Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Rupture of an abdominal aortic aneurysm (RAAA) conveys a mortality risk 5- to 10-fold higher vs. elective repair. A major decline in RAAA incidence is now evident in both North America and Europe ( Fig. 76.1 ). , Despite improvements in perioperative management, service centralization, open RAAA survival rates have only marginally improved. However, ruptured endovascular repair (REVAR) has been associated with improved outcomes, with reductions in mortality, major adverse cardiac events and improved long-term mortality. , RAAA is an abdominal aortic aneurysm (AAA) with extraluminal blood on computed tomography (CT) or noted clinically at the time of surgery. A contained rupture refers to blood outside the aneurysm sac confined to the retroperitoneal space. A free rupture refers to bleeding directly into the peritoneal cavity. A symptomatic, nonruptured AAA is one with back pain or tenderness over the aorta on deep palpation but with an intact aneurysm on CT and at surgery. The pain is thought to be due to acute wall expansion, intramural hemorrhage, wall degeneration, or bleeding into the thrombus and is considered a prelude to rupture. Symptomatic aneurysms are not associated with hypotension, and survival is much better than for RAAA, but worse than after elective repair. Symptomatic aneurysms require rapid diagnosis and urgent management to prevent rupture. Inclusion within RAAAs data can artificially improve outcomes.
The first successful RAAA reconstruction was by Henry Bahnson with a homograft in 1953. By 1954 Cooley and DeBakey had treated six RAAA patients, with a 50% survival rate. Just as elective open aneurysm reconstruction was rapidly applied to ruptures, so too was endovascular aneurysm repair (EVAR), with Yusuf et al. reporting the first REVAR in England in 1994 (see Ch. 75 , Aortoiliac Aneurysms: Endovascular Treatment).
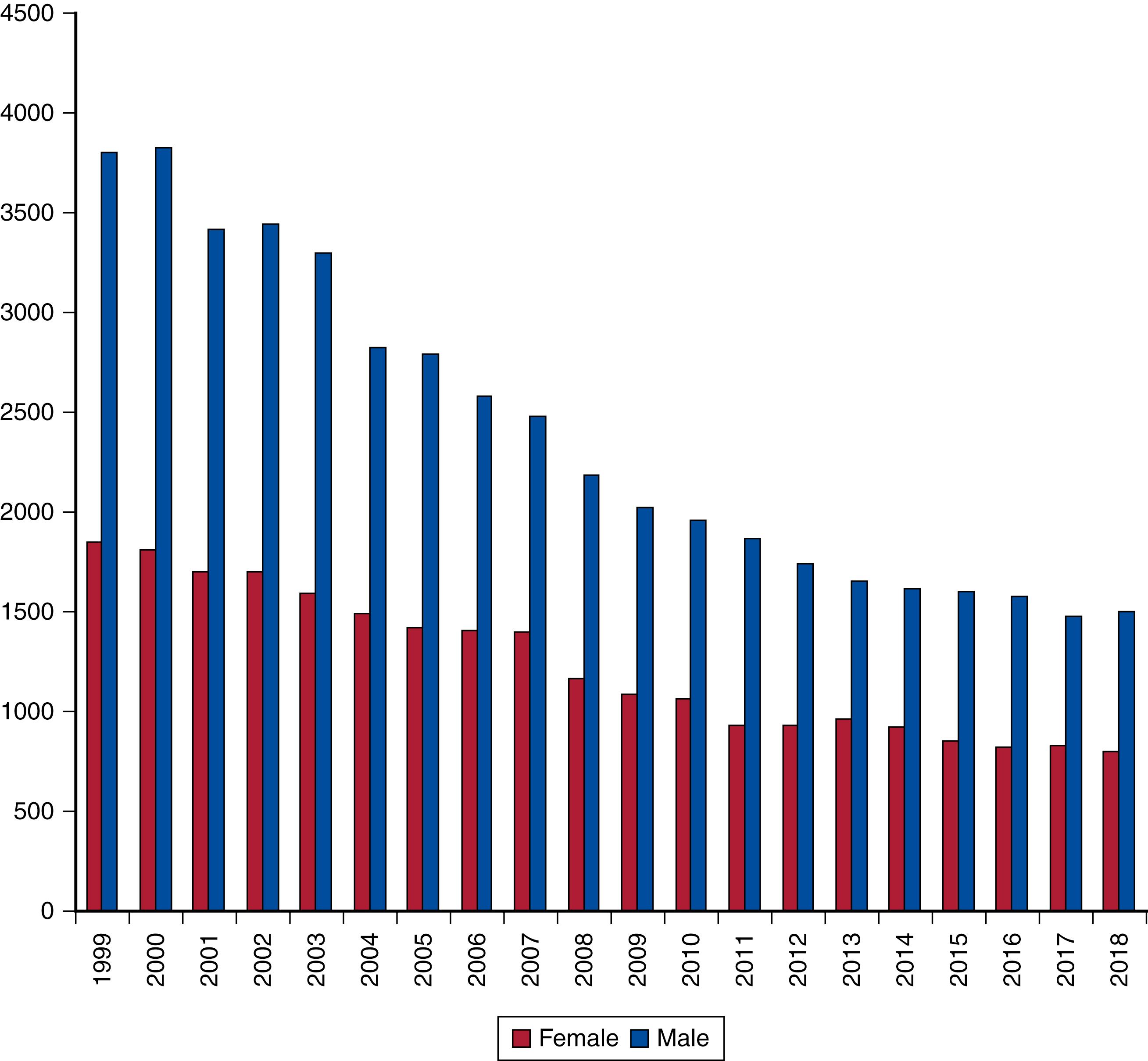
The true incidence of RAAA has historically been difficult to quantify due to the inability to quantify death outside of hospital. The incidence of RAAA was thought to be increasing at the turn of the century based on English, Welsh and Swedish population-based studies. , However, contemporary studies from the past two decades have shown a decline in the incidence of ruptured (and intact) AAA in most Western populations. In Germany, the decline in incidence of RAAA repair was 30% from 2005 to 2014. A population-based analysis of the Swedvasc registry from 1994 to 2014 revealed a 25% decrease in RAAA repairs. Analysis of data from the Swedish National Patient Registry revealed that lower socioeconomic status and educational level were associated with higher odds for presentation with RAAA. Population-based