Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Diabetic kidney disease (DKD) is the leading cause of kidney failure in adults. In the United States, approximately half of patients initiating dialysis have diabetes mellitus, and most of these have type 2 diabetes; patients with youth-onset type 2 diabetes, as shown in the Treatment Options for Type 2 Diabetes in Adolescents and Youth (TODAY) study, exhibit a particularly high risk of progressive DKD after a relatively short duration of disease. Mortality among patients with DKD is high, with cardiovascular diseases predominating. Once overt DKD (as defined by the presence of proteinuria and/or impaired kidney function) is present, kidney failure can often be postponed, but, in most instances, not prevented, by effective antihypertensive treatment and careful glycemic control. Accordingly, there has been intensive research into pathophysiologic mechanisms of diabetic kidney injury, predictors of risk for DKD, and early intervention strategies.
The classic terminology used to describe different states of urinary albumin excretion include normoalbuminuria, moderately increased albuminuria (previously microalbuminuria), and severely increased albuminuria (previously macroalbuminuria or “overt proteinuria”). Normoalbuminuria is typically defined as urinary albumin excretion rate (UAER) <20 µg/min or urinary albumin-to-creatinine ratio (UACR) < 30 mg/g (or 30 mg/day), moderately increased albuminuria as UAER between 20–200 µg/min or UACR between 30–300 mg/g (30–300 mg/day), and severely increased albuminuria as UAER >200 µg/min or UACR >300 mg/g (>300 mg/day). This chapter refers to categories of albuminuria using the updated nomenclature, translating from the original studies.
The pathophysiology underlying DKD is complex and remains incompletely understood. Although other important modulating factors may contribute, the long-term deleterious impacts of hyperglycemia and insulin resistance are central to the development and progression of DKD. Studies in both type 1 and type 2 diabetes have shown that improved glycemic control can mitigate the risk of developing DKD. Moreover, the development of the earliest diabetic kidney lesions can be slowed or prevented by strict glycemic control, as was demonstrated in a randomized trial in type 1 diabetic kidney transplant recipients. Similarly, intensive insulin treatment decreases the progression rates of glomerular lesions in patients with type 1 diabetes and moderately increased albuminuria. Finally, established diabetic glomerular lesions in the native kidneys of patients with type 1 diabetes regress with prolonged normalization of glycemic levels after successful pancreas transplantation. In summary, these studies strongly suggest that hyperglycemia is necessary for the development and maintenance of DKD in type 1 diabetes, as correction of hyperglycemia allows expression of reparative mechanisms that facilitate healing of the original diabetic glomerular injury.
Intraglomerular hemodynamic mechanisms, including hyperfiltration, likely play a significant role in the pathogenesis of DKD through neurohormonal (e.g., renin-angiotensin-aldosterone system activation) and tubular (e.g., tubuloglomerular feedback) pathways. However, patients with other causes of glomerular hyperfiltration, such as unilateral nephrectomy, do not typically develop evidence of kidney disease. Furthermore, it is unlikely that all patients with diabetes and glomerular hyperfiltration develop DKD. Therefore, glomerular hyperfiltration alone cannot fully explain the genesis of the early lesions of DKD. However, previous studies and clinical observations suggest that hemodynamic factors are important in modulating the initiation of nephropathy, and the rate of progression of diabetic lesions that are already well established. Studies in adults with type 1 and type 2 diabetes have also demonstrated that glomerular hyperfiltration is associated with a greater risk of experiencing a rapid glomerular filtration rate (GFR) decline. However, these findings were not recapitulated by an analysis in the Diabetes Control and Complications Trial (DCCT) where no association was found between glomerular hyperfiltration and the development of stage 3 CKD. It is worth noting that the presence of impaired GFR in normoalbuminuric patients with type 1 diabetes is associated with more severe glomerular lesions of DKD, and these patients may be at magnified risk of further progression, possibly on the basis of ischemia and inflammation-related mechanisms.
Systemic hypertension and a lack of normal nocturnal blood-pressure dipping may both be implicated in the pathogenesis of DKD. Supporting this hypothesis is the association between intensive blood-pressure control and decreased rates of progression from normoalbuminuria to moderately increased albuminuria and from moderately to severely increased albuminuria in both normotensive and hypertensive patients with type 2 diabetes.
Studies on human genetics offer important mechanistic insights into complex traits such as DKD. A genetic predisposition to diabetic nephropathy is suggested in multiple cross-sectional studies in type 1 and type 2 diabetic siblings concordant for diabetes. Importantly, diabetic sibling pairs that are concordant for DKD risk are also highly concordant for diabetic glomerulopathy lesions, and this risk is in part independent of glycemia. Novel loci associated with albuminuria were identified by genome-wide association study (GWAS) meta-analysis of albuminuria traits in the general population. An association between protein coding gene for cubilin (CUBN) and albuminuria was demonstrated, and gene-by-diabetes interactions were detected and confirmed for variants in HS6ST1 and near RAB38/CTSC. One large GWAS found a common missense mutation encoding for the collagen type IV alpha 3 chain gene and associated with a thinner GBM to be protective against DKD in T1D, a paradoxical finding that may guide new research on the pathogenesis of the disease. Another large GWAS in African American patients with T2D identified eight associations in seven genetic loci for DKD-related ESKD; further studies are required to confirm replicability. Additionally, an integrated biological approach linking clinical phenotyping with histopathological and molecular phenotyping of kidney tissue can enhance our understanding of the molecular pathogenesis engendering DKD.
