Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
ALTHOUGH CHILDHOOD CANCER is not particularly common, it is the second most common cause of death in children younger than 15 years of age. The most common malignancies affecting children are different from those affecting adults. Leukemia, brain tumors, lymphomas, and sarcomas of tissue and bone are the most common pediatric cancers and account for over 50% of all pediatric malignancies ( Table 11.1 ). Embryonal tumors (e.g., neuroblastoma, Wilms tumor, retinoblastoma, medulloblastoma) are unique to early childhood, which underscores the need for these children to be cared for in pediatric centers with sufficient expertise in their management. Survival rates for most pediatric cancers have improved significantly in the past several decades; more than 80% of children diagnosed with a childhood malignancy will become 5-year survivors of their cancers, and 5-year survival rates for acute lymphoblastic leukemia (ALL) have now increased to nearly 90%. The great improvements in survival for many malignancies of childhood are directly related to advances in diagnostic modalities and the large percentage of children treated in cooperative clinical trial protocols. Treatment options in these protocols include chemotherapy, radiation therapy, biologic modifiers, hematopoietic stem cell transplantation (HSCT), and adoptive T-cell therapies.
| Type of Cancer | INCIDENCE (%) BY AGE GROUP | ||||
|---|---|---|---|---|---|
| 0–4 years | 5–9 years | 10–14 years | 15–19 years | 0–19 years | |
| Leukemias | 36.1 | 33.4 | 21.8 | 12.4 | 25.2 |
| Central nervous system tumors | 16.6 | 27.7 | 19.6 | 9.5 | 16.7 |
| Lymphomas | 3.9 | 12.9 | 20.6 | 25.1 | 15.5 |
| Carcinomas and other malignant epithelial tumors | 0.9 | 2.5 | 8.9 | 20.9 | 9.2 |
| Soft tissue sarcomas | 5.6 | 7.5 | 9.1 | 8.0 | 7.4 |
| Germ cell, trophoblastic, and other gonadal tumors | 3.3 | 2.0 | 5.3 | 13.9 | 7.0 |
| Malignant bone tumors | 0.6 | 4.6 | 11.3 | 7.7 | 5.6 |
| Sympathetic nervous system tumors | 14.3 | 2.7 | 1.2 | 0.5 | 5.4 |
| Renal tumors | 9.7 | 5.4 | 1.1 | 0.6 | 4.4 |
| Retinoblastoma | 6.3 | 0.5 | 0.1 | 0.0 | 2.1 |
| Hepatic tumors | 2.2 | 0.4 | 0.6 | 0.6 | 1.1 |
| Other and unspecified malignant neoplasms | 0.5 | 0.3 | 0.6 | 0.8 | 0.6 |
Children with cancer typically undergo many surgical procedures that require anesthesia. The procedures may occur before or during the cancer therapy, years into remission, or during terminal stages of the disease. Certain considerations apply to this population, including the direct effects of the tumor, effects of the chemotherapy and radiation therapy, impact of the surgical procedure, pain syndromes, and psychological vulnerabilities of the child and family. Children undergoing active treatment for cancer range from gravely ill to relatively healthy with excellent functional capacity. Survivors of childhood cancer experience various long-term and often debilitating sequelae after completion of cancer therapy. In one report, 62% of survivors of childhood cancer reported at least one chronic health condition from cancer, and 28% reported a severe or life-threatening condition. These chronic health conditions may impact nearly every organ system and have considerable bearing on any anesthetic plan years into remission. The field of pediatric oncology is extensive, complicated, and ever-changing. Multidisciplinary communication in the perioperative period is crucial to ensure the safe care of these complex patients.
Most pediatric cancers are treated with an aggressive multimodal approach that may include surgical resection, radiation therapy, and chemotherapy for control of local and metastatic disease. Equally important is supportive care to ensure minimization of toxicity from the tumor and treatment therapy, including nutritional, emotional, and psychological support. A brief review of these therapies with pertinence to the perioperative period follows, but additional sources are available concerning the current indications, side effects, and precautions of the available chemotherapeutic drugs.
Conventional chemotherapeutics, along with radiation therapy, have been the mainstay of pediatric antitumor treatment for decades. In the modern era of multidrug combination chemotherapy, molecularly targeted therapies, radiation therapy, rescue therapies, and HSCT, anticipation of precise toxicities from any given therapy in an individual patient is difficult. However, treatment protocols include a potentially vast array of antitumor agents with widely divergent mechanisms of action and toxicity profiles, and the average child with cancer will be at risk for focal or widespread toxicity to nearly every organ system. Most chemotherapeutic agents are cytotoxic to rapidly dividing cells via several mechanisms. Because these agents lack specificity to tumor cells alone, the impact to healthy tissues is unavoidable and often limits effective chemotherapy dosing schedules. Concurrent administration of several chemotherapeutics with non-overlapping toxicity profiles and at lower doses lessens the additive toxicity while also increasing the simultaneous cytotoxic attack on tumor cells. Regardless, bone marrow suppression, immunosuppression, myocardial toxicity, pulmonary toxicity, and dysfunction of nearly every other organ system are possible in children with cancer and must be thoroughly considered when evaluating and caring for these children. These toxicities range from short-term to lifelong effects.
