Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Peripheral artery disease (PAD) is the most common condition requiring treatment by vascular surgeons and vascular specialists. Over the last decade, the global prevalence of PAD has continued to rise, and it is a major contributor to rising healthcare resource consumption. , An estimated 8 to 12 million individuals in the United States are afflicted with PAD, and at least 202 million people suffer from PAD across the globe. A recent metaanalysis of more than 34 studies showed a 23.5% increase in the prevalence of PAD during the first decade of the twenty-first century. The primary drivers of this dramatic rise in PAD prevalence are the underlying increases in the major risk factors for the development of PAD around the world. These risk factors include population aging (i.e., increased longevity); the global epidemic of diabetes, hypertension, and obesity; and the persistence of tobacco smoking in many parts of the world ( Fig. 63.1 ). An increased PAD prevalence has been noted in both high- and low-income countries ( Fig. 63.2 ), although the rise has been more dramatic in low- and middle-income countries (28.7% increase) than in high-income countries (13.1% increase).
The economic impact of PAD has been growing in parallel with its increased prevalence, especially in the United States and in many industrialized countries. In 2001, PAD-related treatments comprised approximately 13% of all Medicare Part A and B expenditures and contributed to an estimated economic burden of over $4.3 billion. By 2004, according to a detailed analysis of the Reduction of Atherothrombosis for Continued Health (REACH) Registry, the total estimated costs of vascular-related hospitalizations in the United States was $21 billion. , The main contributor to these costs was revascularization procedures, particularly rises in the use of endovascular therapy (EVT).
The chapter which follows will review the pathophysiology, anatomy, patient presentations, natural history, diagnosis, and management of lower extremity PAD. While there have been many advances in therapy over the last decade, there is still a striking relative lack of high-level evidence for many of the treatments in common use. The condition is underdiagnosed, medical management is underutilized and often not maximized, and there are wide regional differences and disparities in the application of revascularization procedures and strategies.
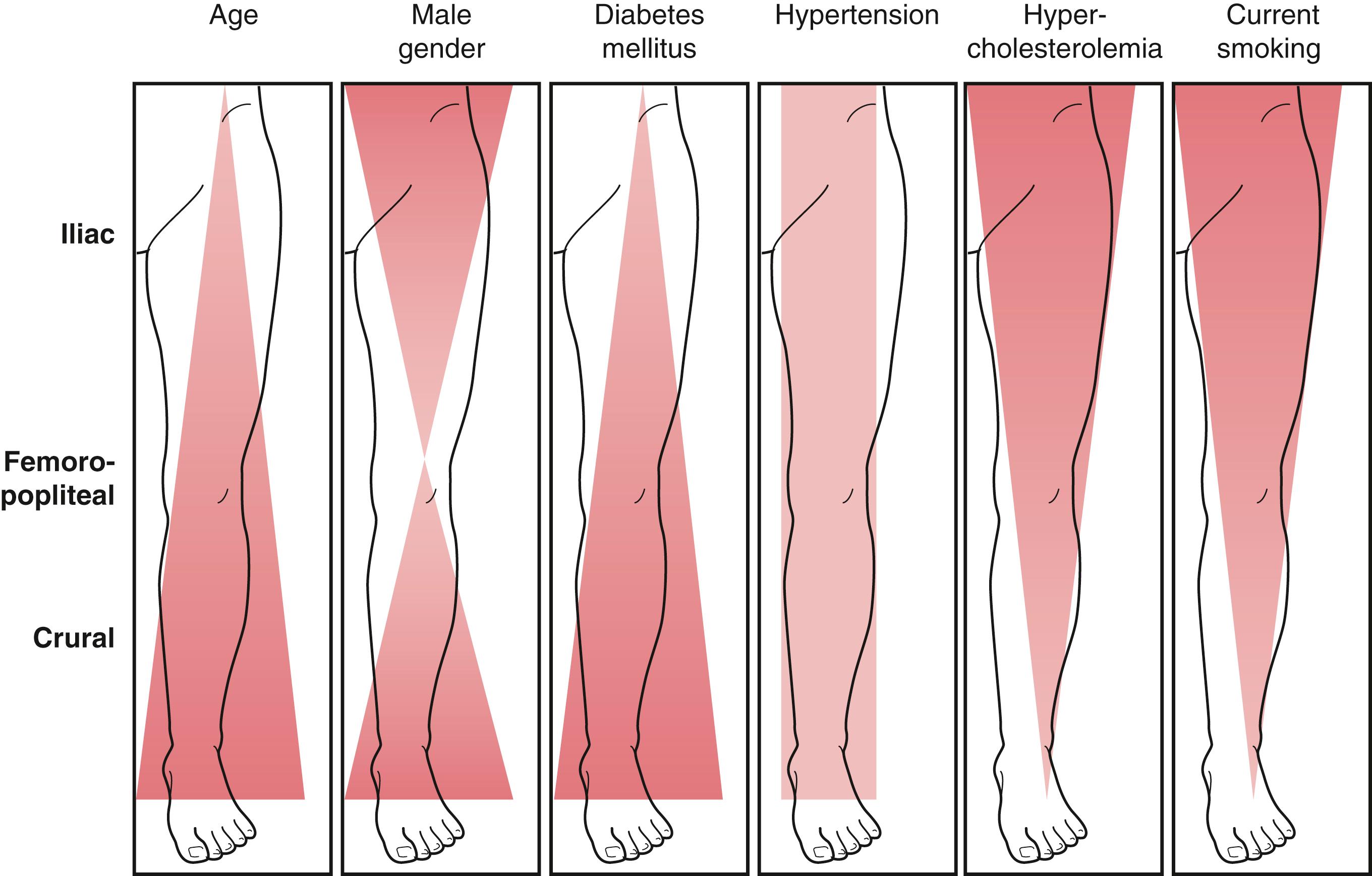
Aging is a major risk factor for the development of PAD. Lower extremity PAD most commonly presents in patients more than 50 years of age and increases markedly with each decade beginning at 60 years of age. The prevalence of PAD has been estimated at 14.5% in patients more than 69 years of age and as high as 20% in patients 80 years of age and older. Cigarette smoking and its intensity is also strongly associated with PAD with one study estimating its population attributable fraction at 44%. In the last 20 years, it has become evident that diabetes has become one of the most prominent risk factors for the development of PAD. There is an ongoing global epidemic of diabetes and currently over 383 million people are affected; this number is expected to almost double in the next 15 to 20 years. Diabetes is strongly associated with PAD; population-based studies have reported odds ratios of 1.9 to 4 ( Fig. 63.1 ). Patients with diabetes are more likely to develop a foot ulcer and to present with chronic limb-threatening ischemia (CLTI). Hypertension and hyperlipidemia are the other major risk factors associated with the development of PAD.
Patients may have detectable PAD and yet be completely asymptomatic; this situation is not uncommon in the aging population as other factors may limit activity levels while PAD lurks in the background. Individuals may also present with atypical leg symptoms that result from other conditions such as lumbar spine disease, neuropathy, degenerative joint disease, and myopathy. Typical symptoms of PAD include vasculogenic claudication and CLTI. The latter category includes ischemic rest pain, ischemic ulcer, and gangrene.
Symptoms typical of vasculogenic claudication are characterized by cramping or aching discomfort in the buttock, thigh, or calf muscles that is induced by walking and relieved by rest. The location of muscular symptoms often correlates with the anatomic site of disease, such that aortoiliac disease produces buttock and thigh claudication, while femoropopliteal occlusive disease results in calf claudication. Such complaints are usually reproducible in onset but may arise sooner by walking at a faster pace or uphill. Vasculogenic claudication typically resolves with a short period of rest (which reduces the muscular metabolic requirement), and in contrast to neurogenic claudication, is neither variable in onset nor does it require a change in position for symptom resolution. Symptoms related to nerve root compression are often variable in onset, can take a long time for recovery, may arise from standing alone (without walking), and are often relieved by changes in spine position (such as spine flexion or sitting down). These two conditions may coexist, and a good history and physical examination will often help the clinician differentiate them.
