Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Rubella (i.e., German measles) is a benign, self-limited, vaccine-preventable viral illness. Acquired rubella is characterized by an exanthem and lymphadenopathy, although infants and children with rubella frequently are asymptomatic. Much of the morbidity attributed to rubella virus (RuV) occurs from in utero transmission, especially during the first trimester, resulting in miscarriage, stillbirth, fetal death, or congenital rubella syndrome (CRS). Sequelae of CRS include growth retardation, deafness, congenital heart disease, and mental retardation.
Routine childhood vaccination with rubella-containing vaccine (RCV) in conjunction with enhanced epidemiologic surveillance has resulted in elimination of rubella and CRS in the World Health Organization (WHO) Region of the Americas in 2009. However, rubella and CRS remains a major public health problem in many low-income countries. The Global Vaccine Action Plan has targeted measles and rubella for elimination in the remaining five WHO Regions by 2020. 1
RuV is the only member of the Rubivirus genus within the Matonaviridae family. The virus particle is spherical, medium-sized (50–70 nm), and contains a single-stranded, positive-sense RNA genome. The nucleoprotein core is surrounded by a glycolipid envelope; the lipid component is host cell derived. There is only one antigenic type of RuV. Genetic characterization of RuV has revealed two clades and 13 genotypes. Humans are the only natural hosts of RuV
RuV contains three major structural proteins: E1, E2, and C. E1 and E2 are glycosylated envelope proteins that are embedded in the envelope as heterodimeric spikes. C is the capsid nucleoprotein. E1 is the primary immunodominant epitope that binds hemagglutination and neutralizing antibodies. E1 plays a major role during virus entry into target cells due to its receptor-binding and membrane-fusion functions. Although no definite cellular receptor has been identified for RuV, the E1 protein binds to myelin oligodendrocyte glycoprotein (MOG), allowing virus entry into certain target cells. Since MOG is expressed primarily on human central nervous system (CNS) cells and placenta, other cellular receptors may be involved in the pathogenesis of RuV. In lymphoid cells and erythrocytes, the binding of RuV E1 to the host cell membrane is Ca 2+ -dependent specifically directed to membranes enriched in sphingomyelin and cholesterol, but RuV also binds to many cell types in a Ca 2+ -independent mechanism via an unidentified RuV receptor(s). Heat shock protein 90 has been identified as a key host factor for RuV genome replication. RuV is readily inactivated by lipid solvents, heat, ultraviolet light, and extremes in pH. RuV grows well in a variety of primary and continuous cell lines.
Rubella is most likely transmitted by direct or droplet contact with upper respiratory tract secretions of infected people. Infectivity, which correlates with virus shedding from the nasopharynx, can occur 1 week before to 2 weeks after rash onset. People are most infectious from 5 days before until 7 days after the onset of rash.
After transmission, the virus replicates in the nasopharynx and then spreads hematogenously to regional lymphatics. Primary viremia occurs 5–7 days after infection. Virus replicates in local and distant reticuloendothelial sites and is followed by secondary viremia at high levels, especially during the second week of infection. Cell-associated virus can be recovered from peripheral blood lymphocytes and monocytes for up to 4 weeks after infection.
Within 8–14 days after exposure, active viral replication occurs throughout the body and can be recovered from numerous sites, including the respiratory tract, skin, lymph nodes, urine, cerebrospinal fluid, and breast milk. Maximal nasopharyngeal shedding, the development of humoral immunity, and clinical manifestations of infection occur during this period.
Humoral immunity to natural and vaccine RuV is characterized by development of a variety of antibody types reactive to distinct viral antigens, including viral hemagglutinins, complement-fixing (CF) antigens, and precipitating antigens. Development of hemagglutination inhibition (HAI) and neutralizing antibodies (NAbs) occurs with the onset of the rash illness, and CF antibodies are detected 1 week later. Peak titers of HAI and NAbs occur about 2 weeks after rash onset; CF antibodies peak about 2 weeks later. The HAI and NAbs persist for life.
