Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
body mass index
coronary artery disease
hepatitis B virus
hepatocellular carcinoma
hepatitis C virus
human immunodeficiency virus
locoregional therapy
liver transplantation
Model for End-Stage Liver Disease
nonalcoholic steatohepatitis
primary biliary cholangitis
pediatric end-stage liver disease
primary sclerosing cholangitis
simultaneous liver and kidney
transarterial chemoembolization
transarterial radioembolization
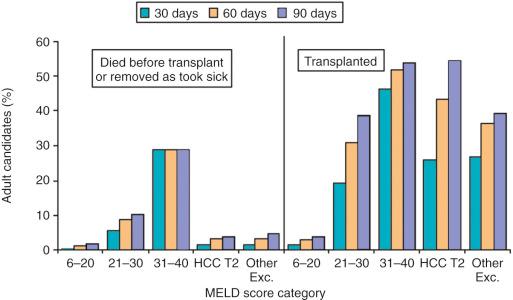
Liver transplantation (LT) has revolutionized the care of patients with end-stage liver disease. Since the first successful human liver transplant performed in 1967 by Thomas Starzl at the University of Colorado, advances in surgical technique, immunosuppression, and patient selection have made the procedure a standard life-saving treatment for patients with decompensated acute and chronic liver diseases. In the United States, LT resulted in more than 400,000 life-years saved from 1987 to 2012. Liver transplantation is available in most regions of the world, generally with excellent patient and graft survival. U.S. national graft survival rates as of 2013 at 1 year, 3 years, and 5 years after LT are roughly 85%, 75%, and 68% respectively ( Tables 50-1 and 50-2 ). Ubiquitous application of LT is limited by the scarcity of available donor liver grafts. Despite efforts to expand the donor pool and improve systems to triage liver grafts, death rates of patients while they are waiting for liver transplant remain high. Fig. 50-1 shows the waiting list outcome for U.S. liver transplant candidates after 30 days, 60 days, and 90 days. Of the more than 15,000 patients on the waiting list in the United States in 2013, there were 5763 liver transplants and 1767 deaths, with 1223 patients reported as too sick for transplant. For patients with end-stage liver disease, the stakes are high, and their healthcare providers' familiarity with the appropriate pretransplant evaluation and management is of utmost importance.
| Primary Diagnosis | PATIENT SURVIVAL (%) | |||
|---|---|---|---|---|
| 3 mo * | 1 yr * | 5 yr † | 10 yr ‡ | |
| All diseases | 95 | 90 | 74 | 61 |
| Noncholestatic liver disease/cirrhosis | 95 | 89 | 73 | 58 |
| Cholestatic liver disease/cirrhosis | 95 | 92 | 83 | 72 |
| Acute hepatic necrosis | 92 | 89 | 77 | 64 |
| Biliary atresia | 96 | 94 | 90 | 86 |
| Metabolic diseases | 95 | 93 | 84 | 78 |
| Malignant neoplasm | 96 | 90 | 68 | 55 |
| Other diseases | 95 | 93 | 76 | 67 |
* 10,394 patients during the years 2009 to 2010.
| Primary Diagnosis | GRAFT SURVIVAL (%) | |||
|---|---|---|---|---|
| 3 mo * | 1 yr * | 5 yr † | 10 yr ‡ | |
| All diseases | 92 | 86 | 70 | 56 |
| Noncholestatic liver disease/cirrhosis | 92 | 86 | 69 | 53 |
| Cholestatic liver disease/cirrhosis | 92 | 88 | 76 | 63 |
| Acute hepatic necrosis | 90 | 85 | 71 | 58 |
| Biliary atresia | 91 | 88 | 81 | 76 |
| Metabolic diseases | 93 | 88 | 78 | 71 |
| Malignant neoplasm | 94 | 87 | 65 | 52 |
| Other diseases | 88 | 83 | 66 | 56 |
* 11,142 patients during the years 2009-2010.
