Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
SYNOPSIS
Patient satisfaction and health-related quality of life are essential outcomes to consider in addition to traditional clinical outcomes (e.g., flap survival, postoperative infection, length of hospital stay) for many plastic and reconstructive surgery procedures.
Patient-reported outcome measures (PROMs) are valuable tools for evaluating the benefits of treatments and newly developed surgical techniques.
PROMs developed using modern psychometric methods, such as Rasch Measurement Theory, represent highly calibrated measurement instruments.
The creation of a PROM involves mixed-methods research (i.e., qualitative and quantitative methods) that usually takes place in several phases, and its development is often a multi-year process.
Validity and reliability are psychometric properties of a PROM. Validity refers to whether a PROM measures the concept or attribute it intends to measure. Reliable refers to whether a PROM yields consistent and reproducible results.
PROMs can and should be routinely included in clinical care and research to better understand the effects of surgical care from the patient’s perspective.
Future priorities for patient-reported outcomes (PROs) in plastic surgery are: (1) incorporation of PRO data in the decision-making process for patient care, ( 2 ) integration of PRO data in the electronic health record, and (3) use of PRO data to measure and improve quality of care.
Healthcare costs continue to rise, outpacing gross domestic product (GDP) growth in developed countries. Restructured healthcare systems have been proposed, aiming to deliver cost-effective and evidence-based treatments. The desire to provide effective, evidence-based treatments has resulted in a proliferation of value-based healthcare (VBHC) systems in many countries. The concept behind VBHC is that the healthcare system should focus on achieving predetermined outcomes, while optimizing the use of resources through integrated healthcare. A team-oriented approach to patient care is prioritized and the capability for data sharing and measurement of outcomes is optimized. The “value” in value-based healthcare is achieved by measuring health outcomes against the cost of delivering the care. A well-functioning VBHC system ideally aims to result in decreased costs and increased quality of care, benefiting patients, providers, payers, suppliers and society as a whole ( Fig. 12.1 ).
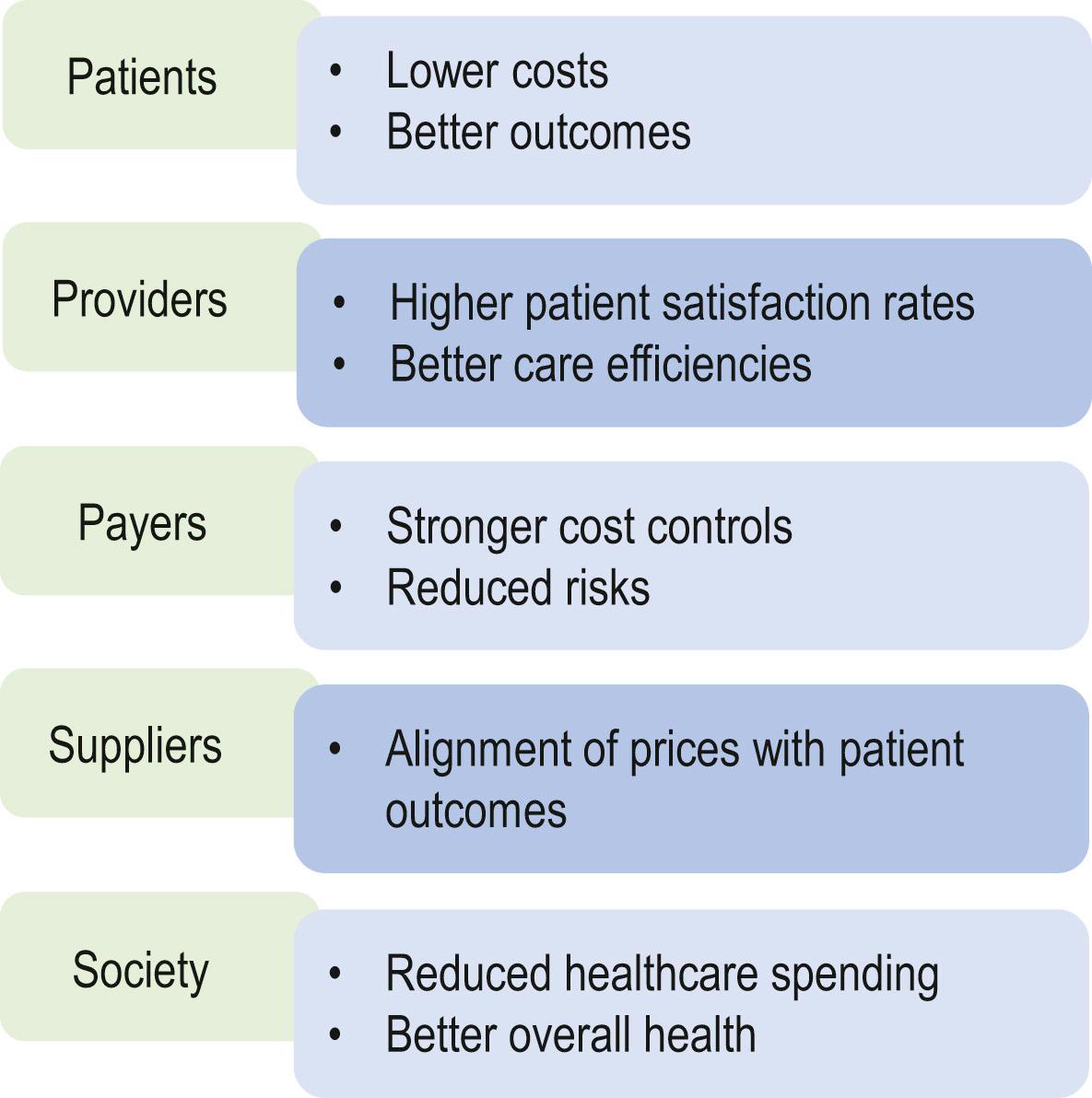
When measuring quality of care and treatment effectiveness, objective or “hard” outcomes, such as survival, reoperation rates, and postoperative complication rates, have traditionally been used. However, such outcomes may not always correlate with a patient’s assessment of the intervention’s effect, or with outcomes that matter to patients on a daily basis. Furthermore, in plastic and reconstructive surgery, most procedures are not life-saving but are performed with the aim to improve patients’ health-related quality of life (HRQL) and to reduce symptoms. Additionally, both reconstructive and aesthetic plastic surgeons are trained to restore or improve patients’ satisfaction with form, function, and/or aesthetic appearance. Therefore, the use of tools capable of measuring HRQL (physical and psychosocial well-being), patient satisfaction (e.g., with outcome or with preoperative information), appearance, and symptoms (e.g., postoperative pain or fatigue) have gained interest within the field. This interest has led to an increased uptake of patient-reported outcome measures (PROMs). Common psychometric terms and their definitions are presented in Table 12.1 .