Although the antibody titers induced by the RA 27/3 vaccine virus currently used in the US are lower than those induced by natural infection, the nature of the antibody response is similar to the response to natural infection. High-avidity IgG antibodies against the neutralizing epitope of E1 glycoprotein is an important correlate of RuV protection. Vaccine-induced immunity is long lasting, with detectable antibodies for 20 years found in 95% of patients. Persistence of immunity may be due to protective anamnestic responses. However, serologically confirmed reinfections (usually asymptomatic) can rarely occur after natural infection or vaccination. , , The role of cell-mediated immunity (CMI) in rubella is unclear. CMI to RuV develops at least 1 week before the development of humoral immunity and likely persists for life.
In contrast to acquired rubella infection, congenital infection results in a persistent, progressive infection. Infants with CRS can shed RuV in nasopharyngeal secretions and urine for up to 1 year and can transmit rubella to susceptible contacts. Placental and fetal infection with RuV occurs most consistently after maternal infection during the first trimester of pregnancy, and early fetal infection, especially during organogenesis, is associated with increased risk of fetal death or teratogenicity. , Maternal RuV infection in the first trimester of pregnancy results in fetal defects in 85%–90% of cases; risk drops to 50% with maternal infection at 13–16 weeks’ gestation and 25% at 15–16 weeks. , Fetal defects are rare if maternal infection occurs after 16 weeks’ gestation. However, sensorineural deafness can occur with maternal infection as late as 20 weeks’ gestation.
Placental infection usually occurs during the period of maternal viremia. The virus crosses the placenta and enters the fetal bloodstream. In CRS, multiple organ systems are affected due to the multifactorial effect of RuV on organogenesis and systemic inflammation. Pathologic effects of congenital infection primarily are the result of generalized virus-induced, progressive, necrotizing vasculitis. Human fetal endothelial cells are highly permissive to RuV. RuV antigen has been detected in capillary endothelial cells and the basal plate of placenta. Neural progenitor cells, myocardial interstitial fibroblasts, and adventitial fibroblasts of the aorta and pulmonary artery are also susceptible to RuV. Fetal histopathology is characterized by noninflammatory damage to endothelial cells of small blood vessels resulting in thrombosis and surrounding tissue necrosis. , Other pathologic effects are focal inflammation, edema, and granulomatous changes. The resulting parenchymal hypoplasia produces characteristic clinical findings associated with CRS.
Mitotic cell arrest, apoptosis, and chromosomal defects have been identified in congenitally infected fetuses and in human embryonic cells, leading to reduced cell numbers and hypoplasia. , Fetal infection is severe with multiple organ involvement if maternal infection occurs early in the first trimester. If maternal infection occurs after 11–12 weeks’ gestation, fetal infection results in more restricted organ involvement (eye, auditory system).
Humoral and CMI responses in congenital rubella infection are distinct from those observed after acquired infection ( Fig. 221.1 ). With congenital infection, fetal immunoglobulin (Ig) M antibodies to RuV can be detected as early as 20 weeks’ gestation, and fetal IgG can be detected by the middle of the second trimester. Concentrations of IgM and IgG continue to rise throughout the first few months of life. IgM levels decline by about 6 months of age, and IgG levels persist for life. Loss of antibody may occur over time leading to reinfection in some children with CRS.
