Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
We thank the previous authors, Yi-Yung Chen and Thomas Jansson, for their foundational work and expertise in writing the previous version of this chapter, many of the sections and figures which have been preserved in this revision.
Intrauterine growth restriction (IUGR) remains one of the leading causes of perinatal and neonatal morbidity and mortality worldwide, with significant implications for the short- and long-term health and development of children. IUGR remains an important public health concern in both developing and well-resourced countries. Recent estimates are that IUGR affects 3% to 7% of all newborn infants, with apparently higher incidence during the last decade. IUGR is typically defined as the failure of the fetus to achieve its genetically determined growth potential. Clinically, this individual genetic growth potential is largely unknown and the diagnosis of IUGR relies on proxies, including the estimation of fetal weight in utero and/or measurements of birth weight. The rate and potential for normal growth during pregnancy is also based on the race and gender of the fetus. Small-for-gestational age (SGA) is typically defined as fetal weight or birth weight less than the 10th percentile on standardized weight-for-gestational age growth curves. , However, only about one-third of infants born at less than 10th percentile have an identified or apparent etiology for pathologic growth restriction, and otherwise healthy, constitutionally small birth weight-for-gestational age infants are usually considered to be at low risk for adverse perinatal or long-term outcomes.
Early, antenatal identification of IUGR is typically achieved with serial assessment of intrauterine growth, using fetal biometric measurements obtained by ultrasound, such as head-to-abdominal ratio and head-to-femur ratio, estimated fetal weight, growth velocity, and abdominal circumference. Historically, abdominal circumference less than 10th percentile was considered the most sensitive biometry marker for diagnosis of IUGR with varying thresholds (<3rd or <5th percentiles) to diagnose IUGR and predict fetal compromise in utero. More recently developed growth curves suggest that fetal biometry alone is not sufficient to discriminate constitutionally small fetuses from those at risk for adverse perinatal outcomes. Doppler ultrasound imaging has improved diagnostic accuracy, as pathologic IUGR is often associated with Doppler abnormalities in the uteroplacental and umbilical circulation, whereas constitutionally small fetuses often exhibit normal fetal Doppler measurements. Repeated fetal growth estimates from ultrasound imaging showing a deviation from previously established intrauterine growth curves and findings of abnormal blood flow patterns in the fetal circulation, such as increased resistance in the umbilical artery, are indicative of IUGR. , Amniotic fluid volume, which is believed to reflect placental and fetal renal functions, provides additional information. Low amniotic fluid volume with intact membranes is commonly associated with placental insufficiency.
Two main patterns of IUGR are observed based upon fetal and neonatal assessment. Symmetric IUGR is characterized by a proportional lack of growth resulting in small head and abdominal size. The inciting cause is thought to begin early in pregnancy (first or second trimester), with reduced fetal cellular proliferation of all organs. Congenital infections and chromosomal abnormalities are typically associated with symmetric IUGR. In contrast, asymmetric IUGR is seen more commonly (70% to 80%) and is characterized by disproportionately larger head size relative to expected abdominal circumference, and is typically associated with failure of nutritional or oxygen supply and/or blood flow in the second half of pregnancy (i.e., placental or maternal vascular factors). In asymmetric IUGR, the growth of the fetal head is relatively spared due to redistribution of cardiac output to preferentially support cerebral blood flow. Changes in placental function have classically been associated with asymmetric IUGR, but with more recent understanding of the role of the placenta in earlier fetal growth and programming, the distinction between symmetric and asymmetric IUGR is not always clear and there is significant overlap between the two groups.
IUGR is associated with increased perinatal morbidity and mortality , and remains one of the most important risk factors for unexplained intrauterine demise. As the causes of IUGR and stillbirth usually overlap, the risk for stillbirth is directly proportional to the severity of the fetal growth restriction. The risk for stillbirth is 1.5% for fetal weights less than 10th percentile and increases to 2.5% if the fetal weight is below the 5th percentile for gestational age. IUGR is also associated with preterm birth, perinatal asphyxia, intraventricular hemorrhage, and infections. , In the neonatal period, the growth-restricted infant is susceptible to abnormal thermoregulation, hypoglycemia, and other metabolic problems believed to be related to limited glycogen and fat stores.
