90% of patients and offers survival comparable to that of amputation without the associated morbidity.
For most patients, local control is improved with preoperative or postoperative radiotherapy. Size and primary site also affecting its use.
The role of chemotherapy for high-risk patients remains controversial, but chemotherapy is used at several major centers for high-risk patients, especially for extremity tumors with known chemotherapy sensitivity, preoperatively when possible.
In addition to surgery, 3 years of adjuvant imatinib is the standard of care for high-risk gastrointestinal stromal tumor (GIST).
Local recurrence rates vary depending on the anatomic primary site and the adequacy of local therapy. For extremity lesions, approximately 20% of patients develop locally recurrent disease.
Systemic cytotoxic chemotherapy with selective use of surgery is the mainstay of therapy for patients with metastatic disease.
Correct histologic diagnosis is important for the selection of palliative chemotherapy—for instance, eribulin for liposarcoma.
Kinase-directed therapy is clinically beneficial for patients with specific sarcoma diagnoses (e.g., GIST). Pazopanib was shown to be active in patients with other types of sarcoma failing other systemic therapy, but has comparatively limited benefit.
For the small subset of patients who develop isolated (solitary) lung metastases, 20% to 50% 3-year survival rates have been reported with metastasis resection.
Trabectedin, eribulin, and olaratumab have been approved by the US Food and Drug Administration since the last edition of this book was published.
Soft tissue sarcomas (STSs) comprise a group of relatively rare, anatomically and histologically diverse neoplasms. Most STSs share a common embryologic origin, arising primarily from tissues derived from the mesodermal or ectodermal germ layers, distinct from carcinomas, which arise from the endodermal germ layer, although some sarcomas such as angiosarcomas also have an endodermal origin. Although the somatic soft tissues account for as much as 75% of total body weight, neoplasms of the soft tissues are comparatively rare, accounting for less than 1% of adult malignancies and 15% of pediatric malignancies. The relative rarity of these tumors, coupled with the histologic diversity of tumors, has led to studies that include diverse STS subtypes in a single study group, making it difficult to develop specific therapies for specific tumor types, especially in adult STSs. The annual incidence of STSs in the United States is about 12,390 new cases, comparable to the incidence of testicular cancer. However, an estimated 4990 patients die annually of STS—a rate nearly 10-fold greater than is seen with testicular cancer—emphasizing the comparatively high overall mortality rate that is seen with this type of tumor.
The first portion of this chapter reviews the available literature on the evaluation and treatment of extremity sarcomas (which account for half of all STS). Other specific anatomic sites are reviewed at the end of the chapter.
No specific etiologic agent may be identified in the majority of patients with STS. There are a number of recognized associations between environmental factors and the subsequent development of sarcoma; these are summarized in Table 90.1 . The development of sarcoma has been reported after the use of ionizing radiation for the treatment of lymphoma ; solid tumors of the head and neck, breast, gynecologic organs, and skin; and benign conditions including endometriosis, tuberculous arthritis, and benign thymic enlargement. The vast majority of radiation-associated sarcomas are high grade (87%), and the predominant histologic types include undifferentiated pleomorphic sarcoma (UPS; formerly termed malignant fibrous histiocytoma [MFH]), angiosarcoma, malignant peripheral nerve sheath tumor (MPNST), osteosarcoma, and others; translocation-associated sarcomas are rare, in comparison, and teleologically it is difficult to understand how random DNA damage leads to a specific chromosomal translocation.
| Factor | Agent | Patient Population | Comment |
|---|---|---|---|
| Radiotherapy | Orthovoltage and megavoltage radiation | Therapeutic radiation patients | Most commonly undifferentiated pleomorphic sarcoma and osteosarcoma; dose-response relationship |
| Chemotherapy | Alkylating agents: cyclophosphamide, melphalan, procarbazine, nitrosourea, and chlorambucil | Pediatric cancer patients | Relative risk of bone sarcoma increased with cumulative drug exposure |
| Chemical exposure | Phenoxyacetic acids: 2,4-dichlorophenoxyacetic acid (2,4-D); 2,4,5 trichlorophenoxy acetic acid (2,4,5-T); 2 methyl-4 chlorophenoxyacetic acid (MCPA) | Forestry and agricultural workers | Phenoxy herbicide and defoliant exposure |
| 2,3,7,8-tetrachlorodibenzo- p -dioxin (TCDD); dioxin | Vietnam veterans | No clear link demonstrable for dioxin (Agent Orange) | |
| Chlorophenols | Sawmill workers | ||
| Thorotrast | Diagnostic x-ray patients | Hepatic angiosarcoma | |
| Vinyl chloride | Vinyl chloride workers | Hepatic angiosarcoma | |
| Arsenic | Vineyard workers | Hepatic angiosarcoma after exposure to arsenical herbicides | |
| Chronic lymphedema | Postsurgery patients Postradiation patients Patients with congenital lymphedema or filariasis |
Stewart-Treves syndrome (lymphangiosarcoma) |
By criteria that were described initially by Cahan and colleagues, radiation-induced sarcomas arise no sooner than 4 years after the therapeutic radiation and often arise decades later. More recently, however, a shorter 2-year period after diagnosis has been used to define radiation-related disease, and the median time from radiation to development of a secondary sarcoma is 8 to 10 years, but it can arise even later. Contrary to early theories, recent studies have suggested that both orthovoltage and megavoltage treatments are sarcomagenic at doses from 8.8 to 70 Gy. In a carefully conducted case-control analysis, the Late Effects Study Group found 64 cases of osteosarcoma in 9170 patients who had survived more than 2 years after the diagnosis of a variety of cancers. A dose-response relationship was found between the radiation dose and the subsequent development of osteosarcoma, with a relative risk ranging from 0.6-fold in patients who received less than 10 Gy to 38.3-fold in those who received more than 60 Gy. A complete understanding of the underlying biology of radiation-induced malignancy remains elusive. Host-related factors (especially young age) and treatment (especially its intensity) both seem to play a role in a complex relationship. Underlying genetic susceptibility is also important and is governed by factors such as deletion or mutation of tumor suppressor and DNA repair genes, which likely means that subgroups of the populations have more vastly enhanced risk than had previously been anticipated, and others are at lower risk than was appreciated in the past. The identification of osteosarcoma as a common second malignancy following radiation therapy for retinoblastoma is an example of a high-risk scenario. It is probable that the greater intensity of multimodality treatments for cancer that have been introduced over recent years with the goal of improving cancer control and survival will result in an increase in the rate of radiation-induced malignancies. For example, the use of radiation after lumpectomy for ductal carcinoma in situ may result in unnecessary second cancers, which are avoidable in over 95% of patients with the use of mastectomy and reconstruction. The most common postradiation cancer appears to be sarcoma, especially considering its baseline incidence rates.
Sarcomas have also been weakly associated with exposure to various chemical agents. A number of conflicting reports have emerged that suggest a relationship between occupational exposure to phenoxyacetic acids (found in some herbicides) and chlorophenols (found in some wood preservatives). Studies from Sweden demonstrated a link between phenoxy herbicide exposure in forestry workers and the subsequent development of sarcoma. However, additional investigations in the United States, New Zealand, and Finland have not confirmed this relationship. Similarly, there has been no demonstrable increase in the risk of sarcoma in Vietnam veterans exposed to Agent Orange (dioxin or TCDD [2,3,7,8-tetrachlorodibenzo- p -dioxin]). Hepatic angiosarcomas have been associated with exposure to a number of compounds, including Thorotrast (a colloidal suspension of thorium dioxide that was formerly used as an intravenous contrast agent in radiologic imaging procedures), vinyl chloride, and arsenic.
