Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The recurrence of anterior glenohumeral instability is not uncommon following primary surgical repair with reported rates of 15% to 30%. Optimal management for recurrent shoulder instability remains controversial; as many acceptable surgical options exist, and the decision-making process is heavily case dependent. Numerous level one studies have looked at risk factors for recurrent instability, consistently reporting a young age and arm position at the time of initial dislocation and repair, being of male gender, a history of multiple preoperative dislocations and instability events, hyperlaxity, and participation in competitive contact/overhead sports as the most common risk factors for recurrence following primary repair. Furthermore, technical errors and failure to address concomitant shoulder pathologies during index surgery can place a patient at risk for recurrence. Perhaps the most important predisposing risk factor in recurrent anterior glenohumeral instability is the presence of glenoid bone loss, as recurrence can be seen in as high as 90% to 100% of untreated cases. The surgeon should consider individual risk factors for revision surgery that may affect outcomes such as patient age, activity level, and presence of coexisting shoulder injuries. Patients with recurrent glenohumeral instability can benefit from either arthroscopic or open treatment.
With patients experiencing recurrent instability, it is crucial to address any bone loss or concomitant injuries in the context of recurrent instability to ensure that the best preoperative decision is made. The goal of treatment is to restore the anatomy and biomechanics of the shoulder and prevent further instability and damage to the joint. This chapter describes the diagnosis, our preferred surgical technique, and the outcomes following revision surgery for patients with chronic/recurrent anterior-inferior shoulder instability that have failed primary repair.
A thorough medical history is an integral component of the preoperative workup. A timeline with details about the patient's initial dislocation (including age at initial dislocation), history of instability events, type of instability (subluxation or dislocation), presence or absence of mechanical catching, previous surgical care and treatment thus far, and any history indicative of concomitant shoulder or connective tissue pathologies should be obtained. Questions with an emphasis on injury mechanics, especially direction of instability (anterior, posterior, or multidirectional), magnitude of trauma required for instability, and ease of reduction (self vs. assisted) are essential for diagnosis of the type and severity of shoulder instability and its management.
The diagnosis of glenoid bone loss in the context of failed shoulder stabilization surgery should be suspected when patients describe a high energy mechanism of injury at initial dislocation, particularly with the arm in abduction ≥70 degrees and external rotation ≥30 degrees. Symptomatic bone loss from patient history may be reported as instability events occurring primarily during mid ranges of motion (20 to 60 degrees of abduction). Patients with glenoid bone loss often notice that dislocation/subluxation of the glenohumeral joint has progressively become easier with a prolonged history of instability symptoms. Instability can present with variable symptoms. Some may report feelings of impending instability or pain at extreme ranges of motion. Others will experience subluxation or obvious dislocation with certain shoulder positions, specifically abduction and external rotation, or overhead activities. In some instances, patients may experience recurrent transient paresthesia and weakness stemming from recurrent subluxation.
Emphasis should be placed on diagnosis of instability and assessment of the bony deficit, but the treating surgeon should consider other pathologies, particularly in patients over the age of 40, who may have concomitant rotator cuff, labral, and cartilage injuries. These injuries, such as superior labrum anterior to posterior (SLAP) tear and rotator cuff tear, are important to diagnose in an initial history, because delaying surgery can lead to tissue of lesser quality and an inferior healing environment. It is also pertinent to obtain information regarding patient activity level and participation in contact/overhead sports while determining realistic treatment goals.
Achieving the right diagnosis in a patient with recurrent instability may be difficult; therefore it is important to assess for other shoulder pathologies along with the standard shoulder exam. Physical exam should begin with inspection to identify for obvious deformity (associated with a history of traumatic instability), rotator cuff atrophy (in patients greater than 40 years of age) and scapular dyskinesia (a history suggestive of a rotator cuff pathology or prolonged instability). Then a careful neurovascular evaluation of the entire upper extremity, assessment of passive and active range of motion (ROM), and rotator cuff integrity and strength should be performed by the examiner. Patients can have normal findings upon inspection, palpation, ROM and axillary nerve testing with preserved shoulder girdle and parascapular muscle strength. Patients may report feelings of shoulder instability and/or pain with overhead motion. Some may have limited abduction and external rotation because of discomfort and/or pain secondary to instability of the humeral head on the anterior glenoid rim. A comprehensive evaluation of the musculature of the rotator cuff and scapula is important as they comprise the dynamic relationship between the glenoid and humeral head and contribute to shoulder stability. The magnitude and direction of shoulder instability should be documented.
Provocative tests specific to assessing anterior shoulder integrity should be performed. These exams are most reliable when the patient is relaxed. Apprehension, rather than pain, should be used as the diagnostic criterion for a positive finding. Patients with recurrent instability may also have significant guarding during special maneuvers such as load and shift test, drawer test and apprehension sign/augmentation test, Jobe's relocation test, and Gagey hyper abduction test. These tests are highly predictive for instability when performed correctly. The shoulder of interest should be compared to the contralateral side for the reproduction of a sense of instability. Grading of instability during the movement of the humerus into an abducted and externally rotated position helps to guide the treatment plan ( Fig. 43.1 ).
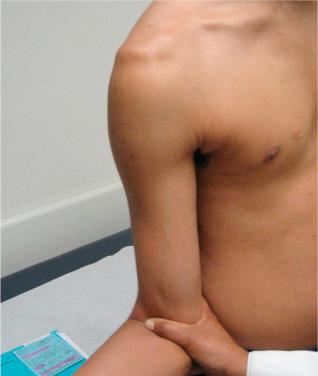
During evaluation, it is imperative to assess for a Hill–Sachs lesion, a SLAP lesion, a Bankart lesion, a humeral avulsion of the inferior glenohumeral ligament (HAGL) lesion, and other capsular injuries. For example, if the humerus begins to dislocate at mid ranges of abduction with external rotation, this may be indicative of an engaging Hill–Sachs lesion or, significant glenoid or humeral head bone loss, and warrants further evaluation. An anterior-inferior labral tear may present with positive load and shift, modified load and apprehension relocation testing, and a sulcus sign. Performing an O'Brien test can be helpful for assessing the superior labral attachment. In a patient with recurrent instability over 40 years of age, the surgeon should evaluate for a rotator cuff tear.
