Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Primary immune defects affect about 1 per 10,000 persons, but the prevalence rises to 1 per 400 to 1 per 5000 persons when selective immunoglobulin A (IgA) deficiency is included. , The prevalence has been increasing as the number of known defects has increased to about 450 different entities. Primary immune defects are now classified into nine categories ( Table 231-1 ). The functions of complement ( Chapter 37 ), disorders of phagocytes ( Chapter 155 ), autoinflammatory syndromes ( Chapter 240 ), and bone marrow defects ( Chapter 151 ) are described elsewhere.
| T- and B-cell combined deficiencies Combined defined defects with syndromic features Antibody deficiencies Complement disorders Phagocyte defects Immune dysregulation syndromes Defects of intrinsic and innate immunity Autoinflammatory defects Bone marrow failure defects |
Because of the number and types of immune deficiencies, clinical recognition of the various phenotypes can be difficult, and diagnosis is often delayed. In general, the spectrum of immune defects varies with the age of the patient. Defects of both T and B cells, phagocytes ( Chapter 155 ), immune dysregulation, and innate immunity are more commonly recognized in early childhood, whereas defects of complement ( Chapter 37 ) and the production of antibodies as well as autoinflammatory diseases ( Chapter 240 ) are more commonly diagnosed in adolescents and adults. However, many exceptions exist to this generalization.
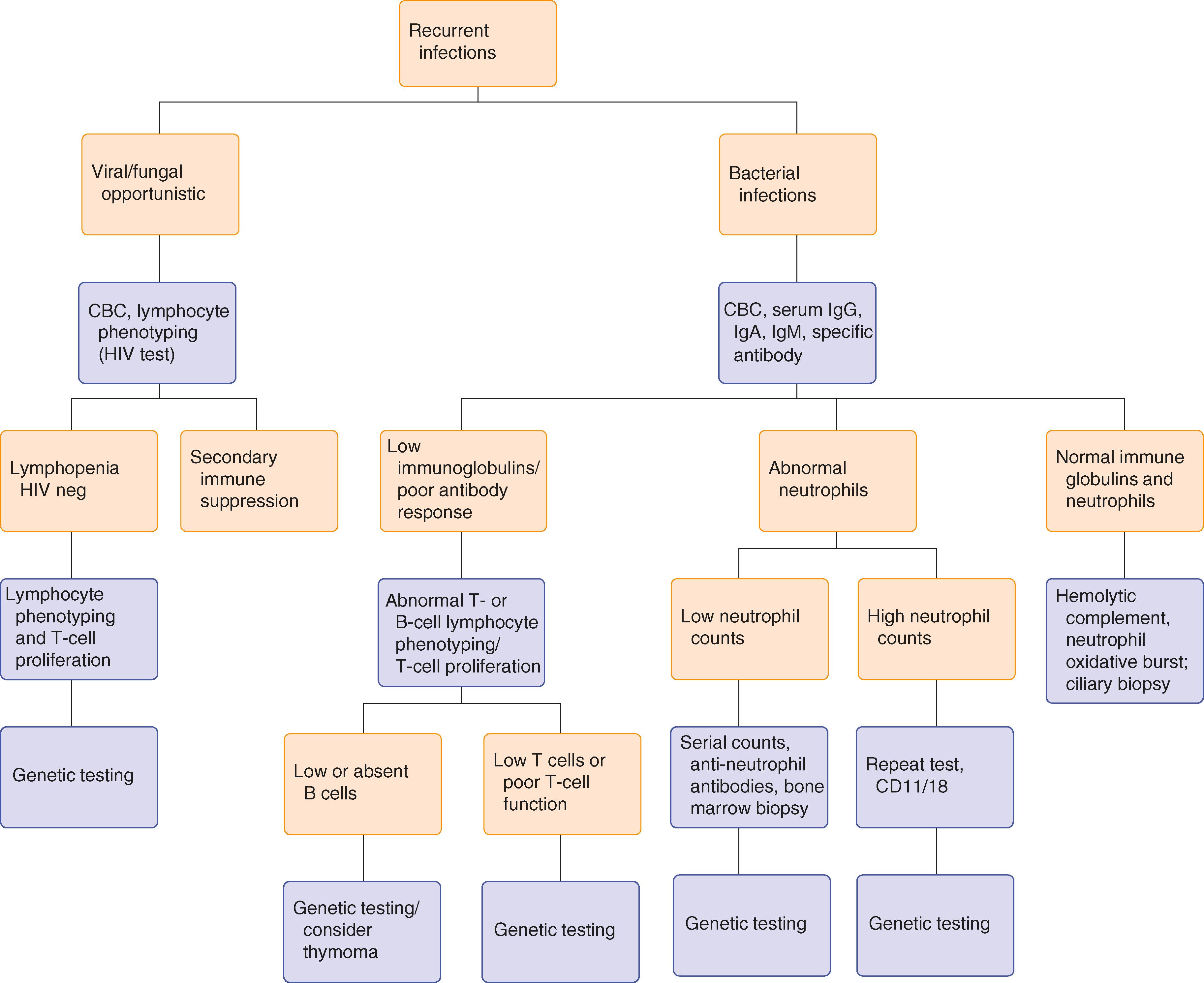
For most patients, the first symptom of an immune defect is a series of relatively common infections, particularly chronic sinusitis ( Chapter 394 ), otitis ( Chapter 394 ), bacterial pneumonias ( Chapter 85 ), meningitis ( Chapter 381 ), or osteomyelitis ( Chapter 251 ). In both children and adults with immune defects, infections are likely to persist longer, may require additional courses of antibiotics, and tend to recur. Infections may also lead to additional complications or procedures, such as empyema after bacterial pneumonia or the need for myringotomy tubes in an adult with chronic otitis. In infants and children, chronic infections lead to poor appetite and failure to thrive. For adults, some weight loss may occur, but it usually is less apparent. Because of lack of immunity, reactivated herpes zoster infection ( Chapter 346 ) is relatively common in patients who have T-cell defects or antibody deficiencies. Other common clinical presentations include acute gastrointestinal infections with characteristic organisms such as Giardia ( Chapter 322 ) or chronic intestinal inflammatory diseases that lead to malabsorption and weight loss that can mimic gluten intolerance or Crohn disease ( Chapters 126 and 127 ). Depending on the clinical presentation ( Table 231-2 ), a systematic approach ( Fig. 231-1 ) can efficiently guide the evaluation of patients who are suspected of having a primary immune deficiency.
