Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
At the end of 2020, approximately 37.6 million people worldwide were living with human immunodeficiency virus (HIV) infection and over 35 million people have died as a consequence of the acquired immunodeficiency syndrome (AIDS). Opportunities to prevent new HIV infection include behavioral modification, preexposure and postexposure prophylaxis, suppression of the viral load in already-infected individuals, and the hope for an effective vaccine ( Fig. 356-1 ). ,
The primary mode of HIV transmission throughout the world is sexual contact. However, the geographic distribution of cases attributable to homosexual or heterosexual transmission varies markedly. In the United States, most sexually transmitted cases of HIV are observed in men who have sex with men (MSM), and heterosexual transmission accounts for a smaller number of new infections except among women. However, heterosexual transmission is the leading mode of transmission worldwide and remains the primary mode of disease acquisition in Africa. Sexual transmission of HIV is relatively inefficient, but behavioral and biologic factors influence the likelihood of HIV transmission in a given sexual encounter. In particular, coinfection with classic sexually transmitted infections (STIs; especially genital ulcerative diseases such as herpes simplex) greatly increases both the infectiousness and the susceptibility of an individual. Sexually transmitted infections increase the concentration of HIV in genital secretions and, as a result, increase the likelihood for transmission.
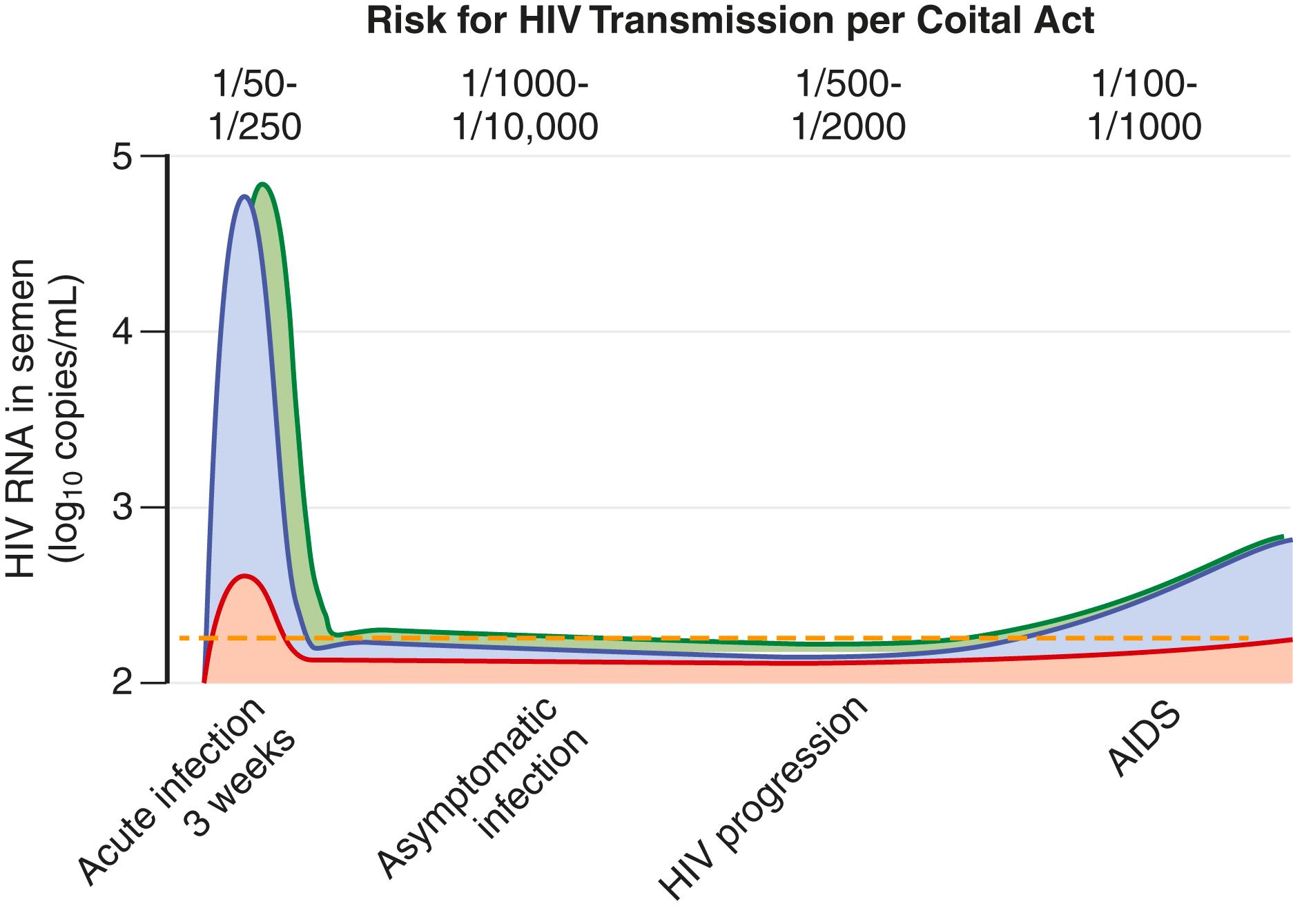
The risk for acquisition of HIV per coital act has been estimated to range from 5 per 10,000 for insertive unprotected penile-vaginal intercourse to 50 per 10,000 for receptive unprotected anal intercourse. However, the risk is not stable and varies depending on the stage of infection and other amplifying cofactors. HIV transmission risk is highest in early HIV infection and in advanced infection ( Fig. 356-2 ), thereby demonstrating that the viral concentration in the genital secretions is the strongest predictor of the risk for transmission.