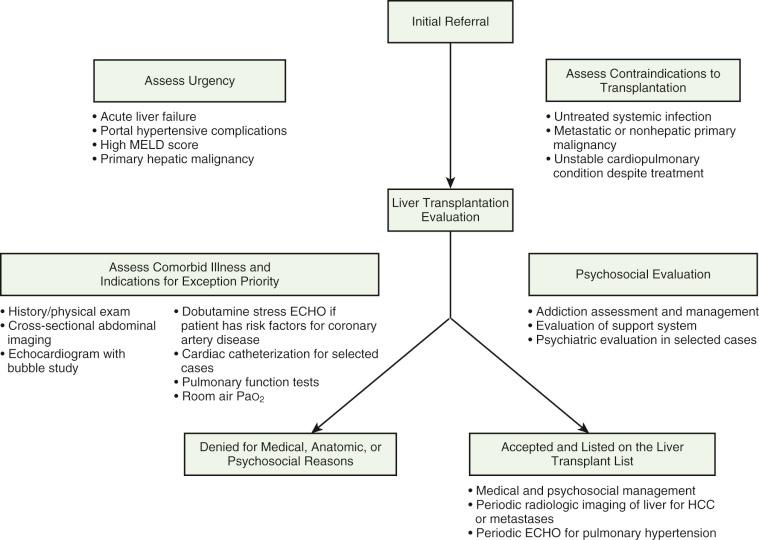
Appropriate evaluation of a patient for LT begins with recognition of a need for the procedure and a timely referral to a transplant center ( Figs. 50-2 and 50-3 ). For many patients the urgency of referral may go unrecognized by their providers (see “ Timing of Referral for Evaluation ”). Once a patient has been referred, most transplant centers have a multidisciplinary approach that includes a detailed history and examination by a transplant hepatologist and transplant surgeon, and assessments by a mental health provider, a social worker, a transplant coordinator, a financial counselor, and other specialists such as pharmacists and nutritionists. Many centers offer patients and their support members counseling, educational sessions, and the opportunity to meet with prior organ transplant recipients to enhance their knowledge and reduce stress related to the process. A large evaluation process, including laboratory, imaging, cardiovascular, and pulmonary tests, is performed to confirm the cause and severity of liver disease, estimate renal function, determine the blood group, evaluate the patient's exposure and immunity to prior infections, screen the patient for and assess the degree of comorbid medical conditions, and assess the technical feasibility of transplant surgery ( Table 50-3 ). Additionally, patients undergo specific screening for concurrent medical conditions that may warrant appeal for exceptional priority for LT, such as hepatocellular carcinoma (HCC) and hepatopulmonary syndrome ( Table 50-4 ). The details of prior therapies for liver disease are collected, especially for patients with hepatitis C virus (HCV), hepatitis B virus (HBV), or human immunodeficiency virus (HIV) infection, whose prior treatments may direct future treatment choices. Age and sex-specific preventive healthcare, dental care, and cancer screening are generally required. Patients should be committed to a healthy lifestyle, including cessation of the use of tobacco products given the link between tobacco smoking and cardiovascular death, hepatic artery thrombosis, and de novo cancers in posttransplant patients. On the basis of the initial evaluation, targeted specialist consultation is requested as needed to evaluate and potentially intervene to mitigate the perioperative and postoperative surgical risk or to further assess medical comorbid conditions. For patients with severe renal insufficiency or failure, the assessment includes whether or not simultaneous liver and kidney (SLK) transplantation may be indicated.
| Standard Blood Tests |
| Complete blood count, liver chemistry, kidney profile, coagulation profile (PT, PTT) |
| ANA, smooth muscle antibody, AMA |
| Iron studies, ceruloplasmin, α 1 -antitrypsin phenotype |
| CMV, EBV, HSV, VZV, HIV; syphilis; toxoplasmosis |
| HAV, HDV serologic tests |
| α-Fetoprotein |
| Other Standard Tests |
| Abdominal ultrasonography with Doppler imaging, electrocardiogram, chest X-ray, pulmonary function tests, endoscopic evaluations |
| PPD skin tests |
| Standard Consultations |
| Dietary |
| Psychosocial |
| Women's health (Papanicolaou test, mammogram in women older than 35 yr) |
| Financial (insurance clearance must be obtained) |
| Overall assessment of patient (clinical judgment in addition to biochemical parameters) |
| Other Optional Tests |
| CT or MRI (to exclude HCC); angiography if needed to exclude vascular abnormalities |
| Carotid duplex scanning (for older or cardiovascular disease patients) |
| Contrast echocardiography (for suspected hepatopulmonary syndrome) |
| Cardiac catheterization (for suspected CAD) |
| Colonoscopy (history of IBD, PSC, polyps, family history of colon cancer), FOBT |
| ERCP (in PSC) |
| Liver biopsy |
| Fungal serologic tests (in areas endemic for dimorphic fungi) |
| Other Optional Measures |
| Pretransplant vaccines, if needed (hepatitis A and B, pneumococcal vaccines, influenza vaccine, tetanus booster) |
| Diagnosis | Criteria |
|---|---|