Diabetic kidney disease is characterized not only by glomerular disease but also by tubulointerstitial injury. While glomerular changes have received more attention than tubulointerstitial changes in DKD research, tubular injury may be more closely associated with kidney function than glomerular injury. Tubular proteinuria predates microalbuminuria in youths with type 1 diabetes, suggesting that tubular damage may occur earlier than glomerular injury in the course of diabetic nephropathy. Tubular changes associated with DKD include basement membrane thickening, tubular hypertrophy, epithelial–mesenchymal transition, glycogen accumulation, and interstitial inflammation. Basement membrane thickening and tubular hypertrophy are mainly related to extracellular matrix (ECM) accumulation, which reflects an imbalance between ECM synthesis and degradation, is the principal cause of mesangial expansion that also contributes to expansion of the interstitium late in the disease. Several mechanisms have been proposed to explain the link between hyperglycemia and ECM accumulation. These include higher levels of TGFβ; activation of protein kinase C, (which stimulates ECM production through the cyclic adenosine monophosphate (cAMP) pathway); increased advanced glycation end products; and increased activity of aldose reductase, leading to accumulation of sorbitol. There is also growing evidence that oxidative stress is increased in diabetes and is also related to DKD, mediated through altered nitric oxide production and action, and endothelial dysfunction. Importantly, many factors associated with DKD may act through both hemodynamic and non-hemodynamic pathways. For example, angiotensin II increases intraglomerular pressure and hyperfiltration and also increases the production of injurious mediators such as protein kinase C. Intraglomerular hypertension, whether a consequence of angiotensin II, other neurohormones, or tubular factors, is associated with increased glomerular wall tension and shear stress, leading to the activation of proinflammatory and profibrotic pathways.
Glycocalyx dysfunction has recently attracted attention as a potential mediator of both diabetic glomerulopathy and tubulopathy. The glycocalyx is a polysaccharide gel that covers the luminal surface of the endothelium, thereby acting as a filtration barrier and regulator of endothelial vascular function. Under exposure to hyperglycemic conditions, the glycocalyx is modified, leading to exposure of heparin sulfate domains that allow chemokine binding and inflammation and result in glycocalyx degradation. Albuminuria is likely to, at least in part, occur as a consequence disruption of the glycocalyx. The presence of overlapping and interrelated injurious pathways that promote diabetic nephropathy highlight the need for a multifaceted therapeutic approach, as outlined below.
In patients with type 1 diabetes, glomerular lesions can appear within a few years after diabetes onset. The same progression time frame occurs when a normal kidney is transplanted into a patient with diabetes. The changes in kidney structure caused by diabetes are specific, creating a pattern not seen in any other disease, and the severity of these diabetic lesions is related to the functional disturbances of the clinical kidney disease as well as to diabetes duration, glycemic control, and genetic factors. However, the relationship between the duration of type 1 diabetes and extent of glomerular pathology is not precise. This is consistent with the marked variability in susceptibility to this disorder, such that some patients may develop kidney failure after having diabetes for 15 years, whereas others escape kidney complications despite having type 1 diabetes for decades.
Kidney hypertrophy is the earliest structural change in type 1 diabetes but is not reflected in any specific light microscopic changes. In many patients, glomerular structure remains normal or near normal even after decades of diabetes, whereas others develop progressive diffuse mesangial expansion, seen mainly as increased periodic acid–Schiff (PAS)-positive ECM mesangial material. In about 40% to 50% of patients developing proteinuria, there are areas of extreme mesangial expansion called Kimmelstiel-Wilson nodules (nodular mesangial expansion). Mesangial cell nuclei in these nodules are palisaded around masses of mesangial matrix material with compression of surrounding capillary lumina. Nodules are thought to result from earlier glomerular capillary microaneurysm formation. Notably, about half of patients with severe diabetic nephropathy do not have these nodular lesions; therefore, although Kimmelstiel-Wilson nodules are diagnostic of diabetic nephropathy, they are not necessary for severe kidney disease to develop.
Early changes often include arteriolar hyalinosis lesions involving replacement of the smooth muscle cells of afferent and efferent arterioles with PAS-positive waxy, homogenous material ( Fig. 26.1 ). The severity of these lesions is directly related to the frequency of global glomerulosclerosis, perhaps as the result of glomerular ischemia. GBM and TBM thickening may be seen with light microscopy, although they are more easily seen with electron microscopy. In addition, tubular glomeruli and glomerulotubular junction abnormalities are present in proteinuric patients with type 1 diabetes and may be important in the progressive loss of GFR in DKD. Finally, usually quite late in the disease, tubular atrophy and interstitial fibrosis occur.
