Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Editors' comment : The role of the renin–angiotensin–aldosterone system (RAAS) in preeclampsia has intrigued investigators for decades. Chesley had a chapter devoted to this topic in his first edition, and he would have been delighted by some of the novel twists that have occurred in this important area, such as the discovery of agonistic autoantibodies (AT1-AA) and that Angiotensin (Ang)1-7 has potent vasodilatory activity. In the current edition, we address salt intake and demonstrate that sodium can be stored at higher concentrations than previously expected in the interstitium and that this influences the immune system. Thus, we combine the subjects of the body fluid volumes, RAAS, and AT1-AA as an intricate regulatory system.
The renin–angiotensin–aldosterone system (RAAS) is one of the most evolutionarily conserved blood pressure and volume regulating systems in vertebrates. Importantly, during human pregnancy, there is a ~12 kg weight gain, as well as a 30%–50% increase in extracellular fluid, plasma, blood volume, and total body water. Also striking is a resetting of the thresholds for vasopressin secretion and thirst, plasma osmolality averaging 10 mOsm below nonpregnant levels (see Chapter 14 ). There is a marked stimulation of the RAAS and other potent mineralocorticoids that accompanies these changes, while opposing salt-retaining influences are increases in glomerular filtration rate (GFR) and a rise in the plasma concentration of several natriuretic hormones. Therefore, pregnancy is a sensitive state relying on a multifactorial autoregulation of blood pressure control mechanisms and body fluid volume homeostasis.
Alterations in the RAAS accompany the development of hypertension. Women with preeclampsia have long been known to have increased vascular sensitivity to angiotensin II in the absence of elevated angiotensin II or plasma renin activity. More recently, alterations in either the vasodilatory Ang 1–7 or activating autoantibodies to the angiotensin II type I receptor (AT1-AA) have been noted to occur during preeclampsia. AT1-AA were found to be present in the serum of preeclamptic women at much higher levels compared with sera from nonpregnant women or pregnant women who went on to have normal pregnancies. Therefore, in recent years, much research has been performed to determine the contribution of AT1-AA to the pathophysiology associated with preeclampsia. AT1-AA binds to and activates the AT1 receptor and induces signaling in vascular cells, including activating protein 1, calcineurin, and nuclear factor kappa-B activation, which can be blocked by an AT1 receptor antagonist. , , This signaling results in increased reactive oxygen species (ROS), sFlt-1 production, and plasminogen activator inhibitor-1 and endothelin-1, all of which have been implicated in preeclampsia. , , More recent studies reveal an important role for AT1-AA in causing the increase in renal and vascular sensitivity to angiotensin II (Ang II). , In addition to being elevated during preeclampsia, AT1-AA has also been reported to be increased in postpartum women. Hubel and colleagues demonstrated that AT1-AA correlated with insulin resistance and sFlt-1 and does not regress completely after delivery. Although these autoantibodies have been linked to poor placentation and abnormal renal function, their role in the hypertensive state of preeclampsia has yet to be fully elucidated. , Furthermore, the importance of AT1-AA after preeclampsia, especially in the context of increased long-term cardiovascular risk, remains to be determined.
Resolving the debate concerning the cause of the increased plasma volume (does it represent “underfill,” “overfill,” or “normal fill”?) could have important implications for management of complications during preeclampsia. Plasma volume generally decreases in preeclampsia (most marked in eclampsia), while interstitial water (edema) may increase further or remain unchanged. Levels of all components of the RAAS are decreased compared to normal pregnancy while the increased incidence of AT1-AA is associated with increased severity of the disease. Along with the controversial AT1-AA, there is an evolving literature on the role of the vasodilating peptide Ang 1–7 in normal pregnancy and preeclampsia. Finally, evolving thoughts regarding treating preeclampsia with sodium loading or plasma volume expansion, a challenging view, were revisited in light of the discovery of genetic mutations leading to inefficient aldosterone production in preeclampsia. This chapter addresses these volume regulatory pathways as we plunge forward to seek better, more innovative, and safe therapeutics for managing edema and alterations in blood pressure during preeclampsia.