| Acute liver failure | Fulminant hepatic failure with onset of hepatic encephalopathy within 8 wk of first symptoms of liver disease, intensive care unit requirement, and one of the following three criteria: (1) ventilator dependence; (2) renal dialysis; or (3) INR >2.0 Primary nonfunction or hepatic artery thrombosis of transplanted liver graft within 7 days of transplant (less priority for those with hepatic artery thrombosis within 14 days of transplant) defined by AST ≥3000 U/L and one of the following: INR ≥2.5 or acidosis (arterial pH ≤7.30, venous pH ≤7.25 or lactate ≥4mmol/L) Wilson disease with acute liver decompensation |
| Hepatocellular carcinoma | Contrast-enhanced cross-sectional imaging documenting hepatic lesion(s) with increased contrast enhancement on late hepatic arterial images with either washout on portal venous/delayed phase imaging, late capsule enhancement, 50% growth in 6 mo or less, or biopsy evidence. A single tumor must be ≥2 cm in the largest diameter but not exceeding 5 cm. For multiple lesions, no more than three lesions, with the largest not exceeding 3 cm Extrahepatic spread must be ruled out with contrasted cross-sectional imaging of the chest, abdomen, and pelvis. Treated lesions can be included if they met the above criteria before treatment |
| Cholangiocarcinoma | Neoadjuvant therapy protocol approved by the UNOS committee Unresectable hilar cholangiocarcioma of ≤3 cm documented by cross-sectional imaging and malignant-appearing stricture on cholangiography and one of the following: carbohydrate antigen 19-9 >100 U/mL or biopsy or cytology results demonstrating malignancy or aneuploidy Exclude intrahepatic and extraheptic metastases by cross-sectional imaging of chest and abdomen initially and every 3 mo Exclude regional hepatic and peritoneal metastases by operative staging after neoadjuvant therapy and before liver transplantation Avoid transperitoneal aspiration or biopsy of primary tumor |
| Hepatopulmonary syndrome | Clinical evidence of portal hypertension, evidence of a right to left extracardiac shunt, Pa o 2 <60 mm Hg with the patient breathing room air, and no significant clinical evidence of underlying primary pulmonary disease |
| Portopulmonary hypertension | Prior MPAP >35mm Hg and elevated transpulmonary gradient >12mm Hg; presently controlled with MPAP <35mm Hg and pulmonary vascular resistance <400 dyn sec/cm -5 |
| Familial amyloid polyneuropathy | Documented amyloidosis, echocardiogram with an ejection fraction of >40%, ambulatory status, identification of gene mutation, and biopsy proven amyloid |
| Primary hyperoxaluria | Documented primary hyperoxaluria, with AGT deficiency proven by liver biopsy, estimated GFR ≤25 mL/mL for 6 wk or more |
| Cystic fibrosis | Documented cystic fibrosis, signs of reduced pulmonary function defined by FEV 1 <40% |
| Metabolic diseases | Urea cycle disorder or organic acidemia |
The initial reason for initiating liver transplant evaluation is the severity of liver complications. Other, also fundamental, considerations for transplant candidacy include the following :
Are significant comorbid conditions present that could limit successful LT with unacceptably high perioperative risk?
Are there significant psychosocial problems, including substance abuse issues, that need to be addressed before LT and/or that could limit successful transplantation and adherence to a long-term complicated medical regimen?
Is there an opportunity to intervene upon medical comorbidities or psychosocial concerns that could result in improved waiting list and posttransplant outcomes?
Patients with decompensated liver disease, regardless of the cause, should be considered as potential candidates for LT. Assessing the urgency of a referral includes the evaluation of (1) whether liver disease is acute or chronic, (2) the severity of complications of cirrhosis that increase the death risk (e.g., ascites, portal hypertensive bleeding, hepatic encephalopathy, hyponatremia, hepatorenal syndrome, and elevated Model for End-Stage Liver Disease [MELD] score), (3) the availability of local expertise to manage liver complications, and 4) the proximity of a transplant center. Several clinical tools exist to assist in assessment of the degree of liver disease, both acute and chronic ( Tables 50-5 and 50-6 ). Nearly all transplant centers will provide triage assistance and emergent management advice at any time of day. A bias toward early referral is generally encouraged.