![Fig. 26.1, Light microscopy photographs of glomeruli in sequential kidney biopsies performed at baseline and after 5 and 10 years of follow-up in a patient with long-standing normoalbuminuric type 1 diabetes with progressive mesangial expansion and kidney function deterioration. (A) Diffuse and nodular mesangial expansion and arteriolar hyalinosis in this glomerulus from a patient who was normotensive and normoalbuminuric at the time of this baseline biopsy, 21 years after diabetes onset (periodic acid–Schiff [PAS] stain, original magnification ×400). (B) Five-year follow-up biopsy showing worsening of the diffuse and nodular mesangial expansion and arteriolar hyalinosis in this now microalbuminuric patient with declining glomerular filtration rate (GFR) (PAS stain, ×400). (C) Ten-year follow-up biopsy showing more advanced diabetic glomerulopathy in this now proteinuric patient with further reduced GFR. Note also the multiple small glomerular (probably efferent) arterioles in the hilar region of this glomerulus (PAS stain, ×400) and in the glomerulus shown in (A). Fig. 26.1, Light microscopy photographs of glomeruli in sequential kidney biopsies performed at baseline and after 5 and 10 years of follow-up in a patient with long-standing normoalbuminuric type 1 diabetes with progressive mesangial expansion and kidney function deterioration. (A) Diffuse and nodular mesangial expansion and arteriolar hyalinosis in this glomerulus from a patient who was normotensive and normoalbuminuric at the time of this baseline biopsy, 21 years after diabetes onset (periodic acid–Schiff [PAS] stain, original magnification ×400). (B) Five-year follow-up biopsy showing worsening of the diffuse and nodular mesangial expansion and arteriolar hyalinosis in this now microalbuminuric patient with declining glomerular filtration rate (GFR) (PAS stain, ×400). (C) Ten-year follow-up biopsy showing more advanced diabetic glomerulopathy in this now proteinuric patient with further reduced GFR. Note also the multiple small glomerular (probably efferent) arterioles in the hilar region of this glomerulus (PAS stain, ×400) and in the glomerulus shown in (A).](https://storage.googleapis.com/dl.dentistrykey.com/clinical/PathogenesisPathophysiologyandTreatmentofDiabeticKidneyDisease/0_3s20B9780323791229000261.jpg)
Diabetes is characterized by increased linear staining of the GBM, TBM, and Bowman capsule, especially for immunoglobulin G (mainly IgG4) and albumin. Although this staining is removed only by strong acid conditions, consistent with strong ionic binding, the intensity of staining is not related to the severity of the underlying lesions.