If preeclampsia is the “disease of theories,” one of the more recently accepted is that agonistic autoantibodies can participate as direct mediators of increased vascular sensitivity and may induce alterations in volume homeostasis. Agonistic autoantibodies directed at the angiotensin II AT1 receptor induce AT1 receptor signaling and were first detected in patients with malignant hypertension. When these were observed in a hypertensive woman with a history of preeclampsia, it prompted a cohort investigation for such antibodies in archived sera from women with preeclampsia. The epitope shows signaling events in vascular smooth muscle cells and trophoblasts that could contribute to the development and signs of preeclampsia. Moreover, AT1-AA has also been observed in a reduced uterine perfusion rat model of hypertension and in pregnant rat models of hypertension induced by elevated cytokines such as TNF-α, IL-6, and IL-17. Once expressed in pregnancy, AT1-AA is capable of eliciting sFlt-1 production in human trophoblasts and in pregnant rodents and causes hypertension by activation of the endothelin-1 and placental oxidative stress pathways. , , , , Newer evidence suggests an interplay between the AT1-AA, Ang II, and the AT1 receptor activating ET-1 and oxidative stress pathways and culminating in hypertension. ,
The seemingly bizarre concept that circulating agonistic antibodies might mediate vasospasm in preeclampsia stemmed from a serendipitous observation in a single patient. When coupled to our knowledge of autoimmunity, immune tolerance, and the remarkable changes in the RAAS during pregnancy, it led to a novel hypothesis. What has evolved in the past decade is an integrated framework of observations that fits into the concept of faulty angiogenesis as a precursor for phenotypic preeclampsia. A goal of this chapter will be to review the notion that such antibodies cause or amplify pathological cardiovascular responses in preeclampsia.
Pregnancy is a physiologic process whereby repeated adjustments in intracellular and extracellular volume occur to maintain the steady state. Each new steady-state value is then held within relatively narrow limits, that is, these changes are sensed as normal and “defended” in face of variations in fluid and sodium intake. There is a 30%–50% increase in extracellular fluid (ECF), plasma, and blood volume, associated with 30%–50% increases in cardiac output, GFR, and renal blood flow.
The cause and significance of such changes have been debated for decades by three schools of thought. One advocates that the alterations are secondary to primary arterial vasodilatation causing “underfill” or decreased effective volume. The second view, called “normal fill,” implies that the gravida senses her volume as “normal” at every new steady state and reacts appropriately to sodium restriction or surfeit. The final concept, “overfill,” views pregnant women as overexpanded with intravascular volume. Each camp agrees, however, that primary renal sodium and water retention is presumably responsible for the volume changes associated with normal pregnancy.
The sources of peripheral edema, a sign of excessive interstitial ECF, are more complex in gravidas than in nonpregnant women. There are two forms of edema in pregnancy—“normal” edema reflects a physiological ECF volume increase, whereas “abnormal” or “pathological” edema in preeclampsia occurs when fluid has shifted from the vascular to the interstitial space (due, for example, to pathological “leakiness” of vessels (see Chapter 9 )—but the two etiologies are currently clinically indistinguishable. In women with “pathological” edema, total ECF is increased. In the second half of gestation, both pedal and pretibial edema can be detected in the majority of pregnant women, occurring more commonly as the day proceeds, often disappearing with recumbency. , In addition to this dependent edema, many women develop edema in the hands and face as pregnancy progresses; again, its frequency reflects the care with which edema is sought, an incidence of >60% being described in the classic 1941 monograph by Dexter and Weiss. Even those women who fail to manifest overt edema have increases in lower limb volume (i.e., subclinical edema). ,
Chesley, in the first edition of this text, suggested that the primary reason for increases in peripheral edema in normal pregnancy was the decrease in plasma oncotic pressure. Values for oncotic pressure decrease from an average level of 370 mm H 2 O (27 mm Hg) in nonpregnant individuals to 345 mm H 2 O (25 mm Hg) in early pregnancy and 300 mm H 2 O (22 mm Hg) in late gestation. Fig. 15.1 combines Chesley's summary during gestation with a study by Zinaman et al. performed at delivery. Note that the lowest values are reached in the hours that follow delivery, the period of time when women with preeclampsia are most at risk of both pulmonary and cerebral edema, if fluid administration is not monitored with appropriate care.