| Criteria of King's College, London |
| Acetaminophen patients |
|
| Nonacetaminophen patients |
|
| Criteria of Hospital Paul-Brousse, Villejuif |
| Hepatic encephalopathy and Factor V level
|
| MELD | |||
| MELD score = 9.57(ln serum creatinine concentration) + 3.78(ln serum bilirubin concentration) + 11.2(ln INR) + 6.43 | |||
| Child-Turcott-Pugh Score | |||
| Points | 1 | 2 | 3 |
| Total bilirubin (mg/dL) | <2.0 | 2-3 | >3 |
| Albumin (g/dL) | >3.5 | 2.8-3.5 | <2.8 |
| INR | <1.7 | 1.7-2.3 | >2.3 |
| Ascites | Absent | Mild/moderate | Severe |
| Encephalopathy | Absent | Grade I-II | Grade III-IV |
| ACLF | |||
| No ACLF, 90-Day Mortality 14% | Grade 1 ACLF, 90-Day Mortality 40.7% | Grade 2 ACLF, 90-Day Mortality 52.3% | Grade 3 ACLF, 90-Day Mortality 79.1% |
| No organ failure or single nonkidney failure or single cerebral failure with serum creatinine <1.5 mg/dL | Single kidney failure or single liver, coagulation, circulatory, or respiratory failure with serum creatinine 1.5-1.9 mg/dL and/or mild to moderate hepatic encephalopathy or single cerebral failure with serum creatinine 1.5-1.9 mg/dL | Failure of two organs | Failure of three or more organs |
| Clinical Cirrhosis Stage (modified Baveno IV) | ||||
| Stage 1 | Stage 2 | Stage 3 | Stage 4 | Stage 5 |
| Compensated cirrhosis without varices | Compensated cirrhosis with varices | Bleeding without complications | First nonbleeding decompensation event * | Any second decompensating event |
* A nonbleeding decompensation event is jaundice, encephalopathy, or ascites.
When severe acute liver injury or acute liver failure occurs, urgent contact with a liver transplant center is indicated. Patients with evidence of severe acute liver injury and signs of hepatocellular insufficiency (encephalopathy and/or coagulopathy with an international normalized ratio >1.5 is an ominous sign) should be admitted to the hospital and monitored closely for further deterioration. Acute liver failure is defined as an international normalized ratio of 1.5 or greater and any degree of encephalopathy without preexisting cirrhosis (excepting hepatitis B, Wilson disease, and autoimmune hepatitis) and with an overall illness duration of less than 26 weeks. Given a high risk of rapid decompensation, transfer to a liver transplant center should be considered early provided there are no obvious contraindications to LT or safe transportation of the patient. Frequent assessment of the patient in the peritransfer period is imperative, with attention to the patient's neurologic status and ability to protect his or her airway before, during, and after transfer. Providers should have a low threshold for intensive care–level monitoring and endotracheal intubation, and even mild hepatic encephalopathy should trigger transfer to a higher level of care. A broad workup for the degree of illness, important comorbid illnesses, and cause should be considered ( Table 50-7 ). Once the patient arrives at the transplant center, consideration for LT should begin immediately.
| Serum Workup for Degree of Illness, Comorbid Illness, and Preparation for Possible Liver Transplantation | Workup for Cause |
|---|---|
|
|
In acute liver failure, the interval from jaundice to encephalopathy is prognostic, with short intervals (<4 weeks) associated with a greater risk of brain edema and intracranial hypertension but an improved spontaneous survival, whereas longer intervals are associated with less brain edema but a lower spontaneous survival. The degree of hepatic encephalopathy correlates with the risk of cerebral edema, which is rarely seen in patients with grade I or grade II encephalopathy, is seen in 25% to 35% of patients with grade III encephalopathy, and is seen in 75% of patients with grade IV encephalopathy. Although there is evidence for a role of ammonia in cerebral edema and herniation in patients with acute liver failure, and lactulose may be associated with a small increase in transplant-free survival, the decision to use lactulose in the patient with acute liver failure specifically should be made with caution out of concern for gaseous abdominal distention causing surgical difficulties during the transplant procedure. Furthermore, limited data on ammonia elimination through the use of l -ornithine l -aspartate are not compelling for reduction of arterial ammonia levels or improvement of survival in patients with acute liver failure. It is not clear if this lack of effect is a failure of l -ornithine l -aspartate therapy and/or evidence against an ammonia reduction strategy in acute liver failure.
Acetaminophen toxicity, the most common cause of acute liver failure in the United States and Europe, has the greatest spontaneous survival rate, whereas idiosyncratic drug-induced liver injury has one of the worst survival rates. Although many prognostic scores exist to predict low transplant-free recovery, the King's College criteria and the Clichy criteria are the most used scores to identify the need for LT (see Table 50-5 ). Although the King's College criteria provide a high accuracy in predicting death without LT (positive predictive value of 80% to 100%), they are less effective in predicting who will recover spontaneously (negative predictive value of 23% to 70%). Therefore, although a patient with acute liver failure meeting the King's College criteria will likely require LT, a patient not meeting these criteria does not accurately predict a lack of a need for LT, limiting their usefulness in optimizing liver transplant decision making. Several other prognostic variables and scoring systems have been studied in acute liver failure, including the MELD score, serum actin-free Gc-globulin concentration, serum phosphate concentration, arterial ammonia concentration, and serum α-fetoprotein concentration.
