Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Rabies virus and 17 bat lyssaviruses are members of the Rhabdoviridae family, genus Lyssavirus, which is derived from the Greek word lyssa, meaning “madness.” Rabies derives from the Sanskrit word rabhas, which means “to do violence.” Rabies virus and many lyssaviruses cause human encephalitis through zoonotic infection.
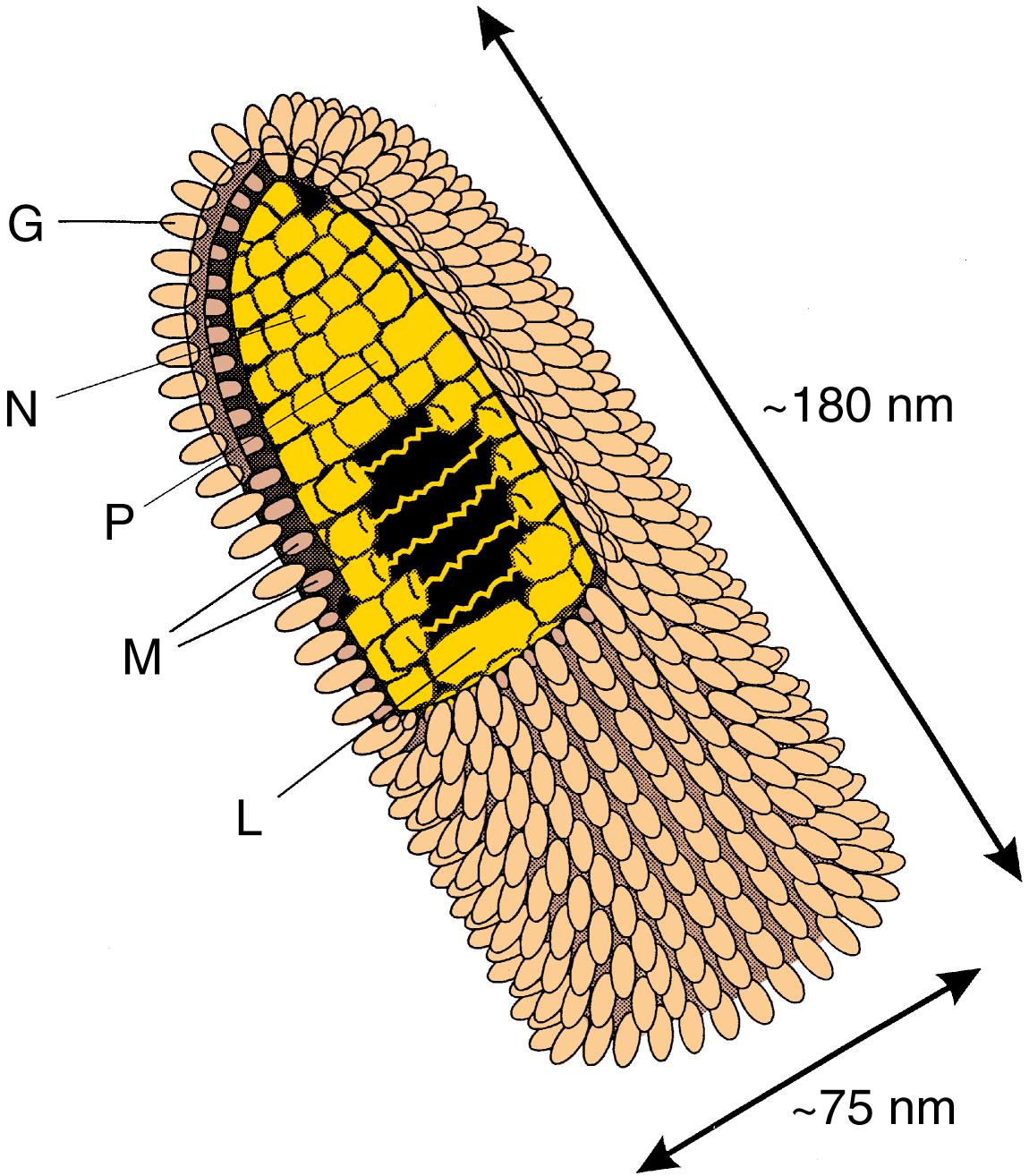
The lyssaviruses differ antigenically but are morphologically similar and neurotropic. Rabies virus is an enveloped, bullet-shaped virus that is 180 nm long and 75 nm wide and composed of five structural proteins ( Fig. 228.1 ). Rabies virus contains one copy of a single-stranded, nonsegmented, negative (noncoding) RNA of approximately 12,000 nucleotides. The virus envelope contains glycosylated G-protein spikes that bind to cells. The matrix (M) protein is located on the inner virus envelope, inside which the virus nucleoprotein (N) tightly binds the viral RNA to form the nucleocapsid core. This core, along with a large transcriptase protein (L) and a phosphorylated protein (P), is the rabies virus nucleocapsid (RNP).