The adverse consequences of IUGR are not limited to the fetal and neonatal period but can also influence lifelong health by developmental programming. This concept is centered around research of the fetal origins hypothesis , which proposes that exposures during gestation have significant impact on the individual’s functional capacity, metabolic competence, and responses to the later environment, likely due to epigenetic programming of the fetal phenotype. These concepts have extended to include neurodevelopmental outcomes and cardiopulmonary health. For example, an altered intrauterine environment, accompanied by changes in nutrient availability, is associated with increased risk for chronic diseases, including coronary artery disease, hypertension, and type II diabetes later in life. , The mechanisms underlying developmental programming remain to be established but may involve epigenetic regulation of key genes or permanent changes in organ structure.
Fetal growth and development are the result of complex interactions between the genetic growth potential of the fetus, the maternal environment, and placental function. Primary conditions affecting the mother, fetus, or placenta may interfere with fetal growth and result in IUGR. Common origins of IUGR are therefore typically categorized into three groups: maternal, fetal, and placental causes ( Fig. 14.1 ).
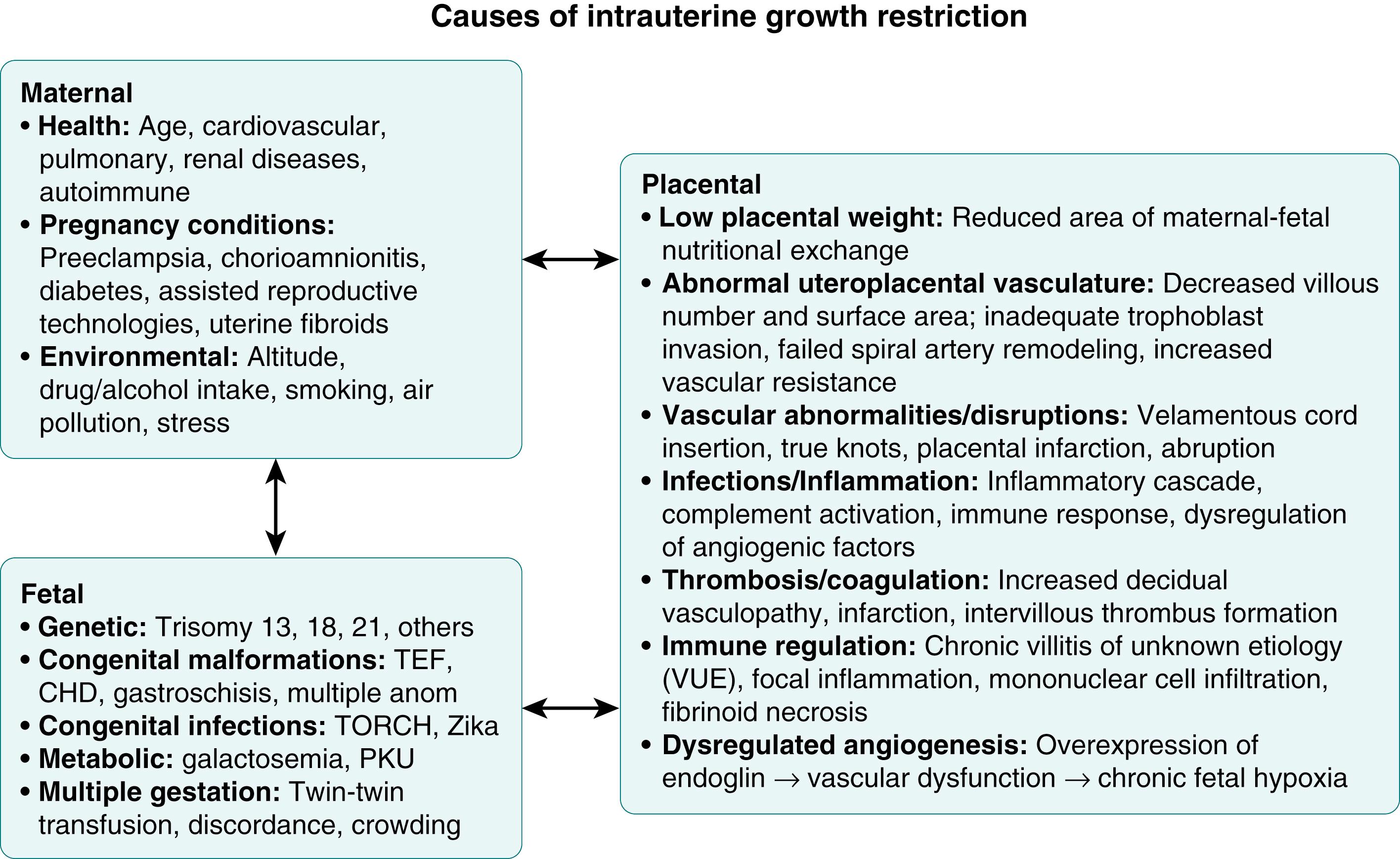
Certain baseline aspects of prepregnancy maternal health and conditions during pregnancy are known to impact fetal growth. Maternal extremes of age (<16 and >35 years), race/ethnicity, and low socioeconomic status are epidemiologic associations that appear to play a biologic role in IUGR. , Maternal undernutrition is a major public health concern worldwide and constitutes the most common cause of IUGR in developing countries and remains a significant problem in the United States. Maternal residence at high altitude during pregnancy is associated with IUGR due to exposure to chronic fetal hypoxia. Maternal vascular diseases, such as gestational hypertension, preeclampsia, and chronic hypertension with or without superimposed preeclampsia, are common maternal causes of IUGR. , Smoking during pregnancy, an example of a modifiable risk factor, increases the risk for growth restriction 3.5-fold. Other maternal conditions include autoimmune disorders, renal disease, exposure to teratogens, and substance abuse. Maternal endocrine disorders and internal/extrinsic factors that alter homeostatic mechanisms (e.g., psychosocial stress) have been shown in animal models to impact fetal growth through deviations in maternal cortisol, placental glucocorticoid, and epigenetic processes. ,