When chronic liver disease manifests itself as signs of decompensation, referral for LT should be considered. Such clinical signs include the presence of encephalopathy, portal hypertensive gastrointestinal bleeding, moderate ascites, spontaneous bacterial peritonitis, or hepatorenal syndrome, or the development of HCC, each of which is associated with significantly increased mortality without LT when compared with compensated cirrhosis.
Several general and disease-specific prognostic tools exist to predict the natural history of chronic liver disease. Among these models, the Child-Turcotte-Pugh score and the MELD score are most commonly used (see Table 50-6 ). The MELD score, using serum total bilirubin concentration, serum creatinine concentration, and serum international normalized ratio, is an accurate and objective measure of liver-related mortality and is used to prioritize patients for LT. The addition of serum sodium concentration to the MELD score further improves its predictive ability and serum sodium concentration was formally incorporated into the U.S. allocation system in 2016.
Acute-on-chronic liver failure is an increasingly recognized clinical scenario wherein a relatively well compensated individual experiences an new and rapid decompensation. It is defined by acute development of ascites, encephalopathy, gastrointestinal hemorrhage, and/or bacterial infection in the setting of one or more organ/system failures. Acute-on-chronic liver failure is graded by type (i.e., liver, kidney, cerebral, coagulation, circulatory, and respiratory) and number of organ failures, and is positively correlated with mortality (see Table 50-6 ). When acute-on-chronic liver failure is assessed at 3 days to 7 days after presentation, the acute-on-chronic liver failure score may be useful for determining the urgency of LT.
Another emerging paradigm is a prognostic clinical cirrhosis staging model (modified Baveno IV) wherein patients are classified according to historical occurrence and type of decompensating events (see Table 50-6 ). This cumulative scale quantifies the risk of a patient transitioning to a higher stage of cirrhosis and mortality. The 5-year risk of transitioning to a higher stage for patients with stage 1, 2, 3, and 4 cirrhosis is 35%, 42%, 65%, and 78% respectively. The 5-year mortality rate for patients with stage 1, 2, 3, 4, and 5 cirrhosis is 1.5%, 10%, 20%, 30%,and 88% respectively. A similar scale of clinical cirrhosis stage has been shown to predict transplant waiting list mortality in patients with a low MELD score (≤20).
Guidelines from the American Association for the Study of Liver Diseases recommend patients with cirrhosis be referred for liver transplant evaluation when the MELD score reaches 15, or when the patient develops the first major complication (ascites, variceal hemorrhage, encephalopathy, or type I hepatorenal syndrome). The pediatric end-stage liver disease (PELD) score includes five factors (total bilirubin concentration, albumin concentration, international normalized ratio, age less than 1 year, and growth failure) and, like the MELD score, has been shown to accurately predict short-term waiting list mortality in pediatric patients with chronic liver disease. Children with chronic liver disease are recommended to have a transplant evaluation when they show evidence of poor weight gain or growth failure, or evidence of infectious or portal hypertensive complications.
Despite the success of LT as a treatment for acute and chronic liver disease, transplant surgery and posttransplant immunosuppression are associated with significant morbidity and mortality ; therefore feasible alternatives to transplantation should be duly considered. The challenge of assessing the risk-benefit ratio of LT is most evident in acute liver failure, where the most gravely ill patient can, on occasion, have a dramatic and complete recovery without LT and avoid lifelong immunosuppression and the associated morbidity. Patients with some chronic liver diseases, such as severe autoimmune hepatitis, chronic Wilson disease, and HBV or HCV cirrhosis, may avoid the need for LT with timely and appropriate intervention (steroids, chelation therapy, or antiviral agents respectively). However, concurrent pursuit of transplantation for critically ill patients with decompensated liver disease where the response to therapeutics is uncertain is reasonable.
Options exist to reduce risks for patients with chronic liver disease and delay transplantation. Among them are means of reducing portal pressure, and the most commonly used is transjugular intrahepatic portosystemic shunting, which is frequently used to address portal hypertensive bleeding and ascites. Patients with difficult-to-control encephalopathy, elevated right-sided heart pressures, portal vein thrombosis, and elevated MELD scores are often not candidates for transjugular intrahepatic portosystemic shunting ; however, the ideal patient is one with a MELD score too low for transplantation but with significant risks from variceal bleeding or refractory ascites.
Improvements in treatment options for HCV infection and evidence that HCV eradication can result in reductions in fibrosis stage and improvements in clinical outcomes have the potential to dramatically impact the management of transplant candidates with HCV infection. The ability to significantly reduce posttransplant HCV infection recurrence with as little as 30 days of viral negativity in patients receiving antiviral therapy before LT is prompting the use of antiviral treatment in transplant candidates. Whether antiviral therapy is applicable when need for LT is otherwise felt unavoidable is less clear. There is a theoretical risk of reducing organ availability to a patient who has been cured of HCV infection and therefore is not accepting HCV-positive donor livers. There is also concern that mild improvements in liver function (and MELD score) after the curing of HCV infection in patients with advanced liver disease may result in patients being too well to be prioritized for transplantation but yet remaining with poor quality of life from their liver disease.