| Term | Definition |
|---|---|
| Health-related quality of life (HRQL) | An assessment of an individual’s health, including emotional, social and physical aspects. |
| Patient-reported outcome measure (PROM) | A scientifically designed questionnaire in which responses are collected directly from patients, without interpretation of the patient’s response by a clinician or anyone else. Outcomes that matter the most to patients, such as health-related quality of life, satisfaction, or functional status, are recorded and quantified. |
| Conceptual framework | A diagram of the expected relationships between domains and the overall concepts measured by the instrument. The validation process confirms the conceptual framework. |
| Domain | A collective word for a group of related concepts. All the items in a single domain contribute to the measurement of the domain concept. |
| Scale | A set of items with response options demonstrating a clinical hierarchy. |
| Item | An individual question or statement that is evaluated by the patient to address a particular concept. |
| Rasch Measurement Theory (RMT) | A mathematical way of separately estimating parameters about test items and the people taking the test at the same time. |
| Validity | The ability of an instrument to measure what it is intended to measure. It is a psychometric property of a PROM. |
| Content validity | The degree to which the content of a PRO instrument is an adequate reflection of the construct to be measured. |
| Construct validity | The degree to which the scores of a PRO instrument are consistent with the hypotheses (for instance with regard to internal relationships, relationships to scores of other instruments, or differences between relevant groups) based on the assumption that the PRO instrument validly measures the construct to be measured. |
| Criterion validity | The degree to which the scores of a PRO instrument are an adequate reflection of a "gold standard". |
| Reliability | The degree to which measurement is free from measurement error, both random and systematic errors. It is a psychometric property of a PROM. |
| Internal consistency reliability | The degree to which items in the PRO instrument are able to measure the same concept of interest. |
| Test–retest reliability | The agreement between observations on the same patient on two or more occasions separated by a time interval under stable health conditions. |
| Responsiveness | The ability of a PRO instrument to detect a meaningful or clinically important change in a clinical state over time. |
| Minimal important difference (MID) | The smallest difference in score in a domain of interest that patients perceive as beneficial and which would mandate, in the absence of troublesome side effects and excessive cost, a change in the patient’s (healthcare) management. |
PROMs are scientifically designed questionnaires in which responses are collected directly from the patient, thus using a patient-centered approach. As defined by the US Department of Health and Human Services Food and Drug Administration (FDA), patient-reported outcomes (PROs) refer to “any report of status of a patient’s health condition that comes directly from the patient, without interpretation of the patient’s response by a clinician or anyone else”.