The first measurable change observed in DKD is thickening of the GBM, which can be detected as early as 1.5 to 2.5 years after onset of type 1 diabetes ( Fig. 26.2 ). TBM thickening is also seen and parallels GBM thickening. A measurable increase in the relative area of the mesangium begins by 4 to 5 years, with the proportion of the glomerular volume that is mesangium increasing from about 20% (normal) to about 40% when proteinuria begins and to 60% to 80% in patients with stage 3 chronic kidney disease (CKD). Immunohistochemical studies indicate that these changes in mesangium, GBM, and TBM represent expansion of the intrinsic ECM components at these sites, most likely including types IV and VI collagen, laminin, and fibronectin.
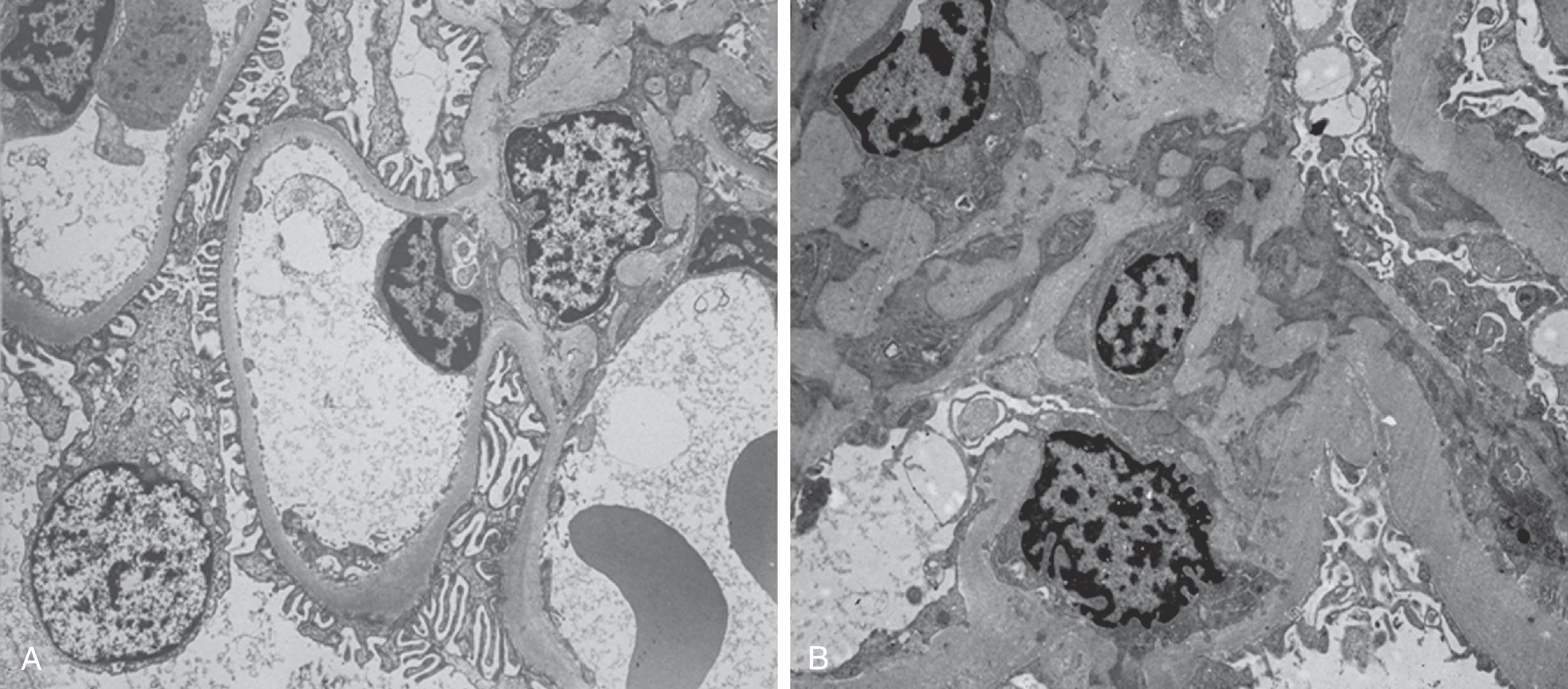
Qualitative and quantitative changes in the kidney interstitium are observed in patients with various kidney diseases. Interstitial fibrosis is characterized by an increase in ECM proteins and cellularity. Preliminary studies suggest that the pathogenesis of interstitial changes in diabetic nephropathy is different from the changes that occur in the mesangial matrix, GBM, and TBM. Whereas, for all but the later stages of DKD, GBM, TBM, and mesangial matrix changes represent the accumulation of basement membrane ECM material, early interstitial expansion is largely a result of cellular alterations and only later, when GFR is already compromised, is interstitial expansion associated with increased interstitial fibrillar collagen and peritubular capillary loss. Consistent with most kidney diseases affecting the glomeruli, the fraction of GBM covered by intact, non-detached foot processes is lower in proteinuric patients with diabetes when compared with either control subjects or individuals with type 1 diabetes with low levels of albuminuria. Moreover, the fraction of the glomerular capillary luminal surface covered by fenestrated endothelium is reduced in all stages of DKD, with increasing severity in normoalbuminuria, moderately increased albuminuria, and severely increased albuminuria in patients with type 1 diabetes as compared with controls.
Glomerular and tubular structures in type 2 diabetes are less well studied but overall seem to manifest in a more heterogeneous fashion than is observed in type 1 diabetes. Between 30% and 50% of patients with type 2 diabetes who have clinical features of DKD have typical pathology findings, including diffuse and nodular mesangial expansion and arteriolar hyalinosis ( Fig. 26.3 ). Notably, some patients, despite the presence of albuminuria, have absent or only mild diabetic glomerulopathy, whereas others have disproportionately severe tubular and interstitial abnormalities and/or vascular lesions and/or an increased number of globally sclerosed glomeruli. Patients with type 2 diabetes with moderately increased albuminuria more frequently have morphometric glomerular structural measures in the normal range on electron microscopy and less severe lesions compared to patients with type 1 diabetes with moderately or severely increased albuminuria. Interestingly, Pima Indians with type 2 diabetes, a high-risk population for kidney failure, have lesions more typical of those seen in type 1 diabetes and were found to have structural defects up to a decade prior to onset of impaired GFR.