Several chemotherapeutic agents are of specific interest to the anesthesiologist caring for a child with cancer. Anthracycline chemotherapeutics (e.g., doxorubicin, daunorubicin, idarubicin, and epirubicin) and mitoxantrone cause cardiomyopathy in a dose-dependent fashion, an effect exacerbated by mediastinal irradiation. l -Asparaginase is associated with a 1% to 2% risk of hemorrhage or thrombosis owing to deficiencies in fibrinogen, plasminogen, antithrombin III, and von Willebrand factor, as well as hepatic dysfunction and acute hemorrhagic pancreatitis. Bleomycin may cause acute pneumonitis with progression to pulmonary fibrosis. Cisplatin and ifosfamide may cause renal tubular damage that can lead to Fanconi syndrome with electrolyte wasting. In large doses, methotrexate (>1 g/m 2 ) may cause renal failure. Corticosteroids may be directly cytotoxic to some hematopoietic tumors by inducing apoptosis, as well as causing adrenal suppression, hypertension, thromboembolism, and obesity. Specific toxicities of chemotherapeutic agents pertinent to anesthesia are discussed in detail below; Table 11.2 provides a brief list of major toxicities of the traditional chemotherapy agents. E-Table 11.1 provides a comprehensive list of traditional chemotherapeutic agents, molecularly targeted agents, and adjuvant medications used in children, with their corresponding toxicities.
| Drug | Adverse Effects |
|---|---|
| l -Asparaginase | Hyperglycemia, hypersensitivity, hepatic dysfunction (secondary hypoalbuminemia and coagulopathies), pancreatitis, thrombosis, stroke |
| Bischloroethyl nitrosourea (BCNU) | Encephalopathy, hepatotoxicity, pulmonary toxicity |
| Bleomycin | Anaphylactoid reactions, fever, hyperpigmentation, nausea, vomiting, oxygen toxicity, pulmonary fibrosis |
| Busulfan | Encephalopathy, hepatotoxicity, pulmonary toxicity |
| Carboplatin | Myelosuppression, nausea, vomiting, nephrotoxicity, neurotoxicity, ototoxicity |
| Cisplatin | Nausea/vomiting, nephrotoxicity, ototoxicity, peripheral neuropathy |
| Corticosteroids | Adrenal suppression, avascular necrosis, cataracts, edema, gastritis, hyperglycemia, hypertension, myopathy, osteoporosis, obesity, osteopenia, tumor lysis syndrome, tumor necrosis, psychosis |
| Cyclophosphamide (Cytoxan) | Cardiotoxicity, hemorrhagic cystitis, myelosuppression, nausea, vomiting, syndrome of inappropriate secretion of antidiuretic hormone (SIADH) |
| Cyclosporine | Cortical blindness, electrolyte disturbances, encephalopathy, gingival hyperplasia, hemolytic uremia, hepatotoxicity, hyperlipidemia, hypertension, hirsutism, myositis, paresthesias, tremor |
| Cytarabine | Myelosuppression, mucositis, hepatitis, nausea/vomiting, neurotoxicity |
| Dactinomycin (Actinomycin D) | Nausea/vomiting, mucositis, myelosuppression, radiation recall? a |
| Daunorubicin (Daunomycin) Doxorubicin (Adriamycin) Idarubicin (Idamycin) | Cardiomyopathy, mucositis, myelosuppression, red-orange urine |
| Etoposide | Hypotension, mucositis, myelosuppression, nausea, vomiting |
| Ifosfamide | Hemorrhagic cystitis, myelosuppression, nephrotoxicity, neurotoxicity |
| Melphalan | Mucositis |
| Methotrexate | Hepatotoxicity, mucositis, myelosuppression, renal failure, neurotoxicity |
| Mercaptopurine (6-MP) | Hepatotoxicity, myelosuppression |
| Mycophenolate mofetil (CellCept) | Electrolyte disturbance, gastrointestinal toxicity, hypercholesterolemia, myelosuppression, rash |
| Procarbazine | Myelosuppression |
| Sirolimus | Hyperlipidemia, myelosuppression |
| Tacrolimus (Prograf) | Anemia, anorexia, back pain, encephalopathy, diarrhea, hyperglycemia, nephrotoxicity, pleural effusion, rash |
| Thiotepa | Neurotoxicity, mucositis |
| Thioguanine (6-TG) | Hepatotoxicity, myelosuppression |
| Total body irradiation | Dental/bony maldevelopment, gastrointestinal toxicity, hepatotoxicity, pulmonary toxicity |
| Vinblastine (Velban) | Myelosuppression, neurotoxicity, SIADH |
| Vincristine (Oncovin) | Neurotoxicity, SIADH |
a Radiation recall: the “recalling” by skin of previous radiation exposure in response to the administration of certain response-inducing drugs.