PROMs allow patients to accurately report on outcomes relevant to their health condition. Rigorous and in-depth qualitative research methods involving patients are required when designing a PROM, as the instrument must be able to measure the concerns that matter most to patients, and thus must include content relevant to patients. PROMs offer the opportunity to ensure that the patient’s concerns are being captured and quantified, thus allowing the patient perspective to help guide clinical care. A well-developed PROM should demonstrate robust measurement properties (validity, reliability, responsiveness to change) and be capable of measuring the impact of interventions on a patient’s daily functioning and satisfaction in a clinically relevant way.
PROMs have proven their worth in comparative research (e.g., evaluating which intervention leads to the most satisfied patients) and in clinical care. The use of PROMs in routine clinical care, and providing feedback to patients using PRO data, was associated with better patient–physician dialogue, improved HRQL, and longer survival of cancer patients. The US National Academy of Medicine considers the use of PROMs to be an essential element of person-centered, high-quality care for patients with cancer, and the best way to quantify patients’ well-being.
Traditionally, the success of plastic surgery was determined by measuring surgical complication rates and/or by having an independent panel of clinical experts evaluate aesthetic results through photographs. The very first PROMs in plastic surgery are considered “ad hoc” questionnaires. Such questionnaires contained questions that were deemed relevant by clinical experts; however, a psychometric evaluation of the instrument’s performance was rarely performed. A limitation of early ad hoc questionnaires is that the majority did not include input from patients in their design. As a result, these questionnaires often contained questions that patients did not understand or concepts they considered to be irrelevant. Furthermore, these instruments often did not perform as consistently as the clinical experts thought they did. Ad hoc questionnaires can be useful if the goal is merely exploratory, i.e., to get a basic sense of answers to the questions posed in the questionnaire. Data from ad hoc questionnaires are not appropriate in the creation of guidelines, to determine treatment effectiveness in research or clinical care, or to determine reimbursement in value-based healthcare systems. Incorrect conclusions may be drawn based on flawed questionnaires; instead of the treatment being unsuccessful it may be that the questionnaire is asking the wrong questions and/or is ineffective in detecting clinical change in a consistent manner.
As psychometric methods advanced, the flaws in ad hoc questionnaires came to light, and PROMs began to be developed using more sophisticated approaches, such as Classical Test Theory (CTT). Instruments based on CTT were initially developed for research purposes, where for example, two groups received different interventions and were compared (e.g., experimental drug versus placebo). Such measures were generally validated to detect large differences between groups but were not capable of detecting smaller clinically relevant changes within an individual patient. Furthermore, most of these “legacy” questionnaires in the field of plastic and reconstructive surgery were developed without patient input. As a result, legacy PROMs have struggled to fully capture what is important to patients in clinical care, resulting in low response rates from patients and low clinical impact. Nonetheless, there was value in the use of CTT-developed PROMs, as they were often the only available tools to measure PROs. Given the length of time they have been available, legacy tools are often well known and frequently used. A large quantity of legacy data for general outcomes is therefore available with which to compare modern-day results.
Limitations of some PROMs developed using a CTT approach relate to the concepts they measure, which were often poorly conceptualized and often use a total score. In some “legacy” PROMs, all items within the PROM need to be completed to get a valid final score. If a patient, for example, completes almost all questions, but decides to skip sensitive questions about their sexual functioning, some PROMs may not be able to provide a (reliable) final score. Furthermore, many legacy PROMs would combine scores from different scales to create a “total score” or would measure multiple distinct concepts (e.g., sexual well-being, anxiety, fatigue) within one scale. In a clinical trial, total scores for scales that measure multiple concepts are not helpful as it is unclear which aspects of health have improved or deteriorated. Understanding in which domain the patient’s issues arise is important to offer targeted interventions to improve a patient’s functioning. The lack of clear concepts in some legacy PROMs is now seen as a flaw in design. New approaches to PROM development take a concept-driven approach to ensure that scales measure a single concept of interest, i.e., unidimensional.