A wide range of fetal chromosomal and genetic abnormalities are associated with IUGR, with autosomal trisomies 13, 18, and 21 being the most common chromosomal disorders. In addition, fetuses with unbalanced chromosomal translocation or deletion are at increased risk. Even in the absence of a chromosomal or identified genetic disorder, structural congenital abnormalities are associated with IUGR. , In a population-based study conducted by the Centers for Disease Control and Prevention, more than 20% of infants with structural malformations had IUGR, corresponding to a 2.6-fold higher risk for growth restriction as compared with infants without malformations. Intrauterine infections, in particular rubella, cytomegalovirus, varicella-zoster, toxoplasma, and malaria have been estimated to cause 5% to 10% of all cases of IUGR.
Changes in placental function, which are often referred to as placental dysfunction or placental insufficiency, are believed to cause or directly contribute to most cases of asymmetric IUGR; however, percentage estimates are difficult to measure epidemiologically due to complex interaction with maternal and fetal causes (see Fig. 14.1 ). Defective placentation that results in placental dysfunction is a fundamental pathophysiologic mechanism of IUGR. , Abnormalities in placental growth and development following early placentation also contribute to IUGR, and these defects may be secondary to maternal and fetal exposures during pregnancy that set the stage for ongoing placental dysfunction. These mechanisms are detailed in this chapter, outlining defects at the various stages and levels of placental development involving structural, cellular, genetic, and epigenetic functions.
Key elements of placental sampling and examination provide valuable information that can help us better understand the mechanisms underlying IUGR. Decreased placental size and weight, usually less than 10th percentile weight-for-gestational age based upon published placental growth curves, is the typical gross placental finding accompanying IUGR. Other macroscopic placental abnormalities include unusual paleness, infarction, and loss of parenchyma. The area of unusual paleness is believed to represent decreased vascularization of chorionic villi, whereas the infarction can be recognized by a pale and indurated region in placental parenchyma.