Studies have suggested a relationship between exposure to alkylating chemotherapeutic agents and the subsequent development of sarcomas. Osteosarcomas have been reported after cyclophosphamide treatment for pediatric acute lymphoblastic leukemia. In the report from the Late Effects Study Group, prior chemotherapy, particularly with melphalan, procarbazine, nitrosoureas, or chlorambucil, was found to be an independent risk factor for the development of sarcoma. The relative risk of sarcoma increased with cumulative drug exposure.
Chronic lymphedema may be a factor in the development of angiosarcoma. These neoplasms have been noted to arise in the chronically lymphedematous arms of women who were treated for breast cancer with radical mastectomy (Stewart-Treves syndrome). Lower extremity angiosarcomas have also been observed in patients with congenital lymphedema or filariasis complicated by chronic lymphedema.
A recent history of trauma is often elicited from sarcoma patients, particularly those with extremity sarcoma. Usually, the interval between the traumatic event and the diagnosis of sarcoma is short, making a causal relationship unlikely. Some reports have suggested, however, that chronic inflammatory processes may be a risk factor for sarcoma. Shrapnel, bullets, intramuscular iron injections, and foreign body implants have been implicated. This effect has been modeled in an acute mouse model of tissue injury.
Germline mutations can play an important role in the development of STSs ( Table 90.2 ). These genetic changes are identified with or similar to the genetic changes that are seen in corresponding sporadic sarcomas ( Table 90.3 ). Mechanistically, the proteins encoded by the altered genes are involved in maintenance of the genome through DNA repair and the cell cycle.
| Syndrome | Inheritance Pattern | Locus | Gene | Associated Soft Tissue Sarcomas |
|---|---|---|---|---|
| Familial gastrointestinal stromal tumor syndrome | AD | 4q12 | KIT | Gastrointestinal stromal tumor |
| Familial desmoid fibromatosis (Gardner syndrome) | AD | 5q21 | APC | Desmoid fibromatosis |
| Li-Fraumeni syndrome | AD | 17p13,22q11 | TP53, CHK2 | Multiple types |
| Neurofibromatosis type I (von Recklinghausen disease) | AD | 17q11 | NF1 | Malignant peripheral nerve sheath tumors |
| Retinoblastoma | AD | 13q14 | RB1 | Multiple types |
| Rhabdoid predisposition syndrome | AD | 22q11 | SNF5/INII | Malignant rhabdoid tumors |
| Werner syndrome | AR | 8p11–12 | WRN | Multiple types |
| Tumor Type | Characteristic Cytogenetic Events | Molecular Events | Frequency (%) | Diagnostically Useful? |
|---|---|---|---|---|
| Alveolar soft part sarcoma | t(X;17)(p 11;q25) | ASPL-TFE3 fusion | >90 | Yes |
| Extraskeletal myxoid chondrosarcoma | t(9;22)(q22;q12) | EWSR1-NR4A3 fusion | >75 | Yes |
| Clear cell sarcoma | t(12;22)(q13;q12) | EWSR1-ATF1 fusion | >75 | Yes |
| Desmoplastic small round cell tumor | t(11;22)(q13;q12) | EWSR1-WT1 fusion | >75 | Yes |
| Dermatofibrosarcoma protuberans | Ring form of chromosomes 17 and 22 | COLIA1-PDGFB fusion | >75 | Yes |
| t(17;22)(q21;q13) | COLIA1-PDGFB fusion | 10 | Yes | |
| Ewing sarcoma, peripheral primitive neuroectodermal tumor | t(11;12)(q24;q12) | EWSR1-FLII fusion | >80 | Yes |
| t(21;22)(q12;q12) | EWSR1-ERG fusion | 5–10 | Yes | |
| t(2;22)(q33;q12) | EWSR1-FEV fusion | <5 | Yes | |
| Fibrosarcoma, infantile | t(12;15)(q13;q26) | ETV6-NTRK3 fusion | >75 | Yes |
| Trisomies 8,11,17 and 20 | >75 | Yes | ||
| Gastrointestinal stromal tumor | Monosomies 14 and 22 | >75 | Yes | |
| Deletion of 1p | KIT or PDGFRA mutation | >25 >90 |
No Yes |
|
| Inflammatory myofibroblastic tumor | 2p23 rearrangement | ALK fusion genes | 50 | Yes |
| Leiomyosarcoma | Deletion of 1p | >50 | No | |
| Liposarcoma | ||||
| Well-differentiated | Ring form involving chromosome 12q | >75 | Yes | |
| Myxoid–round cell | t(12;16)(q13;p11) | FUS-DDIT3 fusion | >75 | Yes |
| Pleomorphic | Complex | EWSR1-DDIT3 fusion | <5 | Yes |
| Undifferentiated pleomorphic sarcoma | Complex | >90 | No | |
| Malignant peripheral nerve sheath tumor | Complex; NF1 loss | >90 | No | |
| Rhabdoid tumor | Deletion of 22q | INI1 inactivation | >90 | Yes |
| Rhabdomyosarcoma | ||||
| Alveolar | t(2;13)(q35;q14) | PAX3-FOXO1 fusion | >75 | Yes |
| t(1;13)(p26;q14), double minutes | PAX7-FOXO1 fusion | 10–20 | Yes | |
| Embryonal | Trisomies 2q, 8, and 20 | >75 | Yes | |
| Loss of heterozygosity at 11p15 | >75 | Yes | ||
| Synovial sarcoma | ||||
| Monophasic | t(X;18)(p11;q11) | SS18-SSX1 or SS18- | >90 | Yes |
| Biphasic | t(X;18)(p11;q11) | SSX2 fusion SS18-SSX1 fusion |
>90 | Yes |
The epidemiologic relationship between the development of STS and inherited syndromes associated with a predisposition to neoplasia (e.g., neurofibromatosis and Li-Fraumeni syndrome) has been appreciated for decades. For example, patients with neurofibromatosis have a 7% to 10% lifetime risk of developing an MPNST. A sudden increase in the size of any neurofibroma suggests malignant transformation. The mechanisms underlying the transformation from a benign neurofibroma to MPNST are not well understood.
However, loss-of-function mutations in the NF1 gene, which are found in patients with neurofibromatosis, result in activation of one of the ras signaling pathways, a well-known mechanism that has been identified in a variety of cancers. It has been observed that secondary MPNSTs (arising from a prior neurofibroma) have deletions of 17p (particularly 17p12–17p13.1) and mutations at the region of the TP53 tumor suppressor gene. Therefore it is postulated that an initial alteration in the NF1 gene contributes to the formation of a benign neurofibroma through activation of the ras pathway and that secondary mutations in the TP53 gene allow the transformation into MPNST. Because mutations in TP53 lead to an inability to control DNA damage via the cell cycle, they also enable rapid accumulation of other mutations, which encode mutant proteins that undoubtedly play an important role in sarcomagenesis.
Li-Fraumeni syndrome was identified when relatives of pediatric STS patients were noted to have an increased frequency of diverse and often multiple primary cancers. The neoplasms that were noted in relatives included some STSs, premenopausal breast cancers, brain tumors, adrenocortical carcinomas, leukemias, and occasional germ cell tumors. Follow-up of Li-Fraumeni kindreds over two decades has revealed that the majority of individuals develop cancer at young ages, with 79% of those affected being younger than 45 years at the time of diagnosis of malignancy. The observed cancer distribution in families is believed to fit a rare autosomal dominant mode of genetic transmission with high penetrance. Molecular genetic studies have identified germline TP53 mutations in the majority of patients with Li-Fraumeni syndrome. Germline mutations in CHK2 , another component of the cell cycle checkpoint machinery encoded by a gene located on 22q11, are responsible for another subgroup of patients with Li-Fraumeni syndrome, and other Li-Fraumeni–like patient kindreds without TP53 alteration have been described.