| Drug a | Myelotoxicity | Cardiotoxicity | Pulmonary Toxicity | Nephrotoxicity | Hepatotoxicity | GI Toxicity | Neurotoxicity | Other |
|---|---|---|---|---|---|---|---|---|
| Alemtuzumab | ++ | − | − | − | − | + (N/V/D/M) | − | |
| Altretamine | ++ | − | − | − | + (LFT↑) | ++ (N/V) | + | |
| Arsenic trioxide | + (Leukocytosis) | ++ (Prolonged QT interval) | + (Effusion) | − | − | + (N/V/D) | +/− | Differentiation syndrome |
| l -Asparaginase | + (Bleeding) | − | − | − | + (LFT↑) | + (Pancreatitis) | − | Coagulopathy |
| 5-Azacitidine | +++ | − | − | − | + (LFT↑) | + (N/V/D) | − | |
| Bevacizumab | − | ++ (HTN, CHF) | − | + (Nephrot i c syndrome) | − | + (N/V/D/M) | + (Asthenia) | |
| Bleomycin | − | − | +++ | + | + | +/− | − | |
| Bortezomib | +/− | + (N/V/D) | + | Fever common | ||||
| Busulfan | +++ | − | ++ | − | ++ (LFT↑, VOD) | + (N/V) | + (Seizures) | Electrolyte abnormalities |
| Capecitabine | ++ | + (In adults) | − | − | + (LFT↑) | ++ (D/M) | ++ | |
| Carboplatin | +++ | − | − | + | − | ++ | ++ | |
| Carmustine | +++ | − | +++ | + | + (LFT↑) | + (N/V) | − | |
| Cetuximab | − | − | + (Rare) | − | − | − | − | Rash, hypomagnesemia |
| Chlorambucil | +++ | − | ++ (Rare) | − | − | − | ++ (Rare) | |
| Cisplatin | − | − | − | +++ | − | ++ (N/V) | ++ | SIADH |
| Cladribine | +++ | − | − | + (At high doses) | − | − | + (At high doses) | Tumor lysis syndrome |
| Clofarabine | +++ | − | − | + (Rare) | ++ (LFT↑) | ++ (N/V) | − | |
| Cyclophosphamide | +++ | ++ (At high doses) | − | ++ (Hemorrhagic cystitis) | − | − | − | SIADH |
| Cytarabine | +++ | − | ++ (At high doses) | − | + (LFT↑) | +++ (Pancreatitis) | ++ | Ara-C syndrome |
| Dacarbazine | +++ | − | − | − | − | ++ (N/V) | ++ (Rare) | |
| Dactinomycin | +++ | ++ (LFT↑, VOD) | +++ (N/V/D/M) | |||||
| Darbepoetin alfa | − | + (HTN) | − | − | − | − | − | |
| Dasatinib | +++ | ++ (Arrhythmias) | − | − | − | ++ (N/V/D) | − | Bleeding disorders |
| Daunorubicin | +++ | +++ | − | − | − | − | − | Skin disorders |
| Decitabine | +++ | − | + (Cough, edema) | − | + (LFT↑) | ++ (N/V/D) | − | |
| Denileukin diftitox | − | ++ (Vascular leak syndrome) | − | + (Rare) | + (Rare) | − | + (Rare) | |
| Dexamethasone | + (Leukocytosis) | − | − | − | − | − | − | Adrenal suppression |
| Docetaxel | +++ | − | − | − | + (LFT↑) | − | + (Uncommon) | Fluid retention |
| Doxorubicin | +++ | +++ | − | − | − | − | − | Skin disorders |
| Epirubicin | +++ | +++ | − | − | − | − | − | Skin rash |
| Erlotinib | − | − | +++ (ILD, rare) | − | + (LFT↑) | + (D) | − | |
| Erythropoietin | − | + (HTN) | − | − | − | − | − | |
| Estramustine | − | +++ (Rare) | − | − | − | +++ (N/V) | − | |
| Etoposide | +++ | − | − | − | + (Rare) | + (N/V) | + (Rare) | |
| Filgrastim | − | − | − | − | − | − | − | Bone pain |
| Floxuridine | ++ | ++ (Rare) | − | − | +++ | ++ (Ulcers) | + (Rare) | |
| Fludarabine | +++ | − | − | − | − | − | +++ (Uncommon) | Autoimmune hemolytic anemia |
| 5-Fluorouracil | +++ | ++ (Rare) | − | − | − | +++ (D/M) | +++ (Uncommon) | |
| Gefitinib | − | + (HTN) | +++ (ILD, rare) | − | + (LFT↑) | − | − | Rash |
| Gemcitabine | +++ | − | +++ (ILD, rare) | + (Hematuria) | + (LFT↑) | + (N/V) | + (Rare) | |
| Gemtuzumab ozogamicin | +++ | − | − | − | ++ (LFT↑, VOD) | − | − | |
| Hydroxyurea | +++ | − | − | − | − | − | − | Skin disorders |
| Ibritumomab tiuxetan | +++ | − | − | − | − | − | + (Asthenia) | Infusion reactions |
| Idarubicin | +++ | +++ | − | − | + (LFT↑) | + (N/V/D/M) | − | Skin disorders |
| Ifosfamide | +++ | − | +++ (Uncommon) | +++ (Hemorrhagic cystitis, Fanconi-like syndrome) | + (LFT↑) | +++ (N/V) | ++ | SIADH |
| Imatinib mesylate | + | − | − | − | + (LFT↑) | + (D) | − | |