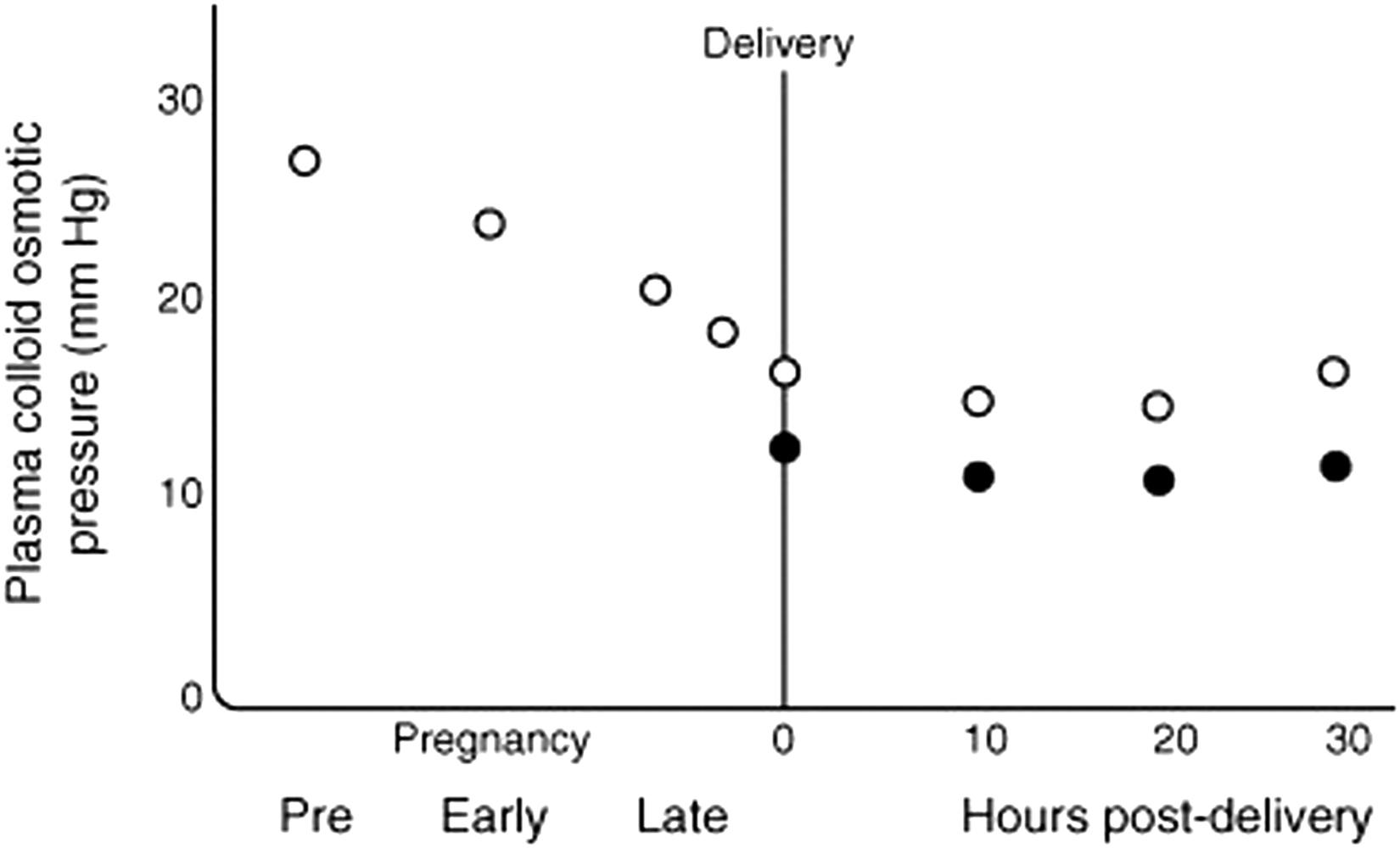
Kogh et al. have shown that reductions in plasma oncotic pressure on the order of that encountered in normal pregnancy can be associated with a significant increase in the rate of fluid extravasation from capillaries. The reduction in plasma oncotic pressure during pregnancy is mainly due to the ~1 g decrement in plasma albumin in normal gestation. The usual explanations of this decline are that it is a dilutional phenomenon related to the increase in plasma water, as well as in the decrease in plasma tonicity of ~10 mOsm. However, such explanations may be simplistic as little is known of the production and disposal of albumin in normal gestation, and while the levels of some circulating solutes decline along with the decrease in tonicity, others do not. Finally, women with “normal” peripheral edema and otherwise normal pregnancies also have the greatest weight gain and plasma volume expansion as well as larger neonates with lower perinatal mortality rates than those with less fluid retention. ,
Appreciation of changes in oncotic pressure during gestation is extremely relevant to clinical practice. Preeclamptic women with peripheral edema appear to be at risk of developing interstitial and frank pulmonary and cerebral edema. This risk is increased by fluid challenges, particularly the intravenous infusion of crystalloids at the time of delivery, and the danger is enhanced in the immediate puerperium when fluid shifts from the expanded interstitial compartment of normal gestation back into the circulation. For example, Benedetti and Carlson have suggested that the risk of pulmonary edema can be predicted by measuring colloid oncotic pressure, a suggestion supported by the observation that the lowest values are measured in the early postpartum period, , the period of greatest risk of this complication. Given that values in preeclamptic women are the lowest, they would naturally be the group at greatest risk of this life-threatening complication.
Finally, Hytten, as far back as 1970, wrote that edema is so common in pregnancy that it is not a useful diagnostic criterion to use in the diagnosis of preeclampsia. , Still, hypertension plus edema continued to be used to diagnose the disease (often compromising the value of research reports), only disappearing from the diagnostic criteria of all major classifications at the commencement of this millennium.