In order to make evidence-based clinical decisions based on PRO data, PROMs need to measure outcomes just as rigorously and reliably as, for example, a ruler is able to measure length. Thus, nowadays the development of PRO-questionnaires needs proper psychometric evaluation and validation before being implemented as part of clinical care or research.
Different types of PROMs exist, and they vary in terms of content and intended application. PROMs can be divided into two main categories: (1) generic and (2) disease-specific (also known as condition-specific ). Generic PROMs can be used for an overall assessment before and after an intervention, and are often applicable to any patient irrespective of their condition or treatment. The scope of a generic PROM is broad and can be applied across many conditions and clinical situations. Commonly used generic PROMs include the SF-36 and PROMIS-29. These PROMs include questions like “Are you able to go for a walk of at least 15 minutes?”. A major advantage of generic PROMs is the ability to compare outcomes across a broad spectrum of health problems (e.g., arthritis vs. cataracts vs. peripheral vascular disease), interventions and patient populations. However, they often are not sensitive enough to measure change in a patient’s particular condition (e.g., breast cancer) after a surgical intervention (e.g., autologous reconstruction). A condition-specific PROM, like the BREAST-Q, is focused on a specific population or condition, and includes content that covers patient’s concerns within this specific setting, providing a precise measure of the effect of the intervention. These instruments can be used in clinical care within a specific patient group, or in clinical trials. They can detect small, but important changes related to the intervention being studied. However, they do not allow for comparisons of a patient’s health-state between different conditions or diseases. When undertaking clinical research, it is often recommended to incorporate both a generic and a disease-specific PROM as outcome measures. Table 12.2 lists some commonly used disease-specific and generic PROMs in plastic surgery.
| Questionnaire | Year of publication | Authors | Details |
|---|---|---|---|
| Disease-specific PROMs | |||
| Breast | |||
| BREAST-Q | 2008 | Pusic AL, Klassen A, Scott A, Klok J, Cordeiro PG, Cano SJ | Pre- and postoperative questionnaires for breast reconstruction, reduction mammaplasty, augmentation and breast-conserving surgery. Scale from 0–100 |
| BRECON-31 | 2012 | Temple CL, Cook EF, Ross DC, Bettger-Hahn M, MacDermid J | 31-item questionnaire using a 4-point Likert scale for patients who have undergone breast reconstruction. Assesses self-image, arm concerns, intimacy, satisfaction, recovery, self-consciousness, expectations, appearance, nipple, abdomen |
| Breast Evaluation Questionnaire | 2006 | Anderson RC, Cunningham B, Eskinder T | 55-item questionnaire using a 5-point Likert scale for patients undergoing augmentation mammaplasty. Assesses breast satisfaction and quality of life outcomes |
| Breast Reduction Assessed Severity Scale Questionnaire | 2007 | Sigurdson L, Kirkland SA, Mykhalovskiy E | 42-item questionnaire using the 5-point Likert scale for patients with mammary hypertrophy. Five domains assessed: physical implications, poor self-concept, body pain, negative social interactions, and physical appearance |
| Head and neck | |||
| FACE-Q | 2013 | Pusic AL, Klassen AF, Scott AM, Cano SJ | To assess impact and effectiveness of facial aesthetic procedures. Four scales examined: satisfaction with facial appearance, health-related quality of life, negative sequelae, and satisfaction with process of care |
| CLEFT-Q | 2018 | Klassen AF, Wong-Riff KW, Longmire NM, Albert A, Allen GC et al . | For people aged 8–29 with cleft lip and/or palate. Includes 12 scales that fall under 3 domains: facial appearance, facial function, and quality of life |
| Hand surgery | |||
| Disabilities of the Arm, Shoulder and Hand Questionnaire | 1996 | Hudak P, Amadio PC, Bombardier C and the Upper Extremity Collaborative Group | 30 base questions using a 5-point Likert scale. Score 0–100. Composite measure of upper extremity disability with additional module regarding work and sport |