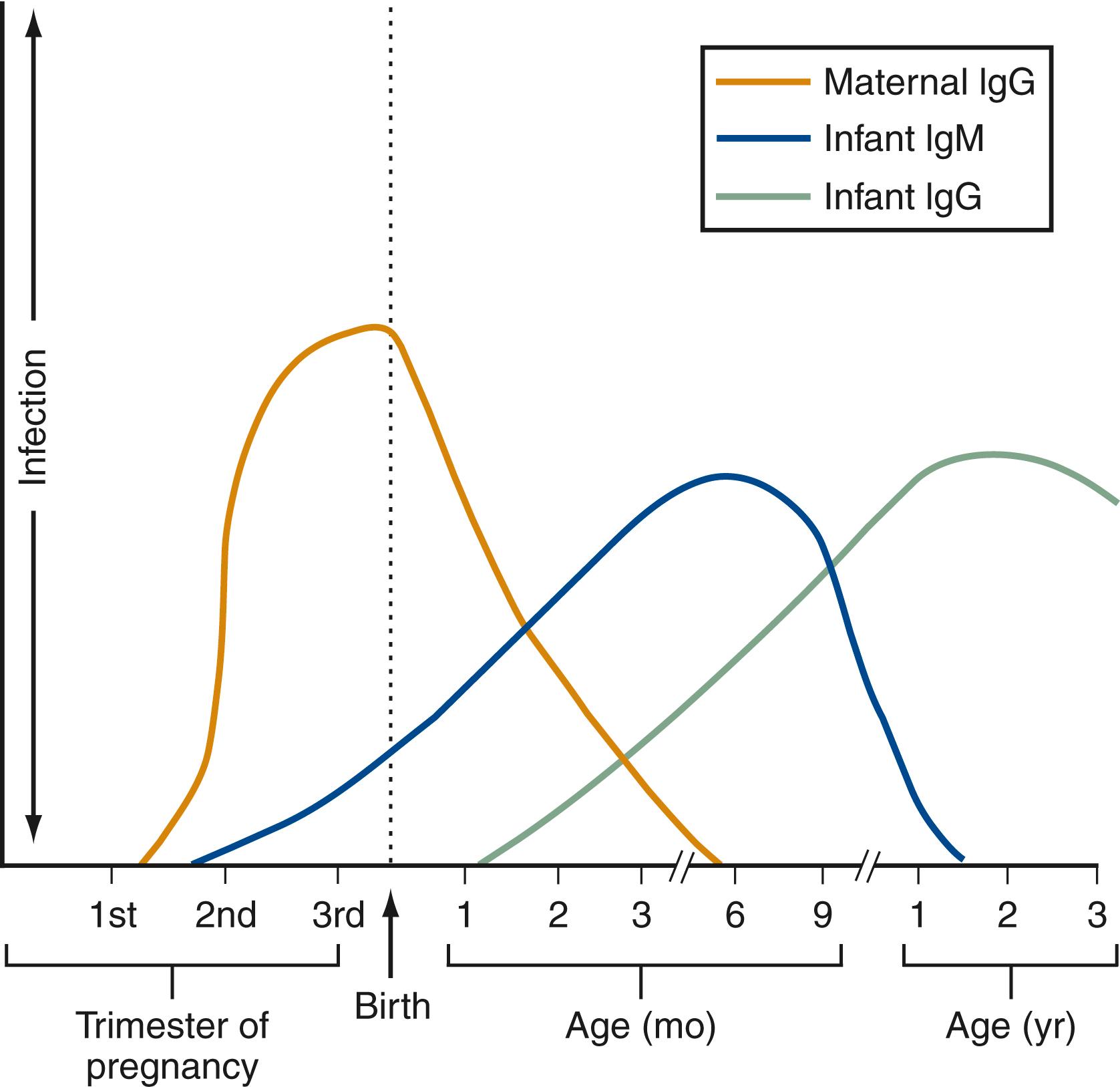
Children with CRS have low-avidity IgG antibodies in contrast with the high-avidity IgG antibodies of their mothers or cases of postnatal infection. , CMI responses to congenital rubella, including delayed-type hypersensitivity reactions, lymphocyte-mediated cytotoxicity, phytohemagglutinin-induced lymphocyte transformation, and interferon production, are decreased compared with responses after acquired infection. The most depressed CMI responses are observed in infants infected earliest in gestation.
