Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
On completion of this chapter, you should be able to:
Describe embryogenesis of the placenta
List the functions of the placenta
List and describe imaging techniques and sonographic findings of the placenta
Identify placental position and describe its importance
Describe the sonographic findings and clinical significance of placental pathologies
Recognize placental abruption with diagnostic ultrasound
The placenta is a maternal-fetal organ developed during pregnancy. Sonography is used to assess information regarding placental configuration, location, pathology, and maturation. The anatomic components of the placenta are discernible from as early as the seventh or eighth week of gestation. By the end of the first trimester, sonography can determine the location and position and identify specific components of the placenta. The placenta can be effectively evaluated during a second-trimester and third-trimester sonographic examination.
The major role of the placenta is to permit the exchange of oxygenated maternal blood, which is rich in oxygen and nutrients, with deoxygenated and nutrient-depleted fetal blood. Maternal blood flows through the decidual spiral arteries, circulating blood into the intervillous space within the placenta. The umbilical arteries, two of the vessels within the umbilical cord, carry the deoxygenated blood from the fetus to the placenta.
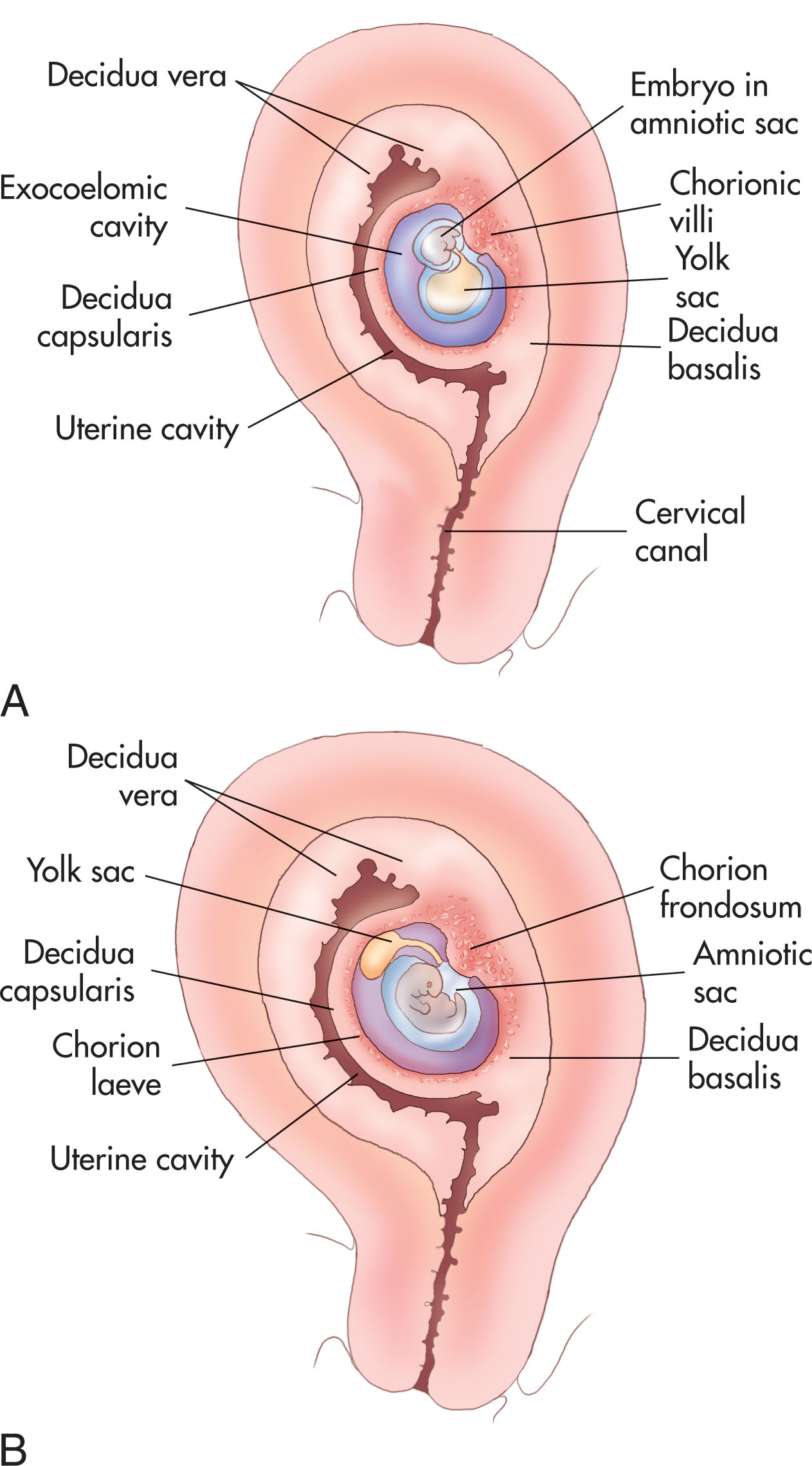
The development of the placenta begins after fertilization when the trophoblastic cells of the blastocyst implant into the uterus. Implantation of the blastocyst occurs 6 to 7 days after fertilization. The enlargement of the trophoblasts helps to anchor the blastocyst to the endometrial lining, or decidua . The development of the placenta is seen in the changes in the decidua ( Box 56.1 ).
Decidua basalis: the decidual reaction that occurs between the blastocyst and the myometrium
Decidua capsularis: the decidual reaction that occurs over the blastocyst closest to the endometrial cavity
Decidua vera (parietalis): the decidua reaction except for the areas beneath and above the implanted ovum
There are two types of trophoblastic cells. The syncytiotrophoblast is the outer layer of multinuclear cells, and the cytotrophoblast is the inner layer of mononuclear cells ( Box 56.2 ). These cells, along with the extraembryonic mesoderm, make up the chorion of the placenta. The chorion, amnion, yolk sac, and allantois constitute the embryonic or fetal membranes.
Chorion frondosum: forms the fetal part of the placenta and contains the villi
Chorion laeve: the nonvillous part of the chorion around the gestational sac
Chorionic plate: the fetal surface of the placenta
Basal plate: the maternal surface of the placenta
The placenta has two components: the maternal portion, the decidua basalis , formed from the endometrial surface ( Fig. 56.1 ), and the fetal portion, which develops from the chorion frondosum . The major functioning unit of the placenta is the chorionic villus ( Fig. 56.2 ). Within the chorionic villus are the intervillous spaces, which the maternal blood enters. As the embryo and membranes grow, the decidua capsularis is stretched. The chorionic villi on the associated part of the chorionic sac gradually atrophy and disappear (smooth chorion or chorion laeve). The chorionic villi related to the decidua basalis increase rapidly in size and complexity (villous chorion or chorion frondosum).