research using Canadian data demonstrated a 69% decrease in RAAA repairs from 2003 to 2016, and confirmed significant decreases across all ages, in men, and at non-teaching hospitals. English Hospital Episode Statistics data from 2002 to 2015 confirmed these sex differences in RAAA repair rates. In Medicare beneficiaries from 1995 to 2008, AAA-related deaths halved, with the greatest decline seen in patients older than 80 years. Data from the Centers for Disease Control and Prevention reported that in 1999–2018 deaths from RAAA dropped by 60% across all ages and in both sexes ( Fig. 76.1 ). Similarly, data from the US National Vital Statistics Registry demonstrated a 70% decrease in incidence of deaths due to RAAA. Mortality varied significantly by geographic region both in Vital Statistics and VQI data. Deaths are more frequent in men compared with women and the peak age for females was older than for men.
Population screening has been shown to reduce AAA-related deaths, but the decline in mortality due to RAAA began before widespread population screening. EVAR may have led to more elective AAA repairs in older and less fit patients, thereby reducing the at-risk population for RAAA. The greatest absolute reduction in RAAA has come from a decline in the prevalence of intact AAA in the Swedish and United Kingdom (UK) screening programs, where the detection rate was 1.7%, less than the 4% expected. This has been attributed to decreased levels of smoking and improved control of cardiovascular risk factors. , , ,
In two population-based series of RAAA, overall mortality rates were 89% and 86%. , A recent population-based Swedish study reported a 76.5% overall mortality from 2011–2015, with a 36% repair rate. The Danish national registry noted a decrease in operative mortality from 51% to 42% from 1994 to 2008, related to centralization of services and better prehospital care. Vascunet outcomes data for 7040 RAAA repairs noted a mortality rate of 31.6%, with the rate declining by 4.2% between 2005 and 2009 attributed to EVAR. German population-based data examining more than 20,000 RAAA repairs from 2005 to 2014 noted operative mortality of 40%. Data from 1994 to 2014 from the Swedvasc registry demonstrated a reduction in RAAA operative mortality from 38 to 28%. Comparison of English and American databases from 2005 to 2010 noted that intervention for RAAA was offered to 80% in the US (21% EVAR) vs. only 58% in the UK (8.5% EVAR). Postintervention mortality was similar at 41.6% and 41.8%. A more recent study examining this comparison revealed intervention rates of 91% and 75% in the US and UK, with in-hospital mortalities of 45% and 35%, respectively. Other studies from the US have confirmed these reductions in operative morality for RAAA.
