Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Chronic childhood arthritis affecting five joints or more during the first 6 months of disease is defined as polyarthritis . , The International League of Associations for Rheumatology (ILAR) classification system for juvenile idiopathic arthritis (JIA) further categorizes polyarthritis as rheumatoid factor (RF)-negative if tests for RF are negative, and RF-positive if RF is detected on two occasions at least 3 months apart ( Table 18.1 ). RF-negative and RF-positive polyarticular JIA subsets have distinguishing clinical features, immunogenetic profiles, and disease courses and outcomes; therefore in this chapter RF-negative and RF-positive polyarticular JIA subsets are considered separately, as distinct clinical entities.
| RF-Negative Polyarthritis |
| Arthritis affecting five or more joints during the first 6 months of disease; a test for RF is negative Exclusions:
|
| RF-Positive Polyarthritis |
| Arthritis affecting five or more joints during the first 6 months of disease; two or more tests for RF at least 3 months apart during the first 6 months of disease are positive. Exclusions:
|
Advancing knowledge about chronic childhood arthritides has prompted proposals for modified classification systems that ignore the number of joints as a categorization criterion. Instead, clinical phenotypes, including distribution patterns of involved joints, would direct classification. , However, as a new classification system has not yet been evaluated, this chapter will adhere to the ILAR-defined JIA taxonomy.
The polyarticular category accounts for approximately 15% to 20% of JIA patients and of these approximately 85% have negative tests for RF, , although RF frequencies vary with ethnicity.
Incidence and prevalence data vary because of differences in diagnostic and classification criteria applied, inconsistency in case ascertainment methods, accessibility to care, and genetic and ethnic characteristics of the respective populations. , Global incidence rates of chronic childhood arthritis range from 0.8 to 23 per 100,000 children. The incidence rate of RF-negative polyarticular disease has been reported as ranging from 0.3 to 6.5 per 100,000. The prevalence of RF-negative polyarticular JIA varies from 1.64 to 33.4 per 100,000. ,
RF-negative polyarticular JIA can begin at any age before 16 years, but onset age displays a biphasic trend with a peak at age 1 to 3 years and another between ages 9 and 14 years. , , RF-negative polyarthritis affects girls four times more frequently than boys. The predominance of females is greater in those with onset during adolescence (female-to-male ratio 10:1) compared with those with a younger onset age (female-to-male ratio 3:1). , Younger-onset RF-negative polyarthritis patients are more likely to be antinuclear antibody (ANA) positive and are at greater risk for developing chronic iridocyclitis, much like oligoarticular JIA. A young onset age of RF-negative polyarthritis is associated with a less favorable long-term outcome. Greenwald et al. found 2-year outcomes in an older onset, RF-negative JIA subgroup to be generally favorable even before the era of biological therapies.
JIA occurs worldwide, but prevalence varies widely among geographical regions. , RF-negative polyarticular JIA is the second most prevalent category of JIA (after oligoarticular JIA) in all regions of the world except in Southeast Asia where systemic JIA and enthesitis-related JIA are most frequently reported. Oen and Cheang reported that polyarthritis accounted for a higher proportion of East Indian (61%) and North American Indian (64%) children with chronic arthritis compared with Caucasian children (27%). Saurenmann et al. analyzed ethnicity as a risk factor for JIA in a multiethnic cohort; among 223 children with RF-negative polyarthritis, no significant differences among European and non-European patients were found. However, there are some reports that at least some North American Indigenous populations had a high relative risk (3.2) of developing RF-negative polyarthritis.
The etiologies of the respective JIA subtypes are unknown but are believed to have complex multifactorial origins.
Environmental and lifestyle influences are proposed as factors promoting arthritis in the context of genetic vulnerability, but the precise underlying immunopathogenic mechanisms in JIA have not been elucidated. In RF-negative polyarticular JIA, as well as in oligoarticular JIA, abnormalities of adaptive immunity mediated by CD4 T lymphocyte responses have been described. Immunity to cartilage-derived self-antigens, such as aggrecan, fibrillin, and matrix metalloproteinase 3, are associated with polyarticular JIA. An imbalance between autoreactive T helper (Th)1/Th2 and T regulatory cells results in T-cell proliferation, a failure of T-cell tolerance to self-antigens, an increase in production of the proinflammatory cytokines like interferon-γ from Th1 cells and interleukin (IL)-17 from Th17 cells, and a decrease in the antiinflammatory cytokine IL-10. Increased levels of IL-17 in seronegative polyarthritis are considered to be of potential pathogenic importance by promoting other proinflammatory cytokines and enhancing matrix metalloproteinases production leading to cartilage degradation.