It is important to distinguish asymptomatic laxity from instability and the perception of laxity by patients during a subluxation or dislocation event. Boileau et al. described the difficulty in distinguishing between laxity and instability on young hyperlax overhead athletes who experienced reproducible pain upon anterior apprehension, which relieved upon relocation but had no true instability. Generalized ligamentous laxity can be assessed using the Beighton Hypermobility Criteria: metacarpophalangeal hyperextension, thumb to forearm sign, elbow or knee recurvatum, and the ability to forward flex the trunk with knee in full extension and palm resting on the floor.
The examiner should assess for glenoid bone loss as part of a comprehensive shoulder exam. Reproducible anterior translation of the humeral head over the glenoid rim is indicative of glenoid bone deficit. While patients with glenoid bone loss usually have a positive apprehension test at 90 degrees of abduction and at 90 degrees of external rotation, some may have positive signs at lesser degrees of abduction and external rotation, between 30 and 90 degrees. This is largely in part due to concomitant injuries leading to disproportionately placed forces causing the feeling of apprehension. Performing shoulder apprehension test in various degrees of shoulder abduction and external rotation may provide additional insight on the extent of bone loss. Patients with positive findings of instability and signs of glenoid bone loss require diagnostic investigation with advanced imaging studies.
Appropriate imaging work up is critical to guiding the clinical decision-making process for the type of revision operative procedure to perform. First, routine radiographs should be obtained, true oblique anteroposterior (AP) view (Grashey view), axillary, lateral, and scapular Y. The true AP view allows assessment of the glenohumeral joint for degenerative joint changes. The scapular Y view allows assessment for glenohumeral joint alignment. Further specialized radiographic imaging of the glenoid rim may be warranted in patients with recurrent instability to assess for bony defects of the anteroinferior glenoid rim: apical oblique, West Point view, or Didiee views. In the context of acute traumatic dislocation, a West Point view or its alternative an apical oblique radiograph allows enhanced visualization of the anteroinferior glenoid to detect for an osseous Bankart lesion. The addition of a Stryker Notch view and a true AP view with internal rotation allows the surgeon to visualize humeral head defects like Hill–Sachs lesions. While initial assessment should begin with radiographs, they often fail to detect or accurately quantify the degree of glenoid bone loss. Up to 60% of bony lesions can be missed upon radiographs alone ; therefore evaluation with magnetic resonance imaging (MRI) and computed tomography (CT) is warranted for potential revision cases to determine the integrity of the surrounding soft tissue and bony structures, respectively. CT with contrast is an especially effective method because it allows enhanced detection of the exact degree and location of osseous defects in comparison with a standard CT scan.
High failure rates following surgery and recurrence of instability are known to be associated with soft tissue incompetence and bony deficiencies. Preoperative diagnostic MRI with intra-articular gadolinium can identify any concomitant pathologies of the labrum, biceps anchor and pulley, glenohumeral (GH) ligaments and rotator cuff. The most common pathologic finding associated with instability is an avulsion of the glenohumeral ligament labral complex from its attachment to the anterior-inferior glenoid. Bony Bankart lesions are reported between 4% and 70% of anterior glenohumeral instability cases. Perhaps the most reliable modality for obtaining detailed shoulder pathologies is a three-dimensional (3D) CT scan as these images are highly correlated with arthroscopic findings. While two-dimensional (2D) CT and sagittal oblique MRI (also 2D) can aid to visualize glenoid bone loss and newly formed glenoid fragments, they are unable to adjust for patient-specific glenoid topography and version. An MRI requires reasonable resolution to differentiate a glenoid bone injury from a soft tissue injury, while 2D CT affects the gantry angle. Furthermore, even with additional MRI slices and views, these images may still underestimate glenoid bone loss.
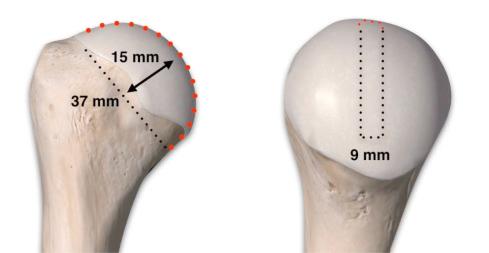
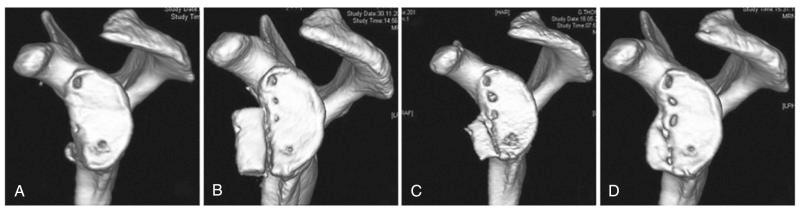
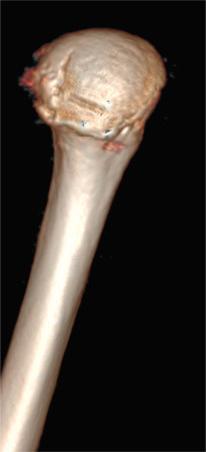
The most accurate method for measuring the magnitude of glenoid bone loss and for assessing the humeral head for a Hill–Sachs lesion is a 3D CT scan with digital subtraction of the humeral head on sagittal oblique imaging ( Fig. 43.2 ). Software for 3D CT allows digital subtraction of the humeral head from the glenohumeral complex resulting in a precise en face view of the glenoid surface and vault and visualization of the scapula. Not only can 3D CT provide information about the extent of glenoid bone injury but also the type of injury (i.e., acute fracture, partial attritional bone loss, or complete attritional bone loss) ( Figs. 43.3–43.5 ). As glenoid bone stock loss usually occurs along a line parallel to the long axis of the glenoid from 12 o'clock to 6 o'clock, several studies have utilized this information to geometrically quantify bone loss from 3D CT images. The inferior aspect of the glenoid on an en face view has been described as a shape of a true circle, with the circle drawn with the center point roughly at the glenoid bare spot in the inferior two-thirds of the glenoid. The glenoid bare spot can be identified upon CT as the area of cartilaginous thinning and increased subchondral density over the tubercle of Assaki. The location of the bare spot is at the intersection between the longitudinal axis and the widest AP diameter of the glenoid. Using this point as the center, a circle is drawn and the radius is determined. The distance from the bare spot to the anterior edge is then measured. One method is to use the ratio of the radius to the previous distance in order to quantify the percentage of glenoid bone loss. A simpler method also relies on the bare spot and quantifies bone loss as the difference between distance from the bare spot to the anterior edge from the bare spot to the posterior edge over twice the distance of the bare spot to the posterior edge. Another important factor of instability is the length of the bony defect; it has been reported that defects greater than half the widest AP diameter results in 70% or less resistance to dislocation. Perhaps the simplest technique of all for determining glenoid bone loss is a linear measurement in millimeters of the defect from the circle.