| Interferonalfa | ++ | ++ (Rare) | − | − | + (LFT↑) | − | − | Autoimmune symptoms |
| Interleukin 2 | + | ++ (Vascular leak syndrome) | − | − | + (LFT↑) | − | + | |
| Irinotecan | +++ | − | − | − | + (LFT↑) | ++ (D) | + (Asthenia) | Electrolyte abnormalities |
| Lapatinib | + | − | − | − | + (LFT↑) | ++ (N/V/D) | − | |
| Lenalidomide | ++ | − | − | − | − | + (D) | − | Thrombosis |
| Leucovorin | − | − | − | − | − | + (N/V) | − | Rash |
| Lomustine | +++ | − | +++ (Uncommon) | +++ (Uncommon) | − | + (N/V) | − | |
| Mechlorethamine | +++ | − | − | − | ++ | +++ (N/V) | + (Rare) | |
| Melphalan | +++ | − | ++ (Rare) | − | − | + (N/V/D/M) | − | SIADH (Rare) |
| Mercaptopurine | +++ | − | − | − | + (LFT↑) | + (N/V/D/M) | − | Skin disorders |
| Mesna | − | − | − | − | − | + (N/V) | − | Rash, arthralgias |
| Methotrexate | +++ | − | +++ | ++ (Uncommon) | − | ++ (M/D) | ++ (At high doses) | |
| Mitomycin C | +++ | − | +++ (Rare) | +++ (HUS, rare) | +++ (VOD, rare) | + (N/V/M) | − | |
| Mitoxantrone | +++ | +++ | − | − | + (LFT↑) | + (N/V/D/M) | − | |
| Nelarabine | +++ | − | + (Uncommon) | − | − | + (N/V/D) | +++ (Common) | Edema |
| Oprelvekin | − | +++ | +++ (Pleural effusions, dyspnea) | − | − | + (N/V/D/M) | + | Edema |
| Oxaliplatin | ++ | − | +++ (Rare) | +++ (Rare) | +++ (VOD, rare) | ++ (N/V/D) | +++ (Common) | Hypersensitivity reactions |
| Paclitaxel | +++ | + (Arrhythmia) | − | − | + (LFT↑) | + (N/V/D/M) | +++ (Common) | Arthralgias |
| Panitumumab | − | − | +++ (Rare) | − | − | + (N/V/D) | − | Infusion reactions |
| Pegaspargase-asparaginase | − | − | − | − | +++ (Pancreatitis, uncommon) | − | − | Thrombosis, glucose intolerance |
| Pegfilgrastim | − | − | − | − | + (LFT↑) | − | − | Bone pain |
| Pemetrexed | +++ | − | − | − | − | ++ (N/V/D/M) | + | |
| Pentostatin | +++ | + (Rare) | − | − | + (LFT↑) | + (N/V) | + | Hypersensitivity reactions |
| Prednisone | + (Leukocytosis) | − | − | − | − | − | − | Adrenal suppression |
| Procarbazine | +++ | − | +++ (ILD, rare) | − | − | + (N/V) | ++ | Hypersensitivity reactions |
| Rituximab | + | + (Rare) | − | − | − | − | + | Infusion reactions, tumor lysis syndrome |
| Sargramostim | − | + (Arrhythmias, uncommon) | − | − | + (LFT↑) | − | − | Bone pain, infusion reactions |
| Sorafenib | + | ++ (Adults, rare) | + (LFT↑) | + (N/V/D) | + | Skin disorders | ||
| Streptozocin | + | − | − | +++ (Azotemia) | + (LFT↑) | +++ (N/V) | − | Glucose imbalance |
| Sunitinib | +++ | + (HTN ↓) | − | − | + (LFT↑) | ++ (N/V/D/M) | − | Bleeding disorder |
| Temozolomide | +++ | − | − | − | + (LFT↑) | ++ (N/V/D) | − | |
| Teniposide | +++ | − | − | − | − | + (N/V/M) | + | Hypersensitivity reaction |
| Thalidomide | − | − | − | − | − | + | +++ | Thrombosis, teratogenesis |
| Thioguanine | +++ | − | − | + (Rare) | +++ (VOD) | +++ (D/M) | + (Rare) | |
| Thiotepa | +++ | − | − | − | − | + (N/V) | − | |
| Topotecan | +++ | − | − | + (Hematuria) | + (LFT↑) | ++ (N/V/D) | − | |
| Tositumomab | +++ | − | − | − | − | + (N/V) | − | Infusion reactions |
| Trastuzumab | − | ++ (CHF) | + (Rare) | − | − | + (N/V/D) | − | Infusion reactions |
| Tretinoin (all- trans retinoic acid) | +++ (Leukocytosis) | +++ (RAS) | +++ (RAS) | + (Rare) | + (LFT↑) | + (N/V) | + (Common), +++ (rare) | RAS |
| Vinblastine | +++ | ++ (Rare) | +++ (Rare) | − | − | ++ (N/V/D/M) | + (Common), +++ (rare) | SIADH (rare) |
| Vincristine | ++ | ++ (Rare) | +++ (Rare) | − | − | ++ (N/V/D/M) | +++ | SIADH (rare) |
| Vinorelbine | +++ | − | − | − | + (LFT↑) | ++ (N/V/D/M) | + | SIADH (rare) |
| Vorinostat | + | − | − | − | − | ++ (N/V) | + (Uncommon) | Thrombosis (rare) |
a Data are from pediatric studies whenever possible, but when pediatric data were incomplete or absent, adult data were included.