Complementing the history, qualitative components of a physical examination form the cornerstone of PAD diagnosis. These components include pulse examination (brachial, radial, femoral, popliteal, posterior tibial, and dorsalis pedis), observation for lack of distal hair growth on the involved extremity and dry skin which may result from apocrine gland dysfunction. The measurement of ankle-brachial index (ABI) forms the objective, quantitative basis of PAD assessment. An ABI of less than 0.9 has a sensitivity of 79% to 95% and a specificity exceeding 95% to establish the diagnosis of PAD in patients in whom it is suspected. , In some individuals, particularly those with diabetes or the aged, medial calcinosis will result in falsely elevated ABIs. Because the toe arteries are often spared calcification, a toe-brachial index may be measured to quantitate PAD in individuals found to have noncompressible ankle arteries. A toe-brachial index less than or equal to 0.7 is abnormal and indicates hemodynamically significant PAD. If the diagnosis of PAD is still in doubt, particularly when compelling symptoms are present in the setting of palpable pulses, an ABI test with exercise can be helpful. This test and other useful studies will be subsequently discussed in more detail under “Evaluation of the Patient with PAD.”
Ischemic rest pain has long been recognized as a classic symptom of advanced PAD and is one manifestation of CLTI. It is more common in smokers than patients with diabetes mellitus, likely masked in the latter by peripheral sensory neuropathy related to underlying diabetes. It occurs in the forefoot and is typically described as having its onset with leg elevation or recumbency (i.e., when going to bed at night) and is relieved by dependency (i.e., dangling the foot off the bed at night). The increase in pedal blood pressure related to gravity is sufficient to relieve the pain. Affected patients lack pedal pulses and usually suffer from distal hair loss in the affected extremity. Pallor on elevation and dependent rubor are common physical findings. The diagnosis is confirmed by one or more of several hemodynamic parameters, including an ABI less than 0.4, an ankle systolic pressure less than 50 mm Hg, a systolic toe pressure less than 30 mm Hg, a transcutaneous partial pressure of oxygen (TcPO 2 ) less than 30 mm Hg, and flat or minimally pulsatile pulse volume recording waveforms in the forefoot. It is simple and important to objectively confirm the diagnosis with hemodynamic testing, as other conditions such as diabetic neuropathy, night cramps, degenerative joint disease, and gout may be confused with rest pain.
Tissue loss (lower leg or foot ulcer) and gangrene can be obvious manifestations of CLTI. The strict definition of CLTI-related tissue loss requires that it be present for at least two weeks (to exclude minor traumatic lesions that heal spontaneously) and that it be accompanied by objective evidence of PAD of sufficient severity to impede wound healing. This topic will be addressed in more detail below when the wound, ischemia, and foot infection (WIfI) classification of CLTI is reviewed as one of the key management concepts recently recommended by the Global Guidelines Committee on CLTI.
This chapter focuses on PAD due to atherosclerotic occlusive disease. Other uncommon arteriopathies and vasculitides that may produce peripheral ischemia are beyond its scope. These nonatherosclerotic conditions include giant cell arteritis, Takayasu arteritis, polyarteritis nodosa, Wegener granulomatosis, thromboangiitis obliterans (Buerger disease), Behcet disease, pseudoxanthoma elasticum, iliac artery endofibrosis, popliteal entrapment syndrome, and cystic adventitial disease.
Arteries are generally grouped into three types: elastic, muscular, and arterioles. The elastic arteries are the aorta and the pulmonary arteries. They need to be elastic because they receive blood directly from the heart and are relatively thin compared to their diameters. With each contraction of the heart, blood is forcibly ejected into the elastic arteries, whose walls must stretch to accommodate this systolic force. During diastole, their elastic walls recoil, thus continuing to propel blood forward while the heart refills. The muscular arteries are distributive in nature and include the coronary and peripheral arteries. These arteries are thicker relative to their diameters than the elastic arteries, with reduced sheets of elastin and characteristically well-defined longitudinal and circular smooth muscle layers. Contraction and relaxation of the muscular arteries alter the amount of blood flow delivered depending on local requirements (e.g., increased peripheral arterial flow is induced by exercise). Arterioles are the vessels of blood delivery to the capillary bed. Arterioles are characterized by concentric rings of smooth muscle whose contraction and dilation control blood flow into the capillary bed; they are generally less than 300 microns in diameter.
Arteries consist of three layers: the endothelium, media, and adventitia (with vasa vasorum). These layers vary in composition and thickness depending on location and health/disease state. The endothelium is considered an organ. As such, it has autocrine, paracrine, and endocrine functions that regulate blood flow and thrombogenicity. The endothelium is remarkable in that it synthesizes multiple compounds that regulate vascular tone and provide vascular homeostasis. Dysfunction of the endothelium is the earliest hallmark of vessel injury (Ross hypothesis of atherosclerosis) and it can be detected before histologic changes associated with atherosclerosis are evident. The injury response is currently thought to be similar in many ways to a chronic inflammatory response. After initial epithelial dysfunction, changes in the arterial wall permeability occur and in response to a multitude of growth factors, stimulatory factors, and interactions between smooth muscle cells (SMCs), monocytes, lymphocytes and platelets, a fibroproliferative response takes place that results in plaque deposition. There are three stages of plaque, with the earliest stage termed the fatty streak. Fatty streaks are focal, yellow, usually linear streaks that can be seen on the luminal surface of arteries and are evident in most individuals after three years of age. These streaks are microscopically macrophages (foam cells) full of lipid that accumulate in the intima. They often occur at branch points. Atherosclerotic plaque also tends to develop at branch points. The intermediate stage is a fibrofatty lesion characterized by increased deposition of layers of matrix around layered macrophages, T lymphocytes, and SMCs. The most advanced stage is the complicated or fibrous plaque. Such plaques have begun to compromise the arterial lumen and protrude into it. On their surface is a fibrous cap beneath which lie dense layers of connective tissue and SMCs with a core containing lipid and necrotic debris. Rupture of the cap characterizes an unstable plaque, which exposes the vessel lumen to lipid and cellular debris, leading to the thrombotic complications associated with atherosclerotic plaque ( Fig. 63.3 ).