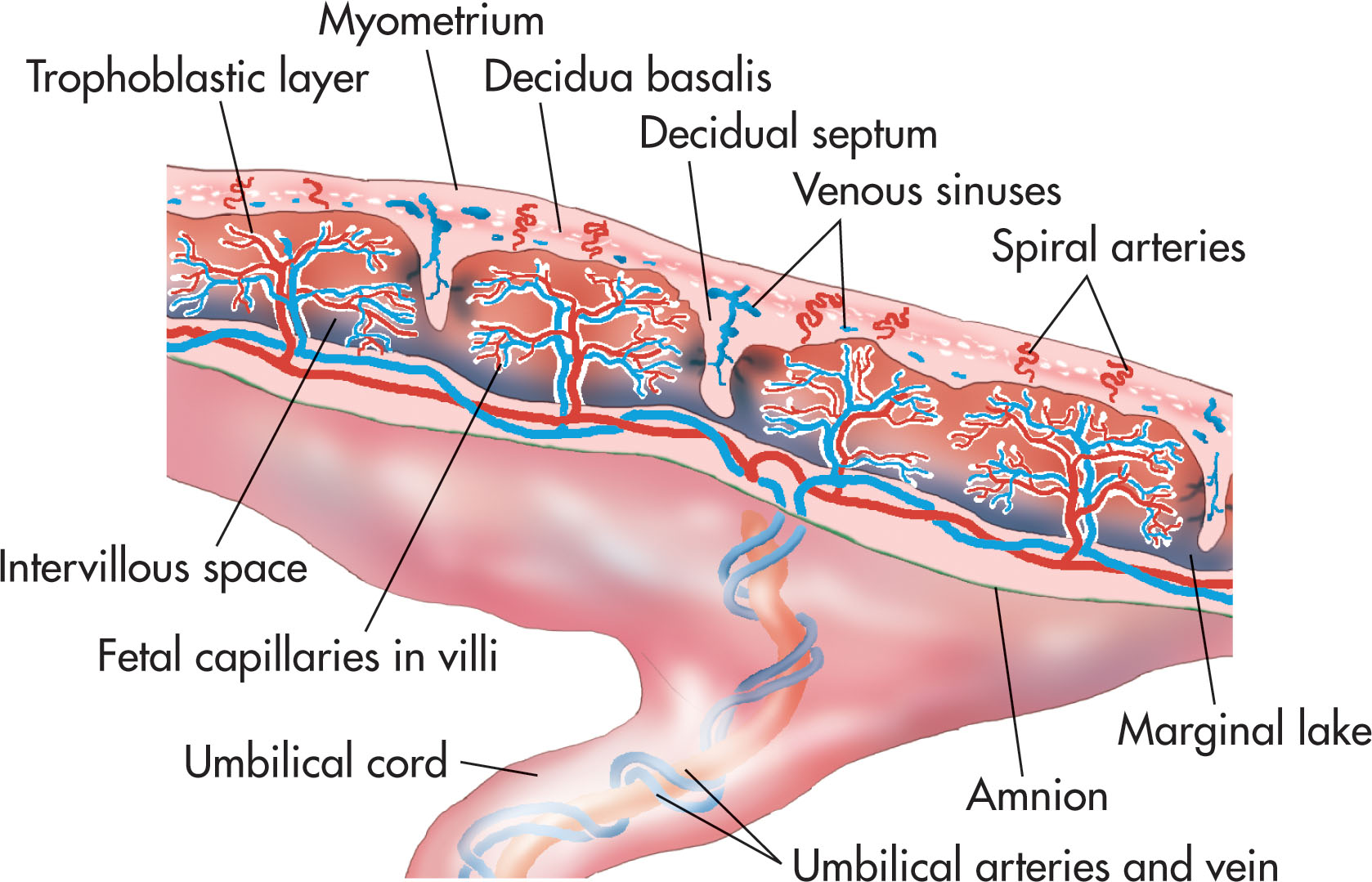
The maternal surface of the placenta, which lies contiguous with the decidua basalis, is termed the basal plate . The fetal surface, which is contiguous with the surrounding chorion, is termed the chorionic plate . The cotyledons are cobblestone in appearance and composed of several main stem villi and their branches. They are covered with a thin layer of the decidua basalis.
During pregnancy, the fetal membranes and placenta perform the following functions and activities: protection, nutrition, respiration, and excretion ( Box 56.3 ). The placental membranes are often called a barrier because a few compounds are unable to cross the placental membranes. At birth, or parturition , the placenta and membranes separate from the fetus and are expelled from the uterus as the afterbirth.
Transfer of oxygen from maternal blood across the placental membrane into fetal blood is by diffusion. Carbon dioxide passes in the opposite direction. The placenta acts as “fetal lungs.”
Water, inorganic salts, carbohydrates, fats, proteins, and vitamins pass from maternal blood through the placental membrane into fetal blood.
Waste products cross membrane from fetal blood and enter maternal blood. Excreted by maternal kidneys.
Some microorganisms cross the placental border.
Carbohydrates, proteins, calcium, and iron are stored in placenta and released into fetal circulation.
Produced by syncytiotrophoblast of placenta: human chorionic gonadotropin, estrogens, progesterone.
Oxygenated maternal blood is brought to the placenta through 80 to 100 end branches of the uterine arteries, the spiral arteries. Maternal blood enters the intervillous space near the central part of each placental lobule, where it flows around and over the surface of the villi. Maternal blood returns to the maternal circulation through the endometrial and then uterine veins. A very thin layer normally separates the fetal blood from the maternal blood. This layer is composed of the capillary wall, the trophoblastic basement membrane, and a thin rim of cytoplasm of the syncytiotrophoblast. The fetal placenta is anchored to the maternal placenta by the cytotrophoblastic shell and anchoring villi. It provides a large area where materials may be exchanged across the placental membrane and interposed between fetal and maternal circulation.
Oxygen- and nutrient-rich blood is carried back to the fetal systemic circulation through the umbilical vein. In the fetal abdomen, some of the blood is distributed to the liver, whereas the rest passes through the ductus venosus into the inferior vena cava and continues to the heart through the right atrium across the foramen ovale and into the left atrium. This oxygen-rich blood then passes into the left ventricle and the ascending aorta and supplies the brain and upper part of the body through the brachiocephalic circulation. Unoxygenated blood from the superior vena cava passes into the right atrium, through the right ventricle, and across the main pulmonary artery. A minor portion of blood from the right ventricle supplies the lungs, whereas the majority of the blood passes through a fetal shunt, the ductus arteriosus, into the aorta arch. This shunt protects the lungs against circulatory overload. The fetal blood continues inferiorly through the descending aorta, internal iliac arteries, and umbilical arteries and into the umbilical cord to return to the placenta for oxygen and nutrient exchange. By term, approximately 40% of the fetal cardiac output is directed through the umbilical circulation.