Pediatric patients with familial retinoblastoma have a 13q chromosomal deletion and an increased incidence of osteosarcoma and other neoplasms, including STS. The retinoblastoma (Rb1) protein is expressed ubiquitously in normal cells and is a well-known tumor suppressor, involved in maintaining the integrity of the genome through control of the cell cycle. Interesting to note, not only is the Rb1 gene that is mutated in osteosarcomas associated with retinoblastoma, but abnormality or absence of the Rb1 gene product has also been observed in multiple other malignancies, including sporadic osteosarcomas, breast cancer, small cell lung cancer, and STSs.
Familial adenomatous polyposis (FAP), a subset of which includes Gardner syndrome with its desmoid tumors, is caused by germline mutations in the APC gene. Patients with Gardner syndrome develop desmoid fibromatosis at a younger age than do patients with sporadic desmoids. With the use of prophylactic colectomy, progressive mesenteric desmoid tumor is now a significant cause of death of patients who have FAP, as are second neoplasms such as ampullary and duodenal cancers. The APC gene is involved in the Wnt cell-signaling pathway. One of the normal functions of the APC protein is to bind β-catenin. Thus loss-of-function mutations of APC result in the activation of transcription of oncogenes by β-catenin. Consistent with the importance of this pathway in desmoid tumors, loss of APC and mutations in CTNNB1 (encoding β-catenin) are also identified in sporadic desmoid fibromatosis.
Rhabdoid predisposition syndrome is due to inactivating germline mutations in the INI1 gene. Patients with this syndrome develop one or more extrarenal and/or renal rhabdoid tumors. INI1 is a member of the SWI/SNF protein complex, which controls gene expression globally through its ability to alter chromatin structure. Thus loss of INI1 gene expression results in other changes in gene expression, specifically activation of oncogenes. Rhabdoid tumors have loss-of-function mutations in both copies of the INI1 gene.
Werner syndrome is a rare genetic instability syndrome caused by mutations in the WRN gene. Affected patients age prematurely and are at greatly increased risk for a variety of cancers, including STSs. The WRN gene encodes a protein involved in DNA repair and loss of WRN protein function leads to genetic instability, accumulation of genetic mutations, and ultimately predisposition to rapid aging and cancer.
Germline mutations in the KIT oncogene are found in patients with familial gastrointestinal stromal tumor (GIST) syndrome. Activating mutations in KIT or PDGFRA are also identified in over 90% of sporadic GISTs. Patients with the familial syndrome develop, to varying degrees, skin hyperpigmentation, urticaria pigmentosa, and cutaneous mast cell disease in addition to one or more GISTs. Activating KIT mutations have been shown to lead to ligand-independent activation of the KIT receptor tyrosine kinase pathway, which results in dysregulated cell growth, and are thought to be the first step in the pathogenesis of GISTs. Interesting to note, the identification of the important role of KIT in the pathogenesis of GISTs has led to treatment with imatinib, the oral multitargeted receptor tyrosine kinase inhibitor (see Prognostic Factors as Therapeutic Targets , and Gastrointestinal Stromal Tumors ).
CStratakis and Carney reported a syndrome with paraganglioma and wild-type GIST called Carney Stratakis syndrome that arises from germline mutations in the mitochondrial succinate dehydrogenase (SDH) subunit B, C, or D. This trial also included pulmonary chondroma mutations in SDH subunits B. Interesting to note, these GISTs arise without mutation in KIT or PDGFRA.
Sarcomas tend to fall into two major subsets. One group has a complex karyotype that is associated with aneuploidy. In the other group, tumors are cytogenetically simple and are characterized by near diploid karyotypes with few chromosomal rearrangements, resulting in the formation of fusion proteins. These translocations are highly diagnostic of specific histologic subtypes of STSs. Also, included within the cytogenetically simple STSs are GISTs, which are characterized by a specific activating mutation of the KIT or PDGFRA gene, but otherwise relatively limited genetic abnormalities. Finally, other than the common loss of expression of the metabolic enzyme ASS1 that occurs in 88% of sarcomas, most sarcoma subtypes demonstrate their own underlying biology because sarcomas are as diverse a set of tumors as carcinomas.
A large number of sarcomas have been found to have consistent chromosomal abnormalities (see Table 90.3 ). These chromosomal rearrangements are important diagnostically, may be important prognostically (see Potential Molecular Prognostic Factors ), and have shed light on the pathogenesis of sarcomas. Some of the specific translocations, such as those seen in dermatofibrosarcoma protuberans (DFSP) or tenosynovial giant cell tumor (TGCT), provide targets for pharmacologic therapy. Benign soft tissue neoplasms also harbor chromosomal rearrangements.
Chromosomal translocations are the most common cytogenetic abnormality in soft tissue neoplasms and are likely responsible for the initiation of tumorigenesis in most cases. Deletions and trisomies have also been reported and are thought to represent secondary changes involved in tumor progression. Deletions tend to represent loss of tumor suppressor genes, whereas trisomies suggest the presence of an oncogene. Although we know much about the principal tumorigenic events in many sarcomas, defining the secondary changes has been much more problematic and is an area of intense study with the advent of large-scale tumor DNA and RNA sequencing and related genomic techniques. The Sarcoma Tumor Cancer Genome Project provides large comprehensive data set on sarcomas.
Cloning and molecular analysis of the various genetic aberrations that characterize different sarcomas have revealed the different pathogenetic mechanisms that underlie these tumors. Translocations typically create chimeric transcription factors or growth factors that result in deregulation of transcription or growth control. A typical example of a chimeric transcription factor is the PAX3-FOXO1 fusion protein, which has been shown to activate a complex myogenic transcriptional program when the protein is expressed in a fibroblast cell line. Infantile fibrosarcoma is characterized by a translocation involving chromosomes 12 and 15 that encodes a chimeric ETV6-NTRK3 constitutively activated growth factor receptor, which remarkably is found in other cancers such as secretory breast cancer, a subset of acute myelogenous leukemia (AML), and a form of salivary gland carcinoma. Additional NTRK fusions have been identified in other STSs, with therapeutic potential. Other oncogenic proteins appear to act by a mechanism that remodels chromatin structure, which is known to have a profound influence on gene expression (e.g., INI1 mutations in rhabdoid tumors).
Specific chromosomal rearrangements are very useful in the diagnosis of STSs. Beyond the obvious benefit of providing further objective proof of a diagnosis in morphologically typical cases, the detection of chromosomal aberrations may facilitate the diagnosis of lesions that are difficult to characterize with standard histopathologic, ultrastructural, and immunohistochemical techniques. For example, the presence of the translocation t(X;18)(p11;q11) has been used to confirm the diagnosis of synovial sarcoma in poorly differentiated cases that were diagnostically very challenging. Similarly, the finding of the characteristic translocation t(11;22)(q24;q12) in a small round blue cell tumor supports the diagnosis of Ewing sarcoma or primitive neuroectodermal tumor (PNET). By the same token, a morphologic sarcoma subtype without an expected translocation may suggest a variant translocation, as has been observed in Ewing sarcoma–like small round blue cell tumors that lack the EWSR1-FLI1 translocation but rather contain CIC-DUX4 or a pericentric X chromosome.
Translocations may be identified through a variety of genetic analyses, spanning older techniques such as cytogenetic analysis to fluorescence in situ hybridization (FISH), reverse transcriptase polymerase chain reaction, and RNA sequencing. A detailed description of these techniques is beyond the scope of this chapter, but each technique has its advantages and disadvantages. Cytogenetic analysis requires fresh (living) tissue, because the cells need to be cultured before karyotypic analysis, and largely has been supplanted by newer techniques. FISH does not require fresh or frozen tissue and frequently may be performed with paraffin-embedded material; it is a standard diagnostic tool in many pathology laboratories. It is noted that evidence of a translocation from FISH is not absolutely diagnostic of a sarcoma subtype, because one gene such as EWSR1 may be involved in many different types of sarcomas, and such data must be incorporated with other data regarding the tumor sample. Reverse transcriptase polymerase chain reaction is a more sensitive technique that may be performed on fresh, frozen, or paraffin-embedded tissues. The major drawback of reverse transcriptase polymerase chain reaction is the relatively high false-positive rate, which results from its sensitivity. Meticulous care is required to prevent problems from contamination. Sequencing of the RNA complement of the genome allows for detection of mutations, translocations, and to some degree gene amplification, and is expected to be used more frequently over time.