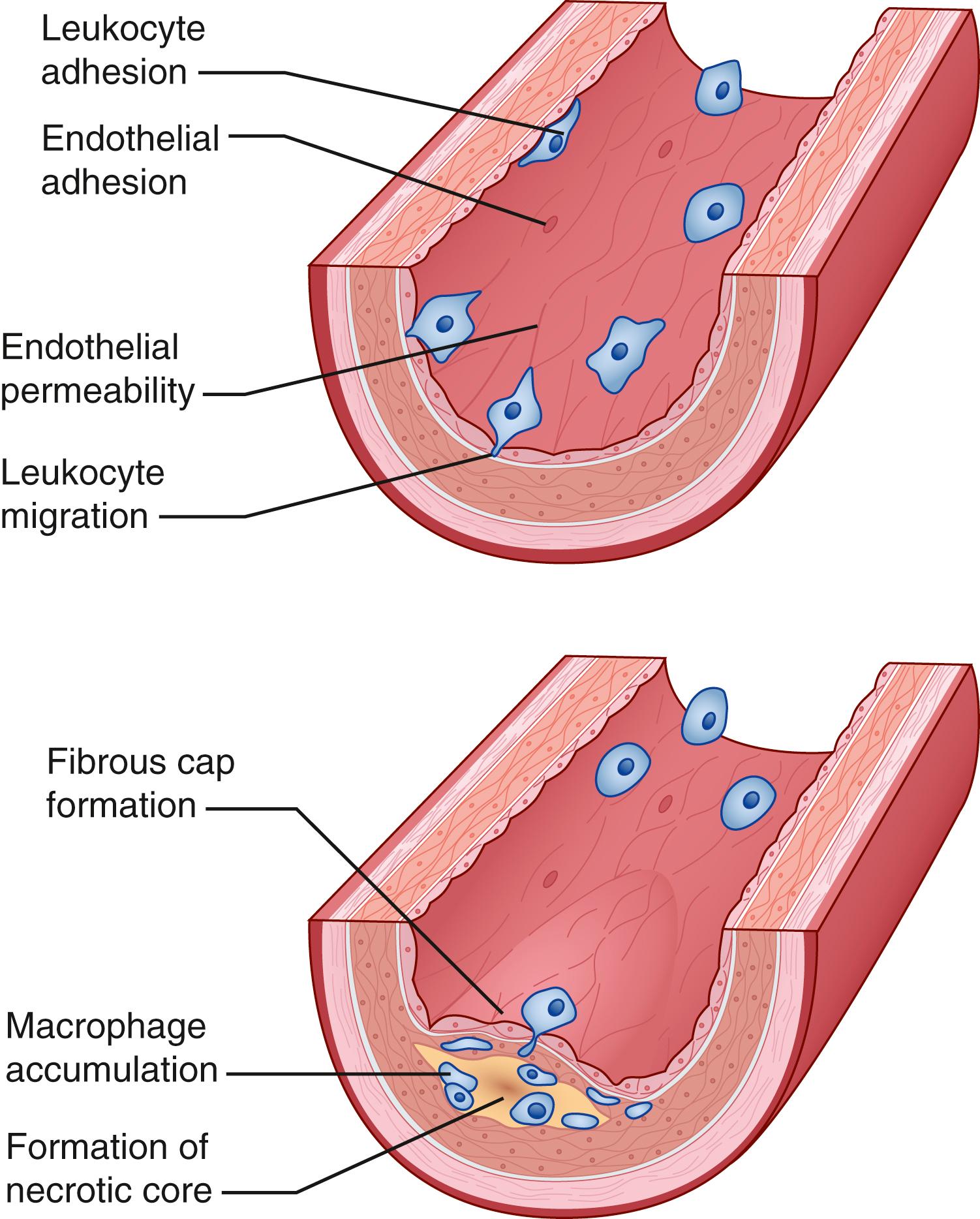
Atherosclerotic disease is a chronic, degenerative, inflammatory process to which the body attempts to adapt to maintain both the structure and underlying function of the arterial circulation. Although systemic in nature, atherosclerosis tends to develop at specific, anatomic locations within the arterial tree. Adaptive responses include compensatory changes in wall thickness and luminal diameter, which are thought to result from changes in shear stress. Glagov and associates were among the first to note that as atherosclerotic plaques enlarge, the lumen enlarges to compensate and maintain similar flow rates. This process has been shown to occur in the coronary, carotid, and superficial femoral arteries as well as the aorta. Such compensatory enlargement may serve to prevent flow-limiting luminal stenosis until the plaque area reaches approximately 40% of the cross-sectional area of the affected lumen. While coronary, carotid, and aortoiliac lesions tend to occur at branch points, obstructive superficial femoral artery plaque tends to develop in the distal portion of the vessel, which is generally straight, with few branches. This predilection for PAD to occur in the superficial femoral artery at the adductor hiatus has been attributed to the anatomic compression by the adductor tendons, which limits compensatory arterial dilation to growing plaque.
In general, patients with claudication will be found to have single level disease, frequently involving either the aortoiliac segment or the femoropopliteal segment, with relative sparing of the distal extremity arteries. These patterns of disease are common in cigarette smokers. Patients with CLTI more often have multilevel disease. Patients with PAD associated with diabetes tend to have patterns of more distal occlusive disease and more frequently have involvement of the deep femoral (profunda) artery and the infrapopliteal arteries. CLTI patients often have femoropopliteal artery stenosis or occlusion along with tibial occlusive disease, but especially in diabetes, they may be found to have isolated infrapopliteal occlusive disease. The pedal arteries are often spared, with more than 85% of patients having a patent pedal vessel, although severe pedal occlusive disease seems to be increasing in frequency, especially among patients with end-stage renal disease. For reasons that remain yet unexplained, the anterior tibial and posterior tibial arteries are most frequently involved, with relative sparing of the peroneal artery. These patterns of disease are important to recognize as they have significant implications for management ( Fig. 63.4 ).
A detailed history and thorough physical examination should be performed in every patient suspected of having PAD. It should include elucidation of pertinent symptoms and the degree of disability associated with them; past medical history (particularly prior surgical operations or revascularization procedures); assessment of all major cardiovascular risk factors (smoking, diabetes, hypertension, hyperlipidemia, obesity, and sedentary lifestyle); palpation of all accessible peripheral pulses (carotid, brachial, radial, ulnar, femoral popliteal, posterior tibial, and dorsal pedal); auscultation of the neck, abdomen and groin for bruits; auscultation of the heart and lungs; and palpation of the abdomen, femoral, and popliteal regions for aneurysm. The extremities should be inspected for temperature changes, color (elevation pallor or dependent rubor), signs of muscle atrophy, distal hair loss, and ulcers of the leg and foot, especially examining all surfaces of the foot and between the toes in patients with diabetes. An adequate extremity examination requires removal of the socks and shoes bilaterally, even if there are only complaints in one limb. The shoes themselves should also be closely examined for signs of uneven wear and foreign bodies within them or stuck in the soles (nails, tacks, screws, etc.). Patients with diabetic neuropathy will not feel these items. All patients with foot ulcers should be tested for neuropathy to detect loss of protective sensation (the Semmes-Weinstein monofilament test is the simplest) and probe-to-bone test should be performed in any patient with a foot ulcer.
PAD is a localized manifestation of systemic atherosclerosis. PAD is associated with high cardiovascular morbidity and mortality from myocardial infarction and stroke. These risks are particularly high among CLTI patients. Major risk factors for the development of PAD include age, sex, hypertension, hyperlipidemia, diabetes mellitus, smoking status, and sedentary lifestyle. Major cardiovascular risk factors should be evaluated in all patients with PAD. Medical management of patients with PAD includes modification of these medical comorbidities when feasible to reduce lower cardiac morbidity and mortality. The Society for Vascular Surgery Global Vascular Guidelines were used as a framework on which the following recommendations were based. ,
Antithrombotic therapy is strongly recommended for all patients with PAD to reduce major adverse cardiac events (MACEs), defined as a composite of nonfatal stroke, nonfatal myocardial infarction, and cardiovascular death. The mainstay of this therapy is low-dose aspirin. Recent data suggests that further benefit might result from the use of alternative antiplatelets agents such as clopidogrel or ticlopidine. The benefit achieved in these patients is a lowering of MACEs. A metaanalysis performed comparing single-agent antithrombotic use in PAD patients suggested that clopidogrel monotherapy was most effective for lowering MACEs. Currently, there is no clear benefit for dual antiplatelet therapy (DAPT) or systemic anticoagulation in patients with PAD to lower MACEs, though there are several ongoing clinical trials to evaluate this issue further.
Lipid-lowering therapy is essential in patients with PAD and has been demonstrated to decrease MACEs. Additionally, there appears to be a direct antiinflammatory effect in PAD patients, which has been postulated to lead to atherosclerotic plaque stability and reduce vascular events. It has been well established that high-intensity statin therapy decreases MACEs in patients with PAD. Specifically, this includes high intensity rosuvastatin (20–40 mg/day) or simvastatin (40–80 mg/day).
Control of hypertension has been shown to decrease MACEs in patients with PAD. Data suggests that targeting systolic blood pressure (SBP) less than 140 mm Hg and diastolic blood pressure (DBP) less than 90 mm achieves optimal reduction in MACEs in patients with PAD. Specific categories of antihypertensives have not clearly been demonstrated to be optimal in PAD patients, with angiotensin-converting enzyme (ACE) inhibitors, calcium channel blockers, beta blockers, and diuretics all being effective to lower MACEs.
Diabetes mellitus is a significant risk factor and contributor to the development of atherosclerosis and PAD. The extent and severity of disease correlates with blood glucose control. Therefore, glycemic control should be a focus of care in patients with PAD. The specific goal is for patients is to maintain a Hemoglobin A1c level of less than 7%. There has been a noted advantage for using metformin as the primary hypoglycemic agent for patients with Type II diabetes and CLTI. Adjunctive medications as well as insulin should be considered to achieve this A1c target.
Tobacco abuse is a frequent comorbid condition for patients with PAD and specifically those with CLTI. The extent of cigarette smoking has been shown to correlate with PAD severity. Tobacco abuse leads to higher MACEs in patients with PAD and also contributes to PAD disease progression. Patients should be asked about the status of their tobacco use at every visit. Clinical support, adjunctive medications, and counseling should be offered to all active smokers with PAD.