The incubation period after rabies virus inoculation begins with ill-defined local replication in muscle or skin for canine or bat rabies variants, respectively. This step in pathogenesis is the basis of the rationale for recommendations of local wound care and instillation of neutralizing antibody to reduce the rabies infectious inoculum. Neutralizing antibodies can penetrate infected cells. Response to the rabies vaccine then generates further neutralizing antibody.
After local tissue replication over weeks to months, the virus attaches to nicotinic acetylcholine receptors that provide access to the peripheral nervous system through neuromuscular spindles or muscle motor end plates. Sensory and autonomic nerves also are infected. Other host receptors for rabies virus include specific carbohydrates, the neuronal cell adhesion molecule (NCAM), and p75 neurotrophin receptor (p75NTR) and integrin β1. ,
Rabies virus moves centripetally along peripheral nerves, without virus replication, at rates of ≥100 mm/day. After ascending to the spinal cord, virus spreads rapidly to the brain, tracing neurons in retrograde fashion across synapses at a rate of one synapse per day. Centrifugal spread occurs contemporaneously along motor, sensory, and autonomic axons into host body tissues (usually restricted to nerves innervating organs, including hair follicles) and particularly into the salivary and lacrimal glands. Pathologic features are minimal. Negri bodies are rabies-specific eosinophilic inclusions of RNPs and chaperonins in the cytoplasm of neurons that vary by brain region and may not be found in 30% of cases. Histology shows perivenular lymphocytosis. Inflammation of the dorsal root ganglia correlates with focal neurologic prodromes. In the less common paralytic form of rabies, axonal loss mimics acute motor axonal neuropathy and other polio-like diseases. Minimal histologic damage is in stark contrast to the severity of clinical rabies. Rabies viremia and viruria are not mechanisms of virus spread.
Host factors that reduce the incubation period are (1) young age, (2) immunocompromised state, including corticosteroid use, (3) greater severity of the wound, (4) closeness of the wound to the central nervous system, and (5) large inoculum. Neuroinvasiveness is determined by the virus G protein. In animal models, a single-point mutation at position 333 of the G protein renders strains avirulent as a result of alteration in the ability of the virus to spread from cell to cell. Overexpression of the G glycoprotein induces neuronal apoptosis and generates effective immunity. ,
The G protein and the RNP are the major targets of the protective host response. The G glycoprotein is the major target of virus-neutralizing antibody and of cellular immunity. Antigenicity of the G protein of rabies virus is broad enough to induce protective immunity to all rabies virus strains, but it may not protect against other lyssaviruses.
Rabies primarily is a mesocarnivore and bat infection. Humans become infected through exposure to infected animals. Worldwide data on the human burden of rabies are incomplete because of the hopelessness, stigma, and extreme poverty associated with populations at greatest risk for rabies. It is estimated that 50,000–60,000 people die annually of rabies. Most cases of rabies occur in males and children.
The control of rabies requires knowledge of the endemic animal sources of rabies (including bats) and the geography of infection and an understanding of virus transmission in nature. Any warm-blooded animal can become infected, but some animal species are more susceptible, and some rabies strains are more infectious than others. , Shedding of virus in saliva and the rapidity of death vary among animal species. Skunks shed virus for up to 18 days before death, whereas foxes succumb rapidly.
The dog remains the principal vector for transmission of rabies virus to humans in most parts of the world outside of North America and Europe. Cats usually are second in importance. In the Americas, with the waning of dog rabies and a broadening of the habitat of vampire bats, most rabies is transmitted by bats. The source of rabies has shifted from domestic animals to wildlife in North America, where 6 separate rabies enzootics exist. Outside of North America, wildlife rabies enzootics involve other animals ( Table 228.1 ). Other bat lyssaviruses have been isolated in Europe, Africa, and Australia and cause sporadic human encephalitis with fatal outcomes. Genetic sequencing and typing by monoclonal antibody panels can distinguish rabies enzootics and whether humans are infected with local or imported rabies virus strains.
| Geographic Region | Principal Terrestrial Species With Endemic Rabies a |
|---|---|
| US and Canada | |
| Central US and Canada | Striped skunk |
| Mid-Atlantic, southeast US | Raccoon |
| New York, Quebec, Ontario | Red fox |
| Northwest territories | Arctic fox |
| Arizona | Grey fox |
| Texas | Grey fox |
| Caribbean | Mongoose |
| Europe | Red fox |
| Iran | Wolf |
| Africa | Jackal |
a The bat is always considered endemically rabid. The dog is the principal vector for transmission of rabies around the world, except for North America and Europe.