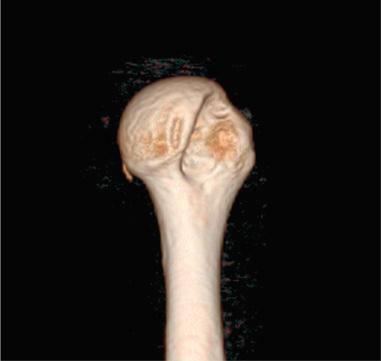
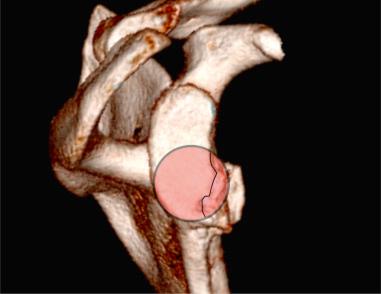
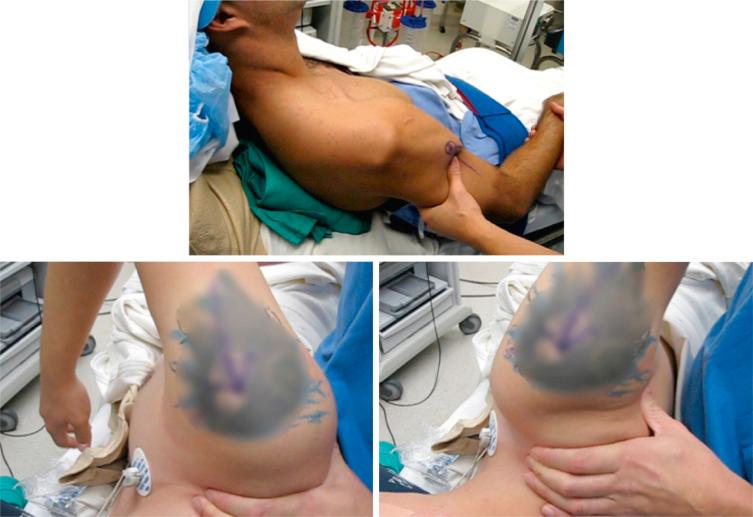
Furthermore, 3D CT imaging provides information about glenoid inversion, inclination, and the relationship between the glenoid and humeral head (engaging or nonengaging, on-track or off-track). Burkhart and DeBeer were the first to define an engaging Hill–Sachs lesion as the humeral head coming into contact with the rim of the anterior glenoid during 90 degrees of abduction with external rotation between 0 and 135 degrees. In their study of 101 athletes undergoing surgical repair for traumatic anterior-inferior instability, a Hill–Sachs lesion was significant only if it was classified as engaging. In the study, they utilized arthroscopic viewing portals to determine the “engagement” status of the Hill–Sachs lesion. After an average follow-up of 27 months (range, 14 to 79 months), they reported that 100% of patients with engaging Hill–Sachs lesions experienced recurrent shoulder instability and that soft tissue repair alone was sufficient without the presence of a concomitant engaging Hill–Sachs lesion. Later studies have also reported that engagement of a Hill–Sachs lesion, rather than the size, has been a greater predictor of recurrent instability. True engagement occurs when the lesion extends over the medial margin of the glenoid track, whereas a nonengaging lesion will pass diagonally across the anterior glenoid, allowing it to make continuous contact with the articular surface of the glenohumeral joint and prevent bone to bone contact. Engagement can be determined either by a CT scan or via arthroscopy while the arm is in abduction-external rotation. An MRI study conducted by Gyftopolous et al. demonstrated that the on-track off-track method was an excellent way to predict engagement, as they found it 84.2% accurate.
When selecting treatment options for recurrent shoulder instability, the most important factors to consider are the magnitude of glenoid bone loss or injury, the patient's expectations for outcomes, and their anticipated postoperative level of activity. The management of recurrent instability following failed stabilization procedure(s) is heavily reliant on clinical assessment and imaging findings. A thorough preoperative evaluation including a clear surgical history with information regarding patient age, sports participation (overhead/contact) if any, lifestyle/activity level, any medical conditions (smoking, current medications), or any voluntary component that may affect the patient's surgery or ability to follow through with rehabilitation can help set reasonable expectations for treatment outcomes. Other important prognostic factors include capsulolabral integrity, concomitant shoulder pathologies, poor tissue quality, and any osseous loss of the glenoid or humeral head.
Nonoperative management can be acceptable for patients with less than 20 degrees of glenoid bone loss, low activity demands, greater age and higher surgical risk, or a history of voluntary glenohumeral dislocation. However, it is imperative to rule out glenoid bone loss as a contributing factor for recurrent instability in these patients with a history of failed shoulder stabilization surgery.