Several non-chemotherapy adjunct medications and therapies are used to provide supportive care and to attenuate the toxicities of chemotherapy. With adequate treatment of toxic adverse effects, larger doses and/or additional chemotherapeutic agents can enhance the rate of remission. Such therapies include antiemetics, hematopoietic growth factors, HSCT, transfusion of blood products, and a number of medications that lessen or block organ-specific toxicity. Many of these therapies also pose acute and chronic risks to children; these risks must be balanced against their benefits.
Commensurate with the growing understanding of cancer biology is the recent development of “targeted” strategies that are directed specifically at cancer cells and thus limit direct toxicity to healthy cells. A commonly known example of these types of agents is the use of tyrosine kinase inhibitors (e.g., imatinib) in BCR-ABL (Philadelphia chromosome)–positive leukemia. As a result of less toxicity, oncologists can combine targeted therapies with full-dose standard chemotherapy for improved outcomes. Targeted agents include antiangiogenic therapies, immunomodulatory therapies, gene therapies, and humanized antibodies. Monoclonal antibodies are targeted to specific, unique tumor cell surface antigenic proteins. Small-molecule drugs, which are designed to target specific genetic signatures and biologic pathways critical to cancer growth and progression, have been developed that target tumor cell apoptotic pathways, histone deacetylation, protein farnesyltransferases, proteasome action, angiogenesis, and inhibition of the epidermal growth factor receptor tyrosine kinase.
Although toxicities of these agents as a whole appear to be far less than those of traditional chemotherapeutic agents, rash, fatigue, alterations to skeletal growth plates (antiangiogenic agents), nausea, diarrhea, hypotension, and anaphylaxis have been reported. The use of investigational and novel targeted antitumor agents amplifies the importance of reviewing cases with the oncologist.
One quarter of all children newly diagnosed with cancer will require radiation as frontline therapy. Photons (e.g., x-rays) and particle radiation (e.g., electrons, protons, neutrons) are the two major types of ionizing beam radiation. Regardless of the particle source, ionizing radiation leads to cell death by damaging cellular DNA. Toxicity from the effects of radiation therapy to surrounding healthy tissues is unavoidable, and the developing tissues of children are particularly susceptible to the acute and late effects of irradiation. The susceptibility of normal tissues depends on the total and fractional dose received, the inherent sensitivity of the tissue to the dose of radiation, the volume of tissue irradiated, and time course of treatment ( Table 11.3 ). Overall, normal host cells have a greater capacity to repair the damaging effects of radiation than cancer cells but require time to recover. To allow sufficient time for the healthy tissue to repair, the total dose of radiation is usually divided into a series of fractional doses over time. Recent technological advances have led to three-dimensional conformal radiotherapy, which closely conforms the radiation dose to the tumor shape and minimizes radiation to the surrounding tissues. Despite these advances to reduce the toxicity of radiation therapy to healthy tissues, children remain at risk for acute and chronic complications. Furthermore, concurrent chemotherapy may potentiate the radiation toxicity, increasing the tissue damage.
| Radiation Field | Late Effects | Risk Factors |
|---|---|---|
| Cranial | Neurocognitive deficits | >18 Gy, IV/IT methotrexate |
| Leukoencephalopathy | >18 Gy with IT methotrexate | |
| Growth hormone deficiency | >18 Gy | |
| Panhypopituitarism | >40 Gy | |
| Large vessel stroke | >60 Gy | |
| Second cancers | Variable | |
| Dental problems | >10 Gy | |
| Cataracts | >2–8 Gy single dose, 10–15 Gy fractionated dose | |
| Ototoxicity | >35–50 Gy | |
| Chest | Cardiac disease | |
| Coronary artery disease | >30 Gy | |
| Cardiomyopathy | >35 Gy, >25 Gy with anthracyclines | |
| Valvular disease | >40 Gy | |
| Pericardial disease | >35 Gy | |
| Arrhythmias | Unknown | |
| Thyroid disease | ||
| Hypothyroidism | >20 Gy local, >7.5 Gy TBI | |
| Hyperthyroidism | >20 Gy local, >7.5 Gy TBI | |
| Thyroid nodules, cancer | Any dose | |
| Pulmonary disease | ||
| Pulmonary fibrosis | >15–20 Gy | |
| Restrictive lung disease | Unknown | |
| Obstructive lung disease | Unknown | |
| Abdomen/Pelvis | Chronic enteritis | >40 Gy |
| Gastrointestinal malignancy | Unknown | |
| Hepatic fibrosis/cirrhosis | >30 Gy | |
| Renal insufficiency | >20 Gy | |
| Bladder disease | ||
| Fibrosis | >30 Gy prepubertal, >50 postpubertal | |
| Hemorrhagic cystitis | Enhances cyclophosphamide and ifosfamide effect | |
| Bladder cancer | Unknown | |
| Gonadal dysfunction | ||
| Ovarian failure | 4–12 Gy | |
| Testicular failure | >1–6 Gy | |
| Any Radiation | Skin cancer | |
| Musculoskeletal changes | ||
| Bone length discrepancy | >20 Gy | |
| Pathologic fractures | >40 Gy | |
| TBI | All the above |
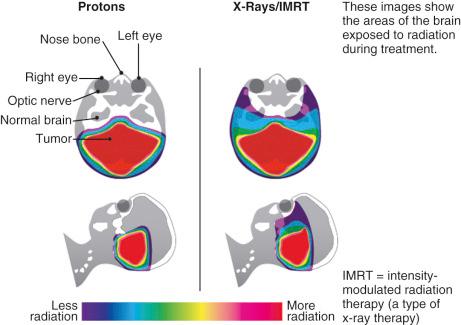
Proton radiation therapy (PRT) is a newer modality in pediatric radiation oncology and has become an established alternative to traditional photon therapy; however, PRT centers remain relatively uncommon (two dozen in the United States in 2016 [ http://www.proton-therapy.org/map.htm ]), which limits rapid access to frontline PRT for many children worldwide. In contrast to the destructive impact to all tissues in the path of a traditional photon beam, the specifically charged velocities of the proton beams are targeted to the calculated depth and shape of the tumor. By modulating the energy of the proton beam to become maximally energized at a specific depth of tissue, the relative dose to the healthy tissues in the plane of entry is as low as 20% to 30%, increases to 100% at the depth of tumor, and then decays to nearly no further penetration beyond the tumor ( Fig. 11.1 ). As such, the advantages of PRT include limited damage to healthy tissues surrounding the tumor and enhanced cell killing of the targeted tumor tissues. Despite these beneficial physical properties, the overall advantage of PRT over conventional radiation therapy is not yet uniformly supported by clinical trials, randomized pediatric trials are lacking, and long-term outcomes are yet to be realized. However, there are recent reports suggesting decreased neurotoxicity, improved health-related quality of life, and reduced neuroendocrine deficits with PRT compared with historical data with conventional photon radiation therapy.
