Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
autoimmune hepatitis
alkaline phosphatase
alanine aminotransferase
antimitochondrial antibody
antinuclear antibody
aspartate aminotransferase
carbohydrate antigen
endoscopic retrograde cholangiography
fluorescence in situ hybridization
genome-wide association studies
human leukocyte antigen
inflammatory bowel disease
immunoglobulin G4
interleukin-6
magnetic resonance cholangiography
natural killer
perinuclear antineutrophil cytoplasmic antibody
primary biliary cholangitis
primary sclerosing cholangitis
anti–smooth muscle antibody
steroid and xenobiotic receptor
tumor necrosis factor α
ulcerative colitis
Sclerosing cholangitis refers to a broad array of diseases that cause fibrosis of the bile ducts, usually of medium-sized and large ducts, leading to a segmental pattern of narrowing with proximal dilatation. It has classically been divided into primary and secondary types. Secondary sclerosing cholangitis may be a complication of a number of different injuries, inflammatory reactions, or malignancies. Included in this group is a newly defined group of diseases with elevated levels of immunoglobulin G4 (IgG4) that can involve the bile ducts, so-called IgG4-related sclerosing cholangitis. Primary sclerosing cholangitis (PSC) is a heterogeneous, idiopathic, inflammatory disorder of the bile ducts frequently associated with inflammatory bowel diseases (IBDs) of the colon resulting in strictures of the intrahepatic and/or extrahepatic bile ducts. Although most PSC patients have IBD, only approximately 5% of IBD patients will develop PSC, and despite intensive investigation the underlying causes of PSC and the IBD association remain poorly understood. PSC can affect all age groups and has been described in a variety of ethnic/racial groups but is best characterized in populations of northern European descent. The natural history of PSC is variable in terms of liver disease progression, with numerous possible clinical outcomes. In addition to progression to portal hypertension, cirrhosis, and its complications, PSC patients may also have bacterial cholangitis, cholangiocarcinoma, and colorectal adenocarcinoma. Clinical trials focusing primarily on bile acid therapy and immunosuppression have not yet proven beneficial effects. Liver transplant remains the best option for those with advanced disease or in select patients with localized cholangiocarcinoma. However, recurrent PSC occurs at a low rate and may require repeated liver transplant. Increasing interest in PSC and international collaboration has led to improved understanding of the heterogeneity of the disease and its underlying genetic structure, and has opened new opportunities to pursue effective therapeutics.
I shall not today attempt further to define the kinds of material I understand to be embraced within that shorthand description; and perhaps I could never succeed in intelligibly doing so. But I know it when I see it. JUSTICE POTTER STEWART
The first description of PSC has been attributed to Hoffmann in 1867, and in the 1920s the description of an obliterative cholangitis of the extrahepatic biliary tree with diffuse thickening of the wall and narrowing of the lumen was described by the French surgeons Delbet and Lafourcade. However, the moniker primary sclerosing cholangitis (PSC) was first used in 1954 by Castleman and later in a 1958 review of six cases by Schwartz and Dale. Early descriptions of PSC noted the predominance of men in the third and fourth decades of life and the association with ulcerative colitis (UC). However, increasingly the features of PSC are seen in a variety of clinical settings with variable subphenotypes, suggesting that what is often described as PSC may be a syndrome arising from several different underlying pathologic processes ( Table 43-1 ). The distinction of sclerosing cholangitis as primary versus secondary will likely become more irrelevant as we better understand the mechanisms leading to PSC and define strict diagnostic criteria. Nevertheless, for current purposes, we will maintain the distinction between PSC and secondary sclerosing cholangitis ( Fig. 43-1 ).
