Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Rheumatology is the study of rheumatism, arthritis, and other disorders of the joints, muscles, and ligaments. , Rheumatologic conditions are highly prevalent. Nearly one-fifth of the United Kingdom population reports a chronic musculoskeletal system condition. Rheumatologic conditions commonly begin during working life, with the peak incidence of gout and rheumatoid arthritis in late working life, although many people are afflicted by inflammatory arthritis in early adulthood or even childhood. Rheumatologic conditions are complex and heterogeneous and require multi-disciplinary approaches to their management. Health systems across the world structure rheumatologic care in different ways, often involving physicians with rheumatologic and general medical expertise, orthopedic surgeons, experts in metabolic bone disease, nurses, physiotherapists, occupational therapists, orthotists, podiatrists, and others. The patient should always be at the center of the team. Pain is the main, although not the only, problem described by people with rheumatologic conditions. Rheumatologic pain management depends on principles and research from other disciplines, incorporating both pharmacologic and non-pharmacologic approaches. Specific aspects of the function of the musculoskeletal system and the nature of rheumatologic conditions may require disease-specific approaches to pain management. Pain and function are closely integrated, and physiotherapy and occupational therapy aim to address both concurrently. Common chronic conditions such as osteoarthritis, rheumatoid arthritis, and gout illustrate persistent chronic and episodic acute pain associated with musculoskeletal disease.
Osteoarthritis is the most common form of arthritis, and its prevalence increases with age. Osteoarthritis subgroups may be defined based on the joint distribution (e.g. knees, hips, or small joints of the hands) or precipitating factors (e.g. primary, post-traumatic, or secondary to inflammatory arthritis). Osteoarthritis affecting major weight-bearing joints substantially impacts mobility and function, although small joint osteoarthritis in the hands or feet can also cause substantial distress and disability.
Rheumatoid arthritis is the most common form of inflammatory arthritis, affecting approximately 1%-2% of the Western population. People with inflammatory arthritis report pain as their highest priority for improvement. , Inflammatory arthritis is an autoimmune condition characterized by immune-mediated inflammation of the joint lining (synovitis) and, if inadequately controlled, consequent joint damage and secondary osteoarthritis. Seronegative inflammatory arthritis may be associated with psoriasis, colitis, ankylosing spondylitis, or reactive arthritis that may follow infection, and each has its own specific genetic and immunologic characteristics. Rheumatoid arthritis has a predilection for small joints of the hands and feet and often affects the knees, shoulders, ankles, or wrists in a symmetrical polyarticular distribution. Seronegative spondyloarthropathies often affect small numbers of weight-bearing joints (oligoarthritis) and/or the axial skeleton, although polyarticular distributions are not uncommon. Seronegative spondyloarthropathies are characterized by enthesopathy (inflammation at ligament insertions) and tendon sheath inflammation. Therefore inflammatory pain might not always be associated with localized joint swelling, and “tender points” because of inflammation might overlap with those associated with fibromyalgia. Pain and stiffness in inflammatory arthritis vary during the day, with the most pronounced symptoms usually in the morning, explained in part by diurnal variation in endogenous glucocorticoids. Disease onset may be acute or insidious, and early disease may be difficult to diagnose. Classifying these conditions as “inflammatory” does not deny the important contributions of inflammation to joint pain in osteoarthritis and gout.
Crystal diseases are characterized by acute, severe episodes of pain caused by the innate inflammatory responses triggered by crystal deposits. High circulating uric acid concentrations because of inadequate renal clearance or increased production (e.g. during chemotherapy) may favor urate crystal deposition and attacks of gout. In calcium pyrophosphate deposition (CPPD), crystals may trigger attacks that are indistinguishable from gout and are therefore called pseudogout. Calcium pyrophosphate deposition in articular cartilage may be detected radiologically as chondrocalcinosis, even in the absence of acute attacks. In CPPD, crystal formation may be associated with various metabolic disorders involving divalent cations (e.g. Wilson’s disease). Gout and pseudogout may be associated with OA. Attacks typically affect a single joint, such as the knee, or, particularly in gout, first metatarsophalangeal, or interphalangeal joint. However, any synovial joint may be affected, and oligoarthritis or even polyarticular acute presentations may occur.
