Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Pericardial effusion, the presence of fluid within the pericardial space, has a number of causes. As fluid accumulates, a critical point is reached at which pericardial pressure negatively affects cardiac filling and causes circulatory insufficiency. This is called pericardial tamponade. Once compensatory mechanisms begin to fail, obstructive shock ensues and a failure to restore hemodynamics eventually leads to cardiac arrest. Only removal of fluid can stabilize hemodynamics at this point.
The pericardium is a two-layered fibroelastic sac surrounding the heart. The pericardium is avascular but well innervated, so inflammation induces pain. The visceral pericardium is a single-cell layer that adheres to the epicardium. The outer parietal pericardium consists mostly of collagen with some elastin. These two layers create the pericardial space, which normally contains 15 to 50 mL of serous fluid and is under negative pressure to promote filling of the right ventricle (RV) during diastole. Pericardial fluid provides lubrication for cardiac contractility and acts as a “shock absorber” for deceleration forces.
The pericardium is a tense structure, but it also has some elasticity. These properties limit the amount of cardiac dilation that is possible during diastole and enhance mechanical interactions between the atria and ventricles during systole. This semi-elastic property can also tolerate an acute (i.e., over a period of hours to days) accumulation of pericardial fluid (80 to 120 mL) without significantly increasing intrapericardial pressure, which is the flat portion of the pressure-volume curve for pericardial pressure ( Fig. 16.1 ). Once a critical volume is reached, adding as little as 20 to 40 mL can double intrapericardial pressure (the steep portion of the pressure-volume curve [see Fig. 16.1 ]) and cause clinical decompensation from cardiac tamponade. Cardiac tamponade typically occurs with an intrapericardial pressure of 15 to 20 mm Hg.
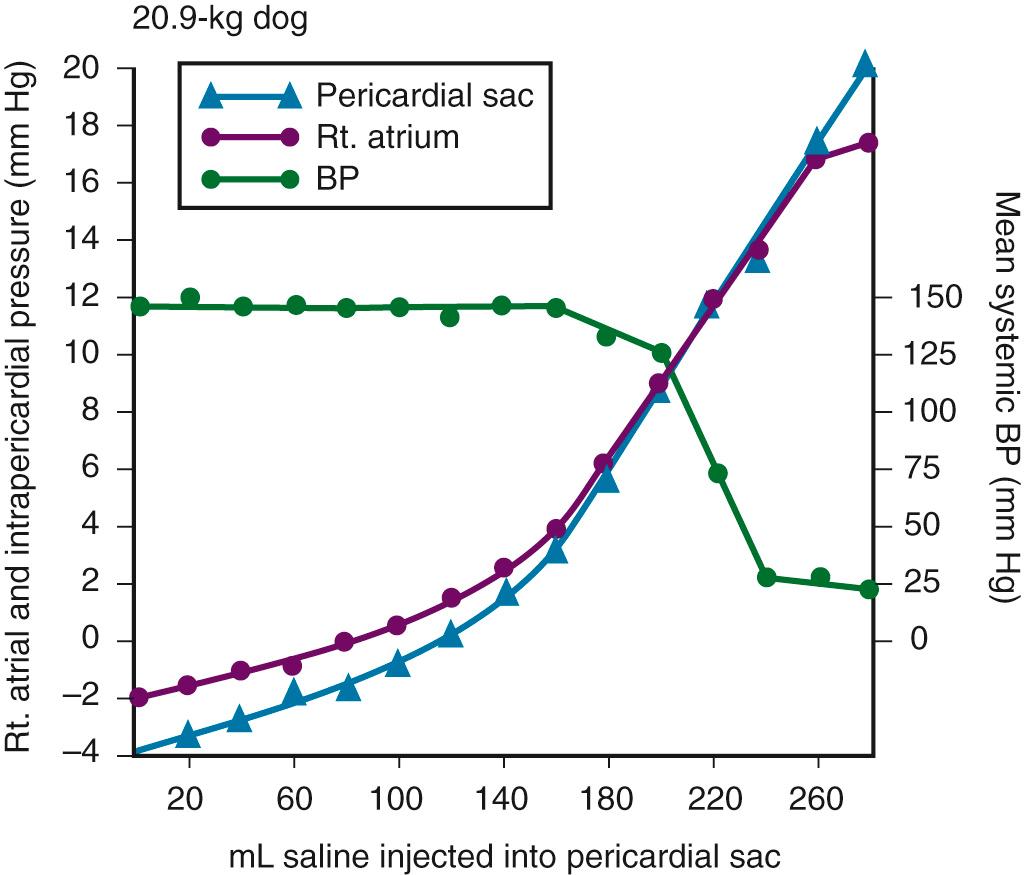
Slow and chronic accumulation of pericardial fluid (over a period of weeks to months) causes the pericardium to expand circumferentially, and it can accommodate several liters of fluid with minimal alteration in intrapericardial pressure. Patients with this condition may be asymptomatic despite large effusions. No specific pericardial volume predicts the hemodynamic consequences of an effusion; such consequences depend on the acuity of the accumulation of the fluid.