| HAND-Q | 2022 | Sierakowski KL, Dean NR, Pusic AL, Klassen AF | The HAND-Q can be used with any type of congenital or acquired hand condition. This PROM includes 14 scales that measure 4 domains: appearance, HRQL, experience of care, and treatment. |
| Michigan Hand Outcomes Questionnaire | 1998 | Chung KC, Pillsbury MS, Walters MR, Hayward RA | 25 questions for the right and left hand each with 16 bilateral assessment. Modules assessed include hand function, ADLs, pain, satisfaction, aesthetics, and work performance |
| Patient-Rated Wrist Evaluation | 1998 | MacDermid JC, Turgeon T, Richards RS | 15 items that explore wrist pain and function. Scores ranging from 0 to 10 and converted to a weighted scale out of 100 |
| Boston Carpal Tunnel Questionnaire | 1993 | Levine DW, Simmons BP, Koris MJ, Daltroy LH, Hohl GG, Fossel AH, Katz JN | Assesses severity of symptoms and functional status associated with carpal tunnel syndrome |
| The Patient Outcomes of Surgery–Hand/Arm (POS–Hand/Arm) | 2004 | Cano SJ, Browne JP, Lamping DL, Roberts AH, McGrouther DA, Black NA | Aims to assess the physical activities, symptoms, psychological and cosmetic functioning, and satisfaction of surgery in patients who have conditions affecting the upper limb |
| Generic PROMs | |||
| SF-36 | 1992 | Ware JE, Jr. and Sherbourne CD | 36-item questionnaire that assesses physical functioning, social functioning, role limitation due to physical health, bodily pain, mental health, role limitations due to emotional health, vitality and general health perceptions. On all SF-36 scale, lower scores (<50) represent worse outcome |
| EQ5D | 1990 | The EuroQol Group | 5-item questionnaire in 2 pages. The first page contains 5 items describing health status across 5 dimensions: mobility, self-care, usual activity, pain/distress, and depression/anxiety. The second page displays a visual analogue rating scale on which the respondent makes an assessment of their overall health. The final outcome consisting of 2 single-index measures of health, the rating scale and the EQ5D index ranging from 0 to 100 |
| PROMIS-29 | 2011 | Hinchcliff M, Beaumont JL, Thavarajah K, Varga J, Chung A, Podlusku S, Carns M, Chang RW, Cella D | 29-item questionnaire, consisting of 4 items each from the 7 core PROMIS domains, as well as one 11-point rating scale for pain intensity. The 7 domains specifically relate to physical, mental, and social health and cover the most relevant areas of self-reported health for the greatest majority of people with chronic illness: pain, fatigue, depression, anxiety, sleep, physical function, and sexual function |
Understanding the methodology of how a PROM was developed is helpful when choosing an appropriate PROM for use in clinical practice or research study. Careful qualitative techniques are key to ensuring that PROMs focus on outcomes that matter most to patients and use language that is easy to understand. In addition, modern methods utilize more robust psychometric approaches in the form of Item Response Theory (IRT), within which a common approach is Rasch Measurement Theory (RMT). IRT is a mathematical framework, rather than a specific technique. The IRT approach focuses on individual items (i.e., questions) within the instrument rather than the overall test score. The major advantages of PROMs developed using RMT and IRT methods are their ability to measure the trait of interest (e.g., satisfaction with breasts) with interval-level measurement estimates, where the numbers generated by the PROM are in fact actual measurements, almost like a ruler ( Fig. 12.2 ). Furthermore, IRT and RMT-based instruments provide the ability to examine change at the individual patient level.
Developing a well-functioning, reliable and validated PROM takes time – often years – and requires psychometric and clinical expertise. To improve the quality of modern-day PROMs, international guidelines for PROM development, including step-by-step guides, have been created. The US Food and Drug Administration (FDA) recommends a multi-stage, iterative process for the development of a PROM, summarized in Fig. 12.3 .