Though primary tumors of the airway are rare in children, various cancer treatment regimens can cause airway changes that challenge the anesthesiologist. Both chemotherapy and radiation treatment can cause mucositis and xerostomia, first appearing soon after initiating treatment. Severe mucositis causes painful and friable oral mucosa and may threaten the airway as a result of pseudomembrane formation, supraglottic edema, bleeding, or aspiration of blood and secretions. Graft-versus-host disease (GVHD) after HSCT may also cause significant mucositis, with up to 30% of those children developing a difficult airway. Chronic radiation therapy to the head and neck may cause fibrosis, which distorts facial tissues and renders them less mobile. Children who have undergone chronic radiation therapy may present a challenge during laryngoscopy, with difficult glottic visualization, poor laryngeal mask airway seal, and subglottic stenosis.
Anesthesiologists caring for children who have undergone chemotherapy, HSCT, or radiation therapy should perform a thorough preoperative airway history and physical examination focusing on symptoms of airway compromise, prior anesthetic history, visualization of oral mucosa, and external assessment of the degree of distortion and immobility of face and neck tissues.
Primary cardiac tumors are uncommon in children, but when they occur (e.g., in children with tuberous sclerosis), they are usually benign and resolve spontaneously. However, cancer-related compromise of the cardiovascular system may occur and includes the effects of chemotherapy and/or radiation-induced treatment, pericarditis, and anterior mediastinal masses.
Anthracycline chemotherapeutic agents (doxorubicin, daunorubicin, idarubicin, and epirubicin) as well as the unrelated agent mitoxantrone, are well known for their cardiotoxic effects. Cyclophosphamide, fluorouracil, vinca alkaloids, cytarabine, cladribine, l -asparaginase, paclitaxel, trastuzumab, etoposide, teniposide, and pentostatin have also been associated with cardiac toxicity. Anthracycline toxicity primarily affects the myocardium, leading to cardiomyopathy and arrhythmias. Acute or subacute toxicity presents with symptoms ranging from only electrocardiographic (ECG) changes to fulminant heart failure shortly after the initial dose. Most children recover from acute toxicity with supportive therapy. Chronic progressive dilated or restrictive cardiomyopathy may develop within a year of treatment. The onset of symptoms can be delayed, and children may present with cardiac failure 20 or more years after treatment. Overall, one in eight survivors of childhood cancer who received both anthracyclines and chest radiation develop a serious, chronic cardiac disease. The risk of cardiotoxicity increases with increasing anthracycline doses, especially when the cumulative dose exceeds 250 to 360 mg/m 2 . However, there is no “safe” threshold dose for anthracyclines, as doses less than 240 mg/m 2 have resulted in cardiac damage. Preventative measures include the use of newer anthracycline analogs, antioxidants, iron chelators, and alterations in the dose. The cardiotoxic effects of chemotherapy are significantly compounded (perhaps tripled) by concurrent chest irradiation.
Unlike anthracycline toxicity that primarily affects the myocardium, radiation therapy can damage all components of the cardiovascular system. Complications from mediastinal radiation include cardiomyopathy, pericardial effusions, pericarditis, valvular fibrosis, conduction disturbances, and accelerated arteriosclerosis. Radiation-related myocardial fibrosis can induce a progressive restrictive cardiomyopathy, leading to pulmonary vascular disease and pulmonary hypertension. Systolic and/or diastolic dysfunction may be present. Premature arteriosclerosis affects the coronary arteries as well as the carotid arteries, pulmonary arteries, renal arteries, and aorta. Fatal myocardial infarction has been reported in children 6 to 22 years of age after mediastinal radiation. Valvular heart disease is predominately characterized by progressive mitral and aortic stenosis and insufficiency. Conduction disturbances range from atrial and ventricular arrhythmias, to right bundle branch block, and occasionally complete heart block requiring pacemaker placement. Radiation damage to autonomic nerves in proximity to the heart can result in tachycardia and loss of phasic respiratory variability similar to a denervated heart. Autonomic denervation can attenuate the perception of anginal pain, so providers should have a low threshold for suspicion of myocardial ischemia in those who have undergone mediastinal radiotherapy in infancy and childhood. With low doses of radiation (<25-30 Gy), short-term radiation toxicity is limited, but late toxicity occurs even at these low doses.