This concept is supported by animal experiments that evaluate relationships between intravascular volume depletion and vasopressin release during pregnancy compared to the nonpregnant state (described below and reviewed in ). Studies in pregnant animals and humans suggest that sodium and water reabsorption in the proximal nephron (determined by indices such as fractional lithium or solute-free water clearances) is unaltered, and these species dilute their urine normally when water-loaded during gestation. , , , Although osmotic thresholds are reset, experimental maneuvers aimed at abolishing “underfill” in both experimental animals and pregnant humans fail to alter the lower steady-state pressure, as well as the decreased osmotic threshold that occurs during gestation. ,
The observations concerning normal dilution and the excretion of water loads are important because failure to dilute the urine maximally and a blunted excretory response to water loading are major pathophysiologic features of the “underfill” status in diseases accompanied by hyponatremia such as cirrhosis, cardiac failure, or nephrotic syndrome. These disorders are considered prototypes of diseases in which absolute extracellular volumes are increased and “effective arterial volume” is low. Finally, most investigators describe similar sodium excretory responses to saline infusions in the pregnant and nonpregnant states in animals and humans. , ,
Controversies about the various “fill” hypotheses are yet to be settled, and perhaps each is correct at particular stages during pregnancy. Hormone-induced vasodilatation creating temporary “underfill” in the early weeks, followed quickly by compensation to a “normal fill” state as pregnancy progresses, fits two of the three theories, whereas during the last trimester, natriuretic factors predominate and restore Na balance, at least in some gravidas. The importance of settling this dispute, however, is not trivial, because a better understanding of how the pregnant woman “senses” her volume changes will impact management, particularly for gravidas with hypertensive complications and cardiac disorders.