Non-inflammatory genetic connective tissue disorders associated with hypermobility are commonly associated with chronic musculoskeletal pain. Hypermobility syndromes include Marfan syndrome, Ehlers-Danlos syndrome, and Stickler syndrome, associated with genetic variation in fibrillin-1, collagen Col3/Col5, or Col2/9/11, respectively. However, most people with joint hypermobility (perhaps 10% of the population) have a medically benign condition on a polygenetic background.
Pain is rarely a single experience, and people with rheumatologic conditions may report multiple qualities of pain. Rheumatologic pain may be described using any of the terms embedded within the McGill pain questionnaire, both those commonly associated with nociceptive and those characteristic of neuropathic pain. The characteristics of osteoarthritis, inflammatory arthritis, and crystal disease often overlap, and these conditions may be comorbid. Evidence and experience from one rheumatologic condition may sometimes be generalizable to others, although caution should be exercised when such generalizations are not based on sound evidence.
In osteoarthritis, pain may occur only on weight-bearing or during joint movement or may be persistent at rest. Intermittent pain may be short-lived but severe, interspersed with periods that are pain-free or with less intense but constant pain. Acute flares may be spontaneous and unexplained or may be attributed to unaccustomed activity. Periods of exacerbation might last weeks or months, and the unpredictability of intermittent pain can prevent forward planning. Pain may be progressive or may subside or resolve.
In rheumatoid arthritis, pain is often associated with joint inflammation, as evidenced by soft tissue swelling, synovial effusion, increased local temperature, and tenderness. Each of these features typically varies over time, either spontaneously or with treatment. However, such variations in inflammation and pain are not always concurrent. An increase in pain may herald an imminent inflammatory flare, and reductions in acute phase reactants on blood tests may precede symptomatic improvement.
Episodic and severe pain characterizes crystal arthropathies, such as gout or pseudogout. During an attack, the affected joint becomes red, hot, and swollen and is exquisitely tender to the extent that movement or weight-bearing might be impossible, and the weight of bed clothes can be intolerable. These signs resemble acute infection, which is an important differential diagnosis that must be excluded. However, between attacks, the joint is typically pain-free.
Pain is not the only symptom of rheumatologic disease, and people differ in how they understand or separate their pain from comorbid symptoms. Pain exists alongside fatigue, stiffness, and disability. Sleep disturbance and depressive symptoms are more common in people with inflammatory arthritis than in the general population. Joint stiffness is particularly a feature of inflammatory joint diseases such as rheumatoid arthritis when it characteristically varies during the day and increases with increased inflammatory disease activity. Morning stiffness lasting more than an hour is not unusual during active synovitis, even though it may ease joint movement and activity. People with osteoarthritis also describe stiffness, which may reflect the inflammatory components of their disease. The reduced movement might also be because of structural changes, such as loss of articular cartilage, bony outgrowths at joint margins (osteophytes), or joint capsule fibrosis and tendon contracture. This type of stiffness, unlike stiffness associated with inflammation, displays slight diurnal variation.
Stiffness is a symptom rather than a sign, and the feeling that a joint does not move freely results from multiple mechanisms in and around the joint and in the central nervous system. Central mechanisms contribute to stiffness. People with rheumatoid arthritis who have undergone limb amputation may describe phantom stiffness, which varies during the day, and with generalized inflammatory flares of their arthritis. People often find it difficult to disentangle their experiences of pain and stiffness. Indeed “stiffness,” which is unpleasant and associated with tissue damage, would satisfy the International Association for the Study of Pain (IASP) definition of pain and might therefore be considered a component of the pain experience.