Many sarcomas are characterized by several different translocations, most of which are mutually exclusive (see Table 90.3 ). For instance, alveolar rhabdomyosarcoma is characterized by a translocation involving chromosomes 2 and 13, which results in fusion of the PAX3 and FOXO1 genes, or by a translocation involving chromosomes 1 and 13, which results in fusion of the PAX7 and FOXO1 genes. Sarcomas within a subtype may also have differences in the specific exons that are involved in each of these different translocations. It has been proposed that this heterogeneity may result in differences in prognosis (see Potential Molecular Prognostic Factors ). For example, Ewing sarcoma or PNET and synovial sarcoma possess genetic variations that have been suggested to have prognostic significance, further underscored by the Ewing-like sarcomas that lack EWSR1 fusions but contain other translocations. Future research, it is hoped, will establish whether cytogenetic and molecular factors may be used as a basis for therapeutic decisions and the prediction and evaluation of response to treatment.
The identification of genetic alterations with high specificity for different sarcomas will also enable identification of specific therapeutic targets. This has already resulted in the successful treatment of a variety of sarcomas—for example, GISTs, DFSP, and TGCT/pigmented villonodular synovitis (PVNS), and to a lesser degree angiosarcoma, desmoid tumor, and Ewing sarcoma. About 90% of GISTs harbor activating mutations in the KIT oncogene, which result in ligand-independent activation of the KIT receptor tyrosine kinase pathway. Imatinib, a small-molecule drug that is administered orally and inhibits the KIT receptor tyrosine kinase, is highly efficacious in the treatment of GIST (see later). Ninety percent of DFSP is characterized by translocations involving the COL1A1 and PDGFB genes, which result in activation of the platelet-derived growth factor-β (PDGFβ) pathway in what is believed to be an autocrine fashion. Imatinib is active against the PDGFβ pathway and has been shown to be effective in the treatment of a small number of DFSPs. In a similar way, TGCT/PVNS with its t(1;2) involving COL6A3-CSF1 is responsive to imatinib or PLX3397 by virtue of its inhibition of the CSF1 receptor.
It is noteworthy to mention two common benign soft tissue tumors—leiomyomas and lipomas—that have a high frequency of chromosomal rearrangements of chromosome 12q that involves the high-mobility protein group gene HMGIC/HMGA2. These translocations are not seen in the corresponding leiomyosarcomas or liposarcomas, indicating that in these tumors, the benign form is not a precursor of the malignant counterpart. Other benign soft tissue neoplasms contain specific translocations, such as nodular fasciitis (t[17;22][p13;q13] MYH9-USP6 ) or angiomatoid fibrous histiocytoma (t[12;22][q13;q12] EWSR1-ATF1 ) , underscoring the idea that translocations can occur in tumors anywhere in the spectrum of connective tissue tumors from benign to malignant.
A second major subset of STSs is characterized by aneuploidy and the lack of specific fusion genes. This group of sarcomas includes such tumors as leiomyosarcomas, MPNST, and fibrosarcomas (see Tables 90.3 and 90.4 ). These tumors tend to occur in older patients and appear to have a relatively high frequency of mutations in the p53 and retinoblastoma (Rb) signaling pathways. These tumors are characterized by chromosomal gains and losses that presumably target tumor suppressor genes (in the event of losses) and oncogenes (in the event of gains). A special case appears to be well-differentiated or dedifferentiated liposarcoma, with amplifications of cell cycle regulatory genes CDK4 and HDM2 and others on chromosome 12q. The cooperation of these genes to create a unique type of aneuploid sarcoma remains a current challenge in the field. Supporting the clinical data from syndromic sarcomas, alterations disrupting chromosomal mechanics and DNA repair appear to be involved in sarcomagenesis.
| Type of Sarcoma | Resembles |
|---|---|
| Fibrosarcoma (other than congenital) | Fibrous tissue |
| Leiomyosarcoma | Smooth muscle |
| Undifferentiated pleomorphic sarcoma | Poorly differentiated |
| Osteosarcoma | Bone |
| Chondrosarcoma (types other than extraskeletal myxoid) | Cartilage |
| Liposarcoma (types other than myxoid) | Fat |
| Embryonal rhabdomyosarcoma | Skeletal muscle |
| Malignant peripheral nerve sheath tumor a | Nerve sheath |
| Angiosarcoma | Endothelium |
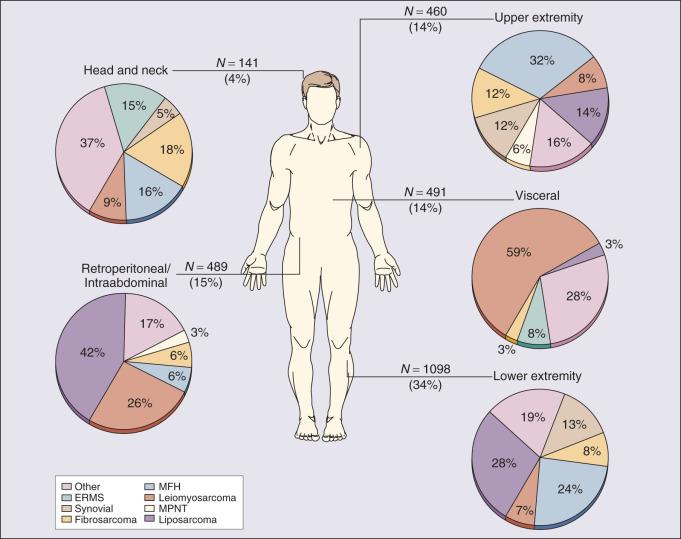
Prospectively collected databases of STS patients, some of which contain data on over 10,000 patients, demonstrated that STSs are described in essentially all anatomic sites. The anatomic sites and site-specific histologic subtypes of 4207 sarcomas treated at a single referral institution are outlined in Fig. 90.1 . Approximately half of all STSs occur in the extremities (lower, 34%; upper, 14%), where the most common histopathologic subtypes are well-differentiated, myxoid–round cell liposarcoma (28%), and UPS (formerly termed malignant fibrous histiocytoma [MFH]) (24%). Retroperitoneal sarcomas make up 15% of all STSs, well-differentiated or dedifferentiated liposarcoma being the predominant histologic subtype (42%). Visceral sarcomas make up an additional 14%, and the head and neck sarcomas make up approximately 4%.
In broad terms, sarcomas may be classified into neoplasms that arise in bone and those that arise from the soft tissues. However, even this seemingly simple distinction is fraught with difficulty, as is illustrated by Ewing sarcoma. In childhood, Ewing sarcomas most commonly arise in close association with bone and are often classified as bone sarcomas. However, it is now clear that these tumors can also arise in soft tissues, which occurs more commonly in adults. So are Ewing sarcomas bone sarcomas or STSs, and does it really matter in terms of classification? Sarcomas of the soft tissues may be further grouped into those that arise from viscera (gastrointestinal, genitourinary, and gynecologic organs) and those that arise from nonvisceral soft tissues (muscle, tendon, adipose, pleura, and connective tissue).