To generate an effective stroke volume, the left ventricle (LV) must be filled during diastole, and this process relies primarily on adequate filling of the RV. Normal inspiration makes the intrathoracic space more negative, which promotes RV filling by increasing venous return and causing dilation of the RV chamber secondary to a reduction in the tension in the RV free wall ( ).
Elevated intrapericardial pressure (i.e., early tamponade) first results in abnormal RV filling followed by abnormal LV filling. In this situation, the free wall of the RV cannot expand against the pericardial fluid during inspiration. To accommodate RV filling, the interventricular septum bows abnormally into the LV, which reduces its volume; this is also known as ventricular interdependence. LV filling, stroke volume, and ultimately distal tissue perfusion is reduced. This phenomenon is responsible for pulsus paradoxus (PP; described later in this chapter), which is sometimes observed with tamponade. LV filling is also reduced by the collapse of right-sided structures. After a critical volume is reached on the pressure-volume curve (see Fig. 16.1 ), the intrapericardial pressure is transmitted to the inferior vena cava (IVC) and right atrium. These thin-walled structures then become compressed and reduce filling of the RV.
The atria and pulmonary circulation are at much lower pressure than systemic arterial pressure and are also vulnerable to rising intrapericardial pressure ( Fig. 16.2 ). Late in tamponade a “pressure plateau” occurs in which right atrial pressure, RV diastolic pressure, pulmonary artery diastolic pressure, and pulmonary capillary wedge pressure are virtually identical. This equalization of chamber pressure leads to a reduction in venous return and the echocardiographic hallmark of tamponade: diastolic collapse of the RV (discussed later in this chapter). At this point, hemodynamic collapse is imminent, with severe hypotension, and potentially pulseless electrical activity (PEA) developing. Unless intrapericardial pressure is decreased immediately, cardiac arrest will ensue.