Fatigue is a common accompaniment of rheumatologic pain and has been reported as the second most troublesome symptom in people with rheumatoid arthritis. People may attribute their fatigue to sleep disturbance, for example, being woken by joint pain when turning during the night. However, sleep disturbance, unpleasant in its own right, is not an inevitable cause of fatigue, and additional biopsychosocial factors are important. Sleep disturbance and fatigue may be associated with evidence of central sensitization in osteoarthritis or rheumatoid arthritis, as it is also associated with widespread chronic pain. Circulating cytokines may contribute to fatigue in people with systemic inflammation. Fatigue is associated with a low mood, even in the absence of chronic musculoskeletal pain. Regardless of its underlying mechanisms, fatigue is associated with chronic musculoskeletal pain, is an important symptom in its own right, and can be a barrier to effective pain management. Overwhelming fatigue challenges the individual’s ability to cope with their pain and limits their ability to exercise as a form of therapy.
Difficulties with acceptance, psychological distress (anxiety and depression), catastrophizing, and cognitive impact are key components of rheumatologic pain, not less than in other chronic pain states. Patients may have difficulty accepting their rheumatologic condition, which in most cases will not be amenable to cure, but instead, require lifelong treatment with associated risks and inconvenience. When newly diagnosed, people often display psychological reactions comparable to grieving, reflecting their permanent loss of good health. Persistent pain may be interpreted (both by patient and clinician) to indicate persistent inflammation, treatment failure, and ongoing joint damage, even when inflammation has been completely suppressed. Fear that pain on activity inevitably indicates ongoing joint damage can be a barrier to rehabilitation and exacerbates declining function.
In addition to constituting the key emotional components of pain, psychological distress may be associated with central sensitization, augmenting nociceptive signaling, and the sensory dimensions of pain. Pain’s ability to distract attention and impair cognitive processing can compromise both work and leisure. With modern immunosuppressive therapy, people now developing rheumatoid arthritis might expect to live normal lives, engage in work, sports, and other activities alongside those without disease. However, the unpredictability of flares can prevent the planning of valued activities, including work and vacations, and undermine an individual’s self-esteem. Self-efficacy and successful self-management require control over persistent and intermittent symptoms and suppression of the underlying pathology.
Suppressing pain in people with rheumatologic conditions is urgently needed. Furthermore, delays in effective pain management can lead to disability and loss of gainful employment, which may be difficult to reverse. Inadequate pain management in people with rheumatologic conditions is a risk factor for future pain, psychological distress, lost work productivity, healthcare utilization, and poor response to later definitive interventions such as joint replacement surgery. , Pain management aims to improve symptoms and modify a key risk for future adverse outcomes, both for the patient and society.
Mechanisms of rheumatologic pain include changes in the peripheral musculoskeletal system, sensitization of peripheral and central nociceptive pathways, changes in brain connectivity, experience, and interpretation within a psychosocial context. Each pain mechanism is modulated by constitutional risk factors and comorbidities. Mechanisms may be shared or may differ between diseases, between individuals, and across time within an individual. Different mechanisms underlie different aspects of the pain experience, including pain quality, periodicity, associated symptoms, impact, and response to treatment.
The musculoskeletal system is designed for weight-bearing and movement, and biomechanical factors contribute significantly to rheumatologic pain. Pain on normal movement might indicate sensitization within either the peripheral or central nervous system. Sensitization might result from actions on peripheral nerves by chemical mediators, including growth factors or cytokines. Structural changes may redistribute forces within the joint. Thinned, irregular, or inelastic articular cartilage may be unable to adequately dissipate force, thereby activating nerves that are normally present in the subchondral bone. Hypermobility, for example, may be associated with patellar maltracking and consequent anterior knee pain. Nociceptive transduction may be increased by abnormal interactions between sensory nerve terminals and adjacent connective tissue molecules.
Loss of osteochondral integrity can expose subchondral nerves to molecules generated by the synovium, which may further stimulate subchondral inflammation and neuronal sensitization. Normal forces may activate nerves that have grown into the articular cartilage or knee menisci during structural disease progression. , These joint structures were not innervated in the normal joints.