Both open and arthroscopic surgery can provide acceptable options for soft tissue or bony repair in revision instability cases when managed appropriately. Although the optimal treatment for glenoid bone loss is not well defined, current literature suggests recurrent instability with less than or equal to around 5 to 7 mm of glenoid bone loss (0% to 20% and up to 25% in some cases) may benefit from soft tissue arthroscopic stabilization with multiple suture anchors and a posterior repair. In these patients with minimal bone loss, studies have reported excellent postoperative outcomes with the incorporation of a Bankart fragment should the operating surgeon choose to do so. Studies with large cohorts of greater than 50 patients comparing the efficacy of open and arthroscopic Bankart repair techniques in preventing recurrent instability demonstrated no significant differences in postoperative shoulder stability outcome scores. While one study noted a significantly higher ( P = .017) ROM in the arthroscopic group ; there is a lack of well-defined outcome scoring systems to quantify the success of either surgical approach. Open stabilization is preferred for patients at a higher risk of recurrent instability, while arthroscopy is acceptable for patients with a lower risk. In general arthroscopy is a less invasive procedure that may allow a greater degree of return of ROM, but the operating surgeon's level of expertise and comfort level with either surgical techniques should be considered.
Bone loss is the most common reason for a failed stabilization surgery. Though it varies in the literature, recurrence of instability can occur with as little as 15% to 20% glenoid bone loss. One study suggested that 13.5% glenoid bone loss might even render surgical intervention with bony reconstruction. To successfully correct instability, bony augmentation of the anteroinferior glenoid rim allows restoration of the anatomical arc of glenohumeral motion. While bony fragment fixation with open capsulolabral repair is considered the gold standard, arthroscopic repair is also acceptable. A 20% to 25% bone stock loss correlates to approximately 6 to 8 mm of the glenoid from anterior to posterior. The bare spot of the glenoid provides a reference point for measuring bone loss, and varies between 23 and 30 mm with the majority of shoulders ranging from 24 to 26 mm. Five percent of glenoid bone loss correlates to approximately 1 to 2 mm of bone, further emphasizing that only a relatively small amount of bony injury is needed for clinical relevance. It is also important for the surgeon to consider attritional bone loss of the remaining fragment in the setting of glenoid defects. McNeil et al. showed that 91.4% of patients exhibited moderate to severe attritional bone loss independent of the initial glenoid bone loss, implying that this fragment is insufficient to reconstitute the glenoid bone stock. Of note, this attritional loss was more pronounced in patients with a longer duration of instability symptoms, which may help further guide the surgeon's decision-making.
Recurrent instability with 25% to 30% glenoid bone loss (~6 to 8 mm) should be treated with open repair with bone augmentation to reconstitute the glenoid arc. Generally, anteroinferior glenoid defect of this size cannot be managed through soft tissue repair alone due to the deficit in articular arc. Furthermore, patients with chronic instability may have attritional bone loss secondary to resorption of the previous osseous Bankart fragment. During the discussion of treatment, patients’ expectations and postoperative desires should be thoroughly addressed in situation of increased glenoid bone loss as recurrence of instability is greater with arthroscopic than open repair. These reconstruction techniques include Bristow and Latarjet. While coracoid transfer has been the standard for over 50 years with excellent postoperative stability and function, there are concerns regarding eventual postoperative arthrosis and limitations in shoulder motion, especially after nonanatomical coracoid reconstruction. When using a different bone block to address the glenoid defect, graft options include autogenous iliac crest graft and various frozen (femoral head and humeral head) and fresh osteochondral allografts (distal tibial plafond). While the most logical reconstruction is to replace the missing glenoid bone with similar bone, glenoid allograft specimens are less commonly available, have been associated with contamination, and do not present proper curvature. Iliac crest autograft may be preferred over a distal tibia allograft (DTA) when an extensive defect is present as it allows a more adequate supply of bone for reconstruction.
Not only should the treating surgeon quantify the amount of glenoid bone loss, but he or she should also assess for the presence of an engaging Hill–Sachs lesion, capsular tissue quality, and associated defects or other soft tissue (rotator cuff, bicep tendon) pathologies. Addressing an engaging Hill–Sachs lesion can prevent extensive stressing of a revision repair on the capsuloligamentous labral complex during abduction and external rotation. A dynamic evaluation of shoulder abduction and external rotation under direct visualization during arthroscopy can provide the most direct view of engaging lesions, and help guide definitive management. When an engaging Hill–Sachs lesion is present, surgical intervention is warranted. The preferred methods for humeral head lesions include direct auto- or allograft transfer, remplissage, disimpaction, and prosthesis replacement. All these techniques increase the bony surface area of the humeral head to prevent further engagement and instability.
When it comes to deciding if a patient is a candidate for humeral head prosthesis (i.e., humeral head hemiarthroplasty), the size of the Hill–Sachs lesion is the most influential factor. Hill–Sachs lesions involving greater than 40% of the humeral head surface are generally managed with prosthetic replacement. Both hemiarthroplasty and total arthroplasty are associated with failure rates, especially in the younger and active patient. Patients greater than 65 years of age may benefit from hemiarthroplasty or total shoulder arthroplasty at a lower percentage of bone loss due to the low physical demands placed upon their glenohumeral joint. The procedure can also be indicated in young patients with chronic defects and significant cartilage degeneration.