| CLINICAL PRESENTATION | DEFECTS | IMMUNE DEFECTS | CONDITIONS | LABORATORY TESTING |
|---|---|---|---|---|
| Recurrent or chronic bacterial, viral, or fungal infections Opportunistic infections |
Cell-mediated immunity | Impaired killing of intracellular organisms Impaired viral immunity Hypogammaglobulinemia |
Severe combined immunodeficiency and other combined syndromes | Absolute lymphocyte count Enumeration of T cells and T-cell subsets Proliferative tests for T-cell function Low immune globulins; no antibody |
| Bacterial infections Viral infections Autoimmunity Lymphoid hypertrophy Inflammatory diseases Enteropathy Giardiasis |
B cells | Hypogammaglobulinemia Impaired bacterial killing Impaired clearance of virus Autoimmunity |
Hypogammaglobulinemia Agammaglobulinemia IgA deficiency Common variable immune deficiency IgG subclass defects Antibody deficiency |
Enumeration of B cells Serum IgG, IgA, and IgM Antibody testing (e.g., tetanus, diphtheria) Vaccine challenge and antibody testing (e.g., tetanus, diphtheria pneumococcal vaccine) |
| Bacterial infections Susceptibility to meningococcal disease Autoimmunity Angioedema |
Complement | Impaired opsonization Impaired bacterial killing Lack of clearance of immune complexes |
Complement C2 deficiency Other complement defects Hereditary angioedema |
CH 50 AH 50 Measure individual complement components C1 inhibitor protein and function |
| Bacterial infections Poor skin healing Fungal infections Stomatitis Periodontal disease |
Phagocytic cells | Impaired neutrophil mobilization Impaired opsonization Impaired bacterial killing |
Chronic neutropenia Cyclic neutropenia Autoimmune neutropenia Leukocyte adhesion deficiency Chronic granulomatous disease |
Absolute neutrophil counts Neutrophil oxidative burst examined by dihydrorhodamine test by flow cytometry Examination of the blood smear Antineutrophil antibodies |
Large gene panels can detect many of the known immune defects. Whole exome or whole genome sequencing can confirm a likely molecular diagnosis in unrelated probands and may influence management in nearly 25% of families. With the advent of genetic testing and its use in clinical medicine, autosomal dominant genes with variable penetrance are increasingly being identified. Importantly, different mutations in the same gene can lead to either loss or gain of function and quite different clinical phenotypes.