The high postoperative mortality after aortic rupture is related to a high incidence of myocardial infarction, colon ischemia, renal failure, and multiple organ failure (MOF). The synergistic effect of the total body ischemia caused by hemorrhagic shock and the lower torso ischemia that occurs during repair, followed by reperfusion secondary to resuscitation and aortic unclamping, has been proposed to explain the high incidence of MOF and mortality after repair. , EVAR minimizes the ischemic insult induced by aortic clamping, and RAAAs anatomically suitable for EVAR have the lowest mortality and complication rates. ,
Aneurysm rupture represents failure of the aortic wall to bear the load of the blood pressure. Aneurysmal aortic wall is weaker than the normal structure (65 N/cm 2 vs. 121 N/cm 2 for aneurysmal vs. normal aortic wall). , Laplace’s law (wall tension is proportional to radius times pressure) relates wall tension to pressure and emphasizes the importance of aneurysm size but does not account for complex AAA geometry or for regional changes in wall strength that may be weakened secondary to localized inflammatory processes that are part of the pathophysiology of aneurysm formation. Approximately 10% of AAAs rupture below the repair threshold.
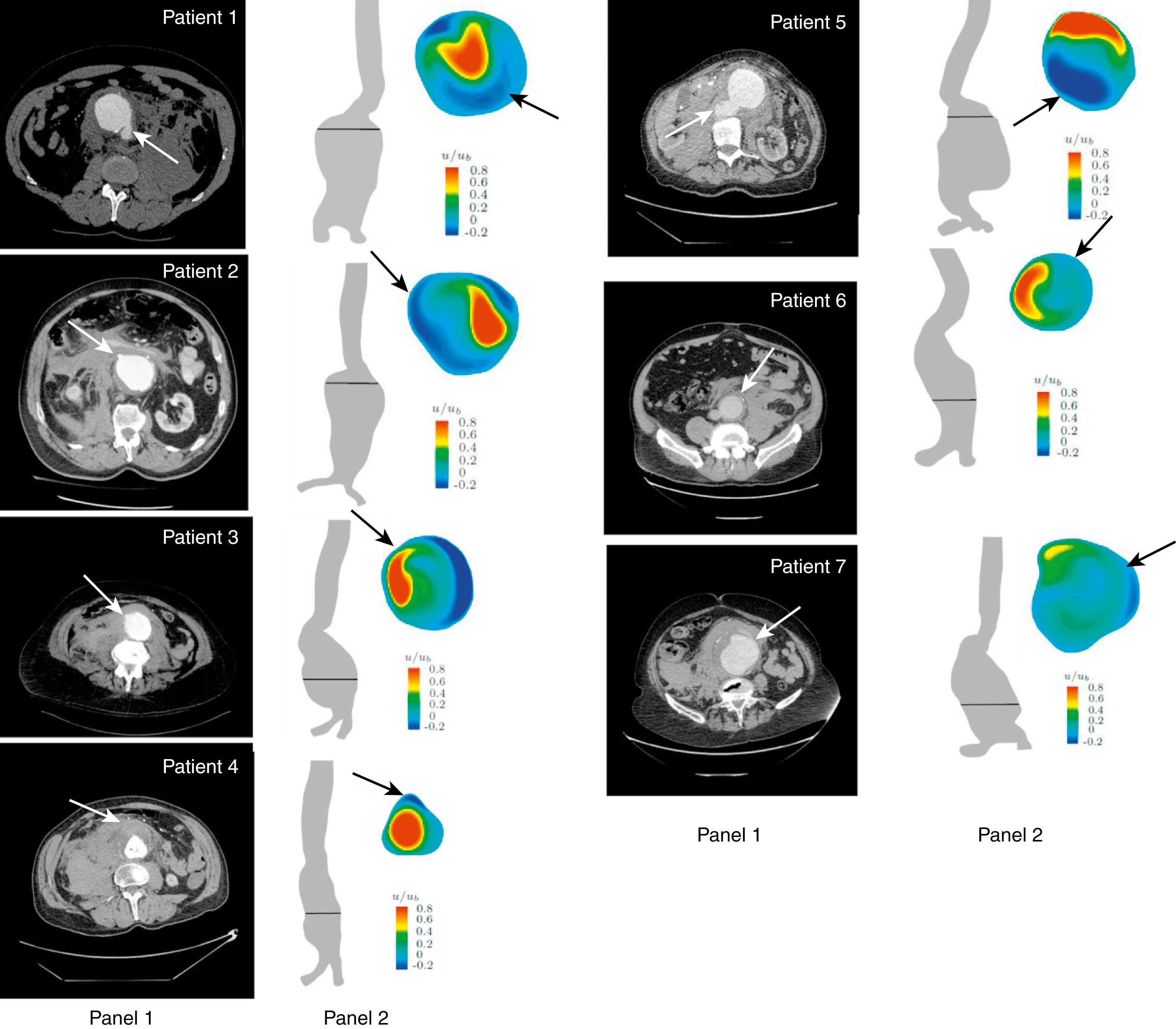
Finite element analysis has been applied to AAAs to estimate regional variations in peak wall stress (PWS) for many small segments of the entire aortic wall using CT scan data, and provides a visual representation of PWS variability. Early 3D representations of PWS noted that the posterior wall of the aorta had higher values corresponding to sites where clinical observations and autopsy studies document the rupture sites. Newer computational models include an analysis of flow, shear stress, growth and remodeling theory that allows evolving wall thickness, stiffness, regional heterogeneity, and fluid–structure interactions within the aneurysm sac to be analyzed. , Areas of low shear stress and increased thrombus deposition have been identified at sites of aortic rupture ( Fig. 76.2 ). Recently both high and low fidelity models of peak wall rupture risk (PWRR) were found to be better than diameter alone in predicting rupture risk. While computational models have increased our understanding of biomechanical factors, they have yet to become routine in clinical practice. ,
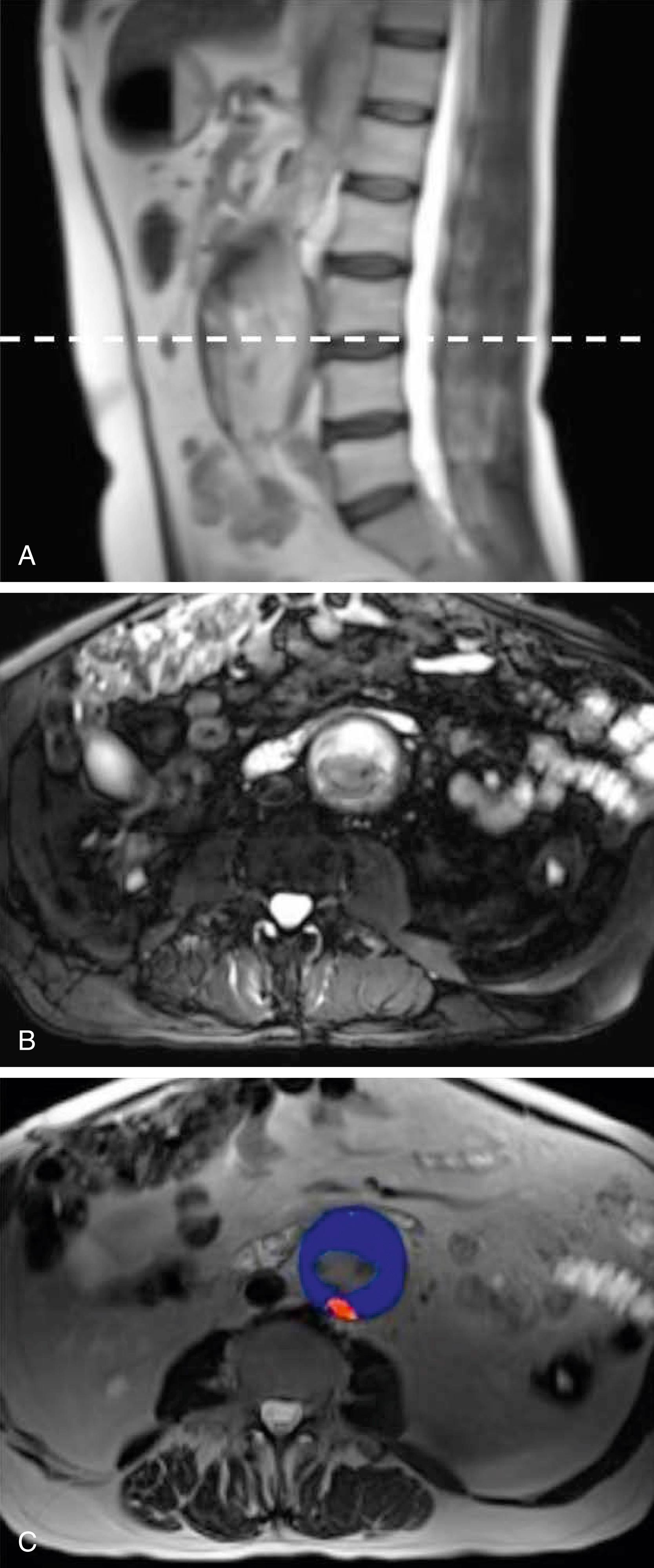