Hill–Sachs lesions are especially clinically relevant in the setting of glenoid bone loss, as there is an increased likelihood of the humeral head engaging the glenoid. When determining treatment of combined lesions, the concept of the “glenoid track” is a prognostic indicator of surgical success. Metzger et al. found that the glenoid track could help predict engagement, as 84.5% of patients with lesions out of the track showed clinical engagement compared to the 12.4% of patients with lesions inside the track that had functionally engaging lesions ( P < .001). Thus the glenoid track can identify lesions at high risk for engagement and help guide treatment options, especially when considering bone augmentation. Prior to the study executed by Shaha et al., most advanced knowledge about the glenoid track was the algorithm developed by Di Giacomo et al., which could predict the track mathematically. The algorithm reads as follows: glenoid track width = .83D − d (D as the diameter of the glenoid and d as the amount of glenoid bone loss [posterior radius–anterior radius]). Shaha et al. found that out of the 49 patients in their cohort that were classified as on-track, only 4 (8%) of the patients experienced any form of instability recurrence. Eight patients were noted to be off-track, and of those patients 6 (75%) were documented to sustain recurrent dislocations due to engagement. They also found the negative predictive value of the study to be 92%, meaning that when the shoulder was on track, arthroscopy failed a mere 8% of the time ( Fig. 43.6 ).
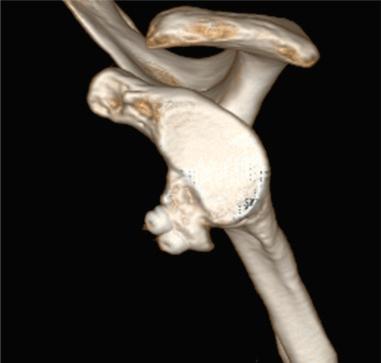
Overall, surgical decision-making is dependent on the extent of glenoid and humeral osseous deficiency, surgeon experience, and training with specific reconstruction techniques and patient-specific factors (occupational and athletic demands). The surgeon must carefully consider the type of procedure and graft if indicated that would be of the most benefit to the patient, and treat concomitant pathologic conditions.
Optimal treatment should be guided by the extent of glenoid bone loss and anticipated patient activity levels. Several surgical options can be considered in revision cases for anterior shoulder instability as both bony and capsuloligamentous pathologies may be present. The goal of a revision stabilization procedure is to restore the anatomy and ROM, and to repair the labrum with appropriate tensioning while preventing further complications (chondral damage, neurovascular injury). Glenoid bone deficit is most commonly addressed with two different methods: a Latarjet or Bristow procedure, or allograft augmentation. Restoration of the surface area can be achieved with an allograft of the iliac crest (bony implant) or distal tibia (bony and articular implant). With the option of utilizing either an autograft or an allograft comes the strategy of deciding which choice is optimal for the patient. Autografts are advantageous because they offer beneficial properties such as being osteoinductive, osteoconductive, and immunogenic. Allografts offer the advantage that they avoid causing donor harvest site morbidity and do not cause pain or sensory disturbance, unlike autograft harvesting. It is imperative to adequately replace the glenoid bony stock, whether it be autograft or allograft, for the success of the revision repair. However, in cases when a graft is insufficient, a hemiarthroplasty or a total shoulder arthroplasty should be considered. Patients may benefit more from a prosthesis in the context of humeral head deficiency or significant glenoid bone loss. Further stabilization in patients with capsular laxity can be achieved through closure of the rotator interval; however, this results in decreased external rotation in adduction and increased potential for posterior inferior translation for some patients.
Removal of symptomatic hardware or osteophytes may improve pain in some cases of arthropathy. Lysis of adhesions should be performed for patients with stiffness secondary to adhesions. Repair of the rotator cuff and débridement of diseased soft tissue may be necessary in some cases of recurrent instability. Revision surgery can be successful with either open or arthroscopic surgery; however, arthroscopic repair requires a higher level of expertise and is preferred for isolated capsuloligamentous repair. An open repair approach better addresses glenoid bone loss, humeral head lesions (Hill-Sachs), subscapularis deficiency, and large capsular defects.
In our experience, an arthroscopic revision shoulder stabilization procedure is best to correct shoulders with inappropriately or untreated capsular laxity/injury and labral injury. An open revision shoulder stabilization procedure, such as a Latarjet or an allograft bony augmentation procedure, is indicated for patients with severe glenoid bone loss, Hill–Sachs lesion or a severe subscapularis deficiency.
While the arthroscopic stabilization can be performed in the beach chair or lateral decubitus position, a lower recurrence rate is seen following surgery with the patient in lateral decubitus. The lateral decubitus position allows the surgeon to obtain circumferential access to the glenoid rim to better addresses the inferior component of anterior glenoid instability. However, arthroscopy in lateral decubitus is technically challenging as the shoulder is in nonanatomic orientation and is at risk for traction-related injury or axillary and musculocutaneous nerve injury during anteroinferior portal placement.
Our preferred arthroscopic soft tissue stabilization is a Bankart repair with an added posterolateral portal for optimal visualization of and access to the anteroinferior glenoid. Following interscalene block and general anesthesia, the patient is placed in the lateral decubitus position and the operative arm positioned in place with a beanbag and padded arm holder. The arm is suspended in gentle traction in slight abduction and forward flexion. Posterolateral, posterior and anterosuperior portals are created. The posterolateral portal is created 4 cm off the posterolateral acromion with the posterior viewing portal 2 cm inferior to the posterolateral corner of the acromion. An anterosuperior portal is created just anterior to the biceps, proximal in the rotator interval. Diagnostic arthroscopy is performed using a 30-degree arthroscopic through the anterosuperior viewing portal. The Bankart lesion and any concomitant injury to the glenohumeral complex, including the long head of the biceps tendon, rotator cuff, humeral head, and glenoid bone stock are identified. The anterosuperior portal allows for optimal viewing of the glenoid. A rasp or burr can be utilized to create glenoid bleeding. The posterolateral portal is used for percutaneous anchor placement to the inferior glenoid through an inside-out technique that follows the trajectory of the posterior border of the clavicle through the teres minor and capsule. This technique allows the trocar to be placed 1 to 2 mm from the glenoid rim and minimizes the risk for movement with drilling and skiving across the cartilage of the glenoid. Sutures are gently pulled to ensure capture prior to trocar removal, and are retained in the posterolateral portal for suture management and knot tying. After successful anchor placement, the posterior portal is cannulated and the anterior capsule is grasped anterior to the anchor to avoid axillary nerve, closest to the 6 o'clock position, and the labrum is brought to the inferior glenoid to enhance plication of the capsular pouch.