Symptomatic acute heart failure after cardiotoxic cancer therapy is uncommon in childhood; however, the potential for cardiovascular compromise in the setting of major surgical stress and anesthesia should be considered. Children who have undergone cardiotoxic chemotherapy or mediastinal radiation should be examined preoperatively for clinical evidence of impaired cardiac function or arrhythmia, including history and physical examination, chest radiography, and ECG. A preoperative or recent echocardiographic evaluation is recommended for the children receiving the following :
Cumulative anthracycline dose >240 mg/m 2
Any dose of anthracycline during infancy
Chest irradiation >40 Gy (or >30 Gy with concomitant anthracycline treatment)
Unknown doses of chemotherapy and radiation
Children with Hodgkin or non-Hodgkin lymphoma often have mediastinal involvement at the time of diagnosis, and half have respiratory symptoms. Less common oncologic causes of anterior mediastinal mass include neuroblastoma, germ cell tumors, and ALL. These children may require anesthesia for biopsy or resection of the mass, intravenous line placement, or radiologic procedures.
Children with clinical findings of superior vena cava syndrome or airway compression from anterior mediastinal mass are at the greatest risk for life-threatening perioperative complications ( Table 11.4 ). Symptoms of orthopnea, upper body edema, stridor, nighttime cough, the need to sleep on one preferred side or position, or wheezing should alert the anesthesiologist of the need for further evaluation. Radiographic or ultrasound evidence of airway or great vessel compression, pulmonary artery outflow obstruction, ventricular dysfunction, or pericardial effusion are particularly concerning findings that necessitate a discussion about the utility of preoperative treatment to reduce the size of the mass. Pretreatment is controversial because either corticosteroids or radiation may alter the tumor histology by causing tumor necrosis and render the precise diagnosis more difficult. However, diagnosis may still be possible in 95% of children after a 5-day course of corticosteroids. A multidisciplinary discussion of the risks and benefits with anesthesia, surgery, and oncology will clarify the optimal management for these children.
| Clinical signs and symptoms |
| Orthopnea |
| Upper body edema (signs of SVCS) |
| Stridor |
| Wheeze |
| Night cough |
| Sleeping in odd or only one position |
| Diagnostic imaging findings |
| Tracheal, bronchial, or carinal compression |
| Great vessel compression |
| SVC obstruction |
| Pulmonary artery outflow obstruction |
| Ventricular dysfunction |
| Pericardial effusion |
Anesthetic considerations for children with an anterior mediastinal mass are presented in detail in Chapter 15 . In brief, local anesthesia and sedation are preferred. However, if general anesthesia is required, crucial perioperative considerations include maintaining spontaneous ventilation (i.e., avoiding paralysis) and being prepared to secure the airway with a tracheal tube and turn the child to the left lateral decubitus or prone position for resuscitation in the face of cardiopulmonary collapse.
Primary and metastatic lung malignancies are uncommon in children. Pulmonary compromise as a direct effect of pediatric tumors is more often the result of a pleural effusion, pulmonary infiltrates, pulmonary embolus, chylous effusions, anterior mediastinal mass, or hyperleukocytosis-induced pulmonary leukostasis.
Therapy-related symptomatic pulmonary dysfunction or abnormal pulmonary function tests occur in 6% of children treated with chemotherapy alone, 20% treated with both chemotherapy and radiation, and 25% after HSCT. Several chemotherapeutic agents—most notably bleomycin—have the potential to cause acute or chronic lung injury, including pneumonitis, pulmonary fibrosis, or noncardiogenic pulmonary edema. Pneumonitis presents insidiously with nonproductive cough, progressive dyspnea, and rales. Although the symptoms usually resolve with completion of the treatment, in some children pneumonitis progresses to irreversible pulmonary disease. In adults, bleomycin-induced pneumonitis occurs in up to 46%, with a 3% mortality. The incidence of pneumonitis in children is less well established.
Pulmonary fibrosis may present acutely during treatment or as a late sequela of chemotherapy, radiation treatment, or GVHD and may be associated with severe morbidity and mortality. The administration of large concentrations of oxygen to children with bleomycin-induced pulmonary fibrosis can acutely or chronically exacerbate their restrictive lung disease. Thus the concentration of inhaled oxygen should be adjusted to the minimum concentration required to ensure adequate tissue oxygen delivery in the perioperative period. Overall, it is important to assess the clinical and functional status of these children for symptomatic or occult pulmonary disease before administering anesthesia. A thorough assessment of baseline pulmonary status is particularly important to assess the need for postoperative ventilatory support or tolerability of thoracoscopy. It is reasonable to seek preoperative pulmonary function testing in those with clinical evidence of pulmonary dysfunction, bearing in mind that formal pulmonary function testing may be a challenge in young children.
Wilms tumor is the most common primary renal tumor in children, followed by clear cell sarcoma of the kidney, malignant rhabdoid tumor, congenital mesoblastic nephroma, and renal cell carcinoma. Each of these tumors can directly impact renal function. Similarly, extrarenal tumors, such as neuroblastoma, can impact the renal system by infiltrating the kidneys, obstructing urinary flow, or compressing the renal vasculature.