In 1965 Martin surmised that as an aneurysm develops, it becomes lined with thrombus, which weakens it by interfering with nutrition of that wall segment. Schurink et al. confirmed that intraluminal thrombus does not reduce pressure near the aneurysm wall, using a pressure transducer during open AAA repair. Vorp et al. noted localized hypoxia in regions of thicker thrombus leading to localized wall neovascularization, inflammation, and regional wall thinning. Hypoxia also affects the function of vascular smooth muscle cells, causing them to secrete more collagenase than in normoxic conditions, with less elastin and collagen production. , , Autopsy studies of patients who died of infrarenal RAAAs noted that 80% of ruptures occurred at the site of mural thrombus. CT studies of patients admitted with rupture showed that most occurred through the thrombus or at its edge. Others argue that ruptures are caused by fissures in the intraluminal thrombus, due to proteolytic enzymes on the luminal surface of the clot. A Dutch report, although accepting a role for fissure formation, confirmed that intraluminal thrombus thickness was associated with vascular smooth muscle cell apoptosis (or cell death), elastin degradation, and high levels of matrix metalloproteinase 2 and correlated with aneurysm rupture. A prospective MRI study of infrarenal AAAs, identified a subset of patients with inflammatory activity noted on the MRI ( Fig. 76.3 ). Those AAAs grew faster and were more likely to rupture compared to those where inflammatory activity was absent. The specific mechanisms that contributed to the activity observed have yet to be defined. ,
The classic presentation of RAAA includes the triad of acute-onset abdominal/back pain, hypotension, and a pulsatile abdominal mass. Severe and unremitting back pain is caused by rupture of the aneurysm into the retroperitoneum. The pain may radiate to the back, testes, inguinal canal, or rectum. On examination, the patient is frequently pale, diaphoretic, and hypotensive with a tender, pulsatile abdominal mass. Flank ecchymosis is a late sign.
In patients older than 50 years with hypotension and/or syncope, consideration of RAAA is critical. The differential diagnosis may include renal colic, diverticulitis, pancreatitis, gastrointestinal hemorrhage, myocardial infarction, and perforated ulcer. The recognition of RAAA in the early stages can be difficult. Presentation can range from sudden onset pain and hypotension to slowly increasing pain over days as the AAA gradually stretches and ruptures. In this age group, chronic back problems are common and vertebral erosion from rupture can be mistaken for exacerbation of long-standing back problems.