![Fig. 26.3, Light microscopy photographs of glomeruli from type 1 (A) and type 2 (B through D) diabetic patients. (A) Diffuse and nodular mesangial expansion and arteriolar hyalinosis in a glomerulus from an individual with type 1 diabetes and moderately elevated albuminuria (periodic acid–Schiff [PAS] stain, original magnification ×400). (B) Normal or near-normal kidney structure in a glomerulus from an individual with type 2 diabetes and moderately elevated albuminuria (PAS stain, ×400). (C) Changes “typical” of diabetic kidney disease (glomerular, tubulointerstitial, and arteriolar changes occurring in parallel) in a kidney biopsy specimen from an individual with type 2 diabetes and moderately elevated albuminuria (PAS stain, ×400). (D) “Atypical” patterns of injury, with absent or only mild diabetic glomerular changes associated with disproportionately severe tubulointerstitial changes. Note also a glomerulus undergoing glomerular sclerosis (PAS stain, ×400) (B through D). Fig. 26.3, Light microscopy photographs of glomeruli from type 1 (A) and type 2 (B through D) diabetic patients. (A) Diffuse and nodular mesangial expansion and arteriolar hyalinosis in a glomerulus from an individual with type 1 diabetes and moderately elevated albuminuria (periodic acid–Schiff [PAS] stain, original magnification ×400). (B) Normal or near-normal kidney structure in a glomerulus from an individual with type 2 diabetes and moderately elevated albuminuria (PAS stain, ×400). (C) Changes “typical” of diabetic kidney disease (glomerular, tubulointerstitial, and arteriolar changes occurring in parallel) in a kidney biopsy specimen from an individual with type 2 diabetes and moderately elevated albuminuria (PAS stain, ×400). (D) “Atypical” patterns of injury, with absent or only mild diabetic glomerular changes associated with disproportionately severe tubulointerstitial changes. Note also a glomerulus undergoing glomerular sclerosis (PAS stain, ×400) (B through D).](https://storage.googleapis.com/dl.dentistrykey.com/clinical/PathogenesisPathophysiologyandTreatmentofDiabeticKidneyDisease/2_3s20B9780323791229000261.jpg)
It is unclear why some studies show more structural heterogeneity in type 2 than in type 1 diabetes whereas others do not. Regardless, the rate of kidney disease progression in type 2 diabetes is related, at least in part, to the severity of the classic changes of diabetic glomerulopathy. Although there are reports that patients with type 2 diabetes have an increased incidence of nondiabetic lesions, such as proliferative glomerulonephritis and membranous nephropathy, this likely reflects biopsies more often being performed in patients with atypical clinical features. When biopsies are performed for research purposes, the incidence of other definable kidney diseases is very low (<5%). It is also noteworthy that a significant proportion of patients with type 2 diabetes exhibit an accelerated GFR decline in the absence of albuminuria. Although this phenotype is not yet completely understood, it has been suggested that this may reflect a predominance of microvascular disease rather than glomerular disease, thereby attenuating albuminuria risk. GFR reduction in the absence of albuminuria highlights the need to identify alternate biomarkers that better capture early DKD risk.
Kidney disease progression rates vary greatly among individuals with diabetes. Patients with type 1 diabetes and patients with proteinuria who are biopsied for research purposes always have advanced glomerular lesions and usually have vascular, tubular, and interstitial lesions as well. There is considerable overlap in glomerular structural changes between patients with long-standing normoalbuminuria and those with moderately increased albuminuria, as some normoalbuminuric patients with long-standing type 1 diabetes can have quite advanced kidney lesions, whereas many patients with long-standing diabetes and normoalbuminuria have structural measurements within the normal range.
Ultimately, expansion of the mesangium, mainly resulting from ECM accumulation, reduces or even obliterates the glomerular capillary luminal space, decreasing the glomerular filtration surface and, therefore, decreasing the GFR. Accordingly, the fraction of the glomerulus occupied by mesangium correlates with both GFR and albuminuria in patients with type 1 diabetes, reflecting in part the inverse relationship between mesangial expansion and total peripheral GBM filtration surface per glomerulus. GBM thickness is also directly related to the albumin excretion rate. Finally, the extent of global glomerulosclerosis and interstitial expansion are correlated with the clinical manifestations of DKD (proteinuria, hypertension, and declining GFR).
In patients with type 1 diabetes, glomerular, tubular, interstitial, and vascular lesions tend to progress more or less in parallel, whereas in patients with type 2 diabetes this often is not the case. Current evidence suggests that, among type 2 diabetes patients with moderately increased albuminuria, those patients with typical diabetic glomerulopathy have a higher risk of progressive GFR loss than those with lesser degrees of glomerular changes. A remarkably high frequency of glomerular tubular junction abnormalities can be observed in proteinuric type 1 diabetic patients. Most of these abnormalities are associated with tuft adhesions to Bowman capsule at or near the glomerular tubular junction (tip lesions). The frequency and severity of these lesions (as well as the presence of completely atubular glomeruli) predict GFR loss.
The data on structural–functional relationships in type 2 diabetes are less abundant. In several small studies, morphometric measures of diabetic glomerulopathy correlated with kidney function parameters similar to those observed in type 1 diabetes, although there seems to be a subset of patients who have normal glomerular structure despite persistent albuminuria. Overall, the relationships between kidney function and glomerular structure are less precise in patients with type 2 diabetes. Importantly the rate of GFR decline significantly correlates with the severity of diabetic glomerulopathy lesions. Thus, kidney lesions different from those typical of diabetic glomerulopathy should be considered when investigating the nature of abnormal levels of albuminuria in type 2 diabetes. These lesions include changes in the structure of renal tubules, interstitium, arterioles, and podocytes. For example, Pima Indians with type 2 diabetes and proteinuria have fewer podocytes per glomerulus than those without evidence of kidney disease, and, in this population, a lower number of podocytes per glomerulus at baseline was the strongest predictor of greater increases in albuminuria and of progression to overt DKD in individuals with moderately increased albuminuria. These results suggest that changes in podocyte structure and density occur early in DKD and might contribute to increasing albuminuria in these patients. More biopsy data are needed in people with type 1 and type 2 diabetes to better characterize DKD and rule out non-diabetes-related kidney disease. Data from genome-wide intrarenal gene expression profiling, morphometric analyses of protocol biopsies, and clinical outcomes were recently integrated to explore novel pathways of early DKD in T2D. This data linked genes associated with cortical interstitial fractional volume (a marker of tubule-interstitial damage) and long-term clinical outcomes of albuminuria and GFR, suggesting potential novel targets for markers of early DKD.