Liver transplantation is indicated for acute or chronic liver failure of any cause. Indications for LT can be categorized as acute liver failure, noncholestatic liver diseases, cholestatic liver diseases, metabolic disorders, vascular disorders, hepatic malignancy, and nonhepatic malignancy. The leading indications for LT in the United States and Europe are HCV infection, alcohol liver disease, and nonalcoholic steatohepatitis (NASH) ( Tables 50-8 and 50-9 ).
| Primary Liver Disease | Number | Percentage |
|---|---|---|
| Chronic hepatitis C | 26,371 | 23.5 |
| Alcoholic liver disease | 14,515 | 12.9 |
| Hepatocellular carcinoma | 13,578 | 12.1 |
| Cryptogenic cirrhosis | 8,912 | 7.9 |
| Primary sclerosing cholangitis | 6,870 | 6.1 |
| Acute liver failure | 6,579 | 5.9 |
| Hepatitis C and alcoholic liver disease | 5,821 | 5.2 |
| Primary biliary cholangitis | 5,610 | 5.0 |
| Autoimmune cirrhosis | 3,640 | 3.2 |
| Metabolic liver disease | 3,809 | 3.4 |
| Chronic hepatitis B | 3,487 | 3.1 |
| Nonalcoholic steatohepatitis | 3,531 | 3.1 |
| Chronic hepatitis C and B | 610 | 0.5 |
| Cholangiocarcinoma | 608 | 0.5 |
| Other diseases | 8,286 | 7.4 |
| Total | 112,227 | 100 |
| Primary Liver Disease | Number | Percentage |
|---|---|---|
| Biliary atresia | 5,699 | 40.0 |
| Acute liver failure | 1,694 | 11.9 |
| Metabolic liver disease | 1,671 | 11.7 |
| Hyperalimentation induced | 704 | 4.9 |
| Hepatoblastoma | 471 | 3.3 |
| Autoimmune cirrhosis | 350 | 2.5 |
| Primary sclerosing cholangitis | 285 | 2.0 |
| Secondary biliary cirrhosis | 237 | 1.7 |
| Cryptogenic cirrhosis | 179 | 1.3 |
| Other hepatic malignancy | 117 | 0.8 |
| Hepatocellular carcinoma | 93 | 0.7 |
| Chronic hepatitis C | 92 | 0.6 |
| Benign neoplasm | 48 | 0.3 |
| Other diseases | 2,597 | 18.2 |
| Total | 14,237 | 100 |
Acute liver failure has a dramatic presentation yet fortunately is a relatively uncommon indication for LT, accounting for only 6% of LTs in the United States and 9% in Europe annually. Because of the rapidly evolving clinical course, patients with acute liver failure require urgent referral to a transplant center for an expedited evaluation. Under most liver graft allocation systems, patients meeting the criteria for acute liver failure are given the highest priority for available liver grafts. In the case of drug- or toxin-induced liver injury, an emergent and thorough psychiatric and social assessment is required for suspected overdose or suicide attempt. This is best performed before the onset of advanced encephalopathy. Even in the absence of a self-inflicted injury, the rapid onset of illness in often previously healthy individuals makes it challenging for both patients and their family to fully grasp the enormity of the transplant evaluation, transplant surgery, and lifelong immunosuppression. Acute liver failure is a clinical entity distinct from chronic or acute-on-chronic liver failure, especially in the risk of cerebral edema and elevated intracranial pressure with resultant hypoxic injury from reduced cerebral perfusion pressure, seizures, and catastrophic herniation. With the exception of N -acetylcysteine therapy for acetaminophen overdose, prompt delivery in women with acute fatty liver of pregnancy, and silibinin therapy for Amanita phalloides poisoning, pretransplant management is predominantly supportive. Use of intracranial pressure monitors is controversial, with concern for complications and variability of utilization across U.S. transplant centers, and it has not been tied to improved outcomes. Management options for patients with clinical evidence of or measured elevated intracranial pressure include intravenously administered mannitol, hypertonic sodium chloride, hyperventilation, barbiturates, and hypothermia. Concerns regarding these strategies include transient effect and metabolic/hemodynamic side effects; nevertheless, with the exception of hyperventilation, their use is supported by the American Association for the Study of Liver Diseases in selected clinical scenarios.