The chorionic villus, the functional unit of the placenta that contains a capillary network derived from fetal circulation, undergoes developmental changes throughout pregnancy. Mediated byprocesses of early spiral artery remodeling and subsequent vascular proliferation and branching, the composition of the placenta shifts from few, large, and poorly vascularized primary villi to numerous, small, and highly vascularized tertiary villi. Histologic lesions accompanying IUGR include those from both vascular and inflammatory domains. The placental domain most commonly associated with IUGR, along with stillbirth and preeclampsia, is maternal vascular malperfusion of the placental bed . In 2016, the Amsterdam workshop recommended renaming this set of criteria from underperfusion to malperfusion; even though many of the effects of inadequate spiral artery remodeling manifest as a spectrum that includes IUGR and preeclampsia, high-velocity malperfusion may be as detrimental as underperfusion to placental function in later pregnancy. While the classic histologic lesions do not reliably distinguish between low- versus high-velocity processes, recent evidence suggests that certain abnormalities in villous architecture may be indicators of dysregulated blood flow more than chronic hypoxia due to persistent underperfusion. For example, three-dimensional reconstruction models of placental intervillous blood flow suggest that increased turbulent jet rates can lead to rupture of anchoring villi, as evidenced by echogenic cystic lesions on ultrasound. Microscopically, high-velocity damage to the villous surface may generate release of syncytial sprouts in early pregnancy and downstream presence of syncytial knots at placental autopsy. Increased shear wall stress at the villous surface has been seen in computer simulation models of turbulent blood flow, combined with elevated blood pressure in the intervillous space. Oxidant stress due to ischemia-reperfusion is another proposed mechanism of villous injury in which retention of smooth muscle predisposes to spontaneous vasoconstriction, leading to high-momentum blood flow entry into the intervillous space and apoptotic changes within the syncytiotrophoblast.
Microscopic findings of maternal vascular malperfusion are indicators of abnormal vascular and villous development involved in the above processes. Decidual arteriopathies, such as mural hypertrophy of membrane arterioles and persistent muscularization of basal plate arteries ( Fig. 14.2A ), are rare but consistently seen lesions in severe IUGR. Presence of these lesions at placental autopsy represent failed early first trimester spiral artery remodeling with persistence of intramural endovascular trophoblasts in the third trimester. Villous changes include accelerated villous maturation (see Fig. 14.2B ) and distal villous hypoplasia (see Fig. 14.2C ). , Placental morphology studies suggest that the presence of more highly branched villi at placental exam of infants with IUGR and ultrasound Doppler evidence of positive end-diastolic flow represent an adaptive response via enhanced branching angiogenesis. In contrast, the pattern of highly branched villi is not present in the growth-restricted placenta with absent or reversed end-diastolic flow in the umbilical artery, suggesting failure of the adaptive process. , Growth-restricted placentas also typically have reduced intervillous space volume, poorly developed peripheral villi, and a thicker trophoblastic epithelium that result in a smaller nutrient exchange area. These changes could also compromise placental oxygen transfer and contribute to fetal hypoxia.
