Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
In addition to causing arthritis by means of direct microbial infection (i.e., septic arthritis; see Chapter 705 ), microbes activate innate and adaptive immune responses, which can lead to the generation and deposition of immune complexes as well as antibody or T cell–mediated cross-reactivity with self. In addition, microbes may influence the immune system in ways that promote immune-mediated inflammatory diseases such as systemic lupus erythematosus (SLE), inflammatory bowel disease (IBD), juvenile idiopathic arthritis (JIA), and spondyloarthritis. Reactive arthritis and postinfectious arthritis are defined as joint inflammation caused by a sterile inflammatory reaction following a recent infection. We use reactive arthritis to refer to arthritis that occurs following enteropathic or urogenital infections and postinfectious arthritis to describe arthritis that occurs after infectious illnesses not classically considered in the reactive arthritis group, such as infection with group A streptococcus or viruses. In some patients, nonviable components of the initiating organism have been demonstrated in affected joints, and the presence of viable, yet nonculturable, bacteria within the joint remains an area of investigation.
The course of reactive arthritis is variable and may remit or progress to a chronic spondyloarthritis, including ankylosing spondylitis (see Chapter 181 ). In postinfectious arthritis the pain or joint swelling is usually transient, lasting <6 wk, and does not necessarily share the typical spondyloarthritis pattern of joint involvement. The distinction between postinfectious arthritis and reactive arthritis is not always clear, either clinically or pathophysiologically.
Reactive arthritis typically follows enteric infection with Salmonella species, Shigella flexneri, Yersinia enterocolitica, Campylobacter jejuni , or genitourinary (GU) tract infection with Chlamydia trachomatis . Escherichia coli and Clostridium difficile are also causative enteric agents, although less common (see Table 181.2 ). Acute rheumatic fever caused by group A streptococcus (see Chapters 182 and 210.1 ), arthritis associated with infective endocarditis (see Chapter 464 ), and the tenosynovitis associated with Neisseria gonorrhoeae are similar in some respects to reactive arthritis.
Approximately 75% of patients with reactive arthritis are HLA-B27 positive. Incomplete elimination of bacteria and bacterial products, such as DNA, has been proposed as a factor in reactive arthritis. A relationship with clinical characteristics of specific infectious disorders is not present. In postinfectious arthritis, several viruses (rubella, varicella-zoster, herpes simplex, cytomegalovirus) have been isolated from the joints of patients. Antigens from other viruses (e.g., hepatitis B, adenovirus) have been identified in immune complexes from joint tissue.