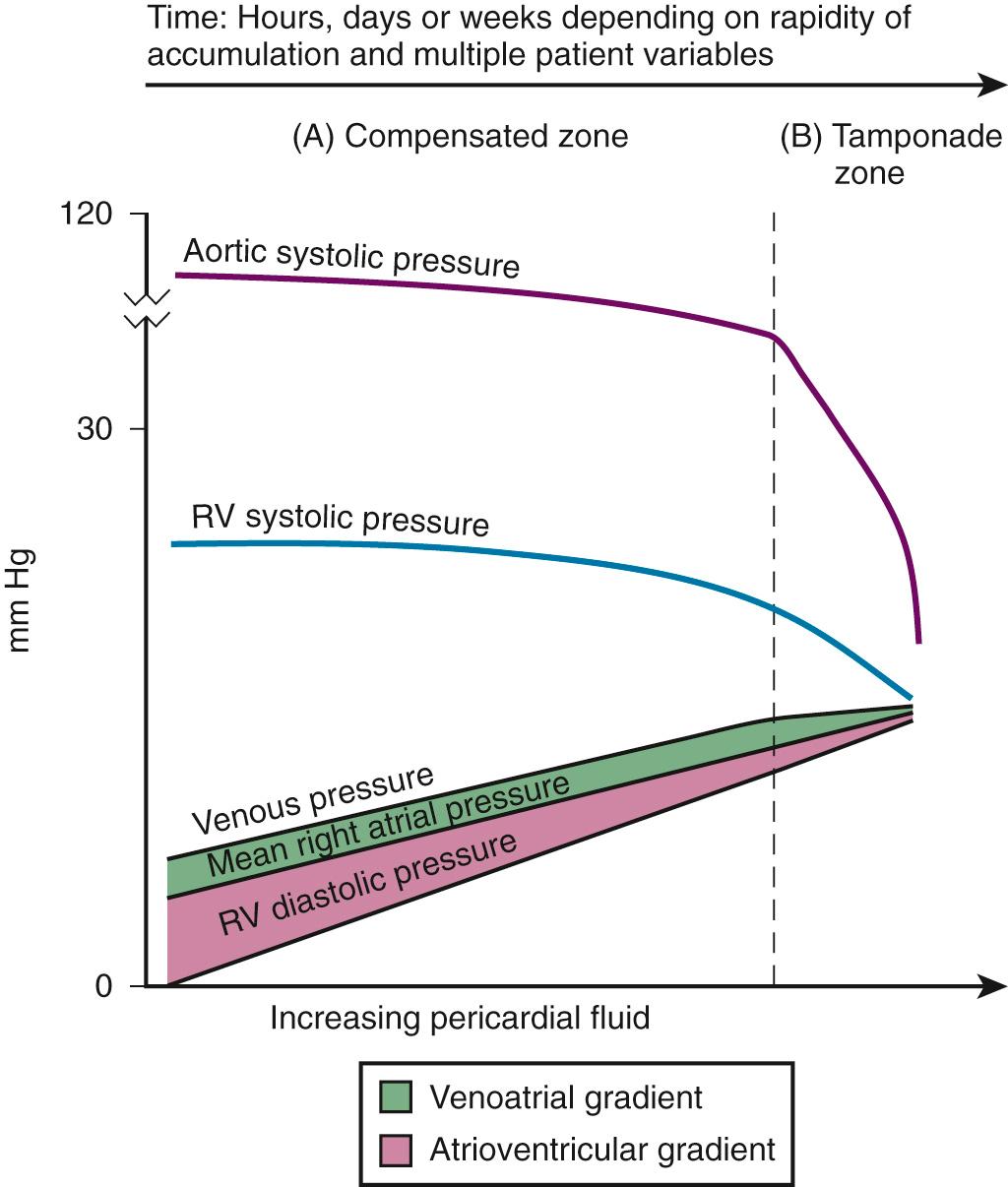
To maintain a physiologic cardiac output early in tamponade, the sympathetic nervous system increases the heart rate, arterial vasoconstriction (to maintain mean arterial blood pressure), and venoconstriction (to maintain normal venous-atrial and atrioventricular filling gradients). Early in tamponade, these compensatory mechanisms are usually effective in maintaining adequate cardiac output.
Compensatory mechanisms also preserve normal cardiac contractility and myocardial perfusion. However, when the pericardial pressure overwhelms the compensatory mechanisms, coronary perfusion pressure is reduced, which leads to myocardial ischemia, systolic dysfunction, and ultimately a reduction in cardiac output. Experimental induction of severe tamponade demonstrated microscopic ischemic cardiac injury. Lactic acidosis (resulting from reduced cardiac output and systemic hypoperfusion) also leads to further reduction in cardiac contractility and, ultimately, cardiac output.
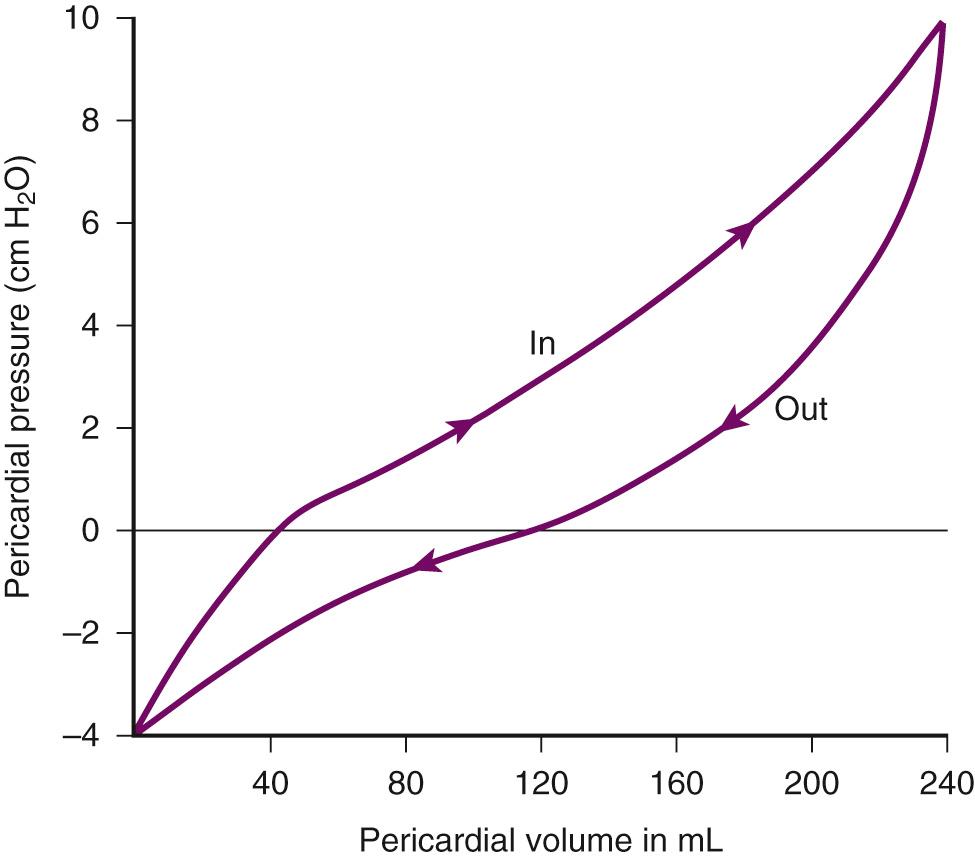
Removal of pericardial fluid (i.e., pericardiocentesis) reverses the pathophysiologic processes just described by improving coronary filling, cardiac filling, and hence cardiac output. Interestingly, the pressure-volume relationship of the pericardial space demonstrates hysteresis; that is, withdrawing a certain quantity of fluid reduces intrapericardial pressure more than addition of the same amount of fluid increases intrapericardial pressure. This effect, however, is not universal and may vary among patients and in various disease states ( Fig. 16.3 ).
Under normal circumstances, positive pressure ventilation (e.g., mechanical ventilation) reduces venous return to the right side of the heart by increasing intrathoracic pressure. This could be detrimental for patients with tamponade because right-sided filling is already compromised and further reductions can lead to severe hemodynamic instability. Therefore positive pressure ventilation (i.e., noninvasive or invasive) should be avoided in patients with known or suspected tamponade unless it is absolutely necessary (e.g., respiratory failure).