The lesions of DKD have long been considered irreversible. Theoretically, if reversal were possible, this would happen in the setting of long-term normoglycemia. Interestingly, in pancreas transplant recipients, the lesions of diabetic injury were unaffected after 5 years of normoglycemia but reversed in all patients by 10 years posttransplant, with a remarkable amelioration of glomerular structure abnormalities evident by light microscopy, including total disappearance of Kimmelstiel-Wilson nodular lesions. The latency needed for diabetic lesions to disappear is consistent with their slow rate of development. The understanding of the molecular and cellular mechanisms involved in these repair processes could provide new directions for the treatment of DKD.
Both kidney and cardiovascular morbidity and mortality are increased in patients with type 2 diabetes, particularly in those with DKD. Accordingly, treatment goals in these individuals focus on slowing the rate of GFR decline and delaying the onset of kidney failure as well as primary and secondary prevention of cardiovascular disease. This is mainly done by targeting multiple kidney and cardiovascular risk factors, such as hyperglycemia, hypertension, and dyslipidemia ( Fig. 26.4 ). Multiple completed cardiovascular outcome trials with glucose-lowering agents have shown substantial cardiac and kidney benefits, altering the clinical approach to anti-glycemic therapies in type 2 diabetes. The remainder of this chapter reviews the traditional and novel therapeutic options to decrease the risk of kidney and cardiovascular morbidity and mortality in patients with diabetes.
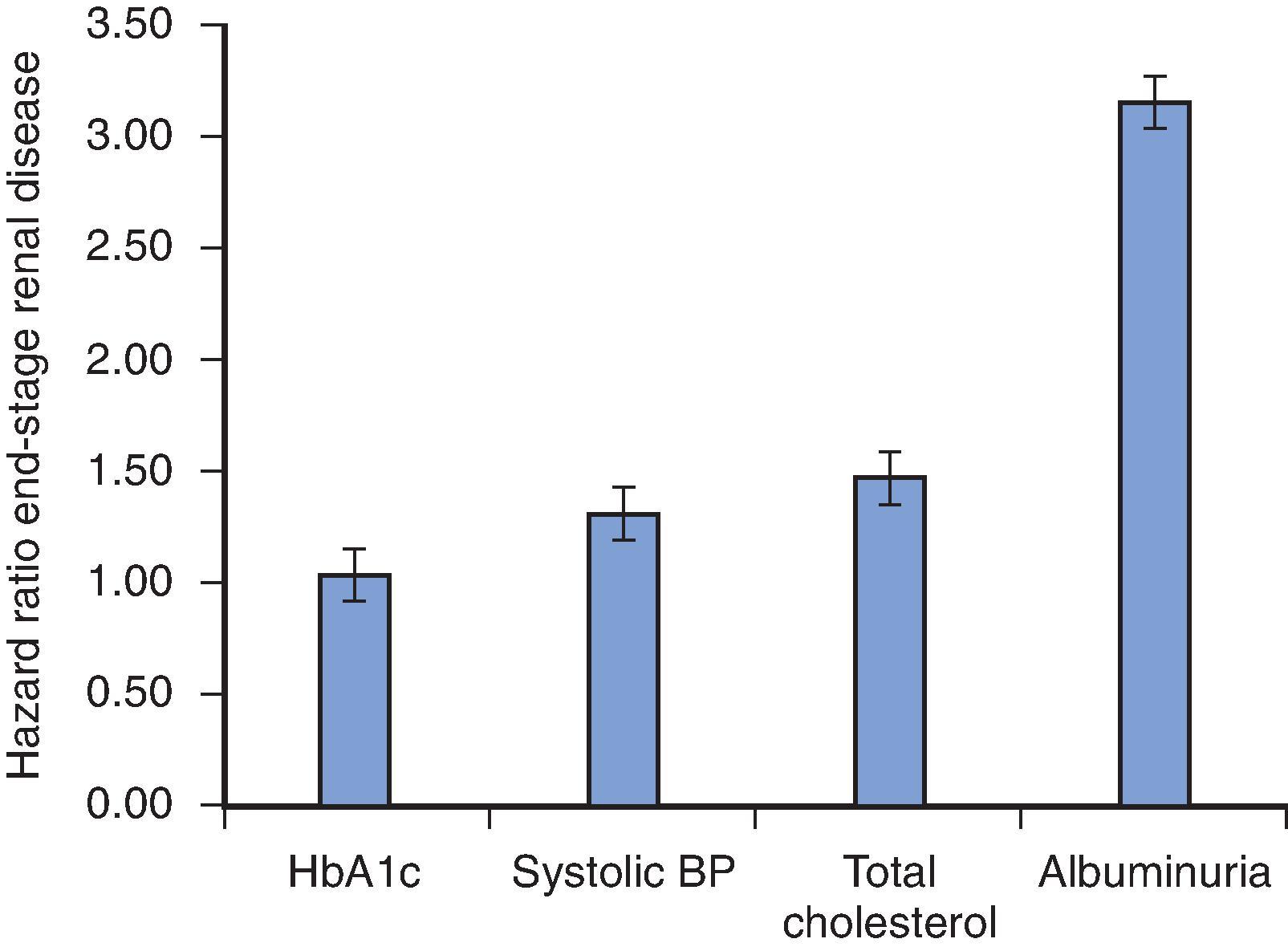
Poor glycemic control, as reflected by higher hemoglobin A1c (HbA1c) levels, is associated with markedly worse kidney and cardiovascular outcomes in observational studies of patients with type 1 and type 2 diabetes, and targeting HbA1c values lower than 7% (53 mmol/mol) may delay the progression of DKD, including development of albuminuria. In patients with type 1 diabetes, the benefit of intensive glucose control in the prevention of microvascular complications (specifically retinopathy or moderately increased albuminuria) was demonstrated in the DCCT, where long-term follow-up showed a significant reduction in the risk of developing reduced GFR among individuals who were treated intensively earlier in the course of diabetes. In type 2 diabetes, the United Kingdom prospective diabetes study (UKPDS) and ADVANCE-ON documented benefit of intensive glucose targeting on microvascular complications. Of note, although most studies of type 2 diabetes have shown a benefit in kidney outcomes, multiple trials failed to show a benefit of intensive glycemic control