Patients with reactive arthritis who are HLA-B27 positive have an increased frequency of acute and symptomatic uveitis and other extraarticular features. In addition, HLA-B27 is a risk factor for persistent gastrointestinal (GI) inflammation following enteric infections, even after resolution of the initial infection, and significantly increases the risk that the individual will develop chronic spondyloarthritis. Nevertheless, reactive arthritis also occurs in HLA-B27–negative patients, emphasizing the importance of other genes in disease susceptibility.
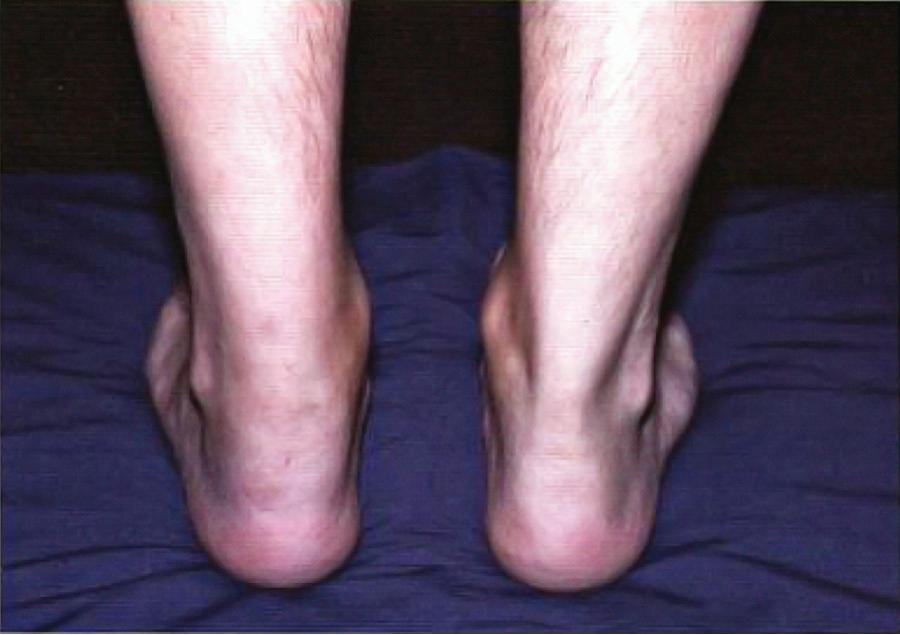
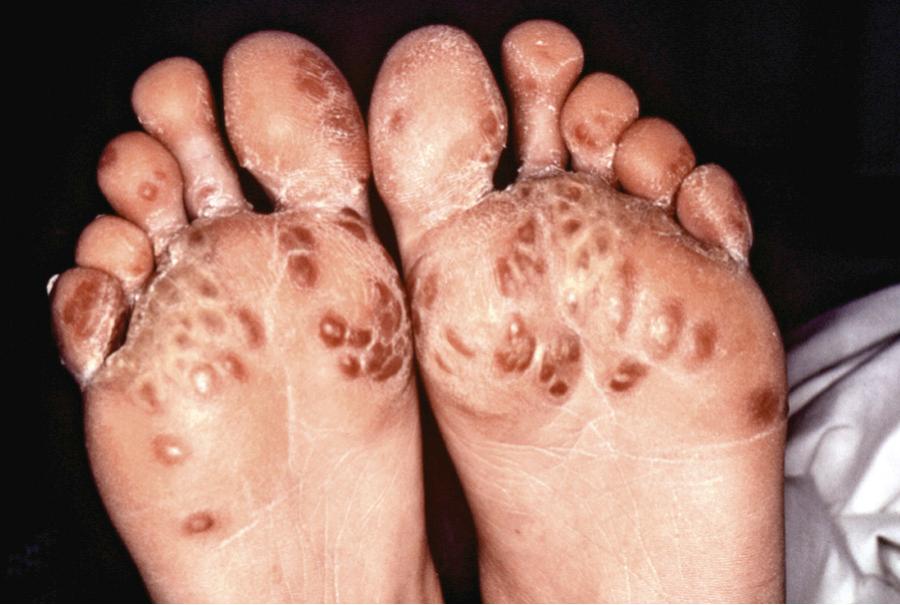
Symptoms of reactive arthritis begin approximately 3 days to 6 wk following infection. The classic triad of arthritis, urethritis, and conjunctivitis is relatively uncommon in children. The arthritis is typically asymmetric, oligoarticular, with a predilection for lower extremities. Dactylitis may occur, and enthesitis is common, affecting as many as 90% of patients ( Fig. 182.1 ). Cutaneous manifestations can occur and may include circinate balanitis, ulcerative vulvitis, erythematous oral macules or plaques or erosions, erythema nodosum, paronychia, painful erosions or pustules on fingertips, and keratoderma blennorrhagica, which is similar in appearance to pustular psoriasis ( Fig. 182.2 ). Systemic symptoms may include fever, malaise, and fatigue. Less common features may include conjunctivitis, optic neuritis, aortic valve involvement, sterile pyuria, and polyneuropathy. Early in the disease course, markers of inflammation—erythrocyte sedimentation rate (ESR), C-reactive protein, and platelets—may be greatly elevated. The clinical manifestations may last for weeks to months.
Familiarity with other causes of postinfectious arthritis is vital when a diagnosis of reactive arthritis is being considered. Numerous viruses are associated with postinfectious arthritis and may result in particular patterns of joint involvement ( Table 182.1 ). Rubella and hepatitis B virus typically affect the small joints, whereas mumps and varicella often involve large joints, especially the knees. The hepatitis B arthritis–dermatitis syndrome is characterized by urticarial rash and a symmetric migratory polyarthritis resembling that of serum sickness. Rubella-associated arthropathy may follow natural rubella infection and, infrequently, rubella immunization. It typically occurs in young women, with an increased frequency with advancing age, and is uncommon in preadolescent children and in males. Arthralgia of the knees and hands usually begins within 7 days of onset of the rash or 10-28 days after immunization. Parvovirus B19, which is responsible for erythema infectiosum (fifth disease), can cause arthralgia, symmetric joint swelling, and morning stiffness, particularly in adult women and less frequently in children. Arthritis occurs occasionally during cytomegalovirus infection and may occur during varicella infections but is rare after Epstein-Barr virus infection. Varicella may also be complicated by suppurative arthritis, usually secondary to group A streptococcus infection. HIV is associated with an arthritis that resembles psoriatic arthritis more than JIA (see Chapter 180 ).
TOGAVIRUSES
Rubella
Ross River
Chikungunya
O'nyong-nyong
Mayaro
Sindbis
Ockelbo
Pogosta
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here