Combined immune defects are early-onset diseases in which both the T- and B-cell compartments are greatly impaired. In some conditions, natural killer cells and other cells of the myeloid linage are also abnormal. These disorders often include additional inflammatory features such as autoimmunity and loss of T-cell regulatory function.
In the United States, universal newborn screening for severe combined immunodeficiency detects these defects in about 1 : 80,000 ( E-Table 231-1 ).
| SCID TYPE | GENES | INHERITANCE | LABORATORY FEATURES | DISEASE AND COMPLICATIONS |
|---|---|---|---|---|
| Defects of V(D)J recombination | RAG1, RAG2 DCLREIC DNA-PKcs |
Autosomal recessive | Very low lymphocyte numbers with loss of T and B cells; hypogammaglobulinemia | Severe infections, failure to thrive; leaky versions may have autoreactive T cells (Omenn syndrome) |
| Adenosine deaminase deficiency | ADA | Autosomal recessive | Variably low lymphocyte numbers with loss of T and B cells; also decreased NK cells; hypogammaglobulinemia | Severe infections, failure to thrive; often with costochondral junction flaring, neurologic features, hearing impairment, lung and liver manifestations |
| X-linked severe combined immunodeficiency | IL2RG | X-linked | Low lymphocyte numbers with loss of T cells; B cells present; markedly decreased NK cells; hypogammaglobulinemia | Severe infections, failure to thrive; leaky cases may present with low T or NK cells or Omenn syndrome |
| JAK3 deficiency | JAK3 | Autosomal recessive | Low lymphocyte numbers with loss of T cells; B cells present; hypogammaglobulinemia | Severe infections, failure to thrive; leaky cases may present with variable T or NK cells |
| IL-7 deficiency | IL7RA | Autosomal recessive | Low lymphocyte numbers with loss of T cells; B cells present; normal NK cells; hypogammaglobulinemia | Severe infections, failure to thrive; leaky cases may present with low T or NK cells or Omenn syndrome |
| T-cell receptor chain defects | γ-, ε-, and ζ-chain mutations | Autosomal recessive | Low lymphocyte numbers due to loss of T cells; normal B and NK cells; hypogammaglobulinemia | Severe infections, failure to thrive; leaky cases may present with low T or NK cells or Omenn syndrome |
The hallmark of combined defects is that they eliminate or greatly impair T-cell development, in most cases leading to profound lymphopenia. Infants with disorders that affect the formation of T- and B-cell receptors, such as when defects of the recombinase activating genes RAG1 and RAG2 impair VDJ recombination, have few if any T and B cells. Similarly, other defects of DNA recombination or repair genes (ARTEMIS, the product of DCLREIC , and DNA-PKcs) will have a similar phenotype. When T-cell immunity is absent, B cells may be present, but they will have no function, as is the case for severe combined X-linked immunodeficiency due to mutations in the cytokine γ chain, which is an essential signaling component of six cytokine receptors (interleukin [IL]–2, IL-4, IL-7, IL-9, IL-15, and IL-21). Defects of the JAK3 gene, which is downstream from the cytokine γ chain, or of the IL-7 receptor itself lead to a similar immune profile.
With loss of both essential limbs of the adaptive immune system, infants with combined immune defects have severe and recurrent infections that are caused by bacteria, viruses, and fungi. Other common features include diarrhea, dermatitis, and failure to thrive. Clinically, most patients present before the age of 3 months, but a significant number of infants may present later, although still usually in the first year of life. Without intervention, severe combined immunodeficiency commonly results in debilitating infections and death by age 2 years. In some cases, the immune defect is such that a few T cells can develop, but these cells are often self-reactive; such cases are often termed “leaky” severe combined immunodeficiency. When the presentation of these cases includes rashes and evidence of autoimmunity, infants are said to have Omenn syndrome.