Exercise has been shown to have clear benefits for patients with PAD and intermittent claudication and should be the attempted prior to revascularization in these patients. This is especially true for patients with stable symptoms that are not lifestyle limiting. Specifically, patients should be referred for a supervised exercise therapy program with an exercise physiologist if possible. There is greater established benefit for supervised compared to nonsupervised programs, but home-based plans have also shown benefit. Exercise has been shown to improve walking distances in claudicants by increasing calf blood flow, improving endothelial function, reducing local inflammation, and inducing angiogenesis. The general recommendations are for these patients to perform a minimum of 45 to 60 minutes of exercise, 3 times per week for 12 weeks, typically walking on a treadmill. The exercise should be sufficiently intense to elicit claudication. No specific randomized controlled trial has been used to evaluate this benefit in CLTI patient symptoms, although the benefit from reduced MACEs associated with cardiac rehabilitation regimens has been demonstrated.
Perhaps the most significant recent change in the diagnosis and management of PAD relates to concepts concerning CLTI. These changes were driven in large part by the global epidemic of diabetes. Diabetes currently affects nearly 400 million people around the world and its prevalence is increasing in virtually every country for which data are available. Due to neuropathy and PAD, about one in four patients with diabetes will develop a foot ulcer during their lifetime; 80% of diabetes-related amputations are preceded by a diabetic foot ulcer (DFU). , A significant fraction (49%–66%) of people with DFUs have detectable underlying PAD. , Even in many, modern, complex healthcare systems, patients with DFU are not routinely evaluated for PAD and the opportunity for diagnosis and revascularization is missed. All too often, DFUs are managed with wound care alone or even amputation (both major and minor) without any evaluation for correctable PAD, despite the well-known association of PAD with delayed wound healing and amputation in such patients. It became apparent that the dated concept of “critical limb ischemia” first proposed in 1982, as well as the most common classification systems used for decades by vascular surgeons (Fontaine and Rutherford ), failed to address numerous issues related to management of DFU. In fact, the authors of the original consensus statement on critical limb ischemia specifically stated that patients with diabetes were to be excluded from the definition as wounds in such patients were often complicated by neuropathy and infection, and the perfusion requirements to achieve healing in patients with diabetes were likely greater than in patients with foot ulcers and gangrene occurring in the setting of pure chronic ischemia from PAD seen in cigarette smokers without diabetes. The hemodynamic parameters for “critical” limb ischemia proposed in the existing classifications were likely too rigid, and both the Fontaine and Rutherford systems lacked sufficient detail about wound characteristics and failed to consider the presence and severity of infection. Both of these factors influence care and outcomes of care, especially in patients with DFU. With these considerations in mind, the Society for Vascular Surgery created and published a new classification system intended to stratify amputation risk and impact clinical management. This classification is applicable to patients with and without diabetes and is based on three major factors: wound, ischemia, and foot infection (WIfI). Since its publication in 2014, the WIfI concept has achieved broad acceptance and has been adopted and recommended by many societies across the globe including, among others, the Society for Vascular Surgery, the European Society of Vascular and Endovascular Surgery, the European Society of Cardiology, the International Working Group on the Diabetic Foot (IWGDF), the American Podiatric Medical Association, and the very recently published Global Vascular Guidelines Committee.
WIfI is a limb staging system. The underlying principle of WIfI is that the limb must be staged at presentation, prior to planning treatment, and in that way, it is analogous to the tumor, node, metastasis (TNM) system for cancer staging. Each of the three factors is graded on an objective scale from 0 to 4, a process which therefore yields 64 potential combinations of WIfI. By Delphi consensus, these combinations were grouped into one of four clinical stages (I–IV), each associated with progressively increasing risk of amputation at one year ( Tables 63.1 and 63.2 ). Although initially based on a consensus approach, when subsequently applied in clinical practice, WIfI has been shown to have considerable prognostic value in predicting amputation risk. A recent metaanalysis of 12 studies comprising 2669 patients with CLTI demonstrated that the likelihood of amputation at 1 year increased progressively with increasing WIfI stage, 0%, 8% (95% confidence interval [CI] 3%–21%), 11% (95% CI 6%–18%) and 38% (95% CI 21%–58%), for WIfI stages I–IV, respectively. Other analyses have yielded similar findings. WIfI may also be used to predict the likelihood of benefit of revascularization, although the data supporting this particular utility of WIfI are less robust. ,
Premises:
Increase in wound class increases risk of amputation (based on WIfI, PEDIS, University of Texas, and other wound classifications systems).
PAD and infection are synergistic (Eurodiale); infected wound + Pad increases likelihood revascularization will be needed to heal wound.
Infection 3 category (systemic/metabolic instability): moderate to high-risk of amputation regardless of other factors (validated IDSA infection guidelines).
|
||
| GRADE | ULCER | GANGRENE |
| 0 | No ulcer | No gangrene |
| Clinical description: ischemic rest pain (requires typical symptoms + ischemia grade 3); no wound. | ||
| 1 | Small, shallow ulcer(s) on distal leg or foot; no exposed bone, unless limited to distal phalanx | No gangrene |
| Clinical description: minor tissue loss. Salvageable with simple digital amputation (1 or 2 digits) or skin coverage. | ||
| 2 | Deeper ulcer with exposed bone, joint, or tendon; generally not involving the heel; shallow heel ulcer, without calcaneal involvement | Gangrenous changes limited to digits |
| Clinical description: major tissue loss salvageable with multiple (≥3) digital amputations or standard TMA ± skin coverage. | ||
| 3 | Extensive, deep ulcer involving forefoot and/or midfoot; deep, full thickness heel ulcer ± calcaneal involvement | Extensive gangrene involving forefoot and/or midfoot; full thickness heel necrosis ± calcaneal involvement |
| Clinical description: extensive tissue loss salvageable only with a complex foot reconstruction or nontraditional TMA (Chopart or Lisfranc); flap coverage or complex wound management needed for large soft tissue defect. | ||
| Grade | Abi | Ankle Systolic Pressure | TP, TcPO 2 |
|---|---|---|---|
| 0 | ≥0.80 | >100 mm Hg | ≥60 mm Hg |
| 1 | 0.6–0.79 | 70–100 mm Hg | 40–59 mm Hg |
| 2 | 0.4–0.59 | 50–70 mm Hg | 30–39 mm Hg |
| 3 | ≤0.39 | <50 mm Hg | <30 mm Hg |
| Clinical Manifestation of Infection | SVS | IDSA/Pedis Infection Severity |
|---|---|---|
| No symptoms or signs of infection Infection present, as defined by the presence of at least two of the following items:
|
0 | Uninfected |
| Local infection involving only the skin and the subcutaneous tissue (without involvement of deeper tissues and without systemic signs as described below). Exclude other causes of an inflammatory response of the skin (e.g., trauma, gout, acute Charcot neuroosteoarthropathy, fracture, thrombosis, venous stasis) |
1 | Mild |
| Local infection (as described above) with erythema >2 cm, or involving structures deeper than skin and subcutaneous tissues (e.g., abscess, osteomyelitis, septic arthritis, fasciitis), and No systemic inflammatory response signs (as described below) | 2 | Moderate |
Local infection (as described above) with the signs of SIRS, as manifested by two or more of the following:
|
3 | Severe ∗ |
TMA, Transmetatarsal amputation.
I: Ischemia
Hemodynamics/perfusion: Measure TP or TcPO 2 if ABI incompressible (>1.3)
SVS grades 0 (none), 1 (mild), 2 (moderate), and 3 (severe).
ABI, Ankle-brachial index; PVR, pulse volume recording; SPP, skin perfusion pressure; TP, toe pressure; TcPO 2 , transcutaneous oximetry.
Patients with diabetes should have TP measurements. If arterial calcification precludes reliable ABI or TP measurements, ischemia should be documented by TcPO 2 , SPP, or PVR. If TP and ABI measurements result in different grades, TP will be the primary determinant of ischemia grade.
Flat or minimally pulsatile forefoot PVR = grade 3.
fI: foot Infection:
SVS grades 0 (none), 1 (mild), 2 (moderate), and 3 (severe: limb and/or life-threatening)
SVS adaptation of Infectious Diseases Society of America (IDSA) and International Working Group on the Diabetic Foot (IWGDF) perfusion, extent/size, depth/tissue loss, infection, sensation (PEDIS) classifications of diabetic foot infection.