The concept of “underfill” in pregnancy partially resembles views of how cirrhotic and heart failure patients “sense” their increased ECF volumes. The following observations suggest that primary arterial vasodilatation causing arterial underfilling with secondary sodium and water excretion occurs in early gestation. Supporters of “underfill” note that systolic and diastolic blood pressures decrease early in the first trimester of pregnancy despite an increase in blood volume. , The RAAS is activated early in pregnancy, an effect that would also occur with arterial underfilling due to peripheral arterial vasodilatation, while primary volume expansion would be expected to suppress these hormones. , , The increase in GFR and renal blood flow may precede the expanded extracellular fluid volume in pregnancy, also suggesting primary vasodilatation. Nitric oxide (NO) is a prime candidate as the mediator of vasodilatation in pregnancy. The resistance to Ang II, norepinephrine, and vasopressin that characterizes normal pregnancy can also be reversed by blockade of nitric oxide synthase (NOS). Additional observations are the resetting of the osmostat and the volume depletion–vasopressin relationships (discussed below) in a direction also consistent with vascular underfilling due to systemic arterial vasodilatation. Other evidence supporting “underfill” includes the observations that pregnant women manifest greater increases in aldosterone release than nonpregnant subjects in response to small quantities of Ang II and display exaggerated decreases in blood pressure when treated with ACE inhibitors.
Of particular interest is a study by Chapman et al. where they serially studied 13 women prior to and immediately following conception, and again during gestational weeks 6, 8, 10, 12, 24, and 36 ( Fig. 15.2 ). Measured were blood pressure, cardiac output, and renal hemodynamics (inulin and para-amino hippurate clearances). Mean arterial blood pressure decreased by 6 weeks of gestation, associated with an increase in cardiac output, decrease in systemic vascular resistance, and an increase in plasma volume ( Fig. 15.2 ). Renal plasma flow and GFR increased by 6 weeks of gestation (see Fig. 14.2, Chapter 14 ). Plasma renin activity (PRA) and aldosterone concentrations increased significantly by 6 weeks while norepinephrine levels did not change throughout pregnancy. Atrial natriuretic peptide (ANP) levels increased as well, first noted at 12 weeks. Plasma cyclic guanosine monophosphate (cGMP) levels decreased and cGMP clearance increased by 6 and 8 weeks, respectively. Chapman et al. strongly suggest that peripheral vasodilatation occurs early in pregnancy in association with renal vasodilatation and activation of the RAAS. Volume expansion occurs early, followed by later increases in ANP, suggesting that ANP increases in response to changes in intravascular volume. The authors also confirmed the decreases in Na, Cl, and HCO serum concentrations, as well as the lowered creatinine, blood urea nitrogen, and hematocrit concentrations throughout pregnancy.