Peripheral sensitization is a key component of rheumatologic pain. Inflamed or damaged tissues generate cyclo-oxygenase products, bradykinin, and nerve growth factor (NGF). These may either activate or sensitize the peripheral terminals of the primary nociceptive afferents. Urate (gout) or calcium pyrophosphate dihydrate (pseudogout) crystals trigger leukocyte degranulation and an acute innate immune response. In chronic rheumatoid arthritis or osteoarthritis, NGF, cytokines such as interleukin (IL)-1 and tumor necrosis factor (TNF)α, and chemokines may be upregulated in the synovium and subchondral bone. NGF has been identified as a key cause of peripheral sensitization in osteoarthritis and may also contribute to pain in rheumatoid arthritis. NGF induces phosphorylation of transient receptor potential cation channel subfamily V member 1 (TRPV1). It increases the expression of substance P, calcitonin gene-related peptide (CGRP), and brain-derived neurotrophic factor in the dorsal root ganglion and increases the levels of transmitters and modulators at the first synapse within the dorsal horn. Systemic factors, such as circulating autoantibodies or immune complexes, might also interact with primary afferent nociceptors to increase sensitization in rheumatologic conditions.
Persistent nociceptive barrage, the release of growth factors, and glial cell activation within the central nervous system, and changes in descending nociceptive control, contribute to central sensitization in rheumatologic conditions. With persistent joint pathology and nociceptive drive, central pain mechanisms become increasingly important. Functional connectivity may be enhanced between brain areas involved in sensory and emotional experiences, aggravating the emotional component of pain.
Pain experience depends on psychosocial context. An increased understanding of the underlying mechanisms of rheumatologic disease has led to major advances in immunologic treatments in recent decades. However, pain and inflammation can be discordant. A patient who is informed that their inflammatory disease is well controlled may feel that their pain experience is invalidated or not believed. Pain does not even need to be present in order to be a problem, for example, if a patient fears future severe or unpredictable flares.
Different individuals experience pain differently, despite apparently similar rheumatologic pathologies. Such diversity depends, in part, on genetic heterogeneity. Genotyping studies in painful osteoarthritis have identified polymorphisms in genes that are anticipated to influence joint shape and structure, but also in genes that regulate neuronal function or analgesic responses. Sex differences in rheumatologic pain similarly reflect both the propensity for joint pathology and differences in nociceptive processing.
Pain is the dominant symptom in most rheumatologic conditions, and patient-centered assessment should include evaluation of the nature, severity, and consequences of pain. Assessment should also address pain mechanisms, such as using biomarkers linked to diagnosis or pathology, to demystify this most bothersome symptom and help select treatments most likely to be of benefit. Careful attention to multimorbidity can inform prognosis, maximize health benefits, and reduce risks from pain treatments. Understanding and informing patients’ expectations and preferences facilitates treatment convergence
Pain assessment aims to identify its nature, severity, and importance to the patient and is essential for evaluating treatment response. Where pain is the presenting problem, assessment is inevitably incomplete without a detailed pain assessment. Pain is a subjective experience, and only patients can provide the gold standard measurement.
Tools for evaluating pain may be generic or disease-specific. Assessment tools used in clinical practice often differ from those used in research because of the different requirements of validity, feasibility, and sensitivity to change. In the clinic, we are interested in the individual’s pain, whereas research often seeks to understand the average effects across populations. Research might seek to explore mechanistic subgroups for whom there is currently no effective treatment, whereas clinical practice aims to identify problems for which interventions may be helpful. Pain is a subjective experience whose mechanisms are complex and are often unknown. Therefore assessment should embrace the patient’s subjectivity rather than seek objective indices that might bear little relevance to the patient’s problem.
In general, patient-centered pain outcome measures focus on the impact of pain on function (physical, psychological, sleep). Putative mechanistic tools, such as measuring neuropathic-like qualities, may inadequately address the problems of people with rheumatologic conditions. Pain rarely occurs as a symptom in isolation, such that rheumatologic-specific outcome measures often include pain alongside other factors such as stiffness and fatigue. , Pain outcome measures might be incorporated into the more generic quality of life tools such as the medical outcomes study short form 36 (SF36) and EuroQol questionnaires. These quality of life measures lack focus on individual joints but instead address the overall burden of commonly comorbid painful conditions. More specific outcome tools might be more able to detect responses to treatments that target a single joint or pain mechanism.