The degree of plication is based on clinical exam or exam under anesthesia (see Fig. 43.5 ). The labrum is repaired anteriorly through the anteroinferior portal and the sutures are shuttled with a single hoop suture configuration as it best allows appropriate tensioning. A sliding knot and alternating half stitches placed away from the articular surface recreates the inferior glenoid bumper. At least three suture anchors between the 3 o'clock and 6 o'clock position should be utilized to recreate the glenoid labrum and appropriate capsular tensioning. The addition of a rotator interval closure can improve anterior instability but not inferior instability specifically. While potentially beneficial for some patients, rotator interval closure can result in a significant loss of external rotation in neutral and abducted arm positions.
Arthroscopic stabilization with bony augmentation should begin with quantification of glenoid bone loss using either a 2 o'clock or 3 o'clock posterior portal. Using the inferior two-thirds of the glenoid cavity as a circle, the glenoid bare spot is the marked center point in order for there to be a consistent radius in the anterior, posterior, and inferior directions. This method of quantification is best for assessing the extent of lesions parallel to the long axis of the glenoid; if the defect occurs at a 45-degree angle relative to the long axis, the bare spot method overestimates the amount of true bone loss. Note that the glenoid bare spot may not be identifiable in all shoulders and therefore findings from preoperative 3D CT may guide the surgical approach.
For smaller glenoid defects (<20% to 25%), a Bankart fragment may be repaired with arthroscopic techniques utilizing various suture anchors and suture constructs if adequate capsulabral tensioning can be achieved. Suture anchor fixation of a Bankart lesion is performed through the use of the posterolateral, transubscapularis, and mid-glenoid portals. The posterolateral portal more readily allows access to the inferior-most aspect of glenoid, especially when the patient is placed in lateral decubitus position. For patients experiencing recurrent instability in the setting of revision surgery, our preferred surgical approach is the Latarjet procedure. The standard Latarjet and its variations have been described in literature with few studies addressing optimal graft orientation and placement. A biomechanical study evaluating the contact pressures and areas of a Latarjet graft demonstrated that the bone block should be oriented with the inferior aspect of the coracoid congruent to the glenoid arc to reconstruct the mean reconstructed glenoid diameter. While some advocate for limited capsular and labral repair, suture anchors can be utilized on the anterior-most aspect of the native glenoid at the coracoid–glenoid interface. Two 3.5 mm metal screws fix the coracoid as flush as possible into position. The previously dissected native capsule from the glenoid neck is repaired with suture anchors and bone tunnels with the coracoid extraarticular. The locally harvested coracoid autograft is positioned to act as an extra-articular extension of the glenoid arc. The addition of an osseous block extends the glenoid rim and allows there to be more articular surface area for the humeral head to make contact with, limiting the opportunity for dislocation. The conjoined tendon acts as a sling to prevent anterohumeral translation when the arm in abduction and external rotation. Both the transferred coracoid process and conjoined tendon over the lower subscapularis tendon reinforce the anteroinferior aspect of the capsule, having an effect like that of a tenodesis.
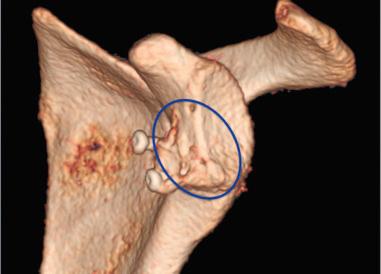
While the gold standard for glenoid bone augmentation is the Latarjet coracoid transfer procedure, we recommend the use of an articular allograft to reconstruct the glenoid when there is greater than 25% bone loss. The Latarjet procedure has low reported long-term instability rates, but there is a concern of graft resorption and early development of glenohumeral arthritis in the high-demand athletic patient population ( Fig. 43.6 ). The preferred technique for open reconstruction of the anterior glenoid rim begins with the patients placed in beach chair with the head elevated to 40 degrees and the operative arm in an arm holder. A modified deltopectoral exposure and subscapular splitting is performed to access the glenohumeral joint, then the joint and viable labral tissue are elevated to expose anterior glenoid. A combination of high speed burr or rasp and an arthroscopic bone cutting shaver (<3.5 mm diameter) is utilized to create a bleeding surface on the anterior glenoid for the incorporation of the allograft. Preparation of the native glenoid and osseous surfaces are key to successful incorporation of the graft and repair. Adequate preparation of the anterior aspect of the glenoid neck is determined when the posterior subscapular fibers can be visualized through the elevated capsule off the glenoid. Recently developed arthroscopic instruments, such as sharp penetrators, suture hooks, or curved suture devices, may be used in conjunction with standard arthroscopic repair devices to punch through bone ( Fig. 43.7 ).