An alternative way to index STSs is by their differentiation. Tumors may be grouped broadly into adipocytic tumors, fibroblastic or myofibroblastic tumors, so-called fibrohistiocytic tumors, smooth muscle tumors, pericytic (perivascular) tumors, PNETs, skeletal muscle tumors, vascular tumors, osseous tumors, and tumors of uncertain differentiation ( Table 90.5 ). Classification is based on clinical, histologic, ultrastructural, immunohistochemical, and genetic features. Electron microscopic evidence of cellular substructures, neurofibrils, microfilaments, actin-myosin complexes, and dense bodies, has historically been used to define the tissue of origin. However, the widespread availability of commercial antibodies for immunohistochemical analysis has all but eliminated the need for electron microscopic examination. Immunohistochemical staining for proteins that are characteristic of smooth muscle (smooth muscle actin and desmin), skeletal muscle (muscle-specific actin, desmin, and myogenin), blood vessels (factor VIII, CD34, and CD31), and epithelial tissue (epithelial membrane antigen and cytokeratins) often facilitates reliable classification, with genetic proof of a sarcoma translocation considered iron-clad evidence of a specific sarcoma subtype. However, because the identical translocation may be found in more than one type of cancer, such as the ASPL-TFE3 translocation of both alveolar soft part sarcoma and papillary renal cancer, translocation data cannot be used in isolation.
| ADIPOCYTIC SARCOMAS |
|
| FIBROBLASTIC AND MYOFIBROBLASTIC SARCOMAS |
|
| SO-CALLED FIBROHISTIOCYTIC SARCOMAS |
| Undifferentiated pleomorphic sarcoma (UPS) |
| SMOOTH MUSCLE SARCOMAS |
| Leiomyosarcoma |
| SKELETAL MUSCLE SARCOMAS |
|
| VASCULAR SARCOMAS |
|
| OSSEOUS SARCOMAS |
| Extraskeletal osteosarcoma |
| SARCOMAS OF UNCERTAIN DIFFERENTIATION |
|
| NOTOCHORD-DERIVED TUMOR BEHAVING–LIKE SARCOMAS |
| Chordoma |
The tissue of origin classification scheme is the basis for the 2013 World Health Organization classification system for sarcomas. The World Health Organization classification system is reproducible for most sarcomas. As the degree of histologic differentiation declines, however, the determination of the tissue of origin becomes increasingly difficult. In particular, despite advanced immunohistochemical and molecular analyses, determining the tissue of origin for some soft tissue tumors may be difficult, occasionally arbitrary, and sometimes impossible. This leads to significant disparities in diagnoses among pathologists. Discrepancies between the original histologic diagnosis and the subsequent diagnosis by an expert reviewer have been noted in as many as 25% of cases, although these data predate modern immunohistochemical and genetic analyses. Review of suspicious tissue specimens at an expert center is therefore wise, because the degree of expertise in correctly diagnosing some of the 50 or more types of rare and unusual sarcomas is directly related to the number of sarcomas that a pathologist has seen, and increasingly the specific molecular diagnostic tools needed to identify rare sarcoma subtypes.
It is important to classify STSs as precisely as possible because of major differences in their clinical behavior and in their susceptibility to different therapies. For example, a few STSs, including epithelioid sarcoma, clear cell sarcoma, angiosarcoma, and rhabdomyosarcoma have a greater risk of regional lymph node metastasis. In one single-institution study, the overall rate of nodal metastasis at the time of sarcoma presentation was under 3%; however, the rate was much higher for angiosarcoma (13%), embryonal rhabdomyosarcoma (14%), and epithelioid sarcoma (17%).
Patterns of distant metastases also differ for subtypes of sarcoma. For example, myxoid–round cell liposarcoma tends to metastasize to soft tissue sites, including the retroperitoneum, and to the spine and pelvis and other bone marrow sites, and patients with myxoid–round cell liposarcoma often present with metastatic disease. If a myxoid or round cell liposarcoma is identified in the abdomen, the thighs should be examined for an occult primary tumor. The majority of what had been suspected to be primary myxoid liposarcomas of the abdomen and retroperitoneum are actually misdiagnosed well-differentiated or dedifferentiated liposarcomas that have myxoid features (and usually metastasize far less often than myxoid–round cell liposarcoma).
Patterns of local spread also differ dramatically among subtypes of sarcoma. For example, DFSP has a propensity to infiltrate subcutaneous adipose tissue in a manner that is very difficult to detect; therefore wide surgical excision of DFSP is essential. Mohs surgery has been used for DFSP, but the concern for recurrences 5 to 10 years after initial diagnosis makes one wary about anything less than a wide excision. When planning an operation for DFSP, the surgeon should regard the grossly observable lesion as the tip of the iceberg. Myxofibrosarcoma and, to an even greater extent, angiosarcoma also spread very diffusely and may be very difficult to resect with negative margins owing to microscopic tumor insinuation 2 cm or more from the gross tumor.
Histologic classification alone does not always provide enough information to predict the clinical behavior of STSs. For many sarcomas, histologic grading provides additional information that can aid in predicting biologic behavior and planning treatment. The spectrum of grades varies among specific histologic subtypes ( Fig. 90.2 ). For example, liposarcomas exhibit wide variations in grade, whereas Ewing sarcomas or PNETs are always considered high grade. In careful comparative multivariate analyses, histologic grade has been the most important prognostic factor in assessing the risk for distant metastasis and tumor-related mortality. Several grading systems have been proposed, but there is no consensus regarding the specific morphologic criteria that should be used in the grading of STSs. The two most important criteria appear to be the mitotic index and the extent of tumor necrosis.
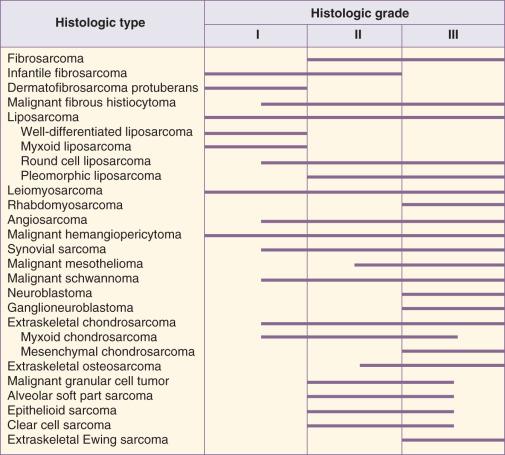
Two of the most commonly used grading systems are the US National Cancer Institute (NCI) system developed by Costa and the FNCLCC system (Federation Nationale des Centres de Lutte Contre le Cancer) developed by the French Federation of Cancer Centers Sarcoma Group. The NCI system is based on the tumor's histologic subtype and amount of tumor necrosis, but cellularity, nuclear pleomorphism, and mitotic index are considered for certain subtypes. The FNCLCC system uses a score generated by evaluation of three parameters: tumor differentiation, mitotic rate, and amount of tumor necrosis. The prognostic values of these two grading systems were retrospectively compared in a population of 410 adult patients with nonmetastatic STS. Significant discrepancies were observed in one-third of cases. An increased number of grade III tumors, a reduced number of grade II tumors, and better correlation with overall and metastasis-free survival were observed in favor of the FNCLCC system. Thus in the absence of other comparative data, the FNCLCC system appears to be the best presently available grading system. The FNCLCC has therefore been incorporated into the American Joint Committee on Cancer (AJCC) version 7 STS staging system.
The majority of patients with STS have a painless mass, although pain is noted at presentation in up to one-third of cases ; synovial sarcoma in particular may be most associated with a painful mass, among the sarcomas. Delay in diagnosis of sarcomas is common; the most common incorrect diagnosis for extremity and trunk lesions is lipoma or hematoma.
Physical examination should include an assessment of the size and mobility of the mass. Its relationship to the fascia (superficial versus deep) and nearby neurovascular and bony structures should be noted. A site-specific neurovascular examination and assessment of regional lymph nodes should also be performed.