∗ Ischemia may complicate and increase the severity of any infection. Systemic infection may sometimes manifest with other clinical findings, such as hypotension, confusion, vomiting, or evidence of metabolic disturbances, such as acidosis, severe hyperglycemia, new-onset azotemia.
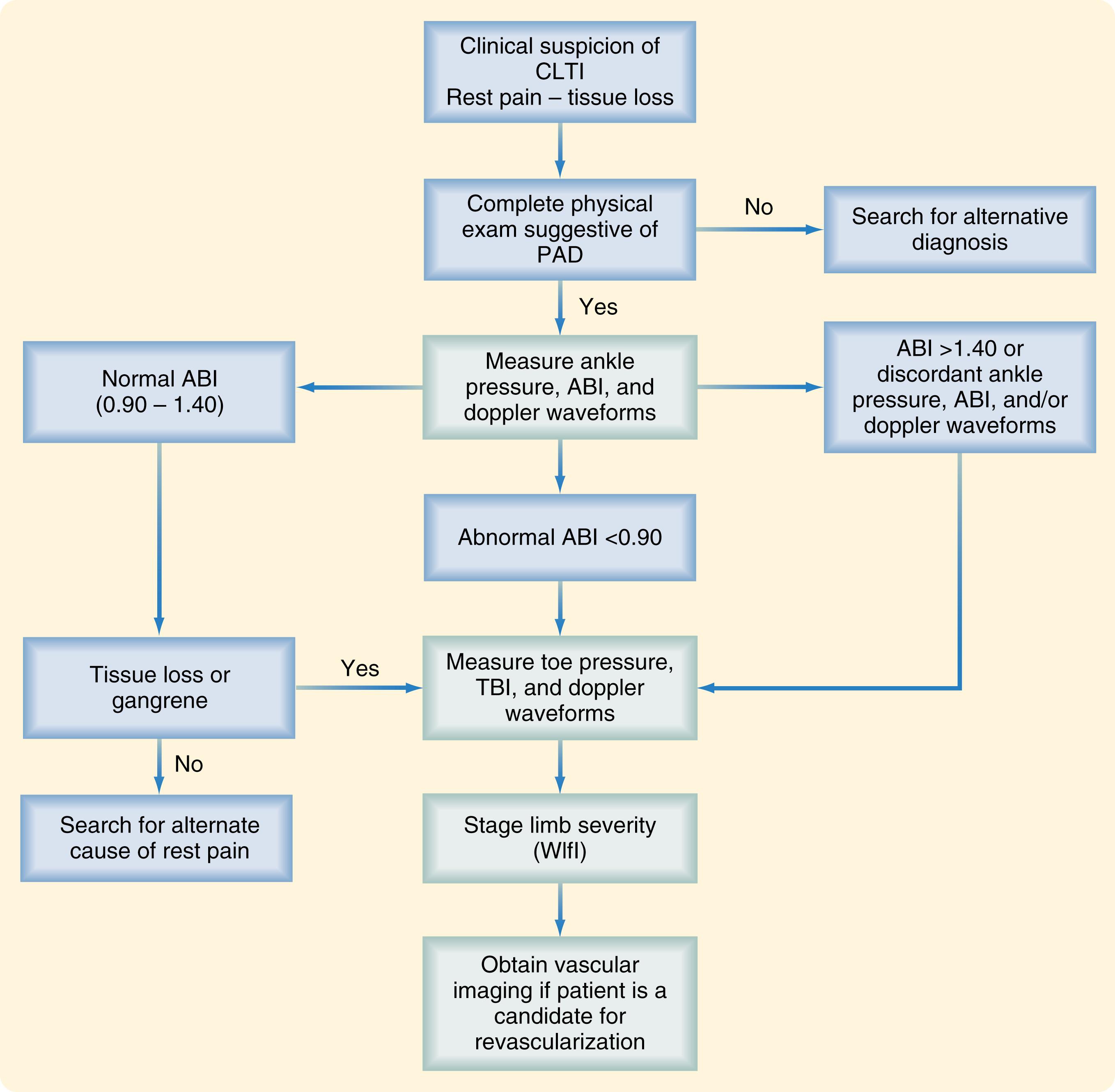
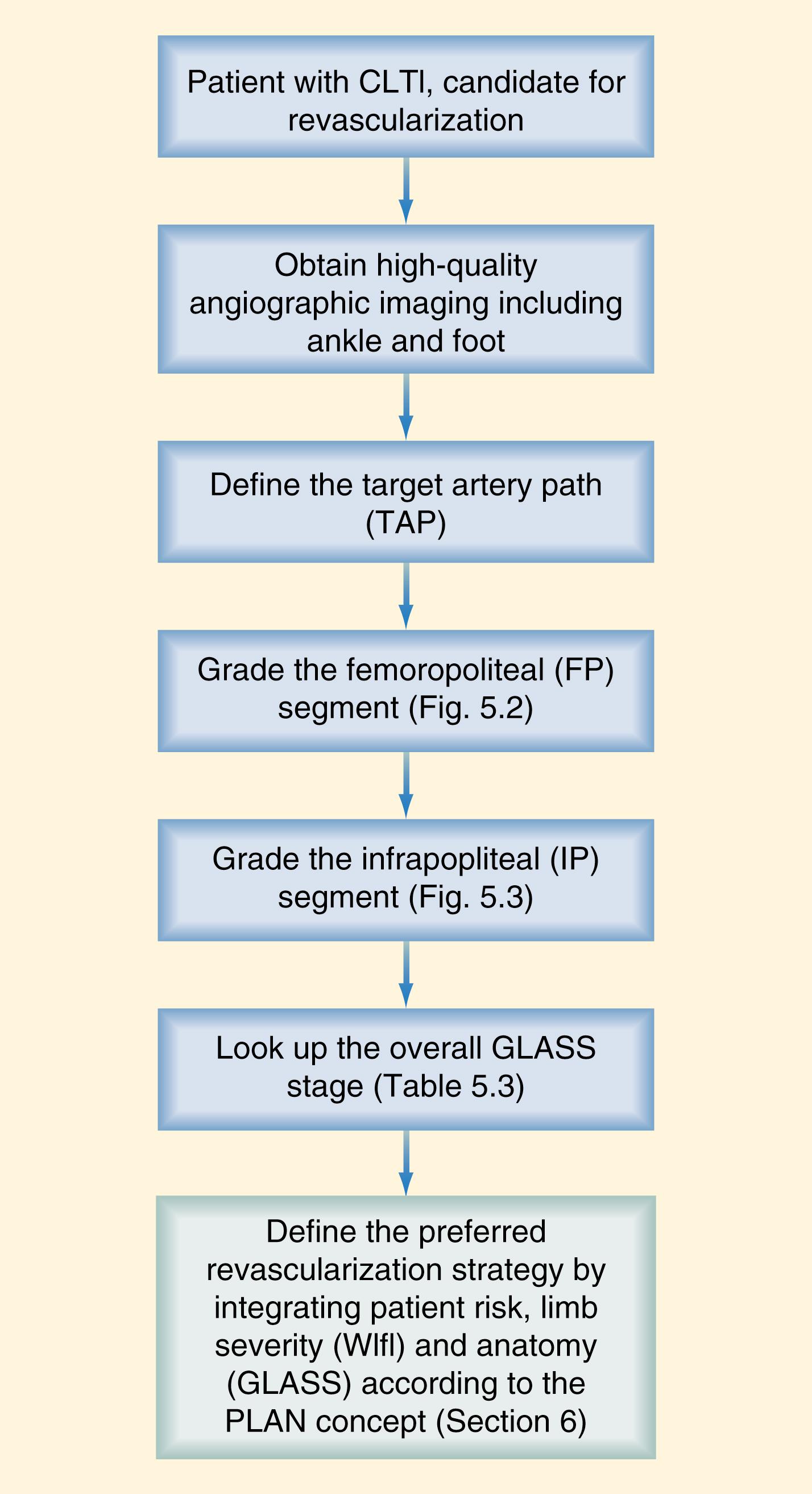
The limb itself, however, is only one issue to be considered in the treatment of patients with CLTI. Patient risk factors, life expectancy, and the underlying vascular anatomy in patients felt to benefit from revascularization also constitute integral components of the decision-making process. The Global Vascular Guidelines on CLTI were recently published in an effort to initiate evidence-based guidelines for the diagnosis, evaluation, and management of such patients. The guidelines include an expanded and more modern definition of CLTI, are based upon a three-step process that includes limb staging with WIfI, and introduce three other new concepts: patient, limb, anatomy (PLAN), target artery path (TAP), and Global Limb Anatomic Staging System (GLASS). These guidelines are succinctly summarized below as they represent an important advance in CLTI care.