on mortality and cardiovascular disease, with some trials actually showing higher mortality with intensive control. In contrast with this older literature, more recent trials with sodium-glucose cotransporter-2 inhibitors (SGLT-2 inhibitors) and glucagon-like peptide-1 (GLP-1) receptor agonists have shown reductions in cardiovascular risk (and kidney benefits in the case of SGLT-2 inhibitors), with only modest reductions in HbA1c. This highlights the importance of non-glycemic mechanisms leading to cardiorenal damage, although the underlying pathways require further study. Accordingly, a careful, individualized approach is required when assigning glycemic targets in individuals with diabetes and kidney disease.
Kidney and cardiac risk should be considered when prescribing glucose-lowering therapies in patients with type 2 diabetes, given the increasing body of evidence for cardiac and kidney benefits with newer drug classes such as SGLT-2 inhibitors and GLP-1 receptor agonists. While dipeptidyl peptidase 4 (DPP-4) inhibitors are frequently prescribed in patients with kidney disease due to safety concerns around hypoglycemia at late stages of CKD with other classes of drugs such as insulin and sulfonylureas, DPP-4 inhibitors have shown cardiac neutrality with no benefit on primary cardiovascular or secondary kidney outcomes.
When prescribed for glycemic control, most drug classes may be used in patients with diabetes, including those with and without kidney function impairment until late stage 3 CKD ( Table 26.1 ). Metformin is cleared by the kidney but is considered safe to use in patients with kidney disease and estimated glomerular filtration rate (eGFR) of ≥30 mL/min/1.73 m2. Reduction in the doses of other oral hypoglycemic agents in later stages of CKD may be necessary, especially for some sulfonylurea compounds that are metabolized by the kidney. In many countries, SGLT-2 inhibitor initiation is currently approved until eGFR<30 mL/min/1.73 m 2 , and similar approaches have been reflected in the KDIGO Clinical Practice Guidelines. An important caveat is that the DAPA-CKD trial enrolled participants with eGFR as low as 25 mL/min/1.73 m 2 , resulting in US FDA approval for initiation at this GFR level. Of note, SGLT-2 inhibitors are authorized to be continued at lower eGFR levels, with these thresholds only for drug initiation. Some GLP-1 receptor agonists such as semaglutide and dulaglutide can be used until eGFR<15 mL/min/1.73 m 2 . Most DPP4i require dose reduction in late stages of CKD, whereas linagliptin is not cleared by the kidney and, therefore, dose adjustment is not required. As insulin is degraded by the kidney, dose reduction of insulin may be needed to prevent hypoglycemia as GFR declines.