The vehicle of virus transmission is infected animal saliva, which can be inoculated by a bite or scratch. Contact of infected animal saliva with human mucous membranes also can transmit infection. Eight of 25 human cases of rabies reported in the US between 2009–2019 resulted from exposure to animals outside of the country. In contrast, 12 of 17 domestic rabies cases were attributed to bats, 4 of these with unknown exposures and 4 by contact without known bites. In addition to animal transmission of rabies virus, there have been 6 outbreaks of solid-organ transplantation-associated rabies and 10 corneal implants transmitting rabies.
Serologic surveys document asymptomatic exposure in high-risk populations. There also are reports of persons meeting laboratory definitions of rabies with milder disease. Survivors of rabies are being increasingly reported, often after partial prophylaxis and with poor outcomes.
Symptoms are caused by accumulation of virus-infected neurons as most patients die before a measurable immunologic response and independent of a glial response. The asymptomatic first phase is the virus incubation period, which usually lasts from 10–90 days but ranges from 4 days to 8 years. The long incubation period lowers the yield of obtaining a history of an antecedent animal exposure. Retaking the history, focused on travel, immigration, occupations, and animal exposures, is critical.
The second, or prodromal, period lasts for 2–10 days and follows considerable virus replication in the central nervous system. Early symptoms, which occur in up to one half of patients, are nonspecific and include fever, dysphagia, and changes in behavior, sleep, and emotions. Local pruritus, pain, paresthesia, weakness, or myoclonic jerks occur at the site of inoculation in approximately 50% of cases and are most helpful in focusing the diagnosis on rabies.
The third stage is the development of central nervous system signs of disease. The furious form of rabies comprises dysautonomia, aggressiveness (e.g., biting, hitting, yelling), excitement, hyperactivity, and hallucinations that last for brief periods and alternate with normal behavior, progressing to confusion or delirium. Fever can be intermittent but is invariably present. More than 50% of patients experience hydrophobia (with attempts to drink liquids) or aerophobia (when air is blown on the face). These reactions result from violent diaphragmatic contractions as an exaggerated respiratory protective reflex, and they are enough to cause pneumothorax. Hypersalivation and dysphagia increase the risk of aspiration. Status epilepticus, marked dystonia, parkinsonian signs, or opisthotonos are uncommon and should suggest other diagnoses. Clinical presentation at this stage can mimic that of intoxication, envenomation, meningitis, encephalitis, cerebral malaria, myoclonic epilepsy, tetanus, polio, or Guillain-Barré syndrome.
The paralytic form of rabies occurs in approximately 20% of patients and is slower to evolve. Flaccid paralysis progresses to quadriparesis, bulbar abnormalities, and respiratory insufficiency. Hypersalivation and hydrophobic spasms can occur. Guillain-Barré syndrome often is considered, but findings of fever, pain, sensory or sphincter disturbance, and more prominent involvement of proximal musculature are more suggestive of rabies. A few developing countries produce tissue-derived rabies vaccines that are associated with vaccine-associated autoimmune encephalitis. Autoimmune encephalitis targeting glutamate (NMDA) receptors have frequently been confused with rabies, particularly after potential animal exposures. Other diagnostic possibilities include hysteria, botulism, and elapid snake bite.
Early deaths can result from respiratory arrest or cardiac dysrhythmias, including asystole. Recognized early, severe dysautonomia responds to neuroleptics or aggressive sedation.
The fourth stage of rabies is coma, usually developing 6–8 days after hospitalization. Coma correlates with generalized spasm of the basilar arteries measured by transcranial Doppler ultrasonography and metabolic changes in cerebrospinal fluid (CSF) measured by nuclear magnetic resonance spectroscopy. , Examination results consistent with brainstem death are distinctively discordant with electroencephalographic (EEG) or magnetic resonance imaging (MRI) findings.
The immune response to rabies usually is detected within 16 days after hospitalization. Complications are associated with the immune response; both vary by phylogeny of rabies viruses. Common complications include complete heart block in dog rabies and cerebral edema in bat rabies. After 12–15 days, intracranial pressure and EEG activity drop acutely, and diabetes insipidus follows. Death usually occurs within 7 days of symptom onset in furious rabies and 21 days in paralytic rabies; critical care can prolong survival to a median of 18 days after hospitalization. Survival is more common in rabies developing after partial postexposure prophylaxis (PEP) than in patients not receiving PEP, but neurologic outcomes are worse in vaccinated survivors. Survival rates are lowest after exposure to insectivorous bats, intermediate with canine rabies, and best after vampire bat transmission.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here