All newborns in the United States are now examined by sensitive and specific DNA-based screening for severe combined immunodeficiency, so the disease is now typically diagnosed before any symptoms have appeared. Newborns normally have a mean absolute lymphocyte count of 4000/μL or higher, but most infants with severe combined immunodeficiency have significant lymphopenia. A flow cytometry panel will enumerate T, B, and natural killer (NK) cells and suggest the genes that may be responsible. Further genetic testing is commonly performed. Less severe forms of combined immune deficiency ( E-Table 231-2 ) are not identified by newborn screening because T cells are present.
| SCID TYPE | GENES | INHERITANCE | LABORATORY FEATURES | DISEASE AND COMPLICATIONS |
|---|---|---|---|---|
| MHC class I deficiency | TAP1, TAP2; TAPBP; β2M | Autosomal recessive | Low CD8, normal CD4, absent MHC I on lymphocytes | Vasculitis, pyoderma gangrenosum; granulomatous disease |
| MHC class II deficiency | CIITA; RFXANK | Autosomal recessive | Low CD4 cells Absent MHC II expression on lymphocytes |
Failure to thrive, respiratory and gastrointestinal infections, liver/biliary tract disease |
| ZAP-70 deficiency | ZAP-70 | Autosomal recessive | Low CD8, normal CD4 number but poor function | May have immune dysregulation, autoimmunity |
| DOCK8 deficiency | DOCK8 | Autosomal recessive | Low IgM, normal to high IgG and IgA, high IgE; poorly functioning Treg | Low NK cells with poor function, eosinophilia, recurrent infections, cutaneous viral, fungal, and staphylococcal infections, eczema and food allergy, virally driven cancers |
| DOCK2 deficiency | DOCK2 | Autosomal recessive | Low lymphocyte numbers; defective T, B, and NK cells | Early-onset invasive bacterial and viral infections |
Early immune reconstitution with stem cells from human leukocyte antigen (HLA)–matched bone marrow or mobilized peripheral blood is mandatory. When the diagnosis is made early and no severe infections have occurred, hematopoietic stem cell transplantation ( Chapter 163 ) is likely to be curative in 90% of selected cases. Gene therapy also can be used for several primary immunodeficiencies, including forms of severe combined immunodeficiency (e.g., adenosine deaminase deficiency) as well as Wiskott-Aldrich syndrome. For infants with severe combined immunodeficiency, lentiviral vector gene therapy combined with low-exposure, targeted busulfan conditioning can result in multilineage engraftment, reconstitution of functional T cells and B cells, and normalization of natural killer cell counts for at least 16 months with low-grade side effects. Gene editing to repair the endogenous gene so it can be expressed under normal regulatory controls is an experimental procedure.
In addition to the severe forms of severe combined immunodeficiency, a number of other genetic defects also impair both T- and B-cell limbs, but newborn screening may not identify these infants because the number of T-cells might not be sufficiently reduced. In these forms, in addition to infections, the phenotype may include not only susceptibility to bacterial infection but also atopy, severe viral infections, autoimmunity, and, in some, cancer ( E-Table 231-2 ).
Examples of these less severe T- and B-cell combined defects include syndromes in which major histocompatibility complex (MHC) class I or class II is not expressed (sometimes called bare lymphocyte syndromes), additional defects of T-cell signaling (such as Zap-70), and syndromes that result when defects of the actin cytoskeleton prevent T-cell activation, including the dedicator of cytokinesis proteins, DOCK2 and DOCK8.
As with the more severe combined immunodeficiency states, these syndromes lead to defects of both T- and B-cell compartments, and infants present with severe and recurrent bacterial, viral, or fungal infections; diarrhea; dermatitis; and usually failure to thrive. One notable example, with atopic disease and high immunoglobulin E (IgE), is DOCK8 deficiency, which was first recognized in babies with autosomal recessive hyper-IgE syndrome ( E-Table 231-2 ).
Hematopoietic transplantation is the only curative measure.