Biopsy of the primary tumor is essential for most patients with soft tissue masses. In general, any biopsy should be performed on any soft tissue mass in an adult that is asymptomatic or enlarging, is larger than 5 cm, or persists beyond 4 to 6 weeks. The preferred biopsy approach is generally the least invasive technique required to allow a definitive histologic diagnosis and assessment of grade. In most centers, core needle biopsy provides satisfactory tissue for diagnosis and has been demonstrated to result in substantial cost savings compared with open biopsy; core needle biopsy (usually with multiple passes) also yields adequate material for molecular testing, with fine-needle aspiration considered inadequate by most practitioners. Direct palpation may be used to guide needle biopsy of most superficial lesions, but less accessible sarcomas often require an image-guided biopsy in order to safely sample the most heterogeneous component of the mass. Needle tract tumor recurrences after closed biopsy are rare but have been reported, leading some surgeons to advocate tattooing the biopsy site for subsequent excision or for inclusion in radiotherapy treatment volumes ( Fig. 90.3 ). Owing to the frequent difficulty in accurately diagnosing these lesions even when adequate tissue is available, fine-needle aspiration is not recommended for initial diagnosis. The major use of fine-needle aspiration in most centers is in the diagnosis of suspected recurrent sarcoma.
Incisional or excisional biopsy is rarely required but may be performed when a definitive diagnosis cannot be achieved through less invasive means. Several technical points merit comment. Biopsy of relatively small, superficial masses that can easily be removed should entail complete excision with microscopic assessment of surgical margins. Incisional and excisional biopsies should be performed with the incision oriented longitudinally (for extremity lesions) to facilitate subsequent wide local excision. The incision should be centered over the mass at its most superficial point. Care should be taken not to raise tissue flaps. Meticulous hemostasis should be ensured to prevent dissemination of tumor cells into adjacent tissue planes by hematoma. All excisional biopsy specimens should be sent fresh, sterile, and anatomically oriented for pathologic analysis. At definitive resection of a previously biopsied sarcoma, the previous surgical biopsy scar should be excised en bloc with the tumor.
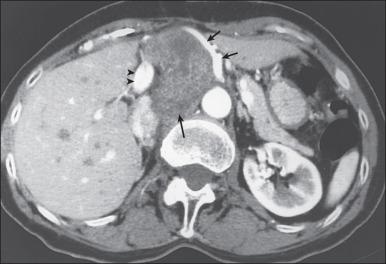
For soft tissue masses of the extremities, magnetic resonance imaging (MRI) has been regarded as the imaging modality of choice ( Fig. 90.4 ). This is because MRI enhances the contrast between tumor and muscle and between tumor and adjacent blood vessels and provides multiplanar definition of the lesion. Despite the fact that a study by the Radiation Diagnostic Oncology Group that compared MRI and computed tomography (CT) in patients with malignant bone ( n = 183) and soft tissue ( n = 133) tumors showed no specific advantage of MRI over CT from a diagnostic standpoint, the majority of musculoskeletal radiologists and almost all surgical oncologists prefer MRI for soft tissue tumors. For pelvic lesions, the multiplanar capability of MRI may provide superior single-modality imaging ( Fig. 90.5 ), although similar techniques are commonly available with CT scans. The multiplanar capability is also helpful for visualization of disease in non-coplanar ways when conformal radiotherapy technique is being used and is an especially helpful adjunct in using image fusion techniques or to visualize peritumoral edema that may harbor sarcoma cells. In the retroperitoneum and abdomen, CT usually provides satisfactory anatomic definition of the lesion ( Fig. 90.6 ) and is a very useful modality for imaging metastatic disease in the lungs and abdomen, given the averaging of signal that occurs with normal patient respiration and peristalsis during the much lengthier MRI procedure. Occasionally, MRI with gradient sequence imaging may better delineate the relationship of the tumor to midline vascular structures, particularly the inferior vena cava and aorta. Older and more invasive studies such as angiography or cavography are almost never used for the evaluation of STSs.

The usefulness of fluorine-18 ( 18 F) fluorodeoxyglucose–positron emission tomography (FDG-PET) with CT registration images (PET-CT) in the evaluation and treatment of STS has been a subject of numerous studies and has been reviewed in detail elsewhere. The technique uses radiolabeled glucose analogues, which are taken up at increased rates by malignant tumors. Pilot studies of PET in STS suggest that by evaluating tumor metabolic activity, PET scans may allow for noninvasive assessment of tumor grade. PET may be helpful in the assessment of locally recurrent STS and in the evaluation of response to therapy. PET may also be useful in differentiating benign peripheral nerve sheath tumors and MPNSTs and plexiforms in neurofibromatosis patients, although more prospective research is needed. The role and cost-effectiveness of PET in the staging of STS remain undefined; further prospective studies will be required in order to fully define the role for FDG-PET in the diagnosis, evaluation, and treatment of STS. For example, for the vast majority of patients with metastatic GIST, PET-CT appears to add very little to the assessment of response to tyrosine kinase inhibitors, given the excellent image quality of contrast-enhanced CT scan.
The relative rarity of STSs, the anatomic heterogeneity of these lesions, and the presence of more than 50 recognized histologic subtypes of variable grade have made it difficult to establish a functional system that can accurately stage all forms of this family of neoplasms. The seventh edition of the AJCC (Union for International Cancer Control [UICC]) system is the most widely used staging system for STSs. This staging system was first published in 1977, and incorporates histologic grade into the conventional TNM system ( Table 90.6 ). The 2010 edition classification functionally eliminated tumor depth in staging, and classifies node-positive disease without metastases (N1M0) as stage III as compared with stage IV in the sixth edition of 2002. In addition, a unique staging system for GIST has been generated. For the detailed background of these changes, the reader is referred to a more complete discussion.
| Stage | Grade | Tumor | Nodes | Metastasis |
|---|---|---|---|---|
| IA | G1, GX | T1a, T1b | N0 | M0 |
| IB | G1, GX | T2a, T2b | N0 | M0 |
| IIA | G2, G3 | T1a, T1b | N0 | M0 |
| IIB | G2 | T2a, T2b | N0 | M0 |
| III | G3 | T2a, T2b | N0 | M0 |
| Any | Any | N1 | M0 | |
| IV | Any G | Any T | Any N | M1 |
| G: PRIMARY TUMOR GRADE | ||||
| The FNCLCC tumor grade is used (three-stage system). | ||||
| G1 | Low grade | |||
| G2 | Intermediate grade | |||
| G3 | High grade | |||
| GX | Grade unknown | |||
| T: PRIMARY TUMOR SIZE AND LOCATION | ||||
| T1 | Tumor ≤5 cm | |||
| T2 | Tumor >5 cm | |||
| a | Superficial to investing fascia (irrelevant in version 7) | |||
| b | Deep to investing fascia (irrelevant in version 7) | |||
| N: LYMPH NODES | ||||
| N0 | No involved nodes | |||
| N1 | Regional nodal involvement | |||
| M: METASTASES | ||||
| M0 | No metastatic disease | |||
| M1 | Metastatic disease present | |||
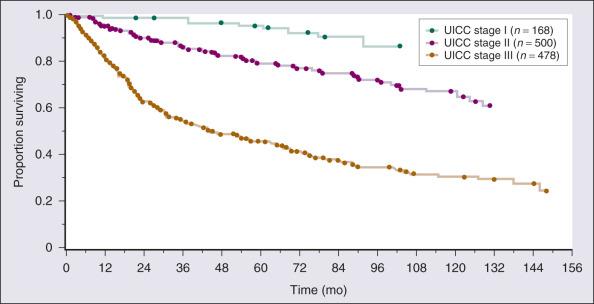
Three distinct histologic grades (G1–3) are recognized according to the French (FNCLCC) grading system, in the interest of consistency among institutions. Histologic grade and tumor size are the primary determinants of clinical stage (see Table 90.6 ). Tumor size is further substaged as “a” (a superficial tumor that arises outside the investing fascia) or “b” (a deep tumor that arises beneath the fascia or invades the fascia), although it does not affect stage in AJCC version 7. Stage-specific survival plots based on prior versions of the AJCC criteria are outlined in Fig. 90.7 .