Traditional strategies for the prevention of sexual transmission of HIV have focused on encouraging abstinence, reducing unsafe sexual behaviors (especially unprotected anal intercourse and concurrent relationships), encouraging proper use of condoms, and treating sexually transmitted infections. These interventions primarily focus on HIV-negative persons.
In situations in which a decision to engage in sexual activity has been made and the HIV serostatus of the partner is positive, unknown, or in doubt, safe sexual practices (“safe sex”) should be implemented. Consistent and proper use of latex but not other types of condoms greatly reduces the transmission of HIV at both the individual and population levels. In particular, natural skin condoms should not be used because they do not prevent transmission of HIV. Petroleum-based lubricants enhance the likelihood of rupture of latex condoms and should be avoided. If needed, water-based lubricants should be used. The effectiveness of condoms in preventing heterosexual transmission of HIV has been estimated to be 87%, but it may be as low as 60% or as high as 96%. The effectiveness of condoms during anal intercourse is probably lower because the frequency of condom breakage and slippage may be considerably higher than during vaginal intercourse. The FDA has recently approved a condom specifically for anal sex.
Circumcision represents an additional strategy to protect men from HIV. Randomized clinical trials have demonstrated a protective benefit of male circumcision, with the risk for acquisition of HIV infection through heterosexual intercourse decreasing by approximately 60%, with increasing reduction in HIV acquisition over time. However, this benefit has not been confirmed for MSM.
For people with HIV, antiretroviral therapy is a powerful tool to decrease HIV replication and thus infectivity and the subsequent risk for transmission through sexual contact. For example, early antiretroviral therapy of people with HIV beginning at a CD4 count of 350 to 550 cells/μL reduces heterosexual transmission of HIV-1 by 96%. Observational data confirm a similar reduction of infectiousness for sexual transmission of HIV-1 when virus loads are suppressed among MSM. The concept that if HIV is untransmittable if it is undetectable is a powerful rationale for achieving an undetectable viral load on antiretroviral therapy.
Antiretrovirals also are effective to prevent HIV when administered prophylactically as preexposure prophylaxis (PrEP) to HIV-uninfected at-risk individuals ( Table 356-1 ), and they are strongly recommended for persons at increased risk of acquiring HIV infection. For example, daily administration of co-formulated tenofovir disoproxil fumarate plus emtricitabine as preexposure prophylaxis can decrease the risk for HIV infection among MSM by about 45 to 85%, with benefits varying across studies depending on adherence. , Subset and post-hoc analyses of the randomized studies to account for adherence to medication suggest greater than 99% efficacy against rectal acquisition and over 94% efficacy against vaginal transmission when taken as prescribed. For MSM and transgender women, daily tenofovir alafenamide in combination with emtricitabine is noninferior to daily tenofovir disoproxil fumarate plus emtricitabine, with a comparable clinical safety profile. For men, “on-demand” pericoital use of a tenofovir disoproxil fumarate plus emtricitabine (a double-dose of tenofovir disoproxil fumarate plus emtricitabine 2 to 24 hours prior to planned intercourse, a single dose 24 hours later, and a final single dose 24 hours later is also of proven efficacy. Individuals with ongoing exposures after the first dose should extend the daily single dosing until 48 hours after their last sexual activity. Randomized clinical trials have also demonstrated superiority of every-2-months injectable cabotegravir compared with daily oral tenofovir disoproxil fumarate plus emtricitabine as preexposure prophylaxis in MSM, transgender women who have sex with men, and cisgender women. , The results strongly support the adherence benefit of an injection rather than oral preexposure prophylaxis.
PrEP should be considered in the following HIV-negative persons:
The preferred regimens are daily oral tenofovir disoproxil fumarate/emtricitabine or “on-demand”/pericoital tenofovir disoproxil fumarate/emtricitabine for men having planned sex and absent active HBV infection.
|
The HIV status of persons using injectable cabotegravir for preexposure prophylaxis should be monitored using the most sensitive testing assay available, ideally an HIV RNA test, owing to alterations in the biology and laboratory testing profiles of newly acquired HIV in persons who are using a long-acting product for preexposure prophylaxis.
Use of a monthly vaginal ring containing dapivirine can reduce HIV infection by about 30%. The ring has been approved by the European Medicines Agency and by the South African government, and it has been endorsed by the World Health Organization (WHO); however, if has been withdrawn from consideration before the U.S. FDA.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here