Among those subsequently diagnosed with an RAAA, only 23% had a definitive and immediate diagnosis of RAAA made by the first examining physician. , The rate of incorrect diagnosis ranges from 16% to 60%, and one study found misdiagnosis is associated with a median 4.8 hour delay to surgical intervention associated with misdiagnosis, but this did not result in excess mortality. , The most common misdiagnoses were renal colic, perforated viscus, diverticulitis, gastrointestinal hemorrhage, and ischemic bowel. The initial manifestation of renal colic occurs infrequently in patients older than 60 years. The classic triad was present in only 9% of the misdiagnosed group compared with 34% of the correctly diagnosed group. The presence of a pulsatile mass was identified in 72% of those correctly diagnosed but in only 26% of the misdiagnosed patients. A recent study of Medicare beneficiaries from 2007 to 2014, including 17,963 with RAAA demonstrated a 3.4% missed diagnosis rate. Significant predictors of missed diagnosis with discharge home included female sex, Black ethnicity and emergency department discharges within 1 year of presentation, and comorbid end-stage renal disease (ESRD), dementia, depression, hypertension, coronary disease and chronic obstructive pulmonary disease (COPD) were associated with missed diagnosis.
Rare presentations include rupture into an adjacent structure. Acute gastrointestinal bleed can occur due to a primary aortoduodenal fistula. An aortocaval fistula presents as a pulsatile abdominal mass, machinery-like bruit, or thrill with elevated jugular venous pressure and new-onset acute congestive heart failure. Cauda equina syndrome, lower extremity motor deficit, incarcerated inguinal hernia, and phlegmasia cerulea dolens are other rare manifestations.
Although the use of plain radiography is currently rare, a retrospective review of plain films of patients with RAAAs showed evidence of the diagnosis on 90% of the films. Enlargement of a calcified aortic wall beyond normal limits was seen in 65%, and loss of a psoas shadow from retroperitoneal hemorrhage was identified in 75%.