Before the introduction of rubella vaccine in 1969, rubella epidemics occurred in 2- to 4-year cycles, most often in late winter and early spring; larger epidemics occurred at 6- to 9-year intervals. Young children were most susceptible to rubella infection (highest attack rate, 5–9 years of age). Most adults were not susceptible during cyclic epidemics. Immediately before the licensure of rubella vaccine in 1969, the annual number of rubella infections reported in the US was 57,600 and there were 62 CRS cases. After vaccine licensure, >99% decline in the incidence of rubella was observed, and in 2004, rubella was no longer endemic in the US ( Fig. 221.2 ). ,
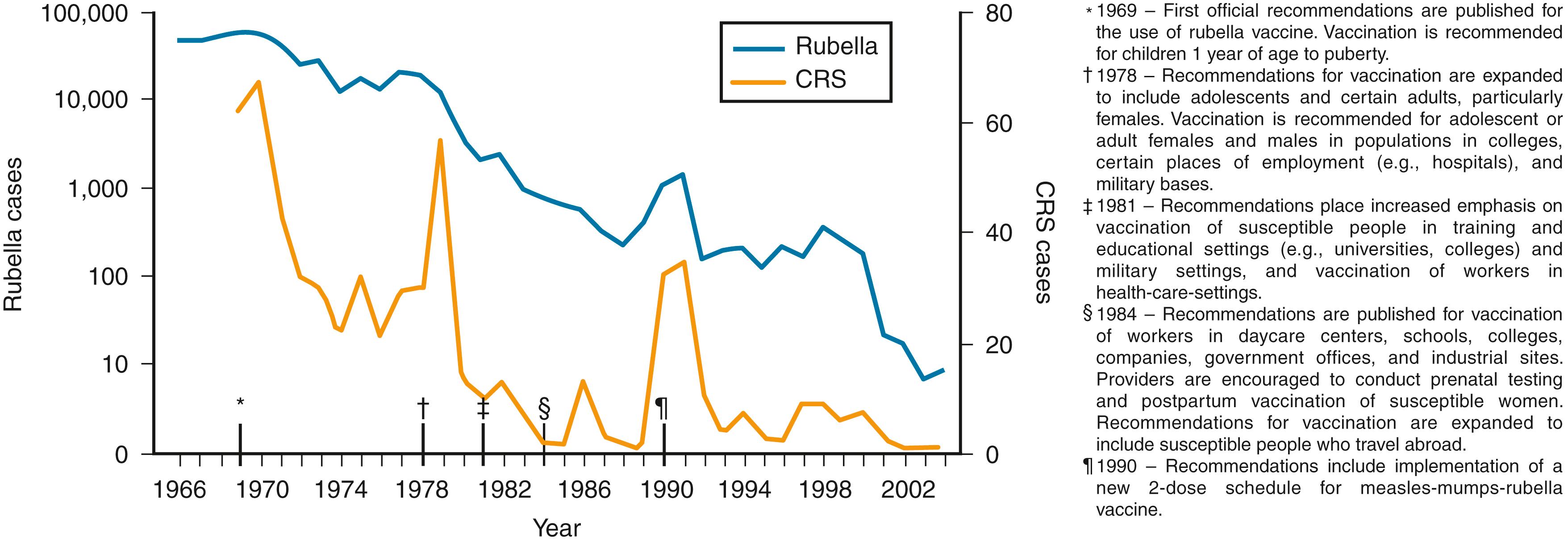
In the post-vaccination era, RuV outbreaks in the mid-1970s and 1980s occurred mostly among unvaccinated adolescents and young adults in college campus and work settings. Between 1989 and 1991, rubella outbreaks occurred among unvaccinated people who belonged to religious groups that traditionally refuse vaccination. Rubella attack rates among susceptible people in closed communities (e.g., college dormitories, military facilities) range from 75% to 90%; however, attack rates can approach 100% among household contacts. In the mid-1990s, reported rubella cases occurred primarily among foreign-born, young Hispanic adults. In recent outbreaks, cases have occurred among people born outside the US or among underimmunized people. ,
In 2004, a CDC panel verified rubella elimination in the US. In the postelimination period from 2004 through 2014, 94 rubella cases and 9 CRS cases were reported in the US; all cases were known importations or from unknown sources ( Fig. 221.3 ). Key strategies for rubella elimination included universal immunization of all infants and targeted vaccination of susceptible prepubertal girls and women of child-bearing age. Success has been achieved primarily with universal childhood immunization. In 2017, national immunization coverage was 91.5% for vaccination with ≥1 dose of measles-mumps-rubella (MMR) vaccine among children aged 19–35 months in the US.