The placenta is dedicated to the survival of the fetus. Even when exposed to a poor maternal environment (e.g., when the mother is malnourished, ill, smokes, or abuses drugs), the placenta can compensate by becoming more efficient. Unfortunately, there are limits to the placenta’s ability to cope with external stressors. Eventually, if multiple or severe enough, these stressors may lead to placental insufficiency, fetal growth restriction, intrauterine death, and pregnancy loss.
During pregnancy, maternal blood volume increases 40% to 50% by term compared with the nonpregnant state. Maternal placental circulation may be affected by maternal conditions that decrease uterine blood flow, such as severe hypertension and renal disease. Intrauterine growth restriction (IUGR) is a fetal change that may be associated with decreased placental flow.
The location of the umbilical cord attachment to the placenta is important. The majority of the time, the umbilical cord inserts on the fetal surface of the placenta more than 3 cm from the margin. Abnormal cord insertions include marginal insertion and velamentous insertion. With a marginal insertion , also referred to as a battledore placenta , the insertion of the umbilical cord is at or close to the placental margin within less than 2 cm of the edge (less than 10%) ( Fig. 56.3 ). A velamentous insertion refers to an umbilical cord that inserts into the fetal membranes and then travels to the placenta within the membranes (1%) ( Fig. 56.4 ). These submembranous vessels are fragile and, in a small number of cases (less than 2%), may be associated with significant fetal hemorrhage, especially if the membrane carrying the vessels is positioned across the internal cervical os (vasa previa).
Placental location should be documented during the second-trimester sonographic examination. Normally the placenta will be located on the anterior, fundal, posterior, or lateral wall of the uterus. The relationship of the placental edge to the internal cervical os should be evaluated during each sonogram. Occasionally, placental implantation will occur within the lower uterine segment (LUS), resulting in a placenta previa or a low-lying placenta .
The fetal membranes consist of the chorion, amnion, allantois, and yolk sac. The chorion originates from the trophoblastic cells and remains in contact with the trophoblasts throughout pregnancy. The amnion develops at the 28th menstrual day and is attached to the margins of the embryonic disk. As the embryo grows and folds ventrally, the junction of the amnion is reduced to a small area on the ventral surface of the embryo to form the umbilicus.
Expansion of the amniotic cavity occurs with the production of amniotic fluid. Usually, by 16 weeks of gestation, fusion of the amnion with the chorion occurs and will no longer be visualized sonographically as two separate membranes. If the amnion/chorion separation extends beyond 16 weeks of gestation, it may be associated with polyhydramnios, aneuploidy, or prior amniocentesis. Placental hemorrhage may mimic the appearance of amnion/chorion separation on sonogram.
The secondary yolk sac forms after the regression of the primary yolk sac on the ventral surface of the embryonic disk at 28 menstrual days. The yolk sac has a role in transferring nutrients to the embryo during the second and third weeks of gestation while the uteroplacental circulation is developing. It is connected to the midgut by a narrow yolk stalk. By 5 menstrual weeks, the amniotic sac and secondary yolk sac are pressed together with the embryonic disk between them. This structure is suspended within the chorionic cavity. The yolk sac becomes displaced from the embryo and lies between the amnion and the chorion (see Fig. 56.1 ). By 9 weeks, the yolk sac has diminished to less than 5 mm in diameter.
The amnion forms a sac that contains amniotic fluid. This sac surrounds the embryo and forms the epithelial lining of the umbilical cord. Most of the amniotic fluid is derived from the maternal blood by diffusion across the amnion from the decidua parietalis and intervillous spaces of the placenta.
During the second trimester, the fetal kidneys begin to function and secrete urine, contributing to the amniotic fluid volume. The fetus swallows the amniotic fluid, and this cycle continues throughout pregnancy. The amniotic fluid has many functions, including permitting the fetus room to move, assisting in maintaining a constant fetal body temperature, serving as a protective buffer for the fetus, and helping in the formation of the gastrointestinal tract and lung development.
The umbilical cord forms during the first 5 weeks of gestation. The cord is surrounded by a mucoid connective tissue called Wharton jelly . The intestines grow at a faster rate than the abdomen and herniate into the proximal umbilical cord at approximately 7 weeks of gestation and remain there until approximately 10 weeks of gestation. The insertion of the umbilical cord into the ventral abdominal wall is an important sonographic anatomic landmark as this area can reveal abdominal wall defects such as omphalocele, gastroschisis, or limb–body wall complex.
The normal umbilical cord has one large vein and two smaller arteries. A single umbilical artery, also referred to as a “two-vessel cord,” is found in approximately 1% of singleton births and 7% of twin gestations. A single umbilical artery may be isolated, but if it is also associated with congenital malformations (renal, cardiac, facial, and musculoskeletal) or aneuploidy, amniocentesis should be offered. There is a reported association with a single umbilical artery and abnormal fetal growth, specifically IUGR. For this reason, a growth ultrasound in the third trimester to evaluate fetal size should be obtained.