Another group of combined primary immune defects has distinctive systemic characteristics, aside from the obvious abnormities in the immune system ( Table 231-3 ). The best known of these are Wiskott-Aldrich syndrome, ataxia-telangiectasia, DiGeorge syndrome, hyperimmunoglobulin E (Buckley-Job) syndrome, cartilage-hair hypoplasia, and purine nucleoside phosphorylase (PNP) deficiency.
| TYPE | GENES | INHERITANCE | LABORATORY FEATURES | ALTERED FUNCTIONS | DISEASE AND COMPLICATIONS |
|---|---|---|---|---|---|
| Wiskott-Aldrich syndrome | WAS | X-linked | Thrombocytopenia, small platelets, low IgM and poor antibody responses to polysaccharides | Impaired cell activation, mobility | Eczema, lymphoma, autoimmune disease, bacterial and viral infections |
| Ataxia-telangiectasia | ATM | Autosomal recessive | Some have IgA deficiency, IgG defects, lymphopenia in some, may have low T cells on newborn screening |
Impaired DNA double-stranded break repair | Ataxia, telangiectasia, pulmonary infections, lymphoreticular and other malignant neoplasms, increased α-fetoprotein, x-ray sensitivity |
| DiGeorge/velocardiofacial syndrome/chromosome 22q11.2 deletion syndrome | 22q11.2 deletion, rarely a deletion in 10p | De novo (majority) or autosomal dominant | Lymphopenia, low T-cell numbers, large deletion in chromosome 22 on fluorescence in situ hybridization | Impaired T-cell immunity | Cardiac abnormalities, hypoparathyroidism, abnormal facies |
| Hyper-IgE syndrome (Buckley-Job syndrome) | STAT3 | Autosomal dominant | Eosinophilia, high IgE | Loss of normal cytokine activation, defective IL-17 | Bacterial infections, eczema, distinctive facial features, osteoporosis, fractures, scoliosis, delay of shedding primary teeth, hyperextensible joints, candidiasis |
| Cartilage-hair hypoplasia | RMRP | Autosomal recessive | Lymphopenia, low T-cell numbers | Impaired processing of mitochondrial RNA | Short-limbed dwarfism, sparse hair, celiac disease, Hirschsprung disease, bone marrow failure, autoimmunity, susceptibility to lymphoma |
| Purine nucleoside phosphorylase (PNP) deficiency | PNP | Autosomal recessive | Progressive T-cell loss, immune globulins normal or low |
Impaired T cell functions | Autoimmune hemolytic anemia, neurologic impairment |
Wiskott-Aldrich syndrome is an X-linked recessive disease that is characterized by eczema, thrombocytopenia, and immune deficiency. Wiskott-Aldrich syndrome is rare, estimated at 1 to 10 cases per million males. Ethnic differences are not known.
The syndrome is caused by mutations in the WAS gene, which codes for the Wiskott-Aldrich syndrome protein that is an intracellular cytoplasmic scaffold protein important for the activation and mobility of all blood cells. This protein is involved in the polymerization of actin and in establishing an interface between immune cells (the immune synapse). Partly depending on the location of the mutation in the WAS gene, milder versions lead to X-linked thrombocytopenia in some cohorts. Another, much rarer version leads to X-linked neutropenia.
The main manifestations in early childhood include eczema, chronic thrombocytopenia sometimes leading to bloody diarrhea, and immune deficiency with recurrent infections. Autoimmunity or inflammatory diseases, including autoimmune hemolytic anemia, splenomegaly, arthritis, inflammatory bowel disease, and vasculitis can be present. The incidence of lymphoma is also increased. Family history may include male relatives with Wiskott-Aldrich syndrome or thrombocytopenia.
The diagnosis is commonly made in the first few years of life in males who have the characteristic eczema as well as thrombocytopenia that leads to petechiae. IgM levels are typically low, whereas IgA (and sometimes IgE) levels are increased. Platelet sizes are smaller than normal, and clot retraction is poor. The diagnosis can be suggested by lack of the Wiskott-Aldrich syndrome protein as detected by flow cytometry, but definitive diagnosis requires gene testing.
Treatment strategies for Wiskott-Aldrich syndrome are diverse and usually determined by experts on a case-by-case basis. Conservative management includes prophylactic antibiotics, immunization with conjugated polysaccharide vaccines, and intravenous or subcutaneous immune globulin for patients who have repeated infections. For eczema, standard measures are used ( Chapter 405 ). Lifelong antibiotic prophylaxis is mandatory. Thrombopoietic agents, such as eltrombopag and romiplostim, can be helpful. Platelet transfusions should be reserved for active bleeding that cannot be managed with usual methods (e.g., aminocaproic acid) and should be avoided in patients for whom hematopoietic stem cell transplantation is being considered. Splenectomy is discouraged because of the risk of post-splenectomy sepsis. Autoimmunity can be difficult to control, and immune suppression should be used with caution. Treatment of lymphomas is by standard regimens ( Chapter 171 ).