De Jager et al. noted increased but comparable plasma levels of IL-6 and IL-12, and chemokines C-C motif ligand (CCL3), C-X-C motif ligand (CXCL)9, and CXCL10 in a small group of children with RF-negative polyarthritis (10 patients) and oligoarthritis with a polyarticular course (five patients). , The role of IL-6 in promoting inflammation in polyarticular and oligoarticular JIA contrasts with its limited expression in psoriatic and enthesitis-related JIA. CCL20, derived from synovial fluid mononuclear cells, is increased in children with polyarthritis (including those with extended oligoarticular JIA); the enhanced production is attributed to hypoxia in the inflamed synovial environment. The hypoxic synovial environment also is a factor that promotes increases in intraarticular vascular endothelial growth factor and osteopontin, both of which enhance angiogenesis in synovial tissue in children with RF-negative polyarthritis and extended oligoarthritis JIA subsets.
An integral role for neutrophils in the pathogenesis of polyarticular JIA is supported by evidence that neutrophils are activated both in active and inactive disease and that these functional changes are a result of abnormal gene transcription and expression linked to fundamental metabolic aberrations.
Intestinal microbiota in patients with autoimmune diseases in general, and with JIA in particular, show deviations from healthy comparison populations. However, there are no JIA category-specific microbiome characteristics identified except in enthesitis-related JIA. Further studies are required to determine whether alterations of gut microbiota in JIA are pathogenically important.
Genetic influence in the pathogenesis of JIA is indicated by ethnic variability in the incidence of certain JIA subsets, female preponderance, increased sibling recurrence rates, and associations with both human leukocyte antigen (HLA) and non-HLA genes.
Class I alleles HLA A2 and class II alleles DRB1∗08 and DPB1∗02 confer susceptibility in RF-negative polyarthritis. , HLA-DRB1∗11:03/11:04 confers susceptibility to both oligoarticular JIA and younger-onset polyarticular JIA but not to older-onset polyarticular RF-negative JIA. HLA-DRB1∗08:01 confers an increased risk in both younger and older-onset RF-negative polyarticular JIA. DPB1∗02:01 in the absence of the predisposing alleles DRB1∗08:01 and 11:03/11:04 is associated with younger onset age in polyarticular RF-negative JIA and both persistent and extended oligoarticular JIA. , The shared HLA associations in RF-negative polyarthritis and oligoarticular JIA suggest that predisposing genetic characteristics are shared between these two JIA categories. , In contrast, comparable overlap between RF-negative and RF-positive polyarticular JIA is not identified.
Beyond the HLA region, non-HLA alleles are associated with polyarticular RF-negative JIA. PTPN22 encodes lymphoid protein tyrosine kinase, an enzyme which negatively regulates T cells. Mutation in this gene is associated with T-cell activation and the subsequent promotion of autoimmune disease. The association between PTPN22 and polyarticular RF-negative JIA, as well as oligoarticular JIA, has been validated in multiple studies. In a cohort comprising 2816 oligoarticular and RF-polyarticular JIA patients, a total of 17 loci reaching the genome-wide level of significance were identified. Of these, six ( C5orf56-IRF1 [rs4705862], ERAP2-LNPEP [rs27290], PRR5L [rs4755450], RUNX1 [rs9979383], RUNX3 [rs4648881], and UBE2L3 [rs2266959]) were replicated in a genotyping study using the Australian Childhood Arthritis Risk Factor Identification Study JIA case-control sample.
The TRAF1/C5 region on chromosome 9 encodes the tumor necrosis factor (TNF)-receptor-associated factor 1 and the complement component 5; an association of a mutation in the A allele encoding the TRAF1/C5 region is noted in RF-negative phenotype. Moreover, an IL-10 single nucleotide polymorphism (-1082 AA) confers susceptibility to polyarticular JIA.
LACC1/FAMIN gene mutations, which result in alterations of macrophage function and promote inflammation, have been identified in a variety of JIA subtypes including RF-negative polyarthritis.