|
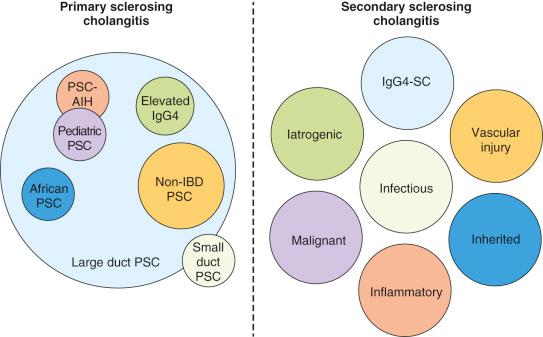
The classic form of PSC as originally described has several characteristic features; namely, large duct PSC occurs predominantly in men (male/female ratio 3 : 2), is coexistent with IBD in 60% to 80% of cases, and typically presents with cholestasis. The IBD typically is a pancolitis with frequent ileitis and rectal sparing. In addition, the IBD is commonly mild and asymptomatic. The association between PSC and IBD appears to be greater at northern latitudes, although even there the frequency of IBD PSC is decreasing. The natural history of large duct PSC ranges from rapidly progressive to indolent. The mean transplant-free survival has been reported to be from 12 years to more than 20 years, with the latter including more recent population-based estimates. This group of PSC is the most common and best described and for which there is the best understanding, including the strong association with the human leukocyte antigen (HLA) haplotype defined by HLA-A*01, HLA-B*08, and HLA-DRB1*03.
In contrast to PSC in the presence of IBD, PSC in the absence of IBD tends to be equally distributed among men and women, is diagnosed in much older individuals, and may have a better prognosis. Although the rarity of PSC without IBD limits the power of genetic analysis, this group of PSC patients appears to share similar HLA risk alleles compared with PSC with IBD.
A small group of adult PSC patients present with clinical and histologic features compatible with PSC, except for the lack of typical cholangiographic findings, and these have been defined as small duct PSC . In some series, IBD was required for the diagnosis. In addition, these patients may have been labeled in the past as having antimitochondrial antibody (AMA)-negative primary biliary cholangitis (PBC), formerly known as primary biliary cirrhosis, or autoimmune cholangiopathy. In most cohorts, small duct PSC constitutes approximately 10% of the total PSC population, rarely progresses to large duct PSC, and has a generally favorable outcome. Recent analysis of the HLA region shows that small duct PSC without IBD is genetically distinct from large duct PSC.
Anywhere between 1% and 53.8% of patients with PSC also have features of autoimmune hepatitis (AIH). Unfortunately, there remain no agreed diagnostic criteria for PSC-AIH overlap, and this is likely why the reported frequency of PSC-AIH differs so widely. These patients may present with significant elevations of liver transaminase concentrations and histologic findings consistent with AIH, or they may initially present with typical AIH that subsequently becomes cholestatic with the development of sclerosing cholangitis. In some cases, PSC-AIH may respond to immunosuppression. Importantly, autoantibodies, including antinuclear antibodies (ANAs) and anti–smooth muscle antibodies (SMAs), are frequent in PSC without evidence of AIH. In addition, 10% or more of patients with AIH may have cholangiographic features on magnetic resonance cholangiography consistent with PSC.
PSC in children appears to have many of the same features as PSC in adults; namely, a male predominance and strong association with IBD. However, in children PSC appears to be much more responsive to therapies and to have a higher frequency of overlap with AIH. Autoimmune sclerosing cholangitis is a term used to designate a group of patients with PSC-AIH features with the exceptional reversal of cholangiographic findings with immunosuppression. Recent case series have also reported marked clinical improvement with orally administered vancomycin in children with PSC. Notably, neither of these therapies has shown similar effects in adults.
Most studies of PSC have been performed in northern European populations or populations which descended from northern Europe, leading some to conclude that PSC is a disease of Caucasians. However, PSC is a modern disease, and given its association with IBD, it is likely that as the geoepidemiology of IBD changes, the frequency of PSC in non-Caucasian populations will change with it. There are few data from Asia, apart from data on the IgG4-related sclerosing cholangitis associated with autoimmune pancreatitis described in Japan, where PSC appears to be extremely rare.
In contrast, studies of a large healthcare organization and U.S. transplant data suggest that the incidence and prevalence rates of PSC among African Americans are at least as great as in Caucasians. In African Americans there is a less striking male predominance and a lower IBD rate. HLA-DR3, which is strongly associated with PSC in European populations, is rare among African Americans and is not associated with PSC in African American patients. However, the HLA-B8 association is shared between both Caucasian and African American PSC patients.