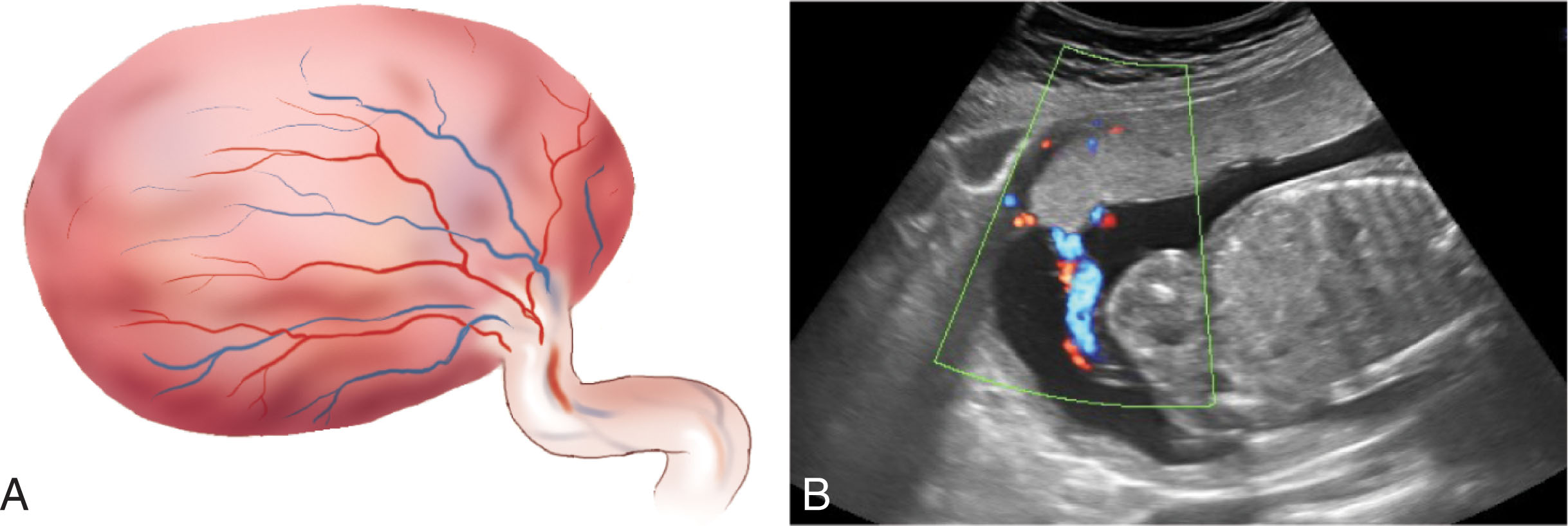
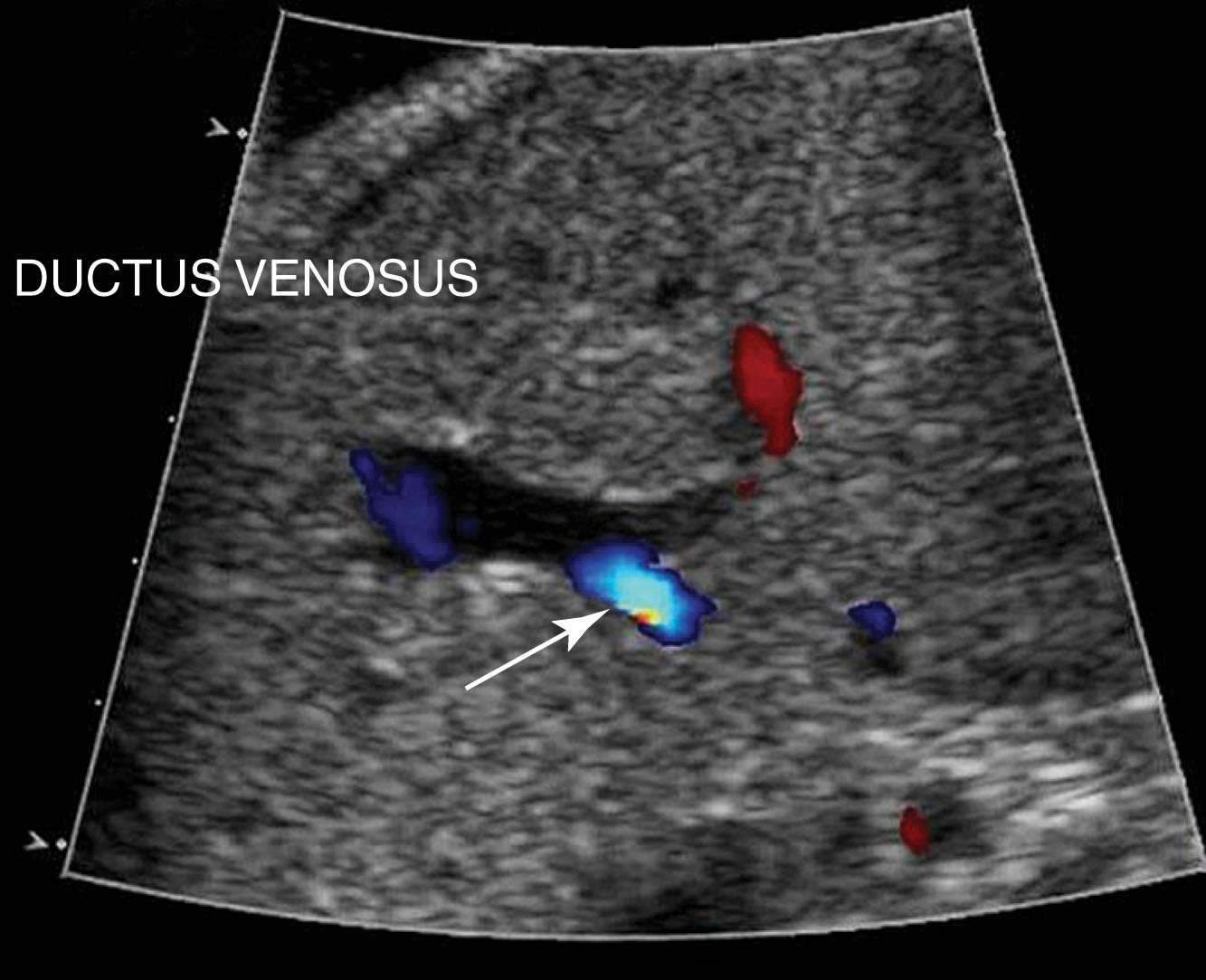
The vessels of the cord may be followed with real-time ultrasound from the placenta to the fetal abdomen, and the sonographer should document the umbilical cord insertion into the placenta ( Fig. 56.5 ) and the fetal abdomen. The intraabdominal portion of the umbilical vein courses superiorly. The ductus venosus shunts a significant amount of the oxygenated blood from the umbilical vein directly into the inferior vena cava. A portion of the umbilical venous blood also supplies the liver ( Fig. 56.6 ).