A major limitation of the present staging system is that it does not take into account the anatomic site of STS, patient age, or histologic type, nor does it is necessarily use enough size categories—all factors that appear to be important determinants of outcome. Patients with retroperitoneal and visceral sarcomas have a worse overall prognosis than do patients with extremity tumors. Although site is not incorporated as a specific component of any present staging system, outcome data should be reported on a site-specific basis when feasible, because this also affects survival.
Nomograms and newer techniques such as bayesian belief networks may better help specify risk for discussion with patients. These have been generated for both specific anatomic sites and for specific sarcoma histologies. It is difficult to envision a unique staging system for each of the more than 50 STS histologies, which argues that a multidimensional risk assessment as may be performed with nomograms should supersede traditional staging systems in the interest of accuracy, although this comes at the cost of simplicity. As the first example of this technique being used in STSs, Kattan and colleagues constructed and validated a nomogram to predict the probability of 12-year sarcoma-specific death based on a prospective series of patients ( Fig. 90.8 ). This tool is useful for individual patient counseling, follow-up scheduling, and clinical trial eligibility assessment and is further facilitated by being also available for personal computers and handheld devices. Nomograms have since been developed for specific anatomic sites such as the retroperitoneum, or for specific sarcoma subtypes, such as liposarcoma. These useful tools may be found online at www.nomograms.org .
A thorough understanding of the clinicopathologic factors that are known to affect outcome is essential in formulating a treatment plan for the patient with STS. Since the late 1970s, multivariate analyses of prognostic factors for patients with localized sarcoma have been reported. Although initial studies were limited by sample size, the largest databases from referral centers and a variety of national registries now contain data from thousands of patients, allowing for increasingly sophisticated analyses of a variety of subsets of sarcoma patients. Nonetheless, many of the primary principles regarding patient outcomes were made evident in those first studies.
The initial study of prognostic factors in extremity sarcoma from Memorial Sloan Kettering Cancer Center evaluated clinicopathologic prognostic factors in a series of 423 patients with localized extremity STS seen from 1968 to 1978. This analysis was among the first to discriminate between specific clinical end points, and clearly established the clinical profile of what is now accepted as the high-risk patient with extremity STS: the patient with a large (in this case >5 cm), high-grade, deep lesion. The adverse prognostic significance of a high tumor grade, deep tumor location, and tumor size over 5 cm was also noted in the report of the French Federation of Cancer Centers study of 546 patients with sarcomas of the extremities, head and neck, trunk wall, retroperitoneum, and pelvis.
A follow-up report evaluated clinicopathologic prognostic factors that had been documented prospectively in a population of 1041 patients with extremity STS. The end points for the multivariate analyses were local recurrence, distant recurrence (metastasis), and disease-specific survival. Results of the regression analyses for each of these end points are summarized in Table 90.7 as an example of the key prognostic factors that are ascertained in such studies. These results, using prospectively acquired data, confirm the initial observations made at that institution using an independent data set. In addition, the previously unappreciated prognostic significance of specific histologic subtypes and the increased risk for adverse outcome associated with a microscopically positive surgical margin or locally recurrent disease were noted. Unlike for other solid tumors, the adverse prognostic factors for local recurrence of STS are somewhat different from those that predict distant metastasis and tumor-related mortality (see Table 90.7 ). Therefore staging systems that are designed to stratify patients according to risk of distant metastasis and tumor-related mortality using these prognostic factors (such as the AJCC/UICC system) do not stratify patients according to risk of local recurrence.
| End Point | Adverse Prognostic Factor a | Relative Risk |
|---|---|---|
| Local recurrence | Age >50 years | 1.6 |
| Local recurrence at presentation | 2.0 | |
| Microscopically positive margin | 1.8 | |
| Fibrosarcoma | 2.5 | |
| Malignant peripheral nerve tumor | 1.8 | |
| Distant recurrence | Size 5–10 cm | 1.9 |
| Size >10 cm | 1.5 | |
| High grade | 4.3 | |
| Deep location | 2.5 | |
| Local recurrence | 1.5 | |
| Leiomyosarcoma | 1.7 | |
| Other nonliposarcoma histology | 1.6 | |
| Disease-specific survival | Size >10 cm | 2.1 |
| Deep location | 2.8 | |
| Local recurrence at presentation | 1.5 | |
| Leiomyosarcoma | 1.9 | |
| Malignant peripheral nerve sheath tumor | 1.9 | |
| Microscopically positive margin | 1.7 | |
| Lower extremity site | 1.6 |
a Adverse prognostic factors identified are independent by Cox regression analysis.
It should be emphasized that the prognostic factors that have been identified have been derived primarily from studies of patients with localized extremity sarcomas. Despite the fact that extremity sarcomas make up the majority of sarcomas, these results do not necessarily apply to the populations of patients with STS in other sites. For example, the AJCC staging system for STSs of retroperitoneum or viscera is nonsensical with respect to primary tumor depth (indicated as superficial or deep), because all tumors from this anatomic site are deep tumors, and staging systems specific for each clinical scenario may be more appropriate means to estimate clinical outcomes. Separate reviews of prognostic factors for sarcomas of the retroperitoneum, head and neck, gastrointestinal tract, colon and rectum, and uterus, synovial sarcomas, UPS (formerly termed malignant fibrous histiocytoma [MFH]), and Ewing sarcoma also have been reported.
As clinicopathologic factors affecting patient outcomes were being identified, examination of the tissue itself for factors predicting outcome became another topic of interest, and continues to this day in the form of more detailed molecular profiling of RNA expression, comparative genomic hybridization, tumor DNA or RNA sequencing, and analysis of DNA modifications such as methylation patterns in specific tumors. Some of the first parameters that were evaluated for prognostic significance included p53, HDM2 (the human version of the murine p53 interacting protein MDM2), Ki-67, altered expression of the retinoblastoma gene product (pRb) in high-grade sarcomas, and the importance of the specific type of SS18-SSX fusion transcripts in synovial sarcoma or EWSR1-FLI1 fusion transcripts in Ewing sarcoma. Notably, although many individual immunohistochemical markers have been examined as prognostic factors in outcome, data are generally conflicting. It is well recognized that there is variability in immunohistochemistry in both technique (which may vary among laboratories) and assessment (what may be 2+ staining to one investigator is 3+ or 4+ staining to another).
As one of the first examples of analyzing multiple immunohistochemical markers with respect to outcome, Heslin and colleagues evaluated the potential prognostic significance of pRb, p53, HDM2, and Ki-67 with immunohistochemical techniques in a population of 121 patients with primary, high-grade extremity sarcomas and compared these factors with conventional clinicopathologic prognostic factors (median follow-up, 64 months). Clinicopathologic and molecular factors that were found to be statistically significant adverse prognostic factors in both univariate and multivariate analyses for the separate end points of distant metastasis and tumor-related mortality included tumor size greater than 5 cm, microscopically positive surgical margin, and a Ki-67 score greater than 20 (>20% nuclear staining). Overexpression of p53 or HDM2 or deletion of pRb did not correlate with an increased risk of distant metastasis or tumor-related mortality. Similarly, data with respect to specific SS18-SSX translocation type (i.e., SSX1 or SSX2 ) and outcome have yielded conflicting results, and the specific EWSR1-FLI1 translocation does not appear to affect overall outcomes for patients. Finally, ASS1 expression was seen to inversely correlate with overall survival (OS) in osteosarcoma and myxofibrosarcoma.
With the increasing use of genomic analyses such as RNA expression arrays, comparative genomic hybridization, or tumor RNA sequencing, it is possible that expression or mutation-specific stratification may be possible within specific histologic types, in a manner analogous to current use of this technique to stratify breast cancer. In addition, these profiles have been applied to determining metastatic potential of primary tumors, and this approach could clearly be applicable to STSs, in which the presence or absence of metastases remains the most important prognostic factor. Phosphoprotein profiling could become useful for patient risk stratification, as has been examined in stage III rhabdomyosarcoma to predict outcome. This approach also holds future promise for both prognostic and therapeutic approaches (see later discussion), although phosphoprotein lability and tissue handling are concerns in the proper conduct of such studies. Specific cellular and molecular parameters have been identified as having independent prognostic significance, but there is currently no consensus on how specific molecular prognostic factors should be used in clinical practice. Validated markers may affect outcome prognostication in the future, but for the time being remain investigational.