The Global Vascular Guideline begins by establishing important definitions and nomenclature. The Global Vascular Guidelines suggest abandoning the outdated term “critical limb ischemia” as it fails to encompass the complete spectrum of patients in modern day practice who are evaluated and treated to prevent limb amputation. Instead, the term CLTI is now proposed so as to include a much broader spectrum of patients with varying degrees of ischemia sufficient to contribute to the development of foot and leg ulcers, delay healing, and increase amputation risk. CLTI includes only patients with chronic atherosclerotic disease and is not intended to be applied to patients with acute thrombotic or embolic leg ischemia, trauma, pure venous disease, or nonatherosclerotic conditions such as the vasculitides and Buerger disease. The target population therefore includes any adult with CLTI, defined as a patient with objectively documented PAD and any of the following clinical presentations: ischemic rest pain with confirmatory hemodynamic measurements; DFU or any lower limb ulcer present for at least two weeks; and gangrene involving any part of the lower limb or foot. CLTI is thus a more inclusive and well-defined term that can be more appropriately applied to the spectrum of patients presenting with limb threat than the dated or imprecise terms, critical limb ischemia or severe limb ischemia.
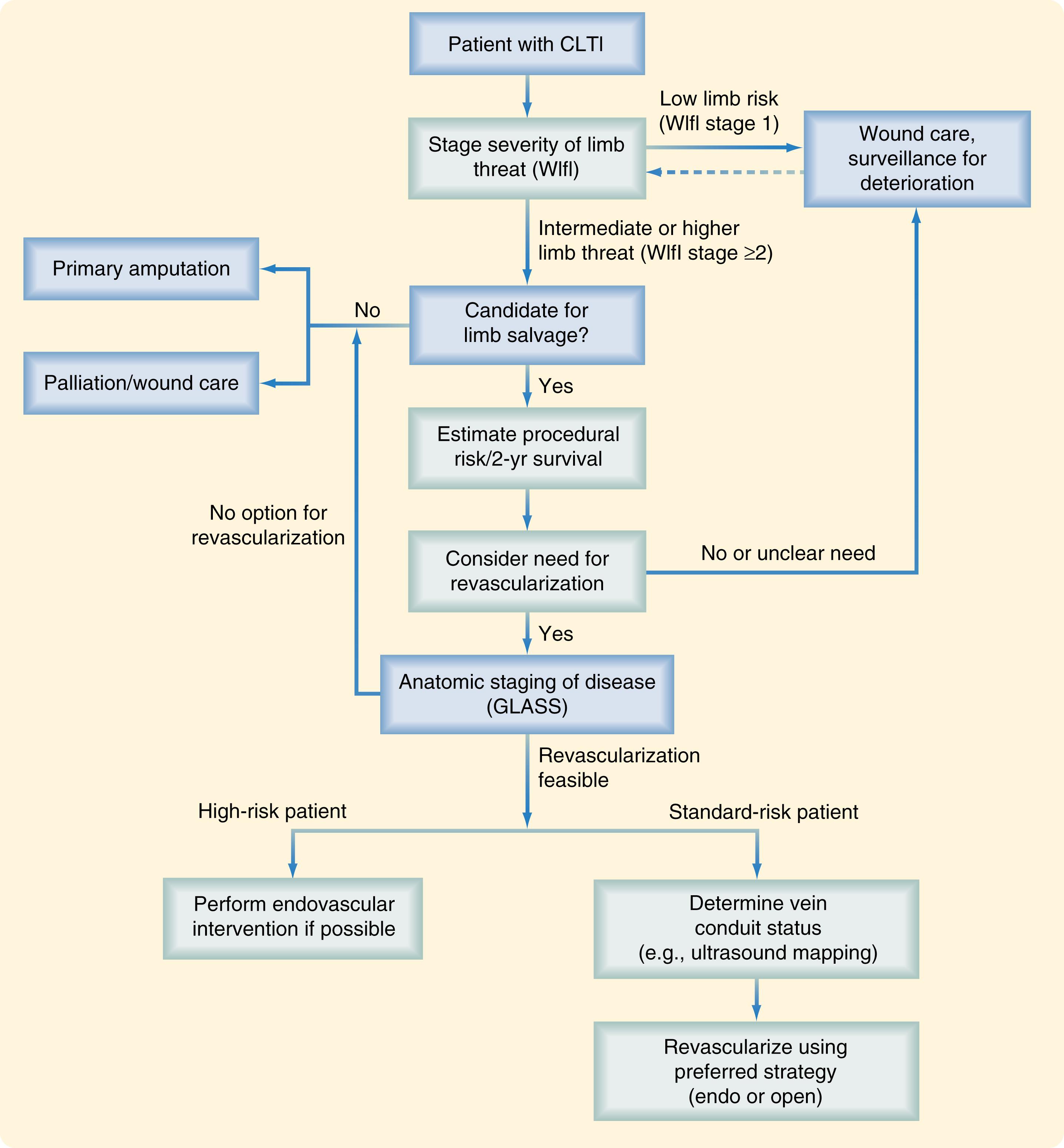
Given a patient presenting with any of the above manifestations of CLTI, the next step is to stage the limb with the WIfI classification system, which provides an evidence-based estimation of the degree of limb threat and helps focus limb salvage efforts.
PLAN includes a focus on the patient and his/her estimated risk for intervention, in particular estimates of short- and long-term mortality. In contrast to patients with CLTI, patients with claudication are generally at lower risk, with a predicted 75% to 80% 5-year life expectancy and a 5-year risk of amputation of only 5%. Interventions should only be undertaken in claudicants after failure of exercise and medical therapy, when anatomic factors are favorable for intervention, and prolonged patency and symptom relief is likely. In contrast, CLTI patients overall have a 50% 5-year mortality, but also a much higher amputation risk, based primarily on the limb stage at presentation. Simons and associates suggested that CLTI patients could be grouped into three risk groups for mortality based on a combination of factors including age older than 80 years, oxygen-dependent chronic obstructive lung disease, stage 5 chronic kidney disease, and bedbound status. Using a predictive model based on these factors from a large cohort of over 38,000 patients derived from the Vascular Quality Initiative, patients were defined as low-risk (30-day survival >97% and 2-year survival >70%), medium-risk (30-day survival 95%–97% or 2-year survival 50%–70%), or high-risk (30-day survival <95% or 2-year survival <50%). These data and those from the BASIL trial were considered in the Global Guidelines and it was recommended that CLTI patients be grouped into average (anticipated periprocedural mortality <5% and estimated 2-year survival >50%) and high-risk groups (anticipated periprocedural mortality ≥5% and estimated 2-year survival ≤50%). One of the reasons for this adjustment was because the BASIL trial had shown that EVT for severe limb ischemia patients had similar outcomes compared to open surgical bypass for patients living less than two years from their initial revascularization attempt, whereas patients living longer than two years appeared to benefit from bypass surgery. , Other factors have also been examined including functional status and frailty, but the data in CLTI patients in predicting perioperative and long-term survival is not yet well defined. The final component of PLAN, after assessing individual patient risk and WIfI limb stage, is to evaluate the arterial anatomy in those patients in need of revascularization and who would be candidates for revascularization. These three pieces are then considered to formulate a revascularization strategy ( Fig. 63.5 ).
To define the arterial anatomy, many groups start with duplex imaging because it can be done in the office, is relatively inexpensive, and requires neither an intravenous catheter nor contrast administration. Computed tomography angiography and magnetic resonance angiography can be considered, especially when inflow disease (aortoiliac disease) is suspected. Most commonly, patients are evaluated with catheter-based digital subtraction angiography because it is the most direct method and offers the best views of the foot ( Fig. 63.6 ). Studies which do not include the foot in CLTI patients are inadequate. , Contrast can be diluted in patients with kidney disease and CO 2 arteriography offers excellent views of the proximal vessels down the popliteal level and is therefore often used to limit contrast in patients with significant baseline renal impairment.