Osteoarthritis pain may be measured using self-report tools such as the intermittent and constant osteoarthritis pain (ICOAP) questionnaire, pain subscales of the Western Ontario and McMaster Universities arthritis index (WOMAC), or knee injury, and osteoarthritis outcome score (KOOS). WOMAC contains five items related to weight-bearing or non-weight-bearing pain and has demonstrated a two factor structure through which weight-bearing and non-weight-bearing OA pain may be associated with different pathologic mechanisms. For example, weight-bearing pain was specifically associated with bone marrow lesions in weight-bearing joint compartments on MRI. KOOS extends the WOMAC items to improve targeting for people with lower pain intensities or musculoskeletal injuries. The ICOAP was developed following qualitative assessment of pain in people with knee or hip OA and displayed a two factor structure corresponding to the intermittent or constant pain. People with early disease may experience predominantly intermittent pain, whereas those with advanced OA report intermittent on a background of constant pain, suggesting changes in pain mechanisms when the structural disease progresses.
The management of rheumatoid arthritis has traditionally focused on suppressing joint inflammation. Pain is assessed as one of the cardinal features of inflammation. However, pain in rheumatoid arthritis may also involve non-inflammatory mechanisms. The 28 joint disease activity score (DAS28) is a frequently used outcome measure in clinical trials of immunomodulatory treatments and clinical practice. This composite tool incorporates tender joint counts (TJC) and swollen joint counts (SJC) based on a standardized set of 28 joints that are relatively frequent and specific for rheumatoid arthritis. TJC is classified by the application of standardized pressure (to blanch the assessor’s nail bed) over each joint line. SJC is determined by the soft tissue swelling observed by a clinician. TJC and SJC are combined with a 100 mm visual analog scale of general health over the past week (VAS-GH) and a laboratory measure of an acute phase response (erythrocyte sedimentation rate [ESR] or C-reactive protein [CRP]). VAS-GH is highly correlated with patient-reported pain. Therefore DAS28 comprises self-reported (VAS-GH and TJC) and observed (ESR/CRP and SJC) components. Synovitis may increase ratings for each component, whereas central sensitization may selectively increase VAS-GH and TJC. Therefore the difference between TJC and SJC has been proposed as an index of non-inflammatory pain mechanisms in rheumatoid arthritis. High DAS28 is used in several countries as a criterion for commencing or continuing immunomodulatory treatments. The use of DAS28 in this way depends on a clear understanding of the contribution of pain to inflammatory disease assessment. Inappropriate escalation of immunomodulatory treatments in people whose pain is attributable to non-inflammatory mechanisms exposes them to risks with little likelihood of benefit.
Biomarkers can help direct the treatment of rheumatologic conditions by indicating diagnosis and identifying and measuring peripheral or central pain mechanisms. Biomarkers may predict the prognosis or treatment outcomes. In general, those who have suffered worse pain in the past may have a worse pain prognosis, and those whose pain will persist or deteriorate stand to gain the most benefit from treatment. Treatments that target a particular mechanism might be expected to work best for those in whom this mechanism drives pain. Biomarker levels might change before patient-reported outcomes deteriorate or improve, opening opportunities for early intervention and accelerating the development of new treatments. Biomarkers may enable early discontinuation to minimize the risk of treatment in those destined to not respond and those who will suffer adverse events.
Despite the overlap in pain mechanisms across diseases, the nature and responses to treatment of rheumatologic pain may vary between diagnoses. Biomarkers may be used to aid diagnosis. Specific connective tissue genetic variants directly cause hypermobility syndromes, such as Ehrlers’ Danlos syndrome. Other genetic variants may predispose to rheumatologic conditions (for example, human leukocyte antigen B27 [HLA-B27] in spondyloarthropathies). Genetic variants encoding neuronal proteins, such as ion channels, may either predispose to or protect against rheumatologic pain. Other genetic variants, such as μ-opioid receptors, might influence the responsiveness to analgesic drugs.