The prognosis of patients with GISTs is often poor when treated with surgery alone, and these tumors do not respond to conventional systemic chemotherapy. Arguably the most exciting discovery in GIST research is the finding of the activity of kinase-directed therapy, which is discussed later in the chapter. The proto-oncogene KIT is the cellular homolog of the oncogene v-Kit (identified in a feline sarcoma virus). KIT (also termed CD117) encodes a transmembrane tyrosine kinase receptor that is structurally similar to PDGF and provides selective targets of key aberrations in the molecular signaling implicated in the pathogenesis of GISTs and other tumors (e.g., DFSP). An interesting additional feature of KIT expression in GIST is that different types and locations of mutations in KIT are independent risk factors in predicting disease-free survival independent of treatment with kinase receptor inhibitors. KIT expression was uniformly evident in a study of GIST cases not treated with imatinib. Of interest, KIT was highly phosphorylated in all cases, even in those samples that lacked demonstrable KIT mutations. Mutations are found in specific regions of the KIT gene (most commonly exon 9 and exon 11), and are associated with differing prognosis (see following discussion). In the era before imatinib, the subset of patients with exon 11 KIT mutations resulting in single amino acid substitutions (i.e., missense codon mutations) fared much better than did patients with deletion or insertion mutations of exon 11 (5-year recurrence-free survival [RFS]) rate of 89% ± 11% versus 37% ± 10%, respectively). A potential explanation for this finding is that exon 11 missense mutations are detected in lower-grade, favorable-outcome GISTs. Against this explanation is the fact that the metastatic GISTs with KIT exon 9 mutations appear to have a lower risk of relapse compared with exon 11 KIT mutation or PDGFRA mutant GISTs. Although it is conceivable that the type of mutation is a surrogate for the behavior of a GIST, it is also plausible that the type of mutation represents the initial pathogenetic mechanism, making it a true prognostic marker and target. It is apparent that the mutation in KIT is context dependent; KIT mutation and ETS transcription factor family member ETV1 overexpression appear to synergize to drive GIST development.
We have just now begun to see the implications and success of targeting genetic alterations in sarcomas that drive oncogenesis. GISTs were previously thought to be gastrointestinal leiomyosarcomas that were particularly resistant to cytotoxic chemotherapy. It was subsequently demonstrated that these tumors were derived from interstitial cells of Cajal or their precursors and were frequently characterized by point mutations in the KIT receptor tyrosine kinase and were clearly distinct from leiomyosarcomas. Subsequently, the tumors were treated with imatinib, which targets the KIT kinase, with dramatic results. Mutations in KIT were observed in exon 11 (juxtamembrane domain, seen in 71% of tumors), exon 9 (the extracellular region, 13%), exon 13 (first lobe of the split-kinase domain, 4%), and exon 17 (phosphotransferase domain, 4%). The subset of patients with exon 11 mutations resulting in point single amino acid substitutions (i.e., missense codon mutations) fared much better than did patients with deletion or insertion mutations of exon 11 (5-year RFS rate of 89% ± 11% versus 37% ± 10%, respectively). In addition, tumors that lack mutations in KIT appear to have mutations in PDGFα receptor gene PDGFRA, and those that have no mutation in KIT or PDGFRA are driven by loss of SDH expression, mutation in BRAF, or other genetic alterations such as gene fusions involving FGFR1 and NTRK3, all changes that are mutually exclusive of mutations in KIT. Finally, the type of mutation that is identified appears not only to predict response to imatinib, but also to suggest whether other kinase inhibitors such as sunitinib may have beneficial effects. Although to date the most common adult STSs have demonstrated few consistent mutations in kinase genes that are amenable to target inhibition, new options for treatment likely will be derived from more careful genetic analysis of specific sarcoma subtypes through use of newer sequencing techniques, comparative genomic hybridization, analysis of tumor microRNA (miRNA), and DNA methylation patterns.
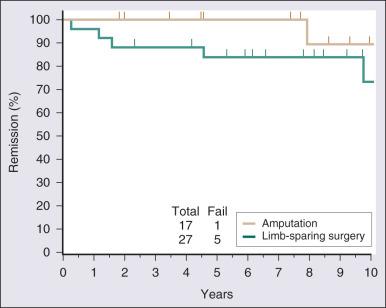
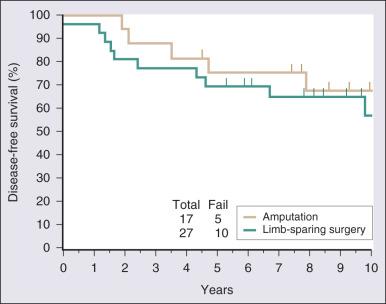
Surgical resection remains the cornerstone of therapy for localized STS, and the prototypical situations concern the management of lesions arising in the extremity, the most common anatomic site. Since the early 1980s, there has been a marked decline in the rate of amputation as the primary therapy for extremity STS. With the widespread application of multimodality treatment strategies, fewer than 10% of patients undergo amputation. The current use of limb-sparing multimodality treatment approaches for patients with extremity sarcoma is largely based on a randomized prospective study from the US NCI in which patients with extremity sarcomas that were amenable to limb-sparing surgery were randomized to receive amputation or limb-sparing surgery with postoperative radiotherapy. Both arms of this trial included postoperative chemotherapy with doxorubicin, cyclophosphamide, and methotrexate. With more than 9 years of follow-up evaluation, 5 (19%) of 27 patients randomly assigned to receive limb-sparing surgery and postoperative radiation with chemotherapy had local recurrences, as compared with 1 (6%) of 17 patients in the amputation plus chemotherapy arm ( P = .22; Fig. 90.9 ). The disease-free survival rate was 63% for limb-sparing surgery versus 71% for amputation ( P = .52; Fig. 90.10 ), and the OS rate was 70% for limb-sparing surgery versus 71% for amputation ( P = .97). This study established that for patients in whom limb-sparing surgery is an option, a multimodality approach using limb-sparing surgery combined with postoperative radiotherapy yields disease-related survival rates comparable to those for amputation while simultaneously preserving a functional extremity.
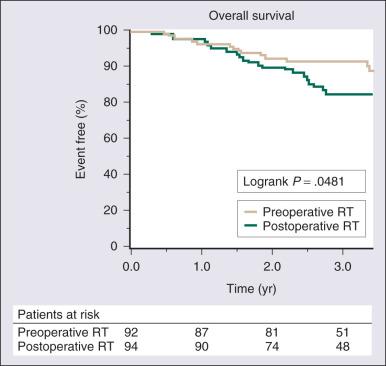
Currently, at least 90% of patients with localized extremity sarcomas can undergo limb-sparing procedures. Most surgeons consider definite major vascular, bony, or nerve involvement to be a relative indication for amputation. Complex en bloc bone, vascular, and nerve resections with interposition grafting may be undertaken, but the associated morbidity is high. Therefore for a few patients with critical involvement of major bony or neurovascular structures, amputation remains the only surgical option but offers the prospect of prompt rehabilitation with excellent local control and survival. An approach approved in Europe for selected patients with locally advanced tumors is limb perfusion, in which a tourniquet is placed proximal to the tumor on a limb, the blood is flushed out, and a perfusate of tumor necrosis factor (TNF) and chemotherapy is administered through a recirculating pump. This technique has allowed for limb salvage even in patients with otherwise unresectable disease; chronic edema is the most common long-term complication. However, TNF is unavailable in the United States, either for patient care or for clinical trials.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here