Continued mining of JIA genome-wide scans is expected to expose additional susceptibility genes allowing for more precise biologically based categorization of JIA, providing insight into mechanisms of disease and informing new treatment strategies for individual patients. ,
Maternal smoking and infection during pregnancy have been invoked as risk factors for the development of polyarticular JIA in the offspring. , A role of stressful life events before the onset of RF-negative polyarticular JIA has been highlighted. A proposed protective role for breast-feeding has not been well established in polyarticular JIA. ,
RF-negative polyarthritis is a heterogenous JIA category having at least three distinct phenotypes. One phenotype, which is characterized by a later onset age, symmetrical polyarthritis, elevated acute phase reactants, negative tests for ANAs, and variable outcomes, is similar to adult-onset RF-negative rheumatoid arthritis. A second subtype is similar to extended oligoarticular JIA; it is characterized by a young onset age, asymmetrical polyarthritis, normal to mild increases in acute phase reactants, ANA positivity, and an increased risk of chronic anterior uveitis. Patients in a third subset of RF-negative children have “dry synovitis,” a condition characterized by polyarthropathy, minimal or absent clinical signs of joint effusion or synovial hypertrophy, joint stiffness, limited range of motion, joint contractures, and often normal laboratory indicators of inflammation. This subtype of polyarthritis, which is often poorly responsive to treatment, can be associated with substantial joint damage. Dry synovitis has been considered a variant of RF-negative polyarthritis, although it also has been suggested to be a forme fruste of scleroderma. This variant of RF-negative polyarthritis can be difficult to diagnose as clinical features can mimic genetic and metabolic diseases such as Farber disease and mucopolysaccharidosis.
The onset of arthritis may be acute, but it is more often insidious, with progressive accumulation of additional affected joints. Morning stiffness, indicative of active arthritis, may persist for hours or occasionally all day. Arthritis may be remittent or indolent. Joint swelling is a result of synovial hypertrophy with or without intraarticular fluid. Joints may be tender and slightlly warm but are not red. Hot, red, and very tender joints suggest infection or malignancy rather than JIA. Stress pain is often identified in active joints. Among children with RF-negative polyarthritis, knees, wrists, and ankles are most commonly affected. Small joint involvement of the hands and feet may occur early or late in the course of the disease; distal interphalangeal joints are seldom affected at onset ( Fig. 18.1 ).
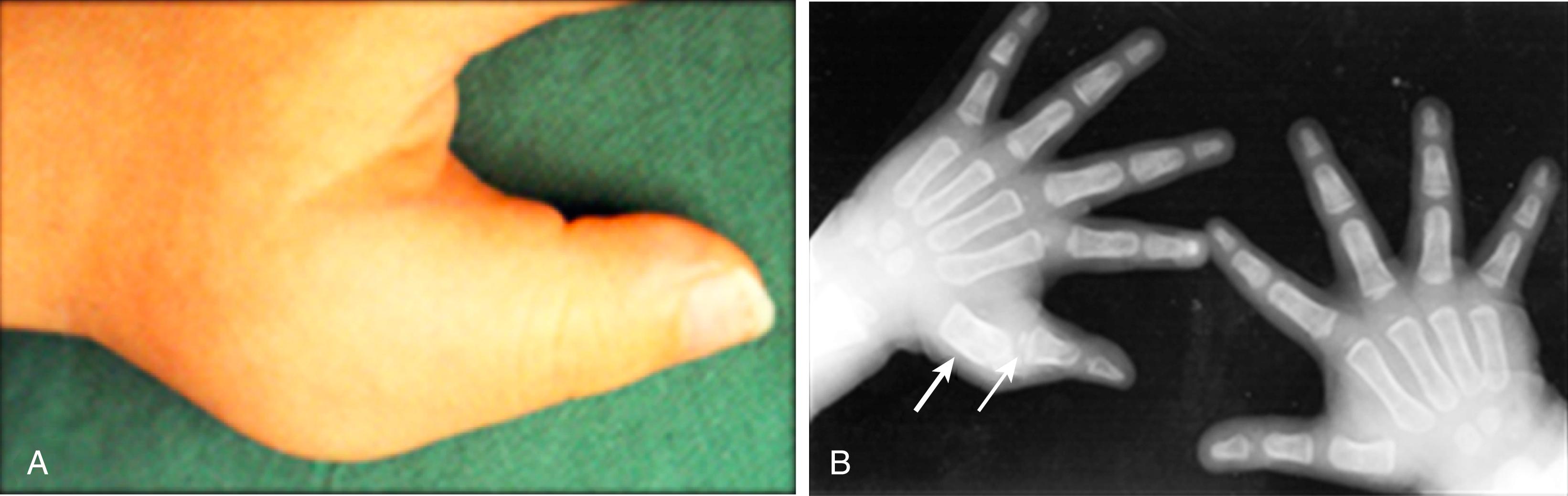
In RF-negative polyarthritis, the number of affected joints tends to be less and the pattern of involvement more asymmetrical than in RF-positive polyarthritis ( Fig. 18.1 ). In RF-negative disease, involvement of wrists and small joints of the hands is less frequent than in those with RF. Clinical signs of hip involvement are present in fewer than 20% with RF-negative polyarthritis at first presentation, but progressive hip abnormalities become evident in the longer term. Oen et al. reported that radiological signs of hip joint involvement during childhood were more likely to occur in RF-negative than RF-positive polyarthritis; the tendency for RF-negative polyarthritis to have its onset at a younger age compared with RF-positive disease might be a factor contributing to the higher frequency of radiographic abnormalities identified during childhood in the seronegative group. Wrist and ankle involvement at first presentation predicts progression to polyarthritis in those first presenting with oligoarthritis.