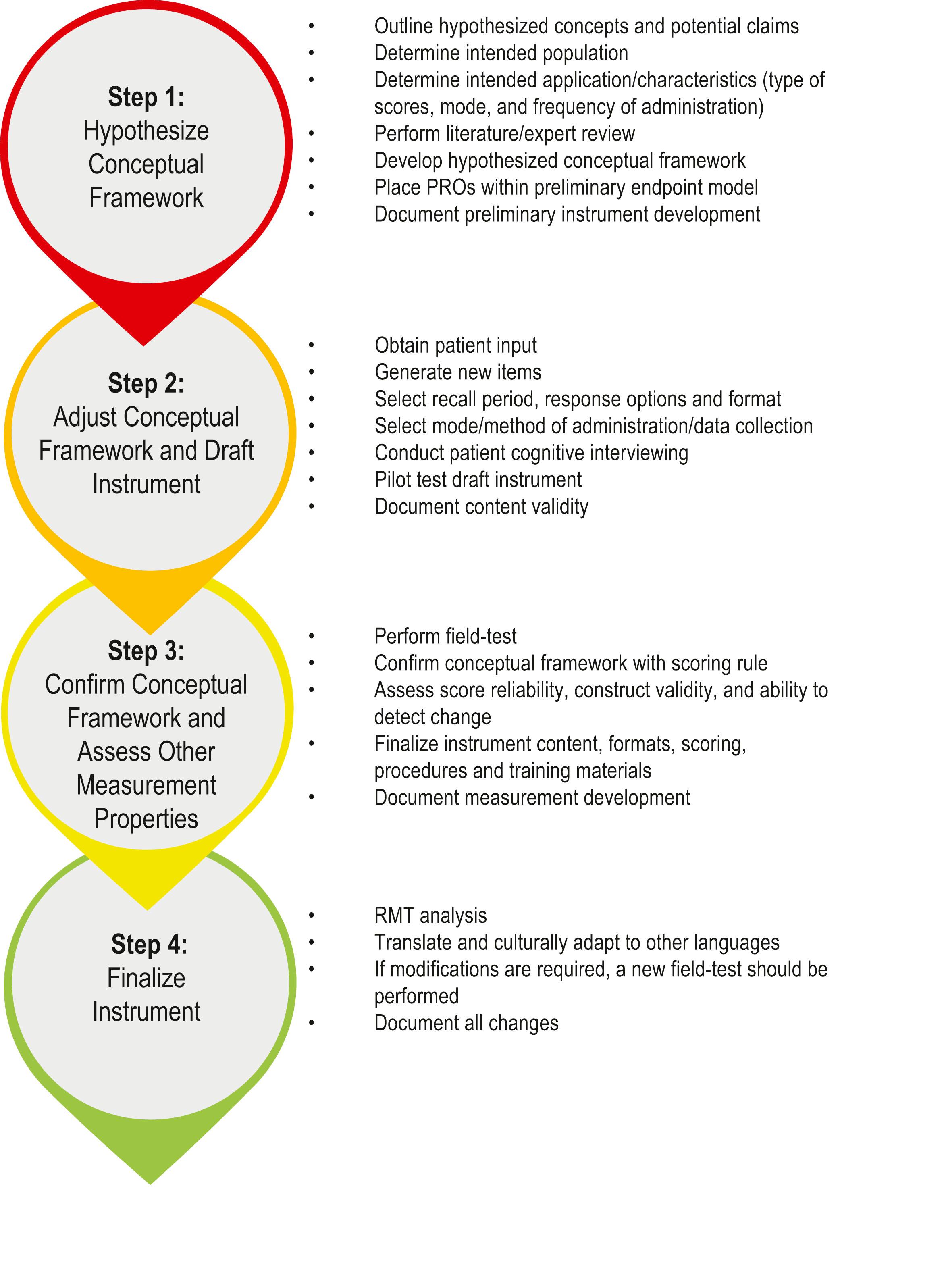
When outlining hypothesized concepts for a PROM, thought should be given to determining what kind of PROM is required, and what the purpose of the PROM will be (i.e., its intended application). The number of PROMs being developed has grown rapidly. A helpful first step before developing a new PROM is to perform a systematic review to identify if any PROMs already exist in the field or topic area of interest. Based on the findings of this systematic review, concepts important to patients can be generated and used to formulate a draft conceptual framework. For example, to develop the BREAST-Q, a literature review of PROMs used in cosmetic and reconstructive breast surgery was performed and 7 existing PROMs were identified. These PROMs were examined to determine their quality and the concepts included. Expert opinions from plastic surgeons, oncologist breast surgeons, psychologists and allied healthcare professionals can also be obtained to determine important concepts from their perspective.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here