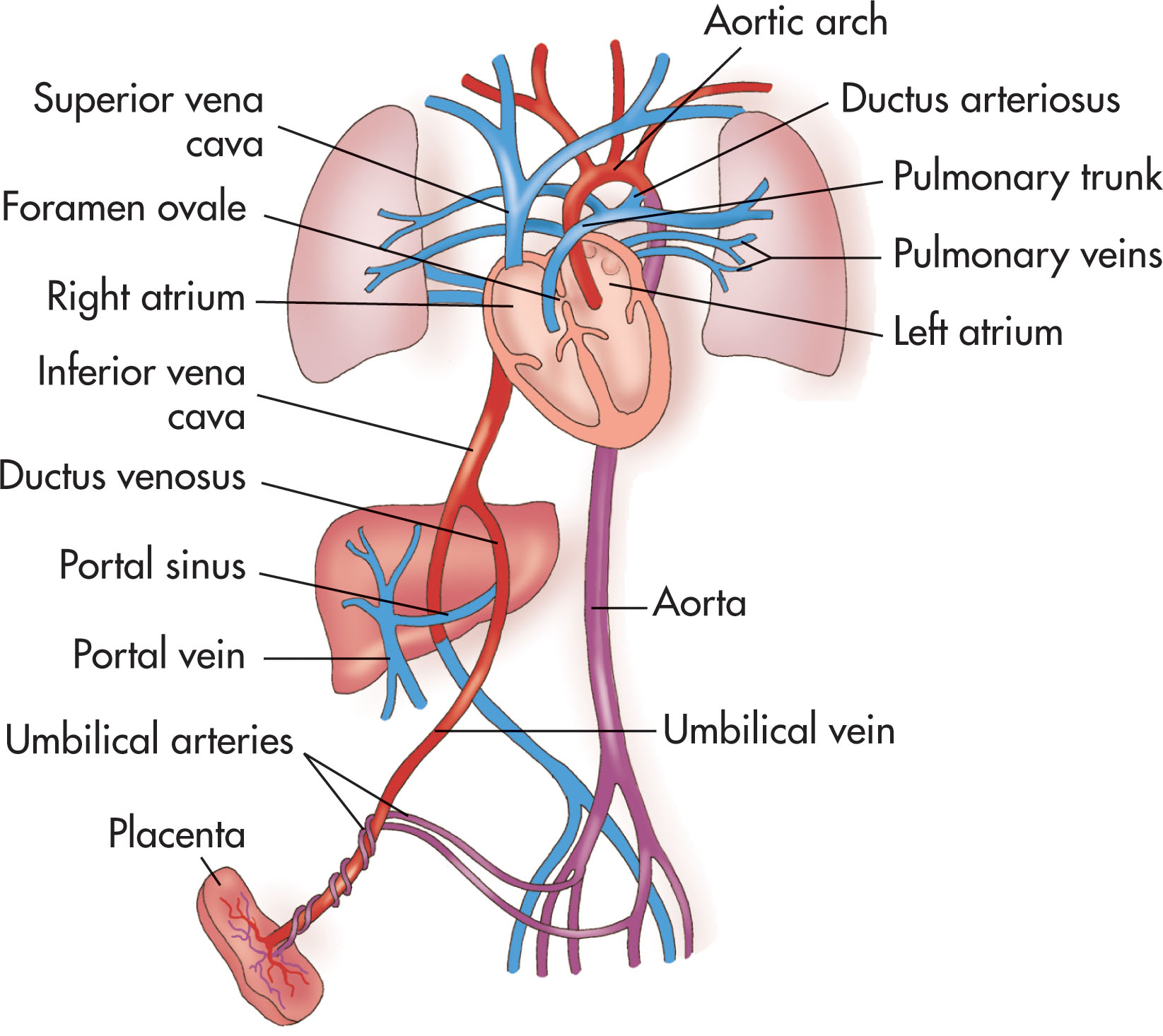
Sonographically, the ductus venous appears as a thin intrahepatic channel with echogenic walls branching from the umbilical vein ( Fig. 56.7 ). It lies in the groove between the left lobe and the caudate lobe of the liver. The ductus venosus is patent during fetal life until shortly after birth, when closure occurs. After closure, the remnant of the ductus venosus is known as the ligamentum venosum .
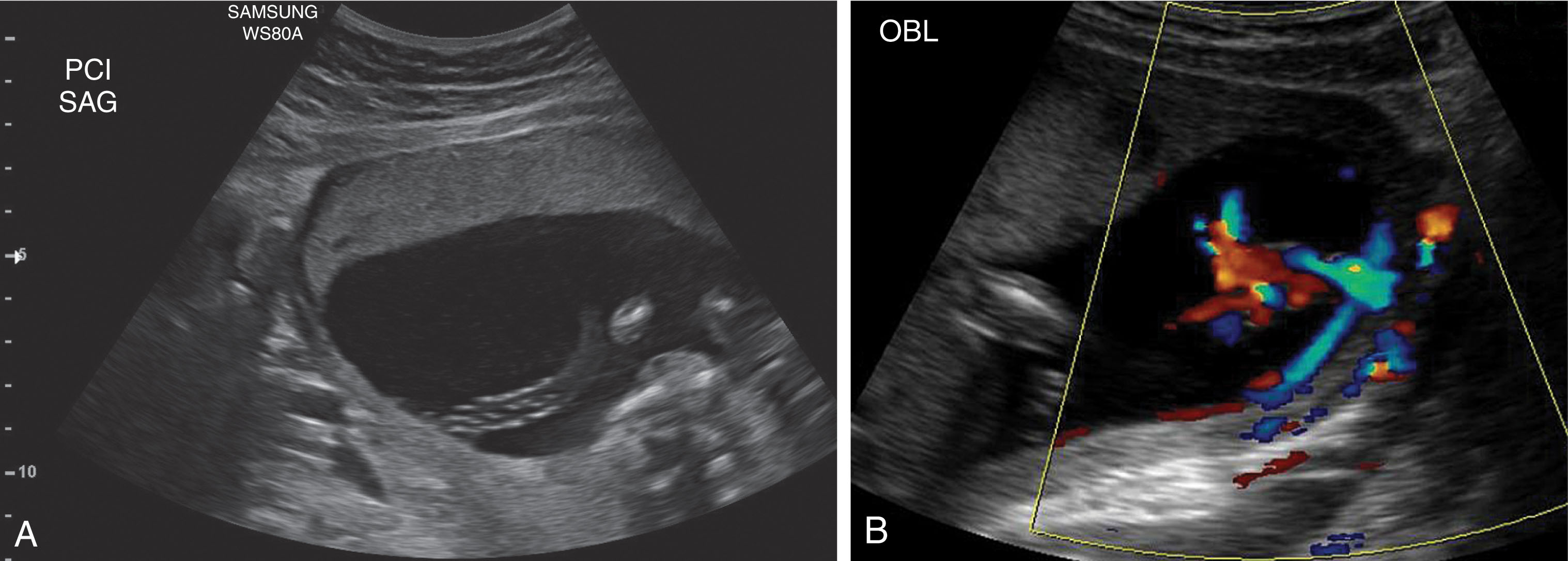
The two umbilical arteries carry deoxygenated blood from the fetus to the placenta. They ascend into the umbilical cord and are a branch of the internal iliac arteries. Their normal position extends around the fetal bladder. There are two acceptable ways to document the umbilical arteries. The first approach is to image the fetal pelvis in an axial scanning plane at the level of the fetal bladder. The umbilical arteries will be visible along the lateral aspect of the fetal bladder and then ascend toward the umbilical cord insertion. Color and power Doppler can be used to visualize the umbilical arteries. The second approach is to evaluate a free loop of cord in a short axis plane to visualize the umbilical vein and two smaller arteries. After birth, the umbilical arteries become the superior vesical arteries.