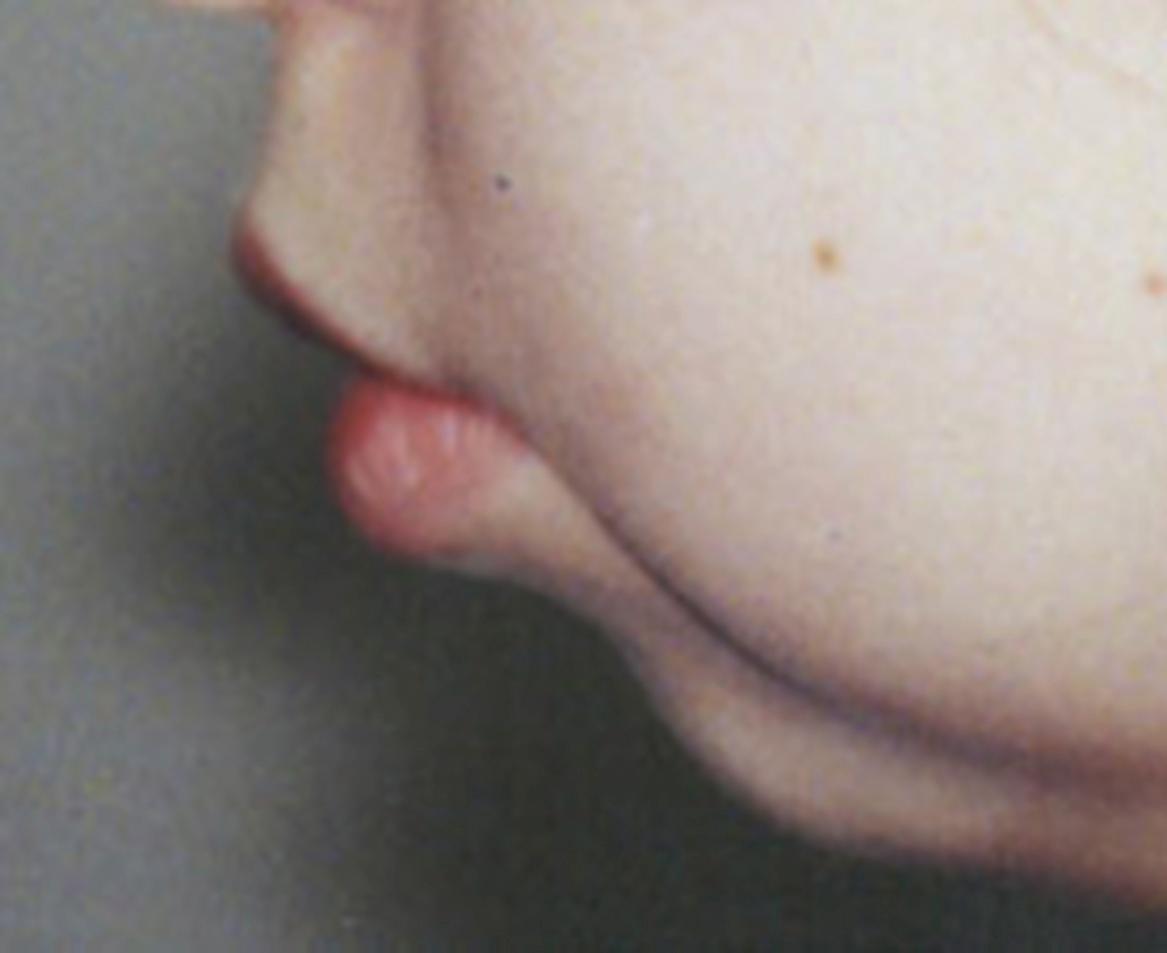
The temporomandibular joint (TMJ) is commonly affected in children with a polyarticular disease course regardless of onset subtype. However, those with RF-negative polyarthritis are more likely to have TMJ involvement, particularly at long-term follow-up, than those who are RF-positive. Growth disturbances of the TMJs are more likely to occur when inflammation starts before the age of 8 years. The earlier age of onset, when the TMJ might be more vulnerable to damage, is thought to be a reason for the greater prevalence of involvement in the RF-negative group. , TMJ arthritis can be present even in the absence of clinical symptoms or signs. As clinical examination has poor sensitivity and specificity to detect TMJ inflammation, delay in diagnosis or overdiagnosis of TMJ arthritis can occur. , Advances in TMJ imaging, including computed tomography and magnetic resonance imaging (MRI), support the view that TMJ arthritis is more common, and the severity more variable, than can be discerned by clinical assessment alone. In experienced hands, ultrasonography can be a suitable method for detecting and monitoring TMJ arthritis, but, as reflected in a meta-analysis of ultrasound as a method to detect TMJ arthritis, further standardization and validation of ultrasonography for assessing TMJs is required. TMJ MRI with contrast remains the gold standard for detection of active inflammation. , Joint damage associated with TMJ arthritis can result in altered physiognomy, including micrognathia, retrognathia, malocclusion, and facial asymmetry ( Fig. 18.2 ). , ,