Most chemotherapeutic drugs are directly nephrotoxic in a dose-dependent manner or lead to physiologic conditions that impair renal function (e.g., sepsis, dehydration, tumor lysis syndrome [TLS]). Cisplatin, carboplatin, and ifosfamide are notorious nephrotoxic chemotherapeutic agents in children and adults, especially when combined. Cisplatin causes a dose-dependent nephrotoxicity and hypomagnesemia. Ifosfamide causes subclinical glomerular toxicity in up to 90% of patients, with clinically apparent toxicity occurring in 30%. Ifosfamide can also induce Fanconi syndrome in up to 7% of children, with a delayed presentation possible up to 18 months after therapy. Methotrexate has the potential to cause severe acute renal failure in children. Many other chemotherapeutic drugs cause nephrotoxicity at large doses. The syndrome of inappropriate antidiuretic hormone (SIADH) is associated with multiple chemotherapeutic agents. Many non-chemotherapeutic drugs (such as antibiotics and diuretics) commonly used in children with tumors also contribute to nephrotoxicity.
Focal abdominal radiation or total body irradiation as preconditioning for HSCT can cause radiation nephritis, which presents with azotemia, proteinuria, anemia, and hypertension. The cumulative dose that leads to renal damage in children has not been established. After HSCT, the incidence of acute renal failure in children is as great as 40%, with chronic renal failure in 18% to 54%.
Anesthesia providers should be aware of the size and location of renal or juxtarenal tumors to determine the risk of significant intraoperative bleeding or great vessel obstruction, particularly in tumors that involve the renal vasculature. Preoperative evaluation of children who have undergone chemotherapy or abdominal radiation treatment should focus on identifying clinical and subclinical renal dysfunction, electrolyte disturbances (hypomagnesia, hypophosphatemia), fluid overload, anemia, and hypertension. The need for preoperative dialysis should be considered in those with profound renal dysfunction. Nonsteroidal antiinflammatory drugs (NSAIDs) should be used with caution in children with renal dysfunction as NSAID-related restriction of renal perfusion may exacerbate any preexisting renal dysfunction.
Primary liver tumors comprise only 1% of childhood cancers, and up to 20% of these are associated with a genetic syndrome, such as Beckwith-Wiedemann syndrome. In young children, hepatoblastomas are the most prevalent primary hepatic tumor, followed by sarcomas, germ cell tumors, and rhabdoid tumors. Hepatocellular carcinoma is occasionally found in older adolescents.
Methotrexate, actinomycin D, 6-mercaptopurine, and 6-thioguanine are associated with acute hepatic toxicity, which can present hours to weeks after a chemotherapeutic dose. Hepatic impairment is typically transient and reversible. Radiation typically causes self-limited acute toxicity, but chronic hepatic fibrosis may follow large doses of radiation (>40 Gy). Most concerning is the potential to develop sinusoidal obstruction syndrome (SOS) in children after HSCT. SOS is characterized by portal hypertension, liver failure, and multiorgan system failure affecting the heart, lungs, and kidneys. Up to 60% of children develop SOS after HSCT, with an associated mortality rate of 19% to 50%.
Both acute and chronic liver disease may be found in children with cancer, associated with a coagulopathy and/or impaired drug metabolism. The dose and timing of drugs that undergo significant hepatic elimination should be adjusted accordingly. Potentially hepatotoxic medications (e.g., acetaminophen) should be used with caution in children who are particularly vulnerable to further hepatic insult. Children exhibiting chronic liver failure may have coexisting genetic syndromes, such as Beckwith-Wiedemann syndrome, which present additional anesthetic challenges.
Primary gastrointestinal (GI) tumors are uncommon in children; however, various intraabdominal malignancies can affect the GI tract by intestinal obstruction, intussusception, erosive perforation, intraabdominal hemorrhage, biliary obstruction, venous or arterial obstruction, and massive hepatomegaly. The most common GI concern in children with cancer pertains to adverse effects of the chemotherapy and radiation treatment.
As chemotherapy and radiation treatments target rapidly proliferating tissues, the gastrointestinal mucosa is particularly vulnerable. Chemotherapeutic agents are well known to cause nausea and vomiting but may also cause more serious GI pathology such as diarrhea, mucositis, stomatitis, and neutropenic enterocolitis. These adverse effects may exacerbate the malnutrition and dehydration that are often found in children with cancer. Similarly, radiation doses in excess of 20 to 30 Gy can cause inflammation and edema of GI tissues. Importantly, acute and chronic GVHD after HSCT (discussed in detail below) commonly impacts the GI tract, although the majority of cases are mild in the current era of prophylaxis with calcineurin inhibitors plus methotrexate or mycophenolate. The incidence of moderate to severe gut GVHD after HSCT is approximately 10%, and the mortality rate is substantial without prompt treatment.
Before induction of anesthesia, children who have been treated for their cancer may have chronic nausea and vomiting or delayed gastric emptying, both of which are exacerbated by opioids. These children may be at increased risk for aspiration and should be managed accordingly. Children with GI dysfunction may also present with malnutrition, dehydration, and electrolyte imbalances that warrant correction before embarking on elective surgical procedures.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here