![Fig. 63.6, Suggested algorithm for anatomic imaging in patients with chronic limb-threatening ischemia (CLTI) who are candidates for revascularization. In some cases, it may be appropriate to proceed directly to angiographic imaging (computed tomography angiography [CTA], magnetic resonance angiography [MRA], or catheter) rather than to duplex ultrasound (DUS) imaging. Fig. 63.6, Suggested algorithm for anatomic imaging in patients with chronic limb-threatening ischemia (CLTI) who are candidates for revascularization. In some cases, it may be appropriate to proceed directly to angiographic imaging (computed tomography angiography [CTA], magnetic resonance angiography [MRA], or catheter) rather than to duplex ultrasound (DUS) imaging.](https://storage.googleapis.com/dl.dentistrykey.com/clinical/PeripheralArterialDisease/4_3s20B9780323640626000633.jpg)
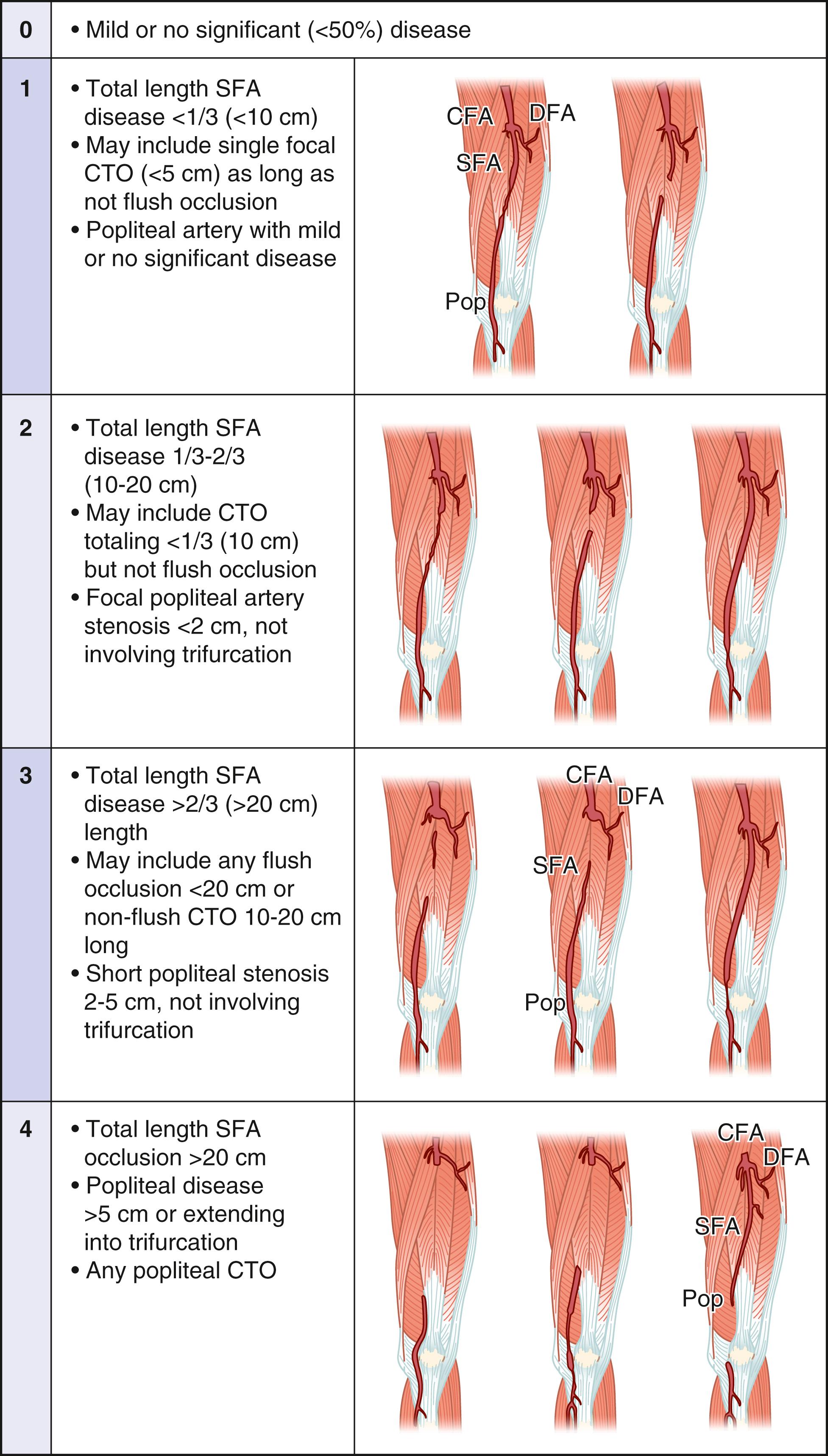
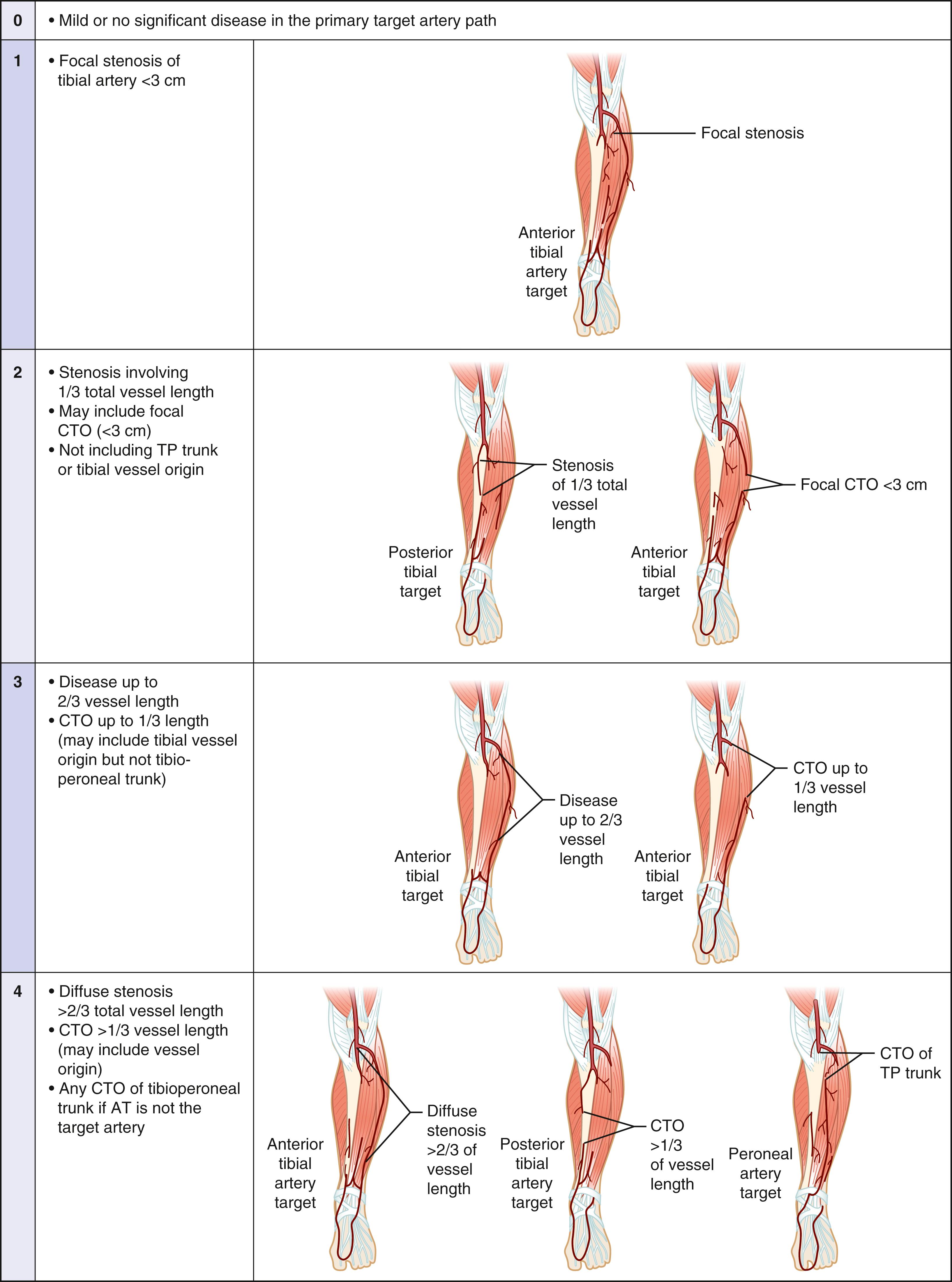
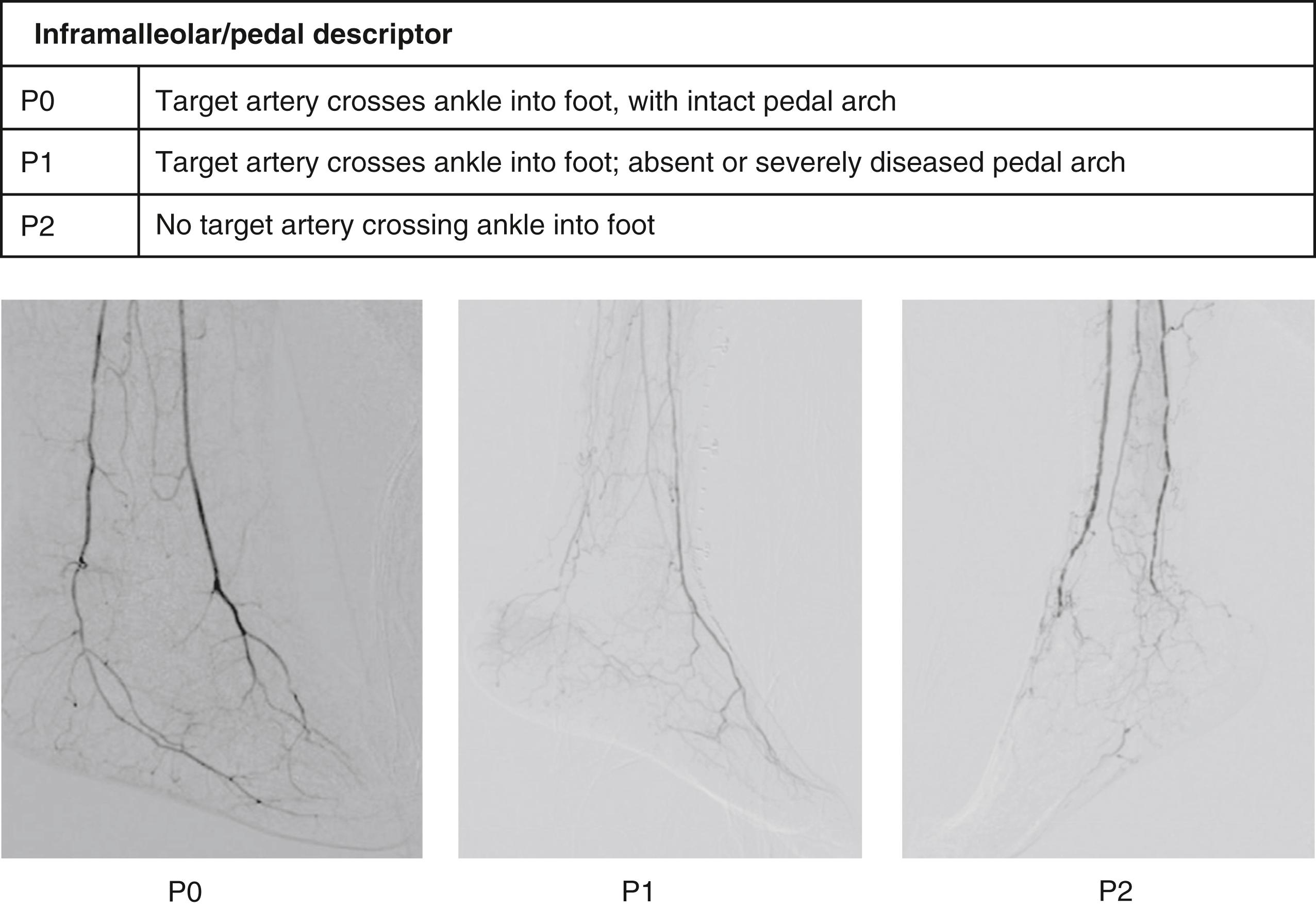
Any inflow disease, if present, must be corrected, most commonly with angioplasty with or without stenting. The entire affected limb is evaluated angiographically and classified using the GLASS. GLASS classifies the limb disease burden from the groin to the foot, with the underlying assumption that inflow disease is not present or has already been corrected. GLASS was designed to address inconsistencies and the overall lack of utility of the older, TransAtlantic Inter-Society Consensus (TASC) I and II anatomic classification systems, which were lesion and arterial segment based and did not correlate with expected patency rates and outcomes of therapy. There are several underlying key assumptions and principles defined by GLASS. The first is that in-line flow to the ankle and the foot is a primary goal of therapy, and to accomplish that, one must select a TAP. The TAP is selected by the operator and is a continuous route of in-line flow from groin to foot. Assessment of patency is limb based, not lesion or segment based. The femoropopliteal and infrapopliteal segments are each graded in severity on a scale from 0 to 4 based on length and other important characteristics of the stenosis or occlusion, such as whether or not the origin of the vessel is involved and whether or not significant calcification is present ( Figs. 63.7 and 63.8 ). Pedal anatomy is used as a modifier/descriptor ( Fig. 63.9 ). CLTI is most often a multilevel disease, so GLASS combines the grades of the infrainguinal segments to create an arterial anatomic stage, analogous to the way in which the WIfI system is used to stage the limb itself. Stages range in progressive severity from I to III, based on consensus