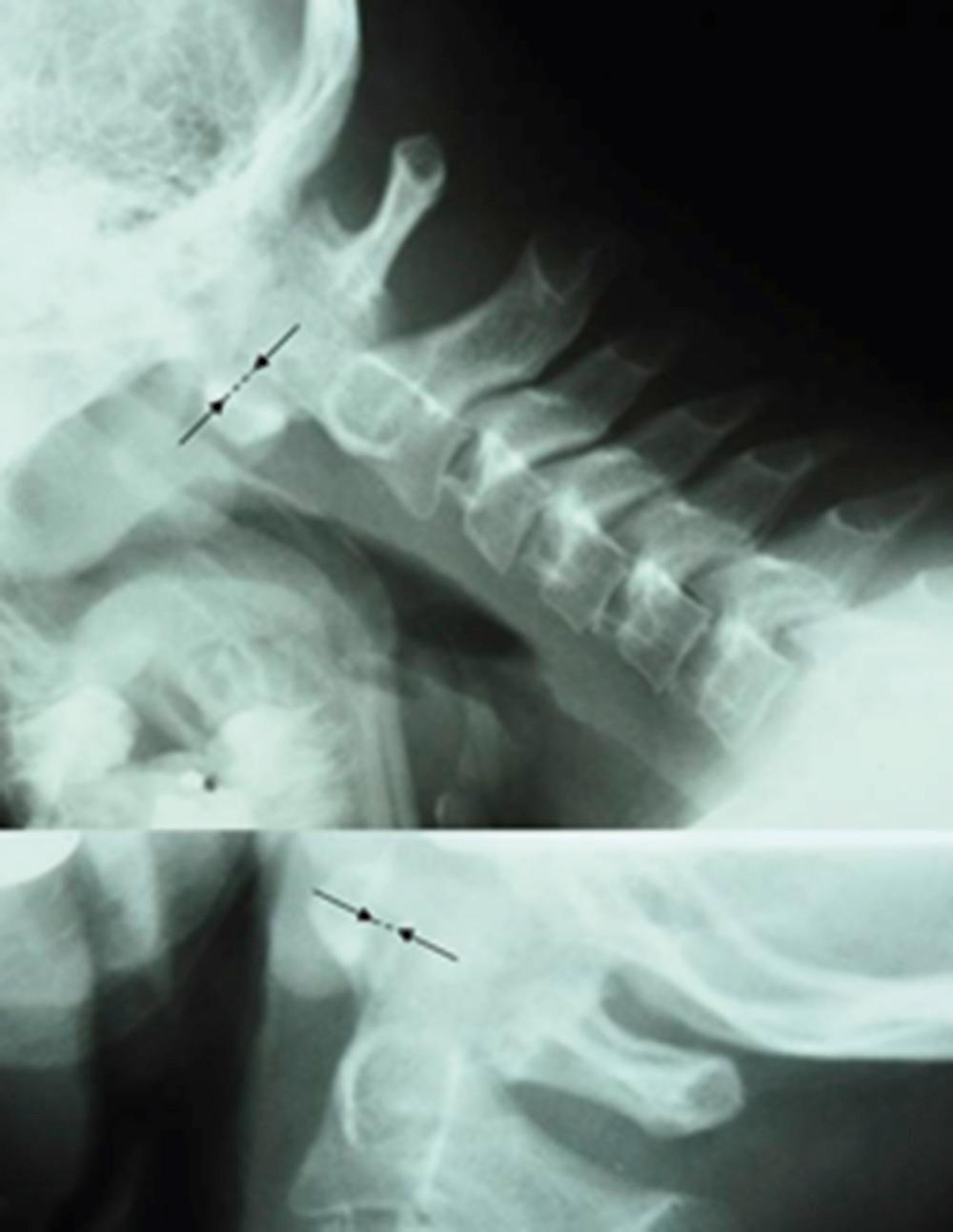
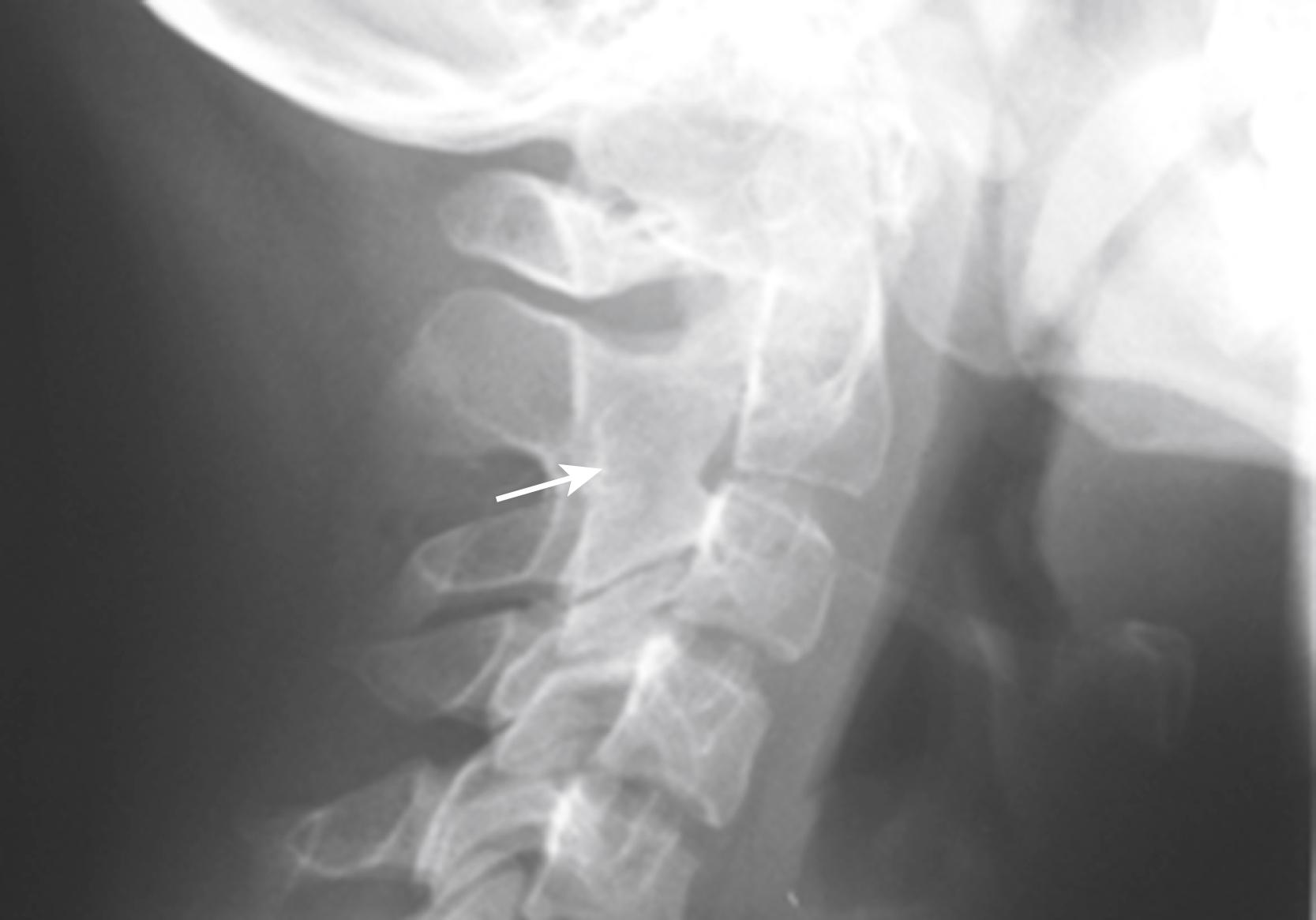
Cervical spine involvement is not commonly recognized early in the course of RF-negative polyarthritis, either clinically or by conventional radiography, but with longer-term follow-up, a decreased range of motion can ensue. , More sensitive imaging modalities, such as contrast-enhanced MRI, reveal that cervical spine involvement is more common than is generally appreciated even in asymptomatic patients. Atlantoaxial subluxation ( Fig. 18.3 ), erosion of the odontoid process, and vertebral ankylosis can occur ( Fig. 18.4 ).
A palindromic course in polyarticular JIA, characterized by episodic arthritis, is not common in children; only 10% of children with a palindromic pattern at onset will progress to persistent, chronic JIA. Some patients can have an oligoarticular onset that progresses to polyarthritis; patients with symmetrical arthritis, wrist and ankle involvement, and elevated erythrocyte sedimentation rate (ESR) are most likely to progress to polyarticular involvement. The term polygoJIA has been proposed as a category to include children with RF-negative arthritis regardless of the number of involved joints, thus incorporating RF-negative polyarthritis and oligoarthritis into one category.