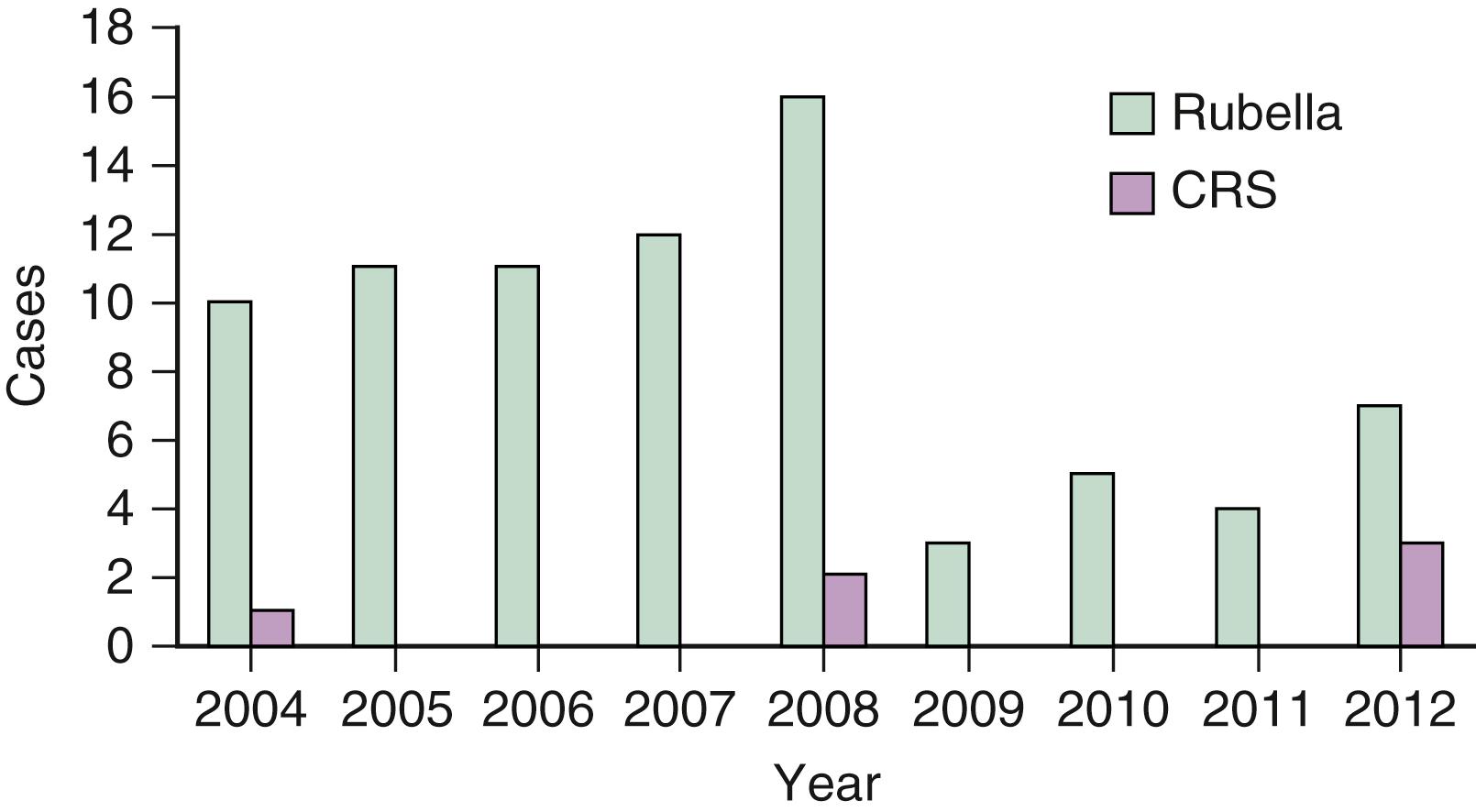
Based on national serosurveys (1999–2004), rubella seropositivity rates were about 95% among children and adolescents between the ages of 6 and 19 years. However, about 10% of women of childbearing age were rubella seronegative. Women born outside of the US or from settings with poor rubella vaccine uptake are more likely to be rubella nonimmune, predisposing their infants to CRS. , ,
In 2015, the Pan American Health Organization verified the elimination of rubella and CRS in the WHO Region of the Americas. The last confirmed case of endemic rubella and CRS in the Americas was reported in 2009. Although progress has been made, the burden of rubella and CRS remains substantial in the African, Western Pacific, and Eastern Mediterranean regions. , , Approximately 100,000 infants are born annually with CRS. Partial vaccination strategies have resulted in large rubella outbreaks in Japan, Philippines, and Romania. During 2016–2018, the predominant circulating RuV genotypes were 1B and 2E.