Low-pressure pericardial tamponade is defined as a hemodynamically significant effusion with an intrapericardial pressure that is lower than expected. This category of tamponade occurs in patients with subacute or chronic effusions, but present with a superimposed hypovolemia (e.g., associated with long-term diuretic use, dehydration, or excessive dialysis). The diagnosis may be challenging because the classic symptoms and findings on physical examination (e.g., distended neck veins) may be absent. Ultrasound is most helpful in making the diagnosis in these patients. Fluid boluses may temporize the hemodynamic compromise while pericardial decompression is being arranged.
The major categories of pericardial effusion include infection, inflammation, malignancy, trauma, and metabolic abnormalities. Effusion may also be associated with aortic disease, connective tissue disease, or idiopathic causes. It is often difficult to report the exact incidence of each type of pericardial effusion because of variations in patient populations, local epidemiology, and the diagnostic protocols used during evaluation. The prevalence of a chronic effusion is also difficult to ascertain because it is often asymptomatic and underreported. General autopsy studies demonstrate an overall prevalence of 3.4%.
Acute hemopericardium, or rapid accumulation of blood in the pericardial space, can have a traumatic or nontraumatic etiology. It is one of the most feared causes of tamponade because the semi-elastic pericardium cannot accommodate acute increases in pericardial fluid and clinical deterioration can be rapid. This diagnosis can be challenging to make because there may be little or no evidence during the initial evaluation (e.g., pericardial size might be normal on a chest radiograph). Common causes include trauma and aortic dissection retrograde into the pericardial sac.