Circulating autoantibodies to citrullinated peptides or double-stranded DNA have moderate to high specificity for rheumatoid arthritis and systemic lupus erythematosus, respectively. Radiographic features contribute to diagnostic classification, such as osteophytes for osteoarthritis, bony erosions at joints affected by inflammatory arthritis, and spinal syndesmophytes or sacroiliac joint fusion in ankylosing spondylitis. Hyperuricemia is a risk factor for gout and radiographic chondrocalcinosis for pseudogout, although the definitive diagnosis of these conditions requires the identification of crystals in joint fluid aspirates. Diagnostic biomarkers might be particularly important in early disease when the link between diagnosis and pain mechanism may be the strongest. Comorbid osteoarthritis or fibromyalgia may reflect increasing mechanistic complexity of pain with increasing time from disease onset.
Diagnostic classification may help predict pain prognosis and treatment response. Radiographic evidence of osteoarthritis predicts pain persistence and persistent pain severity in people with knee pain. Diagnosis may also predict the response to treatment. However, diagnostic uncertainty may prevail, particularly in cases of early disease. The earliest stages of osteoarthritis might not be reliably classified by plain radiographs, and early rheumatoid arthritis may be mimicked by self-limiting parvovirus infection. Diagnostic uncertainty should not prevent adequate pain management.
Biomarkers that may point to pain mechanisms within the joint and are amenable to intervention include indices of inflammation and joint structural pathology.
Acute phase reactants such as ESR or CRP and ultrasound or MRI evidence of inflammation are useful indicators of peripheral drives to rheumatologic pain across multiple diagnoses, such as rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis. Raised acute phase in rheumatoid arthritis predicts worse structural outcomes, but not pain outcomes, perhaps because modern treatments can effectively suppress the inflammatory components of the disease. Similarly, raised acute phase or MRI evidence of spinal inflammation in ankylosing spondylitis predicts better outcomes on biologic therapy. However, precise mechanisms of rheumatologic pain often remain ill-defined, and these “indices” should not be over-interpreted as direct causes of pain. For example, CRP production in the liver is dependent on IL-6, and IL-6 blocking antibodies reduce circulating CRP concentrations irrespective of their effect on pain. Shortly after the onset of polyarthritis, the high acute phase might paradoxically be associated with a good prognosis if, rather than rheumatoid arthritis, the patient has an acute synovitic response to viral infection.