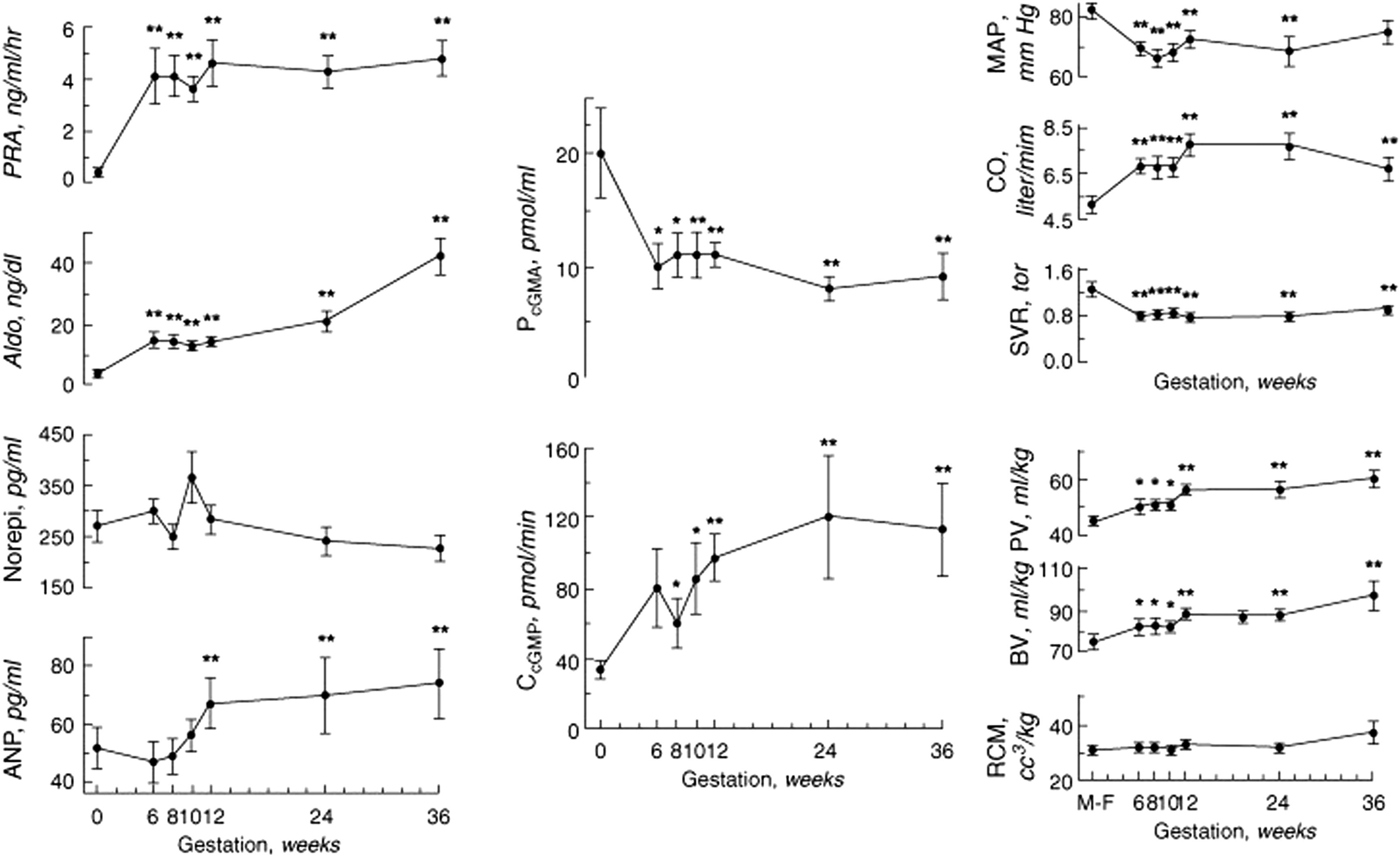
Pronounced placental growth occurs at weeks 6–8 of gestation and is generally complete by week 12. The systemic and renal regulatory changes observed by Chapman et al. occurred well before placentation. The authors speculated that maternal factors related to ovarian function are responsible for the peripheral vasodilatation, suggesting that NO might be largely responsible for the changes they observed. Renal production of NO or other natriuretic substances could in part explain these findings .
Supporters of the “overfill” theory note that there are absolute increments in ECF volumes, suggesting that this expansion may be an epiphenomenon to other changes, primarily a marked increase in mineralocorticoid hormone levels (see below). They also note the high levels of circulating natriuretic factors and various inhibitors of the membrane pump, which are expected responses to hypervolemia. There are also increases in renal hemodynamics, and two groups of investigators , have described an increased sodium excretory capacity in response to saline infusions. Although reports are anecdotal, women with cardiac or renal disorders appear more susceptible to volume overload complications, whereas healthy gravidas seem to tolerate blood loss better than nonpregnant women.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here