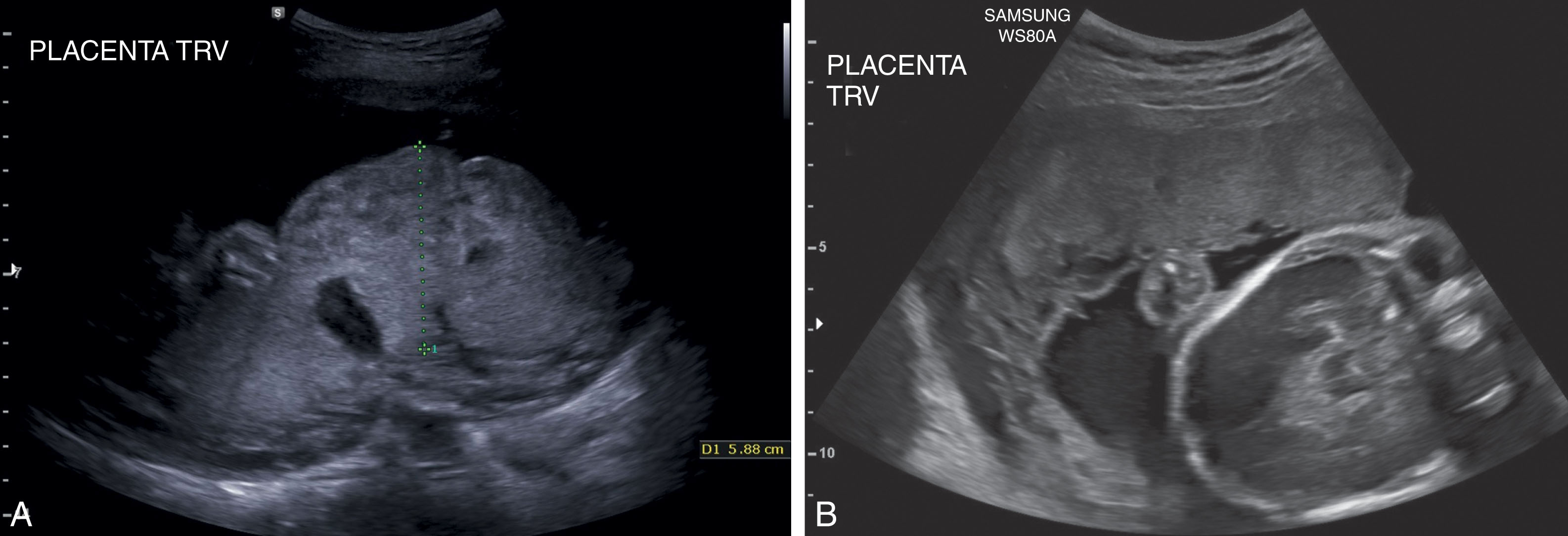
The placenta can be identified with sonography as early as 8 menstrual weeks. The placenta assumes a relatively homogenous midlevel gray appearance between 8 and 20 weeks of gestation and is easily recognized with its characteristically smooth borders. After 20 weeks of gestation, placental lakes (anechoic areas) and placental calcification may begin to appear. The thickness of the placenta varies with gestational age, but an anterior-posterior dimension is usually 2 to 3 cm in fetuses greater than 23 weeks of gestation. The thickness of a normal placenta will rarely exceed 4 cm. When evaluating placental thickness, the sonographer should be perpendicular to the uterine wall and measure through the placenta. Enlarged placentas may be associated with TORCH infections, diabetes, hydrops, and congenital anomalies ( Fig. 56.8A ). A small placenta may be associated with IUGR, intrauterine infection, and aneuploidy (see Fig. 56.8B ).
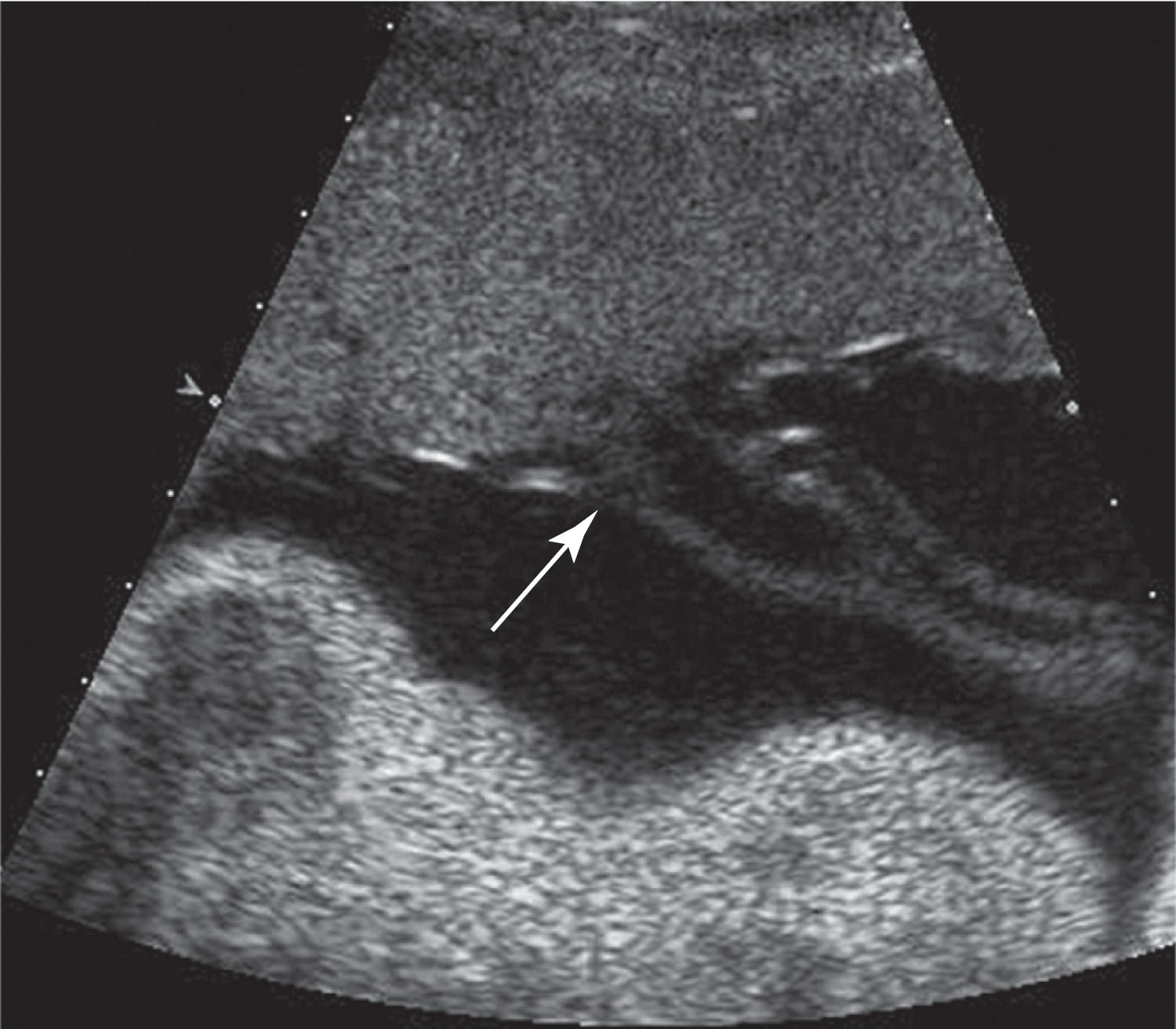
The chorionic and basal plate of the placenta are important in assessing normal placental anatomy and evaluating for placental abruption. The fetal surface of the placenta is the chorionic plate that gives rise to the chorionic villi. Sonographically the chorionic plate appears echogenic and is the portion of the placenta that is adjacent to the amniotic cavity ( Fig. 56.9 ). The basal plate or maternal portion of the placenta, which lies at the junction of the myometrium and the substance of the placenta (see Fig. 56.9 ). Maternal blood vessels reside between the placenta and the myometrium (see Fig. 56.9 ). This represents normal vascularity, which sonographically appears hypoechoic but can be visualized well with color Doppler. The maternal vessels are more apparent sonographically when the placenta is positioned fundally or posteriorly within the uterus. Blood flow can easily be detected within the placenta with color Doppler ( Fig. 56.10 ).