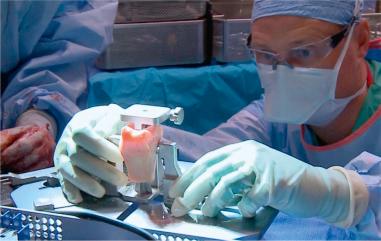
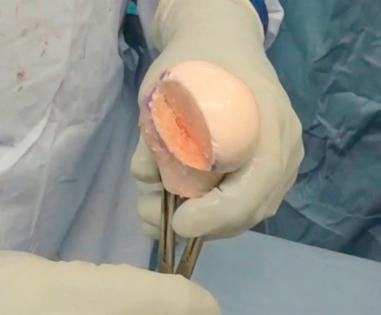
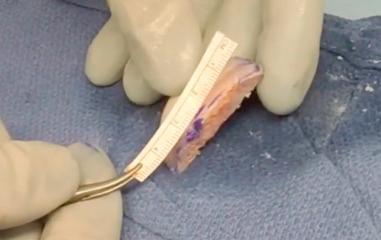
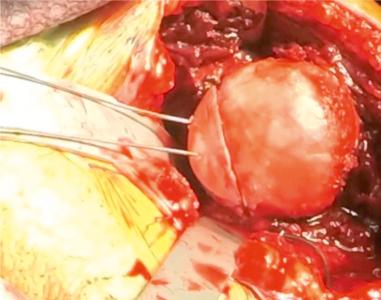
The allograft of choice is a fresh DTA, specifically of the lateral third portion. The lateral aspect of the plafond provides a near anatomical match of the radius of the curvature and articular thickness with dense corticocancellous bone. DTA is readily available from allograft distributors and can be customized to the patient's shoulder. A deltopectoral approach with the subscapularis split longitudinally allows exposure of the capsule. Subperiosteal dissection is performed as medially as possible and tagged with No. 2 nonabsorbable suture. With the glenohumeral joint exposed, the amount of glenoid bone loss is determined. The glenoid is prepared to receive the allograft. Occasionally there is attrition of the labral tissue and it cannot be repaired in these patients. A high speed burr can be used to create a uniform glenoid graft bed that is perpendicular to the articular surface of the glenoid. The DTA is prepared on the back table with a 0.5 inch sagittal saw under continuous irrigation to prevent thermal necrosis. The graft should be secured with pointed reduction clamps or towel clips ( Figs. 43.8–43.10 ). Using a template from a 3D CT scan, the allograft should be measured and cut from superior to inferior (~20 to 25 mm) and then anterior to posterior (~6 to 10 mm). The graft should be approximately 1 cm deep to match the dimensions of the prepped anterior glenoid defect with sufficient subchondral bone for eventual placement of 2 to 3.5 mm screws. The corners of the graft are rounded with pointed the sagittal saw to match the native glenoid morphology. The DTA is then transferred to the operative field and temporarily fixed with two 1.6 mm Kirschner wires at 45 degrees to the articular surface ( Fig. 43.11 ). Washers with 2 to 3.5 mm fully threaded, noncannulated biocortical interference screws are used with the lag technique to achieve adequate graft compression against the anterior glenoid ( Fig. 43.12 ). Capsular and labral repair with sutures attached to screw heads with or without washers may be necessary prior to final tightening of the graft. Prior to final screw tightening, sutures that were used to repair the capsular and labral tissue can be passed under the screwhead and washer for secure fixation. The capsule, subscapularis split, superficial soft tissues, and skin are closed in a standard fashion ( Figs. 43.13 and 43.14 ).


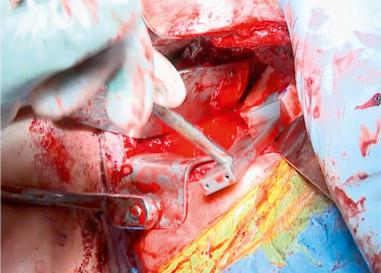
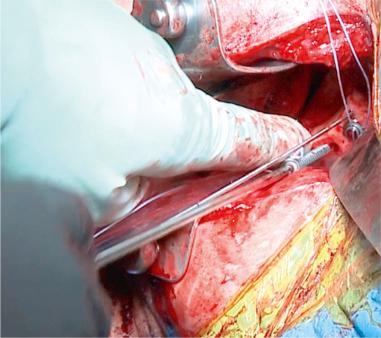
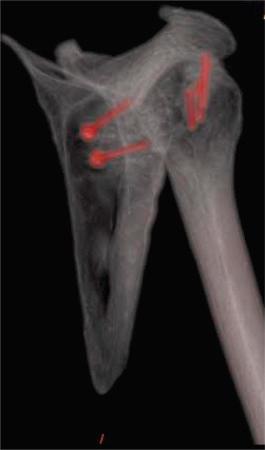
Treatment of other concomitant pathologies is imperative in all revision cases to avoid recurrent instability after revision shoulder stabilization and is possible in multiple surgical approaches. Common pathologic findings include rotator cuff tears (partial and complete), acromioclavicular joint pain, extensive labral tear, SLAP lesion (with possible bicep lesion), anterior labroligamentous periosteal sleeve avulsion (ALPSA) lesion, HAGL lesion and Hill-Sachs lesion. Rotator cuff tear, extensive labral tear, and SLAP lesion require direct repair. Bicep tendon disease may be addressed with débridement and tenodesis. Acromioclavicular joint pain may be alleviated with a distal clavicle excision or a preoperative acromioclavicular joint injection. Important considerations for ALPSA lesion repair are that the labrum and scapular periosteal sleeve are detached medially and inferiorly on the glenoid, and the tear can occur in the anterior band of the inferior glenohumeral ligament. An HAGL lesion may be visualized in the posterior portal with a 30-degree scope in the axillary pouch when the surgical arm is placed in external and internal rotation. An HAGL lesion should be repaired inferiorly to superiorly, and medially to laterally. If engaging, a Hill-Sachs lesion may benefit from remplissage or bone augmentation ( Figs. 43.15–43.18 ).
Rehabilitation plays an important role in patient recovery. Patients are provided a standard abduction sling for arm support during the first 4 to 6 week postoperative period to allow healing of the osseous injury and capsular repair. Early mobilization with gentle passive exercises is encouraged to begin the process of regaining shoulder joint mobility. At 2 to 4 weeks, pendulum exercises and passive ROM exercises in the scapular plane can begin. Patients who underwent a rotator cuff repair may benefit from performing pendulum ROM exercises while supine to maintain scapular stabilization. At 4 weeks, the patient can begin active assisted exercises in flexion, external rotation, and elevation with a goal to gradually achieve full or the patient's normal external rotation in neutral (30 to 40 degrees), 120 to 140 degrees in flexion, and abduction of approximately 45 degrees. Usually around 6 to 8 weeks, the patient can begin strengthening exercises. Patients should not begin strengthening or resistance exercises until their ROM has significantly improved. If a subscapularis split was used, patients may be more aggressive with internal rotation strengthening with less protection of external rotation. Full return to activity is expected around 4 to 6 months postoperatively. Patients who desire to return to competitive contact/overhead sports or occupational activities that frequently places their shoulder in positions of instability, abduction, and external rotation, may benefit from waiting until at least 6 months postoperatively prior to full return of their activities. Patients should be educated on joint protection, such as forceful stretching or manipulation of the shoulder and heavy lifting above shoulder height, until the rehabilitation protocol has been completed.