Bedside ultrasound in the emergency department by trained emergency physicians has led to its use in cases of suspected RAAA. Focused assessment with sonography in trauma (FAST) protocols can rapidly identify fluid collections and direct further trauma care. , When ultrasound is used in patients with back pain, abdominal pain, or other indication of AAA, it can identify aortic aneurysms with high sensitivity, specificity, and positive and negative predictive values. , No large prospective studies have been conducted, although with appropriate training the use of ultrasound to quickly assess patients for the presence of AAA is likely beneficial as it directs rapid investigation and therapy. It is not sufficiently accurate to exclude rupture, , and is consistent with rupture in only 51% of cases. , ,
The most accurate method for diagnosis of RAAA is CT. While non-contrast CT can identify retroperitoneal hemorrhage associated with RAAA, scanning with contrast enhancement is ideal to plan either open surgical repair (OSR) or EVAR ( Fig. 76.4 ). Proper timing of contrast injection reduces the contrast volume required, lessening its renal impact. Suspected RAAA patients going to CT should be accompanied and monitored for acute deterioration.
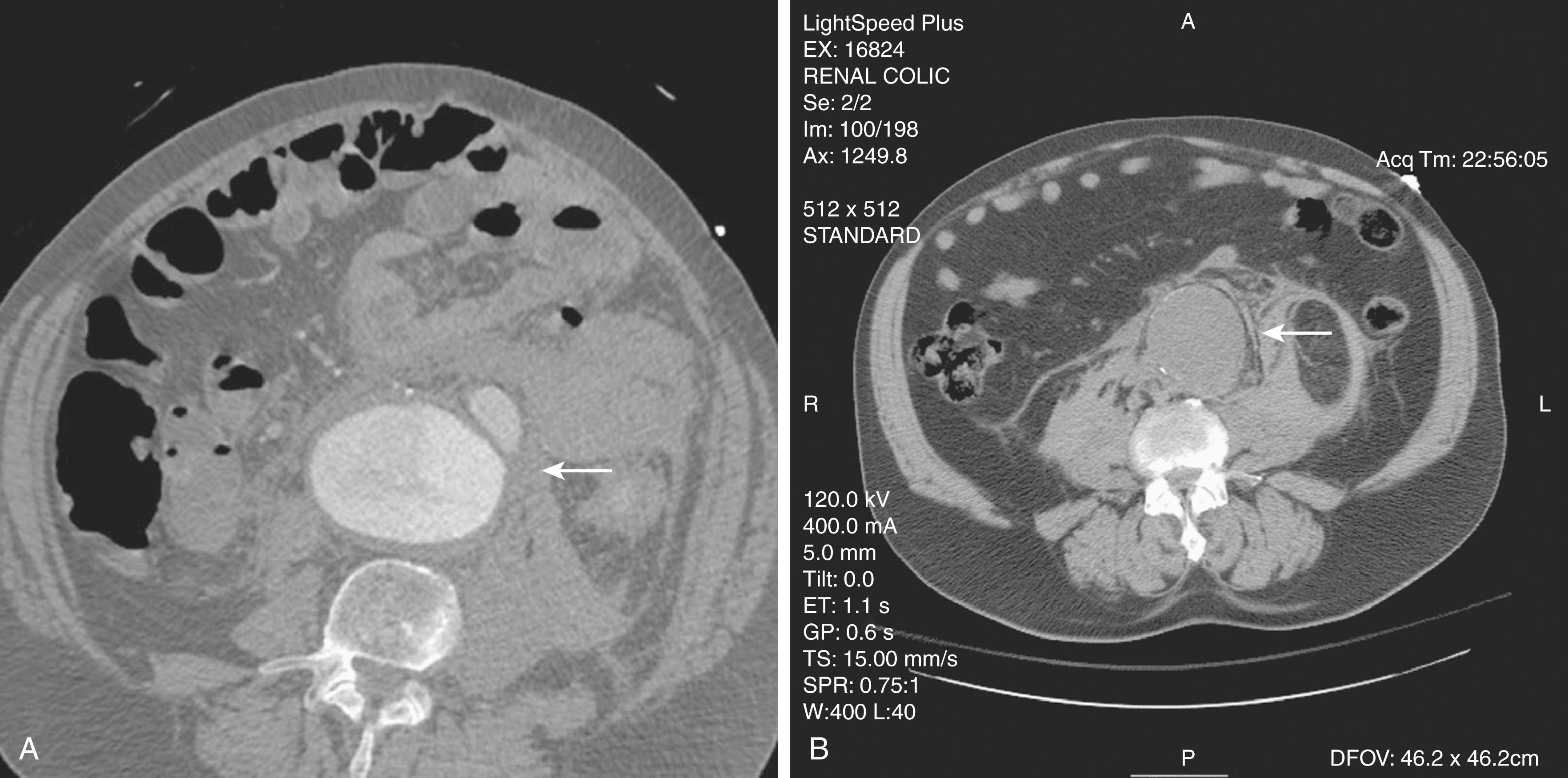
When evidence of retroperitoneal blood with an AAA is used as the “gold standard” for diagnosis of RAAA on CT, it is 77% sensitive and 100% specific. Its positive and negative predictive values are 100% and 89%. In the Amsterdam Acute Aneurysm (AJAX) study, agreement on CT diagnosis of RAAA was only moderate in five observers, agreement on suitability for EVAR was only fair in vascular surgeons and radiologists experienced in EVAR.