estimates of the estimated technical failure rates and 1-year limb-based patency ( Table 63.3 ). The system is geared toward an endovascular approach but should allow meaningful comparison with open bypass surgery for comparable WIfI limb stage and GLASS arterial anatomic stages. Although based on current best evidence and expert consensus, the GLASS classification has not yet been validated. However, neither were the TASC systems it is intended to replace and GLASS makes more clinical sense because the entire limb is staged from an arterial anatomic standpoint and it considers that more than one level of disease may have to be treated to obtain in-line flow to heal the foot ( Fig. 63.10 ). Going forward, it is anticipated that the combination of patient risk, WIfI limb stage, and GLASS anatomic stage can be put together to predict the benefit of revascularization and the best means of accomplishing it (i.e., EVT vs. bypass, see Figs. 63.11 and 63.12 )
| Infrainguinal GLASS Stage (I–III) | ||||||
|---|---|---|---|---|---|---|
| FP Grade | 4 | III | III | III | III | III |
| 3 | II | II | II | III | III | |
| 2 | I | II | II | II | III | |
| 1 | I | I | II | II | III | |
| 0 | NA | I | I | II | III | |
| 0 | 1 | 2 | 3 | 4 | ||
| IP Grade | ||||||
![Fig. 63.12, Preferred initial revascularization strategy for infrainguinal disease in average-risk patients with suitable autologous vein conduit available for bypass. Revascularization is considered rarely indicated in limbs at low risk (wound, ischemia, and foot infection [WIfI] stage 1). Anatomic stage (y-axis) is determined by the Global Limb Anatomic Staging System (GLASS); limb risk (x-axis) is determined by WIfI staging. The dark gray shading indicates scenarios with least consensus (assumptions inflow disease either is not significant or is corrected; absence of severe pedal disease (i.e., no GLASS P2 modifier). Fig. 63.12, Preferred initial revascularization strategy for infrainguinal disease in average-risk patients with suitable autologous vein conduit available for bypass. Revascularization is considered rarely indicated in limbs at low risk (wound, ischemia, and foot infection [WIfI] stage 1). Anatomic stage (y-axis) is determined by the Global Limb Anatomic Staging System (GLASS); limb risk (x-axis) is determined by WIfI staging. The dark gray shading indicates scenarios with least consensus (assumptions inflow disease either is not significant or is corrected; absence of severe pedal disease (i.e., no GLASS P2 modifier).](https://storage.googleapis.com/dl.dentistrykey.com/clinical/PeripheralArterialDisease/10_3s20B9780323640626000633.jpg)
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here