Mesothelioma
Lung
Breast
Melanoma
Lymphoma
Radiation related (especially after Hodgkin's disease)
Viral
Bacterial
Staphylococcus
Pneumococcus
Haemophilus
Fungal
Tuberculosis
Amebiasis
Toxoplasmosis
Idiopathic
Systemic lupus erythematosus
Scleroderma
Rheumatoid arthritis
Acute rheumatic fever
Myxedema
Uremia
Cholesterol pericarditis
Bleeding diatheses
Acute myocardial infarction
Dissecting aortic aneurysm
Congestive heart failure
Coronary aneurysm
Hydralazine
Phenytoin
Anticoagulants
Procainamide
Minoxidil
Doxorubicin
Daunorubicin
Bleomycin
Blunt
Major trauma
Closed-chest cardiopulmonary resuscitation
Penetrating
Major penetrating trauma
Intracardiac injections
Transthoracic and transvenous pacing wires
Pericardiocentesis
Cardiac catheterization
Central venous catheters
Serum sickness
Chylous effusion
Löffler's syndrome
Reiter's syndrome
Behçet's syndrome
Pancreatitis
Postpericardiotomy
Amyloidosis
Ascites
Penetrating cardiac trauma can cause acute hemopericardium by either external forces (e.g., a stab wound to the heart) or internal forces (e.g., iatrogenic injury during placement of a pacemaker). Cardiac perforation can lead to rapid clinical deterioration and PEA.
External cardiac puncture is associated with stab wounds or projectile injuries (e.g., gunshot wounds). Tamponade develops in 80% to 90% of patients with cardiac stab wounds as opposed to 20% of those with gunshot wounds. Stab wounds cause tamponade more frequently because if the pericardial injury is small, it can reseal and trap blood within the pericardial space. On the other hand, a gunshot typically produces both large myocardial and pericardial wounds that allow continuous drainage into the mediastinal and pleural space. Clinical deterioration is usually secondary to hypovolemia. Any penetrating injury to the chest, back, or upper part of the abdomen may injure the pericardium and cause tamponade.
Internal penetrating trauma is typically caused by invasive diagnostic or therapeutic procedures. The procedures most often associated with this injury are cardiac catheterization (angioplasty or valvuloplasty) and pacemaker insertion. Hemopericardium results from puncturing a cardiac chamber, a coronary artery, or a great vessel (e.g., the superior vena cava). Ironically, pericardiocentesis itself (treatment of a pericardial effusion) can cause hemopericardium if a coronary vessel or the myocardium is injured during the procedure.
Internal jugular and subclavian venous catheters (e.g., central venous or hemodialysis catheters) are commonly inserted in the emergency department (ED). During such procedures, hemopericardium results from perforation of the superior vena cava, right atrium, or RV. Hemopericardium can occur immediately or can be delayed for days subsequent to erosion of the catheter through myocardial or vascular tissue. Although this complication seldom occurs, it should always be considered when a patient experiences sudden hemodynamic deterioration following an invasive procedure.
Major blunt chest trauma can cause hemopericardium with or without obvious signs of injury, from rupture of a cardiac chamber or, less commonly, damage to a coronary artery. Myocardial rupture can be uncontained or contained. Patients with uncontained rupture do not typically survive long enough to reach the hospital. Contained rupture may be found soon after injury or may be a late finding. Tamponade can also be caused by a deceleration mechanism of injury that induces either aortic or vena caval disruption. In one case series the incidence of tamponade following deceleration injury was found to be 2.3% (1 in 43 patients).
Chest compressions during cardiopulmonary resuscitation (CPR) can also cause hemopericardium from broken ribs or bleeding intercostal vessels. Hemopericardium following CPR has been described in case reports but is unlikely to be significant, much less to cause tamponade.
Atraumatic hemopericardium is difficult to diagnose. Diagnosis is often delayed because it occurs spontaneously and the clinical findings can be less obvious than those of hemopericardium from traumatic causes. Maintain a high index of suspicion for this condition in patients with risk factors, such as certain malignancies (e.g., lung cancer) or tuberculosis within endemic areas. Common causes of atraumatic hemopericardium are discussed later.
Bleeding diathesis is an important cause of spontaneous hemopericardium and may be associated with the use of anticoagulants (reported incidence of 2.5% to 11%) or thrombolytic therapy (incidence <1%). Patients who have undergone cardiac surgery are at increased risk because of the anticoagulative effects of the cardiopulmonary bypass machine and medications started postoperatively (e.g., clopidogrel, warfarin). Fortunately, tamponade has a low incidence and is generally detected in the postoperative period before discharge. This complication is usually prevented by the intraoperative placement of mediastinal or pericardial drains.