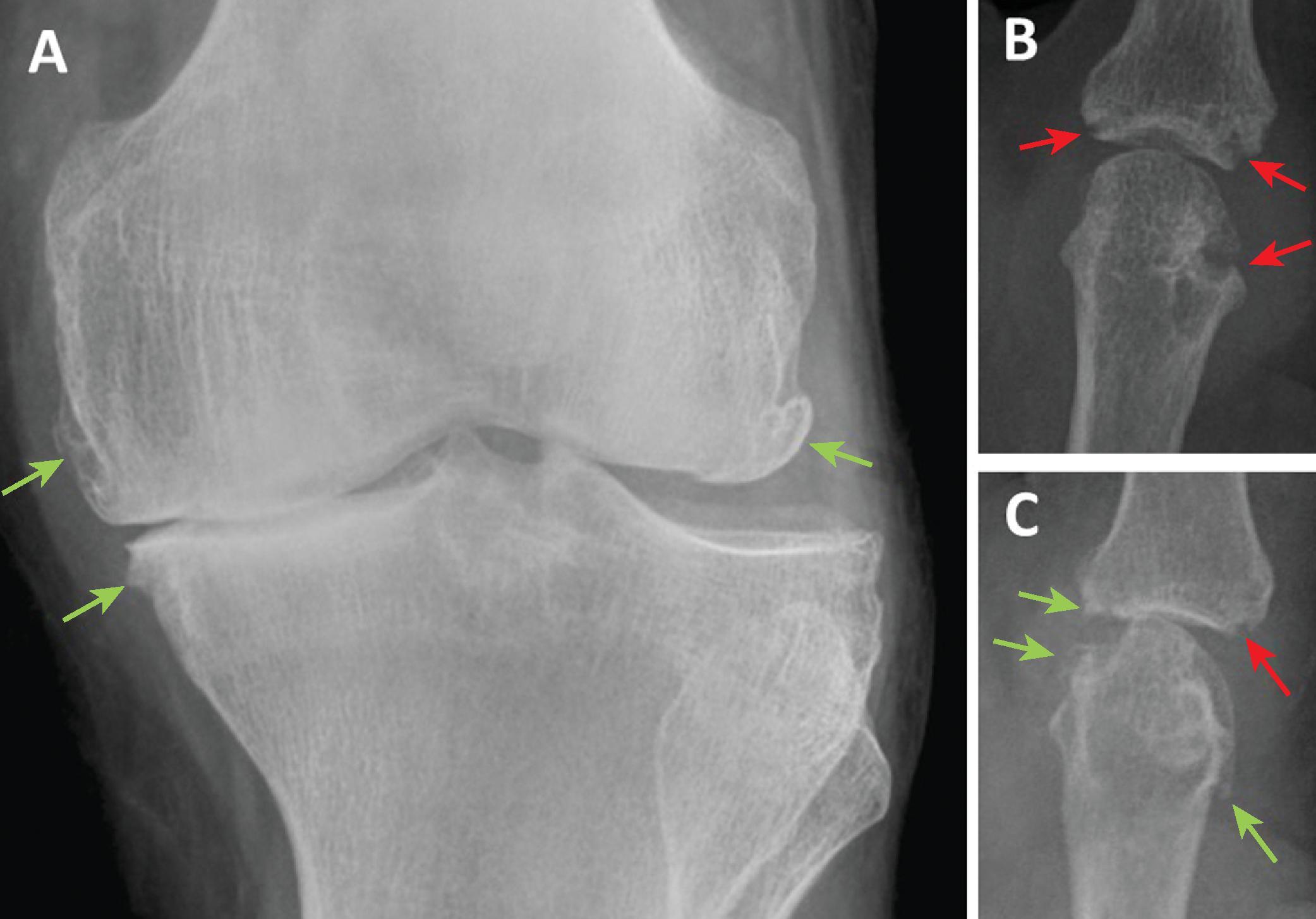
Greater joint space narrowing on weight-bearing knee radiographs is associated with greater pain severity in cross-sectional studies and predicts worse pain outcomes in longitudinal community studies of osteoarthritis but better outcomes from joint replacement surgery ( Fig. 46.1 ). However, the association between radiographic changes and pain in osteoarthritis is weak. Radiographic erosive joint damage or comorbid hand osteoarthritis at presentation with rheumatoid arthritis may be only weakly associated with pain and does not predict chronic pain with long term treatment, consistent with a predominant drive to pain from non-structural mechanisms. MRI may provide more detailed biomarkers of the peripheral pain mechanisms. In osteoarthritis, subchondral bone marrow lesions and synovitis have consistently been associated with pain, both in cross-sectional and longitudinal studies. Other MRI features, including knee meniscal extrusion or cartilage defects, may also be associated with pain, and close associations between different aspects of osteoarthritis structural pathology raise uncertainty about which features directly cause pain.
Quantitative sensory testing (QST) evidence of sensitization is associated with higher concurrent pain severity in osteoarthritis, and rheumatoid arthritis. Mechanical stimulation modalities such as blunt or cuff-induced pressure or punctate stimulation of the skin may be more relevant to rheumatologic pain than thermal modalities, although it is uncertain whether sensitization mechanisms are specific to particular nociceptor subtypes. Baseline QST evidence of sensitization predicts worse pain at follow up and worse pain progression. Much evidence has been derived from static mechanical QST modalities, such as pressure pain detection thresholds (PPT). PPT distal or remote from affected joints has been taken as an indication of central sensitization, whereas PPT at the joint may reflect a combination of peripheral and central sensitization. Dynamic QST modalities such as temporal summation (TS) and conditioned pain modulation (CPM) more directly reflect central sensitization. Greater TS may indicate sensitization at the spinal cord, whereas lower CPM may indicate supraspinal deficiencies in endogenous analgesic mechanisms. Dynamic QST modalities may better predict pain outcomes in rheumatologic diseases than does PPT. , Evidence of central sensitization might predict worse pain outcomes in rheumatologic conditions by indicating resistance to treatments directed at the peripheral nociceptive drive.