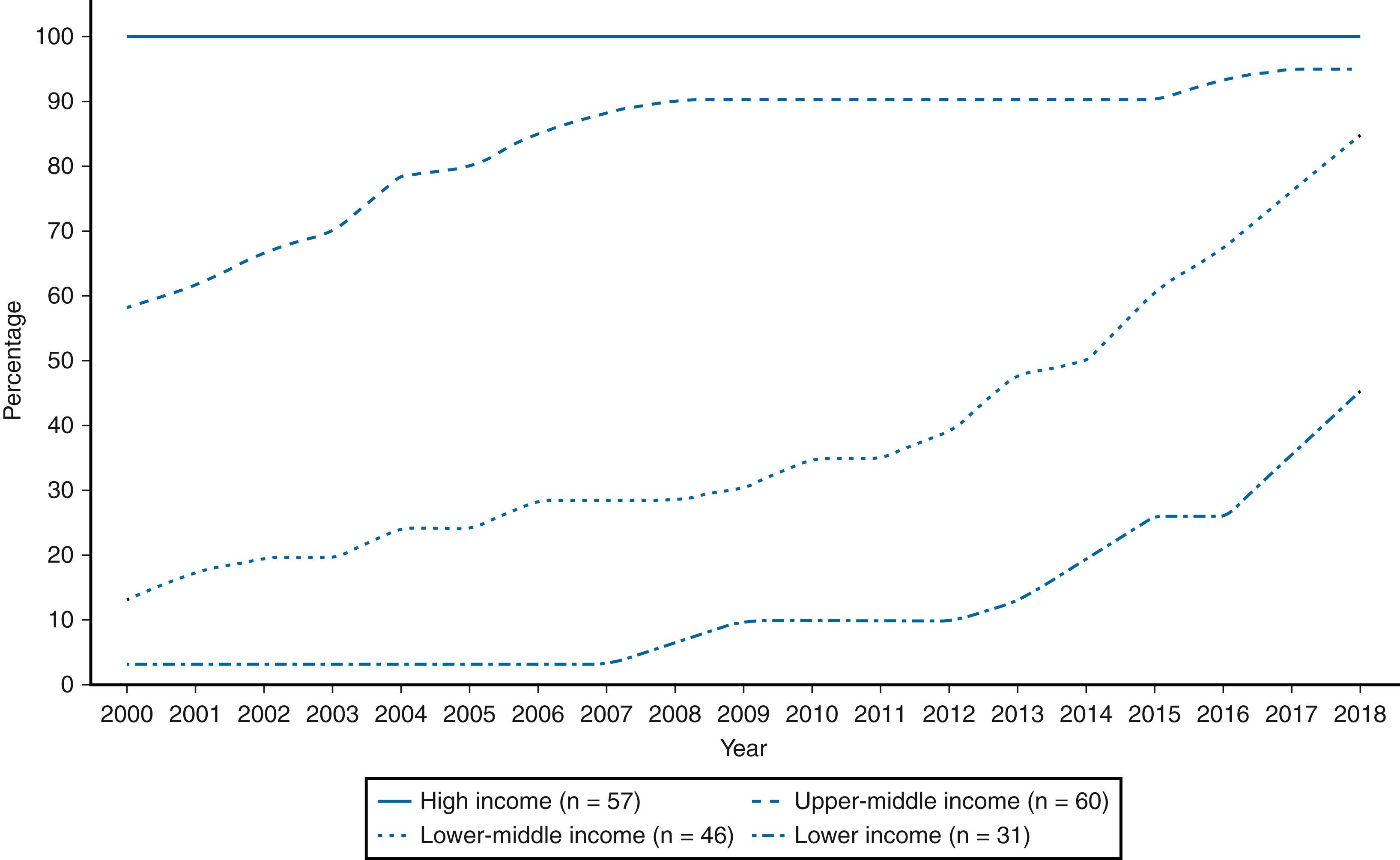
The Global Vaccine Action Plan, 2011–2020, has targeted measles and rubella elimination in the five WHO regions by 2020. In 2011, the WHO recommended that all countries introduce RCV in their routine immunization programs. Uptake of RCV in infants increased worldwide from 21% in 2000 to 40% in 2012 and to 69% in 2018. The verification of rubella elimination was confirmed in 81 (42%) countries. By the end of 2012, 132 (68%) of 194 countries had introduced at least one dose of RCV, and by 2018, 168 (87%) countries introduced RCV, compared with 99 (52%) of 191 countries in 2000 ( Fig. 221.4 ). Reported rubella cases globally declined by 86% in 2012 and by 96% in 2018 compared with 670,894 rubella cases reported in 2000.
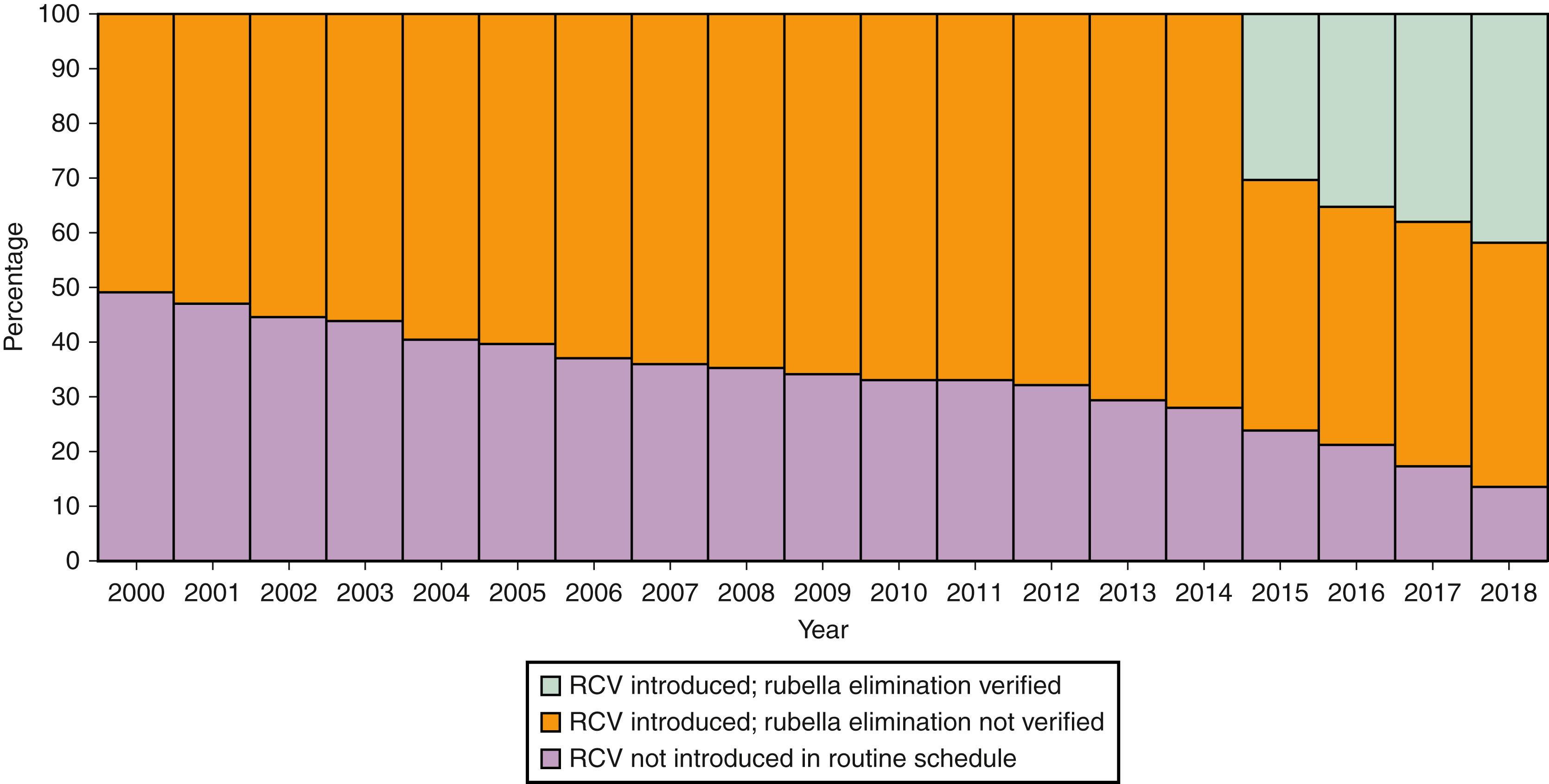