The delay between the initial manifestation of RAAA and the time to death has been studied to determine whether sufficient time exists to perform a CT scan and to evaluate patients’ suitability for EVAR. The time from onset of symptoms to death for patients managed conservatively was a median of 16 hours. Only 13% died within 2 hours of hospital admission, suggesting that sufficient time exists for most patients to undergo CT scanning and evaluation for EVAR. For patients offered intervention, a door to intervention time of less than 90 minutes has not been shown to improve morality. A small number of patients in both the Immediate Management of the Patient with Rupture: Open Versus Endovascular Repair (IMPROVE) and the Amsterdam Acute Aneurysm randomized controlled trials (RCTs) were too unstable for CT scanning and required immediate transfer to the operating room. Prospective data from the IMPROVE trial demonstrated that 90% to 97% of patients had a CT scan prior to intervention and the time to arrival at the operating room was 25 minutes shorter in the EVAR group vs. OSR despite the requirement for a contrast-enhanced CT scan. Thus, for the majority of patients who are stable on arrival at a center where definitive care will be carried out, a prompt CT scan does not adversely affect mortality. , A protocol for priority management of RAAA may significantly shorten the time to definitive therapy despite the CT scan requirement. ,
RAAA is a surgical emergency, and when the diagnosis is being considered, immediate vascular surgery consultation is critical. Rural hospitals without vascular surgery expertise have a higher burden of RAAA patients and more commonly do not offer intervention. , Interhospital patient transfer increases delays to the operating room, although most studies demonstrate that transferred patients do not have a higher but in fact lower mortality. , VQI registry data showed no mortality difference in those with door to intervention times less than 90 minutes compared to those greater than 90 minutes. However, high-volume centers, teaching centers, and high bed capacity hospitals have significantly lower mortality for RAAA patients. , ,
Preoperative resuscitation of RAAA patients with hypotension must be judicious. , Large volumes of intravenous fluids raise blood pressure, causing further hemorrhage by overcoming the tamponade, result in hemodilution, coagulopathy, hypothermia, and acidosis, and further deterioration. Protocols that restrict aggressive fluid resuscitation in hypotensive trauma patients with hemorrhagic shock improve survival so that permissive hypotension for RAAA patients is recommended. Fluid resuscitation should be sufficient to maintain consciousness, minimize organ ischemia, prevent ST depression, and maintain a systolic pressures of 70 to 80 mm Hg. , Contemporary algorithms for management of RAAA patients include permissive hypotensive resuscitation. Aggressive volume resuscitation before proximal aortic control is a predictor of increased postoperative mortality. , The IMPROVE trial demonstrated that those with the lowest BP had the highest mortality and increasing SBP to greater than 70 mm Hg was beneficial. Observational studies also suggest that increased fluid administration prior to aortic control increased mortality. Implementation of a controlled hypotension strategy in a regional ambulance service for patients with hypotension, in which only 27% had a final diagnosis of RAAA, demonstrated the overall risk of harm was low. Evidence regarding resuscitation with blood vs. crystalloids is limited; if blood is available, it should accompany the patient during transfer and can be given to maintain consciousness.