Several phenotypic characteristics have been associated with QST evidence of central sensitization, leading to the proposal that self-report questionnaires might be used to indicate central sensitization in people with rheumatologic conditions. In people with knee pain, psychological distress (depressive or anxiety symptoms), catastrophizing, sleep disturbance, fatigue, cognitive interference, neuropathic-like symptoms, and more widespread pain were each associated with lower PPT distal to the affected knee. These eight characteristics each contributed to a single factor, the central mechanisms trait, which was more strongly associated with PPT than was any one of the eight characteristics alone. Higher central mechanisms trait scores were associated with more severe pain and predicted worse pain outcomes in community dwellers with knee pain. Similar self-report characteristics predict poor pain outcomes in other rheumatologic and non-rheumatologic conditions, and contribute to the diagnostic classification of fibromyalgia.
Comorbid conditions may be associated with rheumatologic disease. Rheumatoid arthritis is associated with cardiovascular diseases, diabetes mellitus, and fibromyalgia. Rheumatologic conditions increase in prevalence with age. Therefore multimorbidity is common, with or without causal association between diseases. Multimorbidity is associated with increased pain severity and worse pain prognosis, and contributes to frailty and loss of resilience to future health challenges. Concurrent morbidities might augment pain from rheumatologic conditions. Therefore treatment of comorbidities might improve pain outcomes. Morbidities or risk factors for future development may influence the choice of pain management strategy. Some, but not all, immunosuppressive therapies help with both cutaneous psoriasis and psoriatic arthritis. Comorbidities might be risk factors for adverse events from treatment, such as gastrointestinal bleeding on nonsteroidal anti-inflammatory drugs (NSAIDs) or analgesic medication abuse. Morbidities that pose an anesthetic or surgical risk, such as hypertension or obesity, should be addressed before elective surgical intervention should be considered. A holistic approach is required to avoid interruption of pain management by intervening in acute medical needs. Morbidities may be painful or non-painful.
Rheumatologic conditions coexist with comorbidities, which further influence the pain experience. Fibromyalgia is a common accompaniment of both inflammatory and non-inflammatory rheumatologic conditions. The extent to which phenotypic similarities between rheumatologic conditions and fibromyalgia reflect shared mechanisms remains uncertain. Chronic widespread pain in people with hypermobility might be associated with comorbid fibromyalgia or central sensitization but might also be an indication of widespread abnormalities in peripheral nerve function. Fibromyalgia may be considered as a comorbid diagnosis, and also fibromyalgianess may be viewed as a continuous trait linked to pain mechanisms. Each may be classified or measured in people with rheumatologic conditions by questionnaires; the Symptom Severity Scale, which addresses fatigue, cognitive, and somatic symptoms, together with WideSpread Pain Index.
Osteoarthritis is a common accompaniment of other rheumatologic conditions. Primary osteoarthritis (OA) is a prevalent condition. Similar to all non-fatal, incurable conditions, osteoarthritis increases in prevalence with age. Rheumatologic conditions may predispose patients to osteoarthritis. Hypermobility may be associated with joint trauma and inflammatory arthritis with erosive joint damage, each of which is a risk factor for secondary osteoarthritis.
Obesity is associated with osteoarthritis and increased pain. The association of body weight with osteoarthritis is multifactorial, partly based on genetic and biomechanical factors. Adipokines may contribute to augmented pain processing. Reciprocally, where arthritis pain is a barrier to exercise, weight reduction may be difficult to achieve. Diabetes mellitus may be associated with obesity and is an autoimmune condition associated with inflammatory rheumatologic conditions. Diabetes mellitus is associated with increased pain, even in the absence of demonstrable neuropathy. A negative association between osteoarthritis pain and hypertension has been attributed to the use of β-blocking medications, although causality awaits demonstration by a randomized controlled trial.
Rheumatoid arthritis and systemic lupus erythematosus are associated with an increased risk of cardiovascular disease, which might be attributed to the effects of systemic inflammation or the adverse effects of treatments such as glucocorticoids or NSAIDs. Cardiovascular risk assessment tools incorporate rheumatoid conditions, including age, sex, smoking status, diabetes mellitus, hypertension, ischemic heart disease, and chronic kidney disease.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here