Accelerated progress has been made towards global rubella elimination since 2011. The progress is largely due to an increase in lower-middle-income and low-income countries introducing RCV in their national immunization programs ( Fig. 221.5 ). To achieve global rubella control and elimination, countries must implement high-quality immunization programs and robust case-based surveillance systems including genetic characterization of RuV isolates. , 2.4
The incubation period from exposure to onset of exanthem is 14–21 days (mean, 18 days). The illness is mild and often subclinical in up to 50% of cases. Nonspecific prodromal symptoms, which occur 1–5 days before onset of exanthem, include fever, eye pain, sore throat, arthralgia, and gastrointestinal complaints. Prodrome is rare in infants and young children.
The characteristic clinical findings are rash and suboccipital adenopathy. Rash usually begins on the face, spreads in a cephalocaudal direction to involve the entire body over the next 24 hours, and fades during the ensuing 2 or 3 days, also in a cephalocaudal direction. The median duration of rash is 3 days. Usually erythematous and maculopapular, the exanthem also can be scarlatiniform, morbilliform, or macular. In adolescents, the rash can be confused with acne, whereas in adults, the rash may be pruritic. Other symptoms include low-grade fever and lymphadenopathy, which can be detected for up to 1 week before rash onset. The posterior auricular and suboccipital lymph nodes are usually involved. Transient polyarthralgia and polyarthritis occur frequently in adults, especially in women.
Reinfection is uncommon. , Reinfection can occur during pregnancy in the presence of natural and vaccine-induced immunity with fetal infection and CRS but is very rare. , , , Reinfection usually is subclinical and nonspecific clinically, and the diagnosis is made only by serologic methods.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here