Hemopericardium can develop following myocardial infarction (MI). Early after a transmural MI (1 to 3 days), the necrotic myocardium causes inflammation of the overlying pericardium and then effusions can form. Late-developing effusions (weeks after an MI) are caused by an autoimmune pericarditis called Dressler's syndrome. Improved reperfusion techniques have drastically reduced the incidence of post-MI pericarditis and effusion.
Ascending aortic dissection causes rapid and usually fatal hemopericardium. The dissection may expand in a retrograde fashion by extending to the base of the aorta and into the pericardial sac. This is a very difficult diagnosis, and best visualized and comfirmed by bedside ultrasound. Risk factors for aortic dissection include hypertension, atherosclerosis, vasculitis (e.g., giant cell arteritis, syphilis), collagen vascular disease (e.g., Marfan's syndrome), and the use of sympathomimetics (e.g., cocaine).
Ventricular free-wall rupture is a rapidly fatal cause of acute hemopericardium that can occur after MI leading to cardiac failure and shock. This complication is less common today than in the past (<1%) secondary to improved revascularization techniques, better therapeutic medications, and faster intervention times (shorter door-to-balloon times) for coronary ischemia. Despite a reduction in its overall incidence, 7% of all deaths related to MI are caused by this complication. Survival is theoretically possible with prompt recognition and treatment, but the prognosis is grim once tamponade occurs.
Nonhemorrhagic pericardial effusions usually accumulate slower than acute hemopericardium does (over a period of weeks to months). Chronic fluid accumulation allows the pericardium to stretch circumferentially and accommodate up to 2000 mL of fluid without any hemodynamic compromise. Effusions that grow slowly allow the circulatory system to adapt to increased intrapericardial pressure, thereby further maintaining hemodynamic stability. Thus, asymptomatic patients with moderate to large effusions may not need emergency pericardiocentesis, in contrast to patients with acute hemopericardium.
Nonhemorrhagic effusions have several causes (see Box 16.1 ), and the exact one may not be obvious during the initial evaluation without diagnostic pericardiocentesis. Common causes of nonhemorrhagic effusions are discussed in the following sections.
Most idiopathic effusions are believed to be viral in origin and most commonly caused by infection with coxsackievirus, echovirus, cytomegalovirus, or human immunodeficiency virus (HIV) (discussed later in the chapter). Idiopathic pericardial effusions may be asymptomatic or have an associated component of pericarditis (e.g., positional pain or diffuse ST-segment changes on the electrocardiogram [ECG]). These effusions are often labeled idiopathic because the diagnosis cannot be made noninvasively (i.e., based on the history, physical examination, or serum testing) and the risk associated with diagnostic pericardiocentesis often outweighs the risk of observation in asymptomatic adults who appear to be well. Diagnostic pericardiocentesis may be recommended for idiopathic effusions that are persistent or symptomatic without a known cause.
Tumors of the pericardium or myocardium may cause nonhemorrhagic effusions. Primary cardiac tumors are less common (0.001% to 0.003%) than metastases from another site (2% to 18%), but either may cause a malignant effusion. Although no malignancy preferentially metastasizes to the heart, certain tumors commonly involve the heart when they metastasize; frequently implicated are lung cancer, breast cancer, mediastinal tumors, malignant melanoma, leukemia, and lymphoma.
Cardiac metastasis is usually a late finding in cancer, as other foci are generally evident first. The classic signs and symptoms of tamponade (e.g., chest pain and dyspnea) may not be obvious with malignant tamponade. When present, they may be mistakenly attributed to the underlying malignancy. Thus, in the relevant clinical scenario, consider screening patients with malignancy for pericardial effusion (e.g., ultrasound) before the clinical findings of tamponade appear.
Pericardial effusions (secondary to radiation-induced pericarditis) can develop acutely during radiation therapy or may be delayed for years. Risk factors include the radiation dose, duration of exposure, and age of the patient. Patients treated with radiation for Hodgkin's disease have the highest association of radiation-induced pericarditis and subsequent effusions. These effusions can be serous, hemorrhagic, or fibrinous.
Congestive heart failure (CHF) is a cause of pericardial effusion. Diagnosis may be difficult because of overlapping signs and symptoms with exacerbations of CHF (e.g., chest pain or dyspnea). Adding to the diagnostic complexity is that 12% to 20% of patients with CHF have a coexisting pericardial effusion. Fortunately, treatment of CHF-associated pericardial effusion does not differ from that for an effusion from other causes: treat the underlying cause unless the patient has evidence of hemodynamic compromise.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here