The goal in managing revision shoulder stabilization is to reconstruct the anatomy through the correction of bony deficits and ligamentous laxity with a secure fixation. Biomechanical and clinical studies have demonstrated that the more anatomical the repair, the better the outcomes in terms of stability and motion. Favorable clinical outcomes have been reported following both arthroscopic and open stabilization repair.
Arthroscopic stabilization with suture anchors is an acceptable revision procedure for anterior shoulder instability. One study reported that patients who underwent arthroscopic stabilization compared to those who underwent open repair have a greater return of external rotation in abduction, 90 degrees and 80 degrees, respectively. Another study on active duty military patients reported a lower failed surgical rate with arthroscopic Bankart repair than with open repair.
Proper treatment for glenoid bone loss is important for the success of revision shoulder stabilization, and a higher recurrence rate of glenohumeral instability is associated with uncorrected glenoid bone loss of 15% to 20%. Glenoid defects less than 21% (mean of 6.8 mm bony loss) may benefit from capsular repair alone ; however, patients with greater than 21% bony loss who undergo capsular repair alone report persistent instability and limited external rotation from inadequate capsular restraints. Arthroscopic repair with a glenoid bone loss between 20% and 25% can be successful with the use of small bony fragments to recreate a more normal anatomy. The outcomes are less predictable if there is significant bone loss with this repair method. One study comparing arthroscopic revisions in either beach chair or lateral decubitus show no statistically significant difference in two postoperative outcome scores: Rowe and Constant-Murley. The primary finding of the study was that performing the repair with the patient in lateral decubitus resulted in a lower recurrence rate than in beach chair; however, the overall recurrence rate was 12%.
Large anteroinferior glenoid defects can be addressed with a Latarjet procedure or bone graft fixation. Allograft augmentation is emerging as a viable option with large glenoid defects, especially in the setting of failed Latarjet. One study reported excellent stability upon clinical exam, minimal graft absorption, and excellent clinical outcomes based on American Shoulder and Elbow Society scores, Western Ontario Shoulder Instability Index, and single numerical assessment evaluation following DTA glenoid reconstruction at an average follow-up of 45 months. A cadaveric study showed the distal tibia's articular surface radial curvature is similar to that of the glenoid, which allows recreation of the full arch ranges of motion intrinsic to the shoulder joint. Furthermore, the DTA is corticocancellous bone that allows for screw fixation. Reconstruction with DTA recreates the anatomy of the glenoid rim better than an iliac crest bone graft or a coracoid process via Latarjet procedure, with a good healing rate and a low lysis rate. Replenishing the glenoid bone stock is imperative to the success of revision instability repairs. Mixed long-term outcomes have been reported following a Latarjet procedure. A retrospective review of 58 shoulder Latarjet stabilizations noted excellent results with no recurrence in dislocations at an average of 14.3 years follow-up. Another study reported 25 shoulders having grade I changes out of 118 treated with a Bristow/Latarjet repair at an average follow up of 15.2 years. While their patients reported an overall satisfaction rate of 98%, a follow up radiographic study for the same cohort found moderate to severe dislocation arthropathy in 14% of the patients. One study on 49 patients treated with a modified Bristow procedure reported nearly a 70% rate of good to excellent results, but 15% of the shoulders had a recurrence of instability at a mean time of 7 years post-repair.
Postoperative complications include recurrence of instability, stiffness, pain, nerve injury, chondrolysis, subscapularis dysfunction, and infection. Inadequate restoration of the glenoid concavity and defect can result in a recurrence of instability. This can be minimized through proper graft choice and avoiding excessive anteroinferior capsular tightening in overhead athletes. Proper tensioning of the capsule can help prevent joint stiffness and shearing of the posterior humeral head on the glenoid surface. Patients may experience pain following revision stabilization. Prolonged pain following surgery has been associated with the use of bioabsorbable tacks. Pain may also be indicative of chondrolysis; however, this is mostly historical and more commonly associated with thermal capsulorrhaphy or the use of an intra-articular pain pump. Hardware failure has been associated with the use of bioabsorbable tacks and improper placement of suture anchors. Three or more anchors should be placed firmly on the subchondral bone below the articular margin under the 3 o'clock position. Should more than three sutures be necessary, the first additional suture anchor should be placed at the 5:30 position and 45 degrees to the articular surface. The use of suture anchors is associated with a risk of glenoid rim fracture, osteolysis, infection, damage of the glenoid articular cartilage and the enlargement of drill holes. Subscapularis dysfunction or insufficiency can occur after open procedures that utilize a subscapularis tendon and capsule incising technique. Tenderness to palpation in the anterior shoulder and pain associated with decreased internal rotation strength or excessive external rotation may be indicative of subscapular dysfunction. Infection is a potential but rare complication following revision repair, particularly with indolent organisms such as Propionibacterium acnes . Joint infection with P. acnes is difficult to diagnose as it may present with nonspecific symptoms, such as generalized pain, up to 2 years following surgery. P. acnes infection has a reported incidence between 0% and 6% after open stabilization and between 0.04% and 0.23% after arthroscopic stabilization. There is a potential risk of damage to the axillary nerve, musculocutaneous nerve, the suprascapular nerve and artery, and the cephalic vein during exposure for arthroscopic stabilization; however, this risk is minimized by using the established portal locations described in our preferred surgical technique. Careful placement of the humeral head to avoid excessive flexion–extension and distraction along with a strong knowledge of shoulder anatomy can further prevent this injury. The axillary nerve is located approximately 1 to 1.5 cm below the inferior aspect of the glenohumeral capsule and the musculocutaneous nerve sits approximately 5 to 8 cm inferior to the coracoid.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here