The recent description of IgG4-related sclerosing cholangitis often found in association with autoimmune pancreatitis as one of many diseases associated with elevated IgG4 serum levels and tissue infiltration of IgG4 plasma cells has led to the recognition that some previously diagnosed cases of PSC were in fact IgG4-related sclerosing cholangitis. Adding confusion to this issue are findings that serum IgG4 levels are often elevated in PSC and IgG4 plasma cells are frequent in PSC liver explants, but the two features do not necessarily correlate. In addition, IgG4-related diseases are typically responsive to corticosteroids, which is not seen in PSC. As such, IgG4-related sclerosing cholangitis fits somewhere between PSC and secondary sclerosing cholangitis. Ultimately, the diagnosis of IgG-4 related sclerosing cholangitis is based on histology, imaging, serology, other organ involvement, and response to steroid therapy, the so-called HISORt criteria that were originally developed for the diagnosis of autoimmune pancreatitis ( Table 43-2 ).
| Histology |
| Periductal lymphoplasmacytic infiltrate with obliterative fibrosis and storiform fibrosis |
| Lymphoplasmacytic infiltrate with storiform fibrosis with abundant IgG4 cells (≥ 10 IgG4 cells per high-power field) |
| Imaging |
| Diffusely enlarged gland with delayed “rim” enhancement, diffusely irregular, attenuated main pancreatic duct Focal pancreatic mass/enlargement, focal pancreatic ductal stricture, pancreatic atrophy, calcification, pancreatitis |
| Serology |
| Elevated serum IgG4 level |
| Other Organ Involvement |
| Hilar/intrahepatic biliary strictures, persistent distal biliary stricture, parotid/lacrimal gland involvement, mediastinal lymphadenopathy, retroperitoneal fibrosis |
| Response to Steroid Treatment |
| Resolution or marked improvement of pancreatic/extrapancreatic manifestation with corticosteroid therapy |
Secondary sclerosing cholangitis may arise from a variety of causes presenting with biliary strictures, both benign and malignant ( Table 43-3 ). The most common benign causes are iatrogenic and secondary to biliary injury after cholecystectomy or liver transplant. Following the inception of laparoscopic cholecystectomy, the rate of iatrogenic biliary injury was reported to be 0.8% to 1.4% compared with rates of 0.1% to 0.2% for open cholecystectomy. However, more recently the rates of biliary injury from laparoscopic cholecystectomy have fallen to rates similar to those for open cholecystectomy. Importantly, biliary strictures represent only a small fraction of all bile duct injuries. In contrast, the rates of biliary strictures following liver transplant appear to be increasing because of the increased risk associated with the use of marginal donors, deceased donors after cardiac death, and partial liver transplant, including living donor liver transplant. Strictures may occur at the anastomotic site or may be nonanastomotic. The incidence of the former has been estimated as 5% to 25% and that of the latter has been estimated as 10% to 15%.
| Pediatric | Benign | Malignant |
|---|---|---|
| Cystic fibrosis Primary and secondary immunodeficiency Histiocytosis X Neonatal sclerosing cholangitis Biliary atresia Ichthyosis with sclerosing cholangitis Congenital bile duct abnormalities Sickle cell disease Progressive familial intrahepatic cholestasis type 3 |
Autoinflammatory: sarcoidosis, eosinophilic cholangitis, mast cell cholangitis Iatrogenic bile duct injury: cholecystectomy; liver transplant (anastomotic stricture, nonanastomotic stricture) Cholelithiasis (Mirizzi syndrome) Chronic pancreatitis Vascular: ischemic cholangiopathy, vasculitis, intraarterial chemotherapy, portal hypertensive biliopathy Infectious: AIDS cholangiopathy, recurrent pyogenic cholangitis Biliary inflammatory pseudotumor |
Cholangiocarcinoma Pancreatic adenocarcinoma Metastatic cancer Gallbladder cancer Ampullary adenocarcinoma Hepatocellular carcinoma Lymphoma |
Several autoinflammatory conditions other than PSC can lead to secondary sclerosing cholangitis. Sarcoidosis is a multisystem granulomatous disease, which frequently affects the liver and in some cases presents with cholestasis and features resembling PBC and in rare cases of PSC. Eosinophilic cholangitis is an extremely rare disorder characterized by a dense transmural infiltrate of eosinophils, which is most frequently part of multiorgan involvement of an eosinophilic infiltration. Mast cell infiltration of the bile ducts is common in a variety of disorders causing secondary sclerosing cholangitis, and in a case report has been associated with systemic mastocytosis.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here