Systemic manifestations in seronegative polyarticular JIA are unusual but can include fatigue and growth failure. Fever seldom occurs and when present is low grade.
Fatigue in children with polyarthritis can be present even in the absence of active joint disease. Ringold et al. studied fatigue in 60 children with polyarthritis, of whom 24 (61.2%) were RF-negative, using a survey tool that includes assessment of general fatigue, sleep/rest fatigue, and cognitive fatigue domains. Both JIA children and their parent/proxies reported less favorable scores in all domains compared with controls. Fatigue in JIA is associated with poor functional ability. Factors contributing to fatigue can include pain and stress, decreased muscle mass, low aerobic and anaerobic capacity, and anemia. , Sleep disturbance is not a common factor contributing to fatigue in JIA.
Growth disturbances are common in JIA. In children with polyarthritis, height-for-age Z scores (reflecting the number of standard deviations from the normal mean) may decline in the first several years of disease but tend to return to normal. In those children without RF, the negative deviation is negligible compared with those with RF, particularly with longer disease duration. , Low growth velocity tends to correlate with disease severity and activity, and with the number of involved joints. , ,
Next to oligoarticular JIA (which accounts for more than half of JIA patients affected by uveitis), chronic asymptomatic anterior uveitis is most common in the RF-negative polyarthritis group. , Approximately 15% to 20% of children with RF-negative polyarthritis have uveitis and account for approximately 20% of all JIA uveitis patients. , ,
Sabri et al. reported that 32 of 142 JIA patients (22.5%) with uveitis had RF-negative polyarthritis; none with RF-positive JIA had uveitis. Similarly, Lee et al. found, as with the oligoarthritis JIA subgroup, uveitis in RF-negative polyarthritis tends to be associated with younger onset age and ANA positivity. Greenwald et al. reported that only 2% of patients with older-onset RF-negative polyarticular JIA had uveitis despite 47% being ANA positive. In an inception cohort of newly diagnosed patients, 18% of RF-negative polyarticular JIA patients had uveitis before enrollment, 25% within the first year, and 45% by the end of the second year.
RF-negative polyarthritis is not typically associated with overt cardiovascular pathology. Bharti et al. reported that children with arthritis, regardless of onset subtype, had significantly greater left ventricular volumes and other abnormalities suggesting abnormal left ventricular diastolic relaxation. Knook et al. demonstrated lower 1-second forced vital capacity and peak expiratory flows in a group of 31 children with chronic arthritis, more than two-thirds of whom had RF-negative polyarthritis. These abnormalities were attributed to impaired respiratory muscle strength rather than intrinsic restrictive or obstructive lung disease.
The differential diagnosis for a child with polyarthritis includes other rheumatic diseases, infections, malignancies, other inflammatory conditions, and metabolic and genetic disorders.
The onset of polyarthritis in a girl later in childhood or during adolescence could suggest the possible diagnosis of systemic lupus erythematosus (SLE). The arthritis of SLE may mimic that of JIA, although it is nonerosive and less likely to be deforming; the presence of other clinical hallmarks and a positive test for anti–double-stranded DNA (anti-dsDNA) antibodies establishes the diagnosis of SLE. Ragsdale et al. described nine children (six of whom were RF-negative) who developed SLE years after an initial diagnosis of polyarticular juvenile arthritis.
The differential diagnosis of RF-negative polyarthritis also includes enthesitis-related JIA. Predominant involvement of large joints of the lower extremities and the presence of enthesitis supports the diagnosis of enthesitis-related JIA, although enthesitis can occur, albeit uncommonly, in other types of JIA. RF-negative polyarticular JIA might be the initial diagnosis when the articular manifestations of psoriatic arthritis appear before skin rash develops.
Scleroderma begins insidiously with joint contractures of the small joints of the hands mimicking features of polyarthritis but ordinarily without associated signs of intraarticular swelling. Children with dermatomyositis may have polyarthritis but can be distinguished from those with JIA by clinical manifestations such as rash and muscle weakness.
Sarcoidosis, Blau syndrome, Sjӧgren syndrome, , and mixed connective tissue disease are additional differential diagnostic considerations for the child presenting with polyarthritis. The early phases of these respective conditions can mimic polyarticular JIA before discriminating clinical, laboratory, and pathological features become evident.
Septic arthritis affecting multiple joints is unusual; only 3% of the 65 children with septic arthritis reported by Al Saadi et al. had more than one involved joint. Lyme disease, typically manifesting as one swollen knee, may be polyarticular but it can usually be differentiated from RF-negative polyarthritis by its intermittent pattern of arthritis activity and accompanying extraarticular abnormalities. Arthritis caused by Neisseria gonorrhoeae may have an early migratory polyarticular phase.
Reactive polyarthritis in response to infection in the respiratory, gastrointestinal, or genitourinary tracts ordinarily can be distinguished from polyarticular JIA by a limited duration of the disease and associated manifestations of the inciting infection. Acute rheumatic fever after group A beta-hemolytic streptococcal pharyngitis is characterized by acute, painful, nonerosive, and migratory polyarthritis, although atypical presentations in which monoarthritis or nonmigratory arthritis of large and small joints can occur. , Poncet disease (reactive polyarthritis with tuberculosis) can sometimes mimic seronegative polyarticular JIA.
Malignant infiltration of bone or synovium can mimic polyarthritis, although in most instances the malignant focus is in juxta-articular bone rather than in the joint. However, joint swelling can occur in lymphoblastic leukemia as a result of leukemic infiltration into the synovium. Joint involvement in malignancy tends to be oligoarticular rather than polyarticular and associated with systemic and laboratory manifestations of malignancy.
Arthritis associated with inflammatory bowel disease or sarcoidosis should be considered in the differential diagnosis of RF-negative polyarthritis. Sickle cell disease causes diffuse, symmetrical swelling of the hands and feet that may mimic true arthritis. Hypermobility syndromes, mucopolysaccharidosis, familial hypertrophic synovitis, , , , familial arthritis and camptodactyly, familial osteochondritis dissecans, Stickler syndrome, velocardiofacial syndrome, Turner syndrome, relapsing polychondritis, idiopathic multicentric osteolysis, Farber disease, familial hypertrophic synovitis, pseudorheumatoid dysplasia, and pachydermodactyly are rare causes of disease that may suggest polyarticular JIA. Scurvy should be considered in the differential diagnosis of polyarthropathy in children at risk for nutritional deficiencies, including those with autism and other developmental disorders.
Laboratory tests can provide evidence of inflammation, are useful in excluding other diagnoses, and are important in classification, prognostication, and guiding therapy.
Children with polyarthritis typically have moderate elevations of the ESR and C-reactive protein (CRP) and may have elevated white blood cell and platelet counts. Normocytic, hypochromic anemia indicative of chronic inflammatory disease can be seen.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here