There is a high burden of HCV disease in the United States and Europe, and HCV disease is the leading indication for liver transplant listing in the United States ; however, new infection rates peaked in the general U.S. population before 1990, and the rate of HCV disease as an indication for LT began dropping after 2004. Whereas the incidence of end-stage liver disease caused by HCV as an indication for LT declined between 1999 and 2006, that for HCC in patients with HCV infection increased over the same time period. Once decompensation occurs, the 5-year survival rate is less than 50% without transplantation. When HCV viremia is present at the time of transplantation, HCV infection of the liver graft is universal and has an accelerated natural history compared with that in nontransplant patients. By 5 years after LT, 10% to 30% of transplant recipients will develop recurrent cirrhosis. Graft and patient survival is reduced in HCV-infected recipients compared with non–HCV-infected patients.
New antiviral treatment promises to substantially impact the burden and clinical history of HCV infection. Achievement of cure in a great majority of treated patients and much improved tolerability over interferon-based regimens broaden the candidate population for new regimens in both the pretransplant and posttransplant populations. Recent projections for the U.S. population suggest hepatitis C could become a rare disease by 2036, predicated on effective population screening and adequate access to the antiviral regimens. In addition to the burden of unknown HCV infection in the population, near-term challenges include financial difficulties from the surge in demand for costly new HCV treatments.
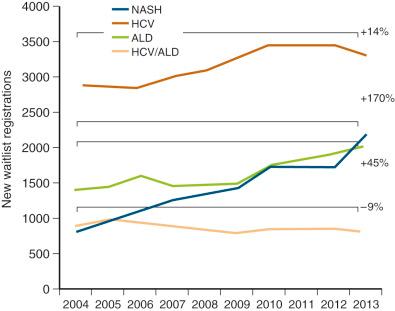
Excess alcohol consumption is very common in the United States, with nearly 5% of the population meeting the criteria for alcohol abuse. Alcoholic liver disease is one of the leading causes of cirrhosis in the United States, accounting for 40% of all deaths from cirrhosis and 28% of all deaths from liver disease, and is the second or third leading indication for LT, and the number of new registrants with alcoholic liver disease grew by 45% between 2004 and 2013. Traditionally, transplant programs required alcohol abstinence, often an arbitrary 6-month period, to allow stabilization of decompensated cirrhosis that may be able to delay or even avert the need for LT and to give the opportunity to address dependency. The aim is to minimize the risk of recidivist alcohol dependence and abuse that can have a negative effect on graft and patient survival ; however, there are insufficient data to support any fixed abstinence period for predicting future drinking after LT, and individual program requirements vary. Although the incidence of perioperative complications may be increased in patients with alcoholic liver disease undergoing transplantation, survival appears to be similar to that of posttransplant patients with nonalcoholic liver diseases.
Historically, patients with acute alcoholic hepatitis were excluded from transplantation. The debate over the acute alcoholic hepatitis population as transplant candidates has recently intensified because of the high mortality of patients with acute alcoholic hepatitis not responding to standard therapy and inability of these patients to survive abstinence periods of 6 months (standard in many programs) before transplantation. Alcoholic hepatitis superimposed on liver cirrhosis does not appear to adversely affect posttransplant outcome. Early transplantation for highly selected patients with acute alcoholic hepatitis not responding to medical therapy was associated with a nearly 50% improved survival rate on 2-year follow-up compared with those who were initially treated without transplantation in a landmark study from France. Only 10% (26 of 264) of patients with alcoholic hepatitis not responding to medical therapy were enrolled in that study. Yet in this highly selected cohort, the recidivism rate was 12% (3 of 26 patients), with none of the alcohol relapsers having graft dysfunction by the end of follow-up. Nevertheless, the low ratio of potential candidates to transplant recipients in that study underscores the high upfront demand likely required to successfully achieve transplantation in acute alcoholic hepatitis patients. The other concern for using deceased-donor liver grafts for patients with acute alcoholic hepatitis is its potential to negatively impact the public's decision to donate. A recent survey study suggests that hesitancy to donate organs to patients with alcoholic hepatitis was not as prevalent as the transplant community might fear, but was still present in roughly 26% of those surveyed.