| Class | Mechanism of Action | Examples of Drugs | Renal Clearance | HbA1c Lowering (%) | Use in Non-Dialysis CKD | Use in Dialysis | Advantage | Disadvantage |
| Biguanides (European Union 1958; United States 1995 a ) | Inhibits hepatic glucose production and increases insulin sensitivity | Metformin | Excreted unchanged in urine | 1.5 | Contraindicated in advanced CKD | Contraindicated | Long-term safety; low costs; weight neutral | Risk of lactic acidosis in CKD patients; gastrointestinal side effects |
| Sulfonylureas (1946 a ) | Binds to SU receptor in β-cells and increases calcium influx followed by insulin release | GliclazideGlipizideGlimepiride Glyburide | More than 90% metabolized in liver to weakly active or inactive metabolites and excreted in urine and feces | 1.5 | May be used | Glipizide may be used; use Glimepiride and glyburide with caution | Long-term safety; low costs | Hypoglycemia; weight gain |
| Meglitinides (1997 a ) | Binds to SU receptor (different from SU site) and increases calcium influx followed by insulin release | NateglinideRepaglinide | Metabolized by liver (100%) and excreted in urine (10%) and feces (90%) | 1.0 | May be used | No data for patients with creatinine clearance less than 20 mL/min | Rapid onset of action and short acting | Few long-term safety data; weight gain |
| Thiazolidinediones (1997 a ) | Decreases peripheral insulin resistance, thus increasing insulin sensitivity | Pioglitazone | Metabolized by liver to weakly active metabolites; excreted in urine (15%) and feces (85%) | 0.6 to 1.5 | May be used; no dose adjustments necessary; caution around edema/heart failure | May be used; no dose adjustments necessary; caution around edema/heart failure | Low-risk hypoglycemia | Pioglitazone is associated with increased risk of bladder cancer; Rosiglitazone withdrawn from the market because of increased cardiovascular risk |
| GLP-1 receptor agonists (2005 a ) | Binds to the pancreatic GLP-1 receptor and promotes insulin secretion, decreases glucagon secretion, gastric emptying, and appetite | Short acting:ExenatideLixisenatideLong-acting:LiraglutideDulaglutideSemaglutideExenatide long acting release | Metabolized by kidney, excreted in urine | 0.7 to 1.2 on top of metformin or SU derivatives | Semaglutide, dulaglutide, and liraglutide may be used | Semaglutide and dulaglutide may be used with caution | Reduced rate of death from cardiovascular causes, nonfatal myocardial infarction, or nonfatal stroke (LEADER, SUSTAIN-6, REWIND trials), weight loss, low risk of hypoglycemia | Subcutaneous administration, cost |
| DPP-4 inhibitors (2006 a ) | Blocks DPP-4, which inactivates endogenous incretins | SaxagliptinSitagliptinLinagliptinAlogliptin | Excreted mostly unchanged in urine and feces (Linagliptin metabolized and excreted in feces) | ∼0.8 (on top of metformin/SU derivatives) | Dose adjustments necessary for saxagliptin, sitagliptin, and alogliptin | Dose adjustments necessary for saxagliptin, sitagliptin, and alogliptin | Weight neutral; low risk of hypoglycemia | No proven cardiovascular benefits, possible risk of heart failure with saxagliptin and alogliptin |
| SGLT-2 inhibitors (2013) | Inhibits proximal tubular glucose reabsorption | EmpagliflozinCanagliflozinDapagliflozinErtugliflozin | Metabolized by liver to active metabolites; excreted in urine and feces | ∼0.8 (on top of metformin) | Less A1c lowering in CKD but kidney and cardiovascular benefits extended to patients with CKDCanagliflozin continued in CREDENCE and dapagliflozin in DAPA-CKD until dialysis | No clinical experience; not recommended | Reduced cardiac endpoints (death from cardiovascular causes, nonfatal myocardial infarction, or nonfatal stroke), renal endpoints (doubling of serum creatinine, ESRD and renal death) (EMPA-REG, CANVAS, CREDENCE, DAPA-CKD trials), and improved heart failure outcomes (cardiovascular death, HF presentation) (DAPA-HF, EMPORER-Reduced) weight loss, low risk of hypoglycemia | Cost, risk of euglycemic diabetic ketoacidosis |
a Year drug became available for clinical us CKD, Chronic kidney disease; DPP-4, dipeptidylpeptidase 4; GLP-1, glucagon-like peptide-1; SGLT-2, sodium-glucose cotransporter-2; SU, sulphonylurea; HF, heart failure
The role of the kidney in maintaining glucose homeostasis is increasingly appreciated. Glucose is filtered at the glomerulus and reclaimed via tubular reabsorption along with sodium through SGLT-2, which is located in the proximal tubule. The SGLT-2 transporter accounts for the reabsorption of approximately 90% of all filtered glucose, whereas the SGLT-1 transporter, located in the more distal proximal tubule, reabsorbs the remaining 10%. SGLT-2 inhibitors reversibly inhibit the SGLT-2 transporter, leading to enhanced glucose and sodium excretion and, in turn, to reductions in plasma glucose, HbA1c, and body weight (see Table 26.1 ).
In addition to improving glycemic control, SGLT-2 inhibitors have heart and kidney protective roles, although the mechanisms for cardiorenal protection remain to be fully elucidated. SGLT-2 inhibitors induce an acute diuretic effect due to both osmotic diuresis and natriuresis, leading to a sustained reduction in plasma volume of approximately 7%. These agents lower systolic and diastolic blood pressure by approximately 3–5/1–2 mm Hg, respectively. As a consequence, modest weight loss (2–3 kg) observed with SGLT-2 inhibitors typically includes both fluid and adipose tissue loss. SGLT-2 inhibition also induces uricosuria via exchange of filtered glucose for uric acid in the kidney, resulting in a 10% to 15% reduction in plasma uric acid levels. While the glycosuric effect and, hence, HbA1c lowering of SGLT-2 is attenuated in patients with GFR <60 mL/min/1.73 m 2 , the beneficial effects on blood pressure and body weight loss are generally preserved in patients with impaired GFR. Initiation of SGLT-2 inhibition is associated with an acute, initial “dip” in GFR of 3-4 mL/min/1.73 m 2, which reverses with cessation of therapy, likely reflecting intrarenal hemodynamic effects.