Hematopoietic stem cell transplantation ( Chapter 163 ) can be curative. Trials with gene therapy are also ongoing.
The prognosis in Wiskott-Aldrich syndrome is highly variable. Some individuals have mild thrombocytopenia and occasional nose bleeds, whereas other patients have inflammatory diseases or other complications that require additional, sometimes intensive, medical management.
Ataxia-telangiectasia is a rare neurodegenerative disease that leads to cerebellar atrophy, skin telangiectasia, and immune defects. Ataxia-telangiectasia is estimated to occur in 1 in 40,000 to 100,000 persons. Males and females are affected equally.
Ataxia-telangiectasia is due to recessive mutations in the gene that encodes the ataxia telangiectasia mutated protein (ATMp), which is important in both cell division and DNA repair. With the loss of ATMp, DNA breakage cannot be repaired, thereby leading to cell death.
The clinical manifestations include progressive difficulty in walking, with ataxia beginning around age 5 years. Skin telangiectasias develop on the bulbar conjunctiva and behind the ears. The immune defects include IgA deficiency, IgG subclass defects, and cellular defects that can lead to recurring pulmonary infections and lung damage. Patients with ataxia-telangiectasia are radiosensitive and commonly develop lymphomas with increasing age.
The diagnosis of ataxia telangiectasia usually can be made by the characteristic clinical phenotype coupled with an increase in the serum α-fetoprotein blood level. Radiosensitivity can be assessed in vitro in fibroblast cell lines. Definitive diagnosis is by ATM gene sequencing.
Treatment for ataxia-telangiectasia includes a medical team that can provide supportive measures and physical therapy as needed. The life expectancy varies greatly, but most patients live into early adulthood.
DiGeorge syndrome is an autosomal dominant defect and one of the members of the 22q11.2 deletion syndrome that includes velocardiofacial syndrome, conotruncal anomaly face syndrome, congenital thymic aplasia, and thymic hypoplasia. DiGeorge syndrome is one of the most common of the immune defects, with an estimated incidence of 1 : 4000. Both sexes are affected equally. The immune defect in DiGeorge syndrome varies widely, from complete loss of thymic development with no circulating T cells to normal T-cell numbers. In most cases, the thymus is hypoplastic.
Although it is classified as an immune defect because of thymic hypoplasia or aplasia, patients with DiGeorge syndrome are likely also to have congenital heart disease ( Chapter 55 ), cleft palate or pharyngeal closure defects, characteristic facies, hypocalcemia due to parathyroid insufficiency, and learning disability. The more common cardiac defects include tetralogy of Fallot, interrupted aortic arch, ventricular septal defects, vascular rings, and anomalous return of brachial arteries. The mnemonic CATCH-22 has been applied: cardiac issues, abnormal facies, thymic aplasia, cleft palate, and hypocalcemia. With loss of thymic tissue, cellular immunity is mildly to moderately impaired, leading to recurrent infections. Hypogammaglobulinemia is not uncommon and may be associated with autoimmune cytopenias, especially thrombocytopenia.
Infants with DiGeorge syndrome may be first identified on a newborn screen when poor thymic function leads to low T-cell receptor excision circles (TRECs). The diagnosis of DiGeorge syndrome can be confirmed on genetic testing with fluorescence in situ hybridization, which detects the loss of the 22q11.2 gene segment or, more rarely, a loss of 10p14-p13. However, about 10% of affected individuals do not have a gene defect but have the syndrome because of maternal diabetes, fetal alcohol syndrome, or prenatal exposure to isotretinoin.
Treatment of DiGeorge syndrome is based on individual needs and may require cardiac surgery, repair of the cleft palate, and supplementation of calcium and vitamin D. Some patients are hypothyroid and require thyroid supplementation ( Chapter 207 ). Whereas the level of T cells may be subnormal for age, sufficient T-cell function remains so that no specific treatment is needed (partial DiGeorge syndrome) because infections tend to be more related to anatomic abnormalities than T-cell hypofunction. If the thymus is totally absent, thymic transplantation can supply sufficient reconstitution.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here