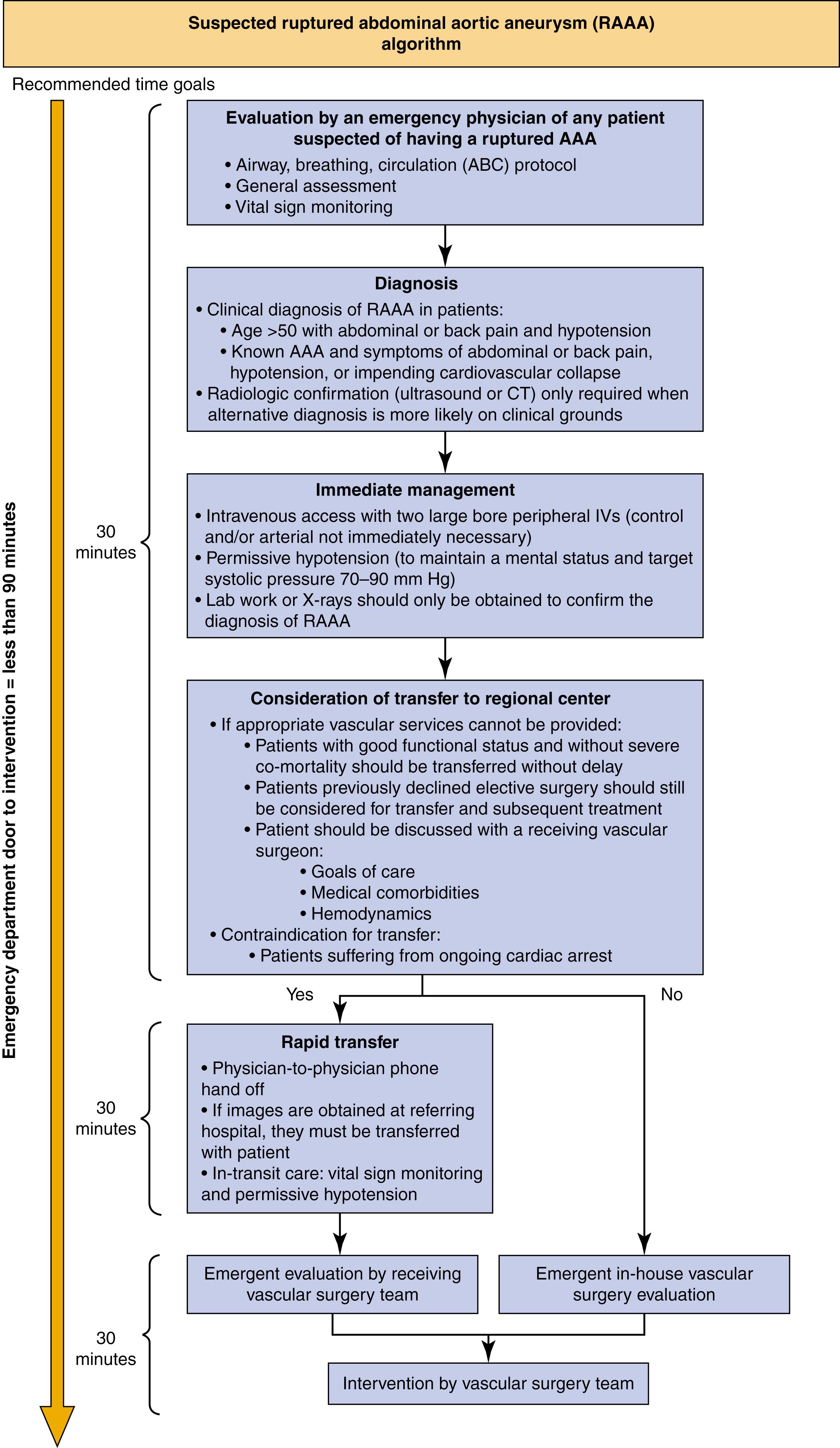
RAAA care begins with early identification by the emergency room staff, and rapid notification of the vascular surgeon/team , , ( Fig. 76.5 ). While an ER ultrasound can identify an AAA and retroperitoneal hematoma, a fine cut, low contrast volume CT scan to assess neck diameter, angulation, and iliac size is of critical importance after blood work including a cross-match. Hemodynamically unstable patients can be directly transferred to the operating room without a preoperative CT scan for balloon control with either an EVAR or conversion to OSR as needed. EVAR for RAAA requires a preoperative contrast CT scan. EVAR obstacles are anatomic suitability, availability of staff and equipment, feasibility of treating hemodynamically stable and unstable patients, and ability of the care team to manage unexpected scenarios under emergent circumstances. One key factor is awareness among the emergency department staff of the diagnosis of RAAA and early notification of the on-call vascular surgeon/team. , , ,
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here