The proportion of people with overweight and obesity in the U.S. general population is now greater than 30%; consequently, NASH is the leading cause of liver disease in the United States. As an indication for LT, NASH increased from 1.2% in 2001 to 9.7% in 2009, the third highest, behind HCV infection and alcoholic liver disease. It is now the second most common reason for waiting list registration. Given the association between metabolic syndrome and NASH, liver transplant candidates with NASH are older, have a higher body mass index (BMI), and have higher rates of diabetes and hypertension. Thus detection of cardiovascular comorbidities and attention to modifiable risk factors is essential in this cohort. Weight loss is encouraged, ideally with a supervised diet and exercise program. Bariatric surgery is associated with reduction of steatohepatitis and fibrosis but is typically limited in the transplant candidate population owing to prohibitive portal hypertension. Conceptually, morbidly obese individuals would be expected to have higher short-term and long-term mortality, with increased risk of recurrent NASH, cardiovascular disease, hypertension, and diabetes. Early studies suggested poorer short-term and long-term outcomes for recipients with BMI greater than 35 kg/m 2 to 40 kg/m 2 but were criticized for not considering nutrition and ascites in their analyses. More recent work suggests no short-term mortality increase. Results on long-term morbidity and mortality are variable. Concurrent bariatric surgery and LT has been studied in patients with BMI greater than 35 kg/m 2 who achieved weight loss before LT, and was promising in reducing posttransplant weight gain, steatosis in the liver graft, and diabetes, and in improving patient and graft survival compared with those who underwent LT alone. A threshold BMI for perioperative and long-term safety of LT is not clear, should it exist at all. Although the American Association for the Study of Liver Diseases guidelines consider morbid obesity a relative contraindication to transplantation, practice differs from center to center. Regardless, attention to healthy diet and exercise both before and after transplantation is likely to improve outcomes.
Worldwide, chronic HBV carriers number roughly 350 million. Although the incidence of HBV infection in the United States is low at 0.4%, it is an important cause of acute and chronic liver disease, particularly in adult Asian immigrants, where prevalence estimates are as high as 25%. Acute infection, flare of chronic infection, complications of cirrhosis, or the development of HCC in HBV-infected patients can precipitate the need for LT. HBV infection accounts for a relatively small proportion of the U.S. liver transplant waiting list. The rate of HBV infection as an indication for LT in the United States remained steady overall at 3% to 4% from 1998 to 2007 ; however, whereas the rate of HBV-induced end-stage liver disease as an indication for transplantation declined between 1999 and 2006, that of HBV-related HCC increased over that period. Chronic carriers whose providers fail to recognize HBV infection before initiation of immunosuppressive or cytotoxic chemotherapy are at risk of devastating reactivation of HBV infection and are a source of avoidable need for LT or death ; following guidelines for HBV prophylaxis in this setting is advised. Candidates with HBV infection die at a comparable rate as patients with other chronic liver diseases. Nucleoside/nucleotide antiviral therapy is usually well tolerated in advanced liver disease. When taken for several months, nucleoside/nucleotide therapy may stabilize decompensated cirrhosis and occasionally avert the need for LT. The combination of hepatitis B immunoglobulin and HBV antiviral agents significantly reduces the recurrence of HBV infection after transplantation. Long-term outcomes after transplantation for HBV infection are excellent.
Liver transplantation is the only effective therapy for end-stage liver disease resulting from primary biliary cholangitis (PBC) and primary sclerosing cholangitis (PSC), and has excellent graft survival rates. Ursodeoxycholic acid is indicated for patients with PBC, improves outcomes, and its use is associated with reduction in need of transplantation for PBC patients. In addition to decompensated cirrhosis, LT may also be indicated in patients with PBC with compensated cirrhosis and severe pruritus refractory to medical therapy. Patients with PSC can develop typical complications of decompensated cirrhosis as an indication for LT, but also are at risk of recurrent cholangitis despite biliary interventions, and frequent episodes of cholangitis (with or without concurrent cirrhosis) can be an indication for transplantation, with the possibility of increased priority by regional MELD exception application. Similarly, PSC patients are at risk of developing cholangiocarcinoma. Whereas performing a transplant in patients with cholangiocarcinoma overall is associated with poor outcomes, careful selection including only patients with localized hilar tumors smaller than 3 cm who undergo protocolized pretransplant neoadjuvant radiation therapy and chemotherapy results in a reasonable 5-year recurrence-free posttransplant survival rate estimated at 65% in a multicenter study. These patients are eligible for exception priority upgrade provided they meet specified criteria, including negative findings on pretransplant staging laparotomy (see Table 50-4 ). In the absence of cholangiocarcinoma, the 5-year patient survival rate post-LT is excellent, approaching 90%. Roux-en-Y choledochojejunostomy with removal of the recipient's distal bile duct is usually used to reduce risk of recurrent PSC.
Biliary atresia is the most common indication for LT in children, accounting for more than 30% of all pediatric transplants, and is universally fatal if untreated. The typical treatment modality is hepatoportoenterostomy, and LT is indicated for those children who continue to have complications of chronic liver disease and/or recurrent cholangitis. The 10-year survival rate after LT for biliary atresia is 86% in the United States. Other pediatric cholestatic indications for transplantation include PSC, Alagille syndrome, nonsyndromic intrahepatic paucity of bile ducts, cystic fibrosis, and progressive familial intrahepatic cholestasis.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here