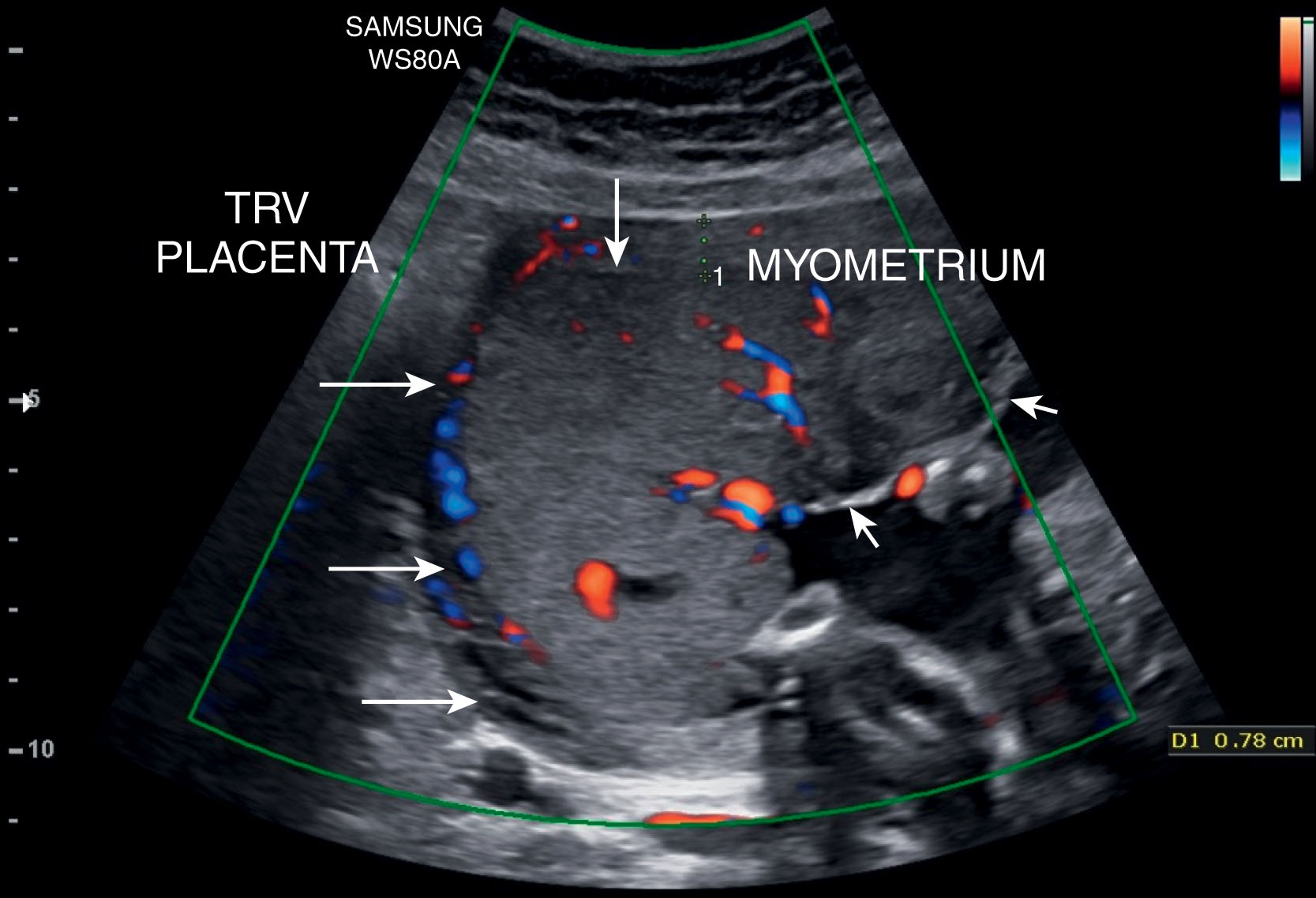
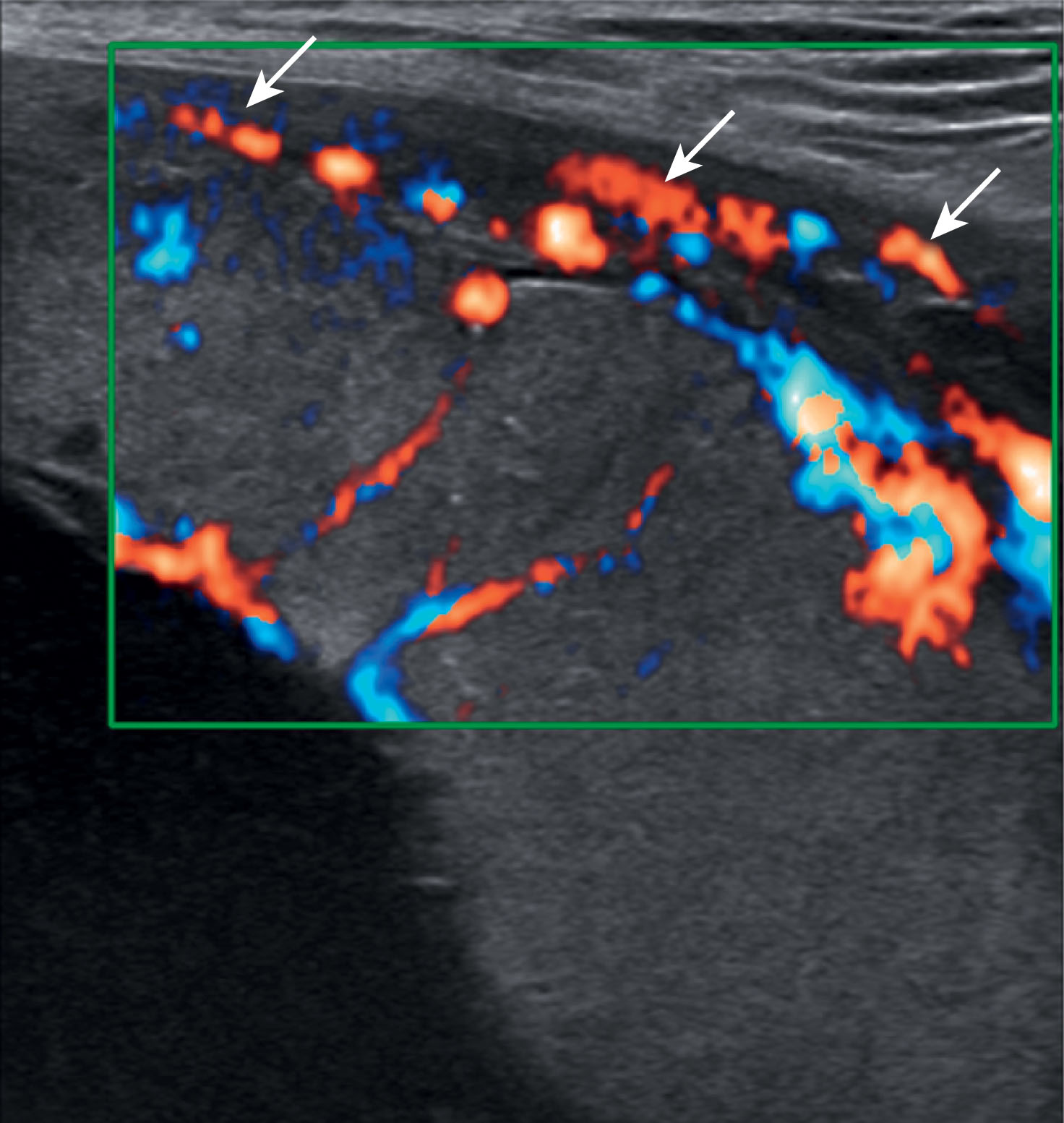
Placental lakes may also be seen within the placenta. These have been referred to as placental sonolucencies and are most often surrounded by normal placenta ( Fig. 56.11 ). Placental lakes located along the fetal surface may appear to bulge into the amniotic cavity. These areas may change dramatically in shape and size during the course of the ultrasound examination, and with real-time sonography, slow swirling flow can be appreciated.
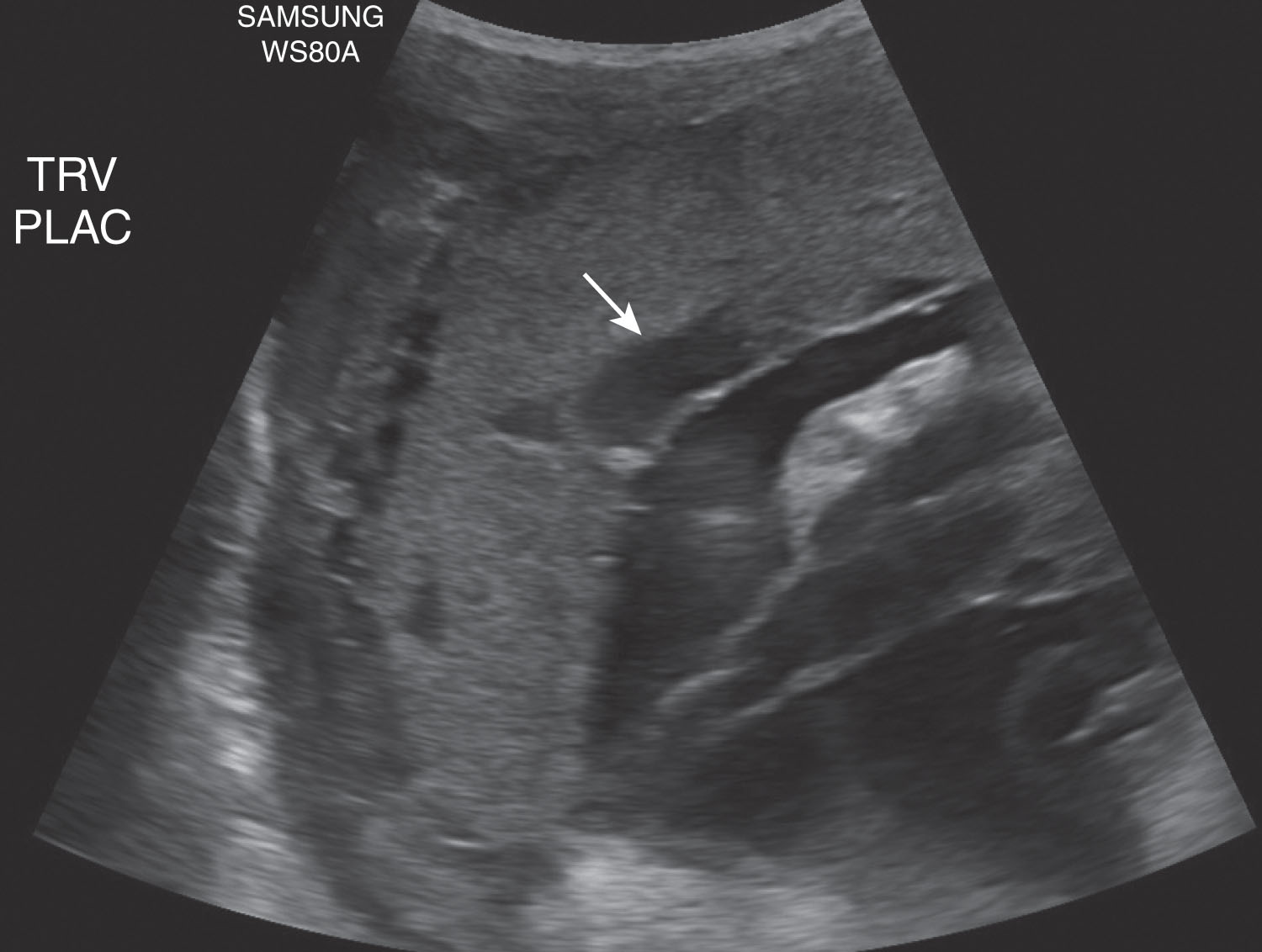
The placenta is separated from the myometrium by a subplacental venous complex. These veins (basilar and marginal) can become very prominent, especially for lateral and posterior positioned placentas, and should not be confused for a retroplacental or marginal hemorrhage. The myometrium appears slightly less echogenic when compared to the echogenicity of the placenta (see Fig. 56.9 ). The basilar veins and the myometrium measure as much as 9 to 10 mm in thickness.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here