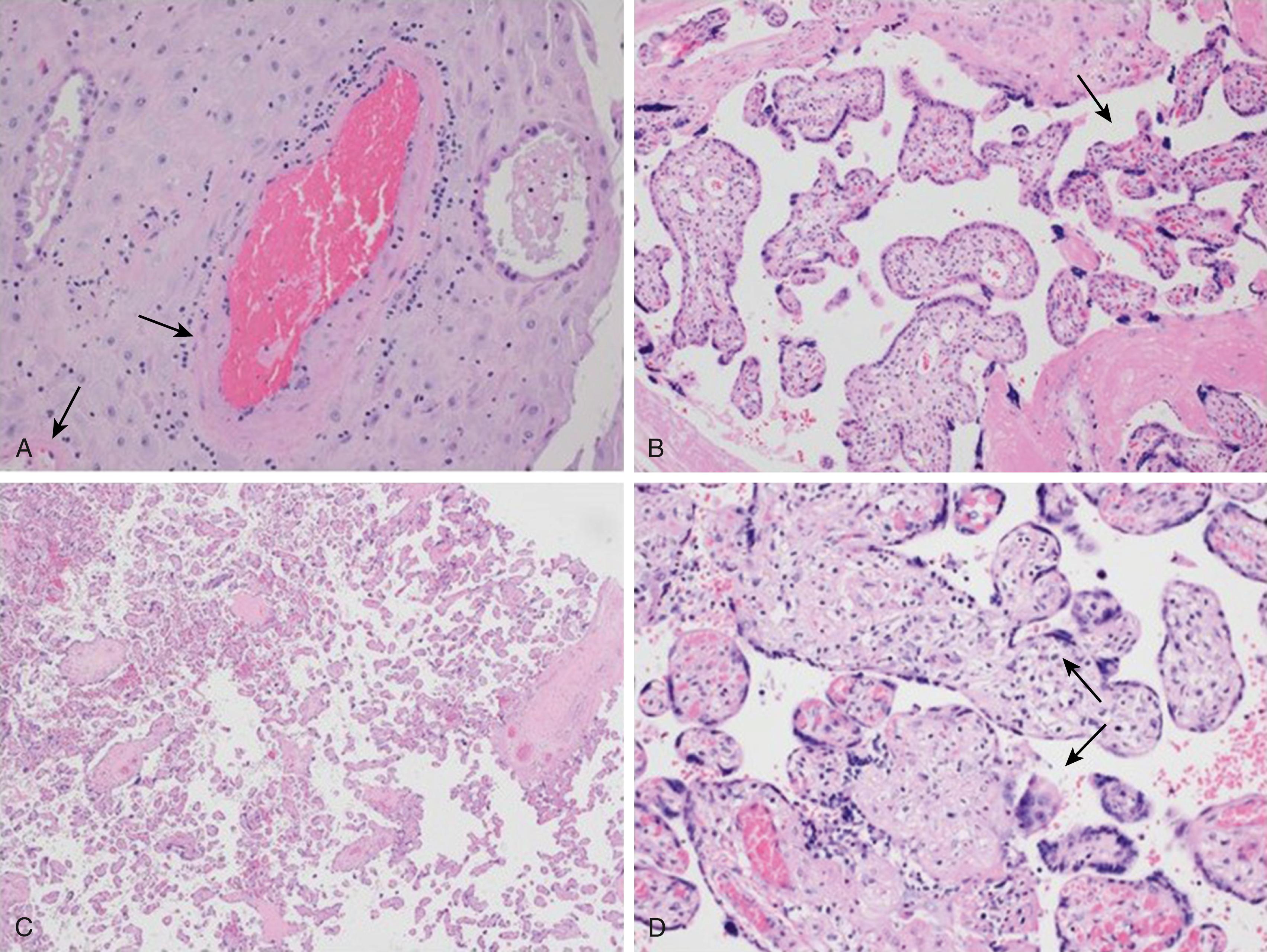
Villitis of unknown etiology (VUE), also known as nonspecific chronic villitis , is an inflammatory condition of the placenta characterized by maternal T-cell infiltrates in the villous stroma (see Fig. 14.2D ). Despite changes over the years in the classification and nomenclature to define this lesion, VUE continues to be a frequent histologic finding in IUGR and stillbirth and may involve vascular damage when inflammatory cells damage muscular wall vessels. Recent studies of coculture of placental explants with maternal leukocytes resulting in increased leukocytes in villous tissues and elevated cytokine levels support mechanisms by which altered placental inflammation has deleterious effects on placental function. Immunohistochemistry of placentas with chronic villitis has revealed cytokine profiles suggesting a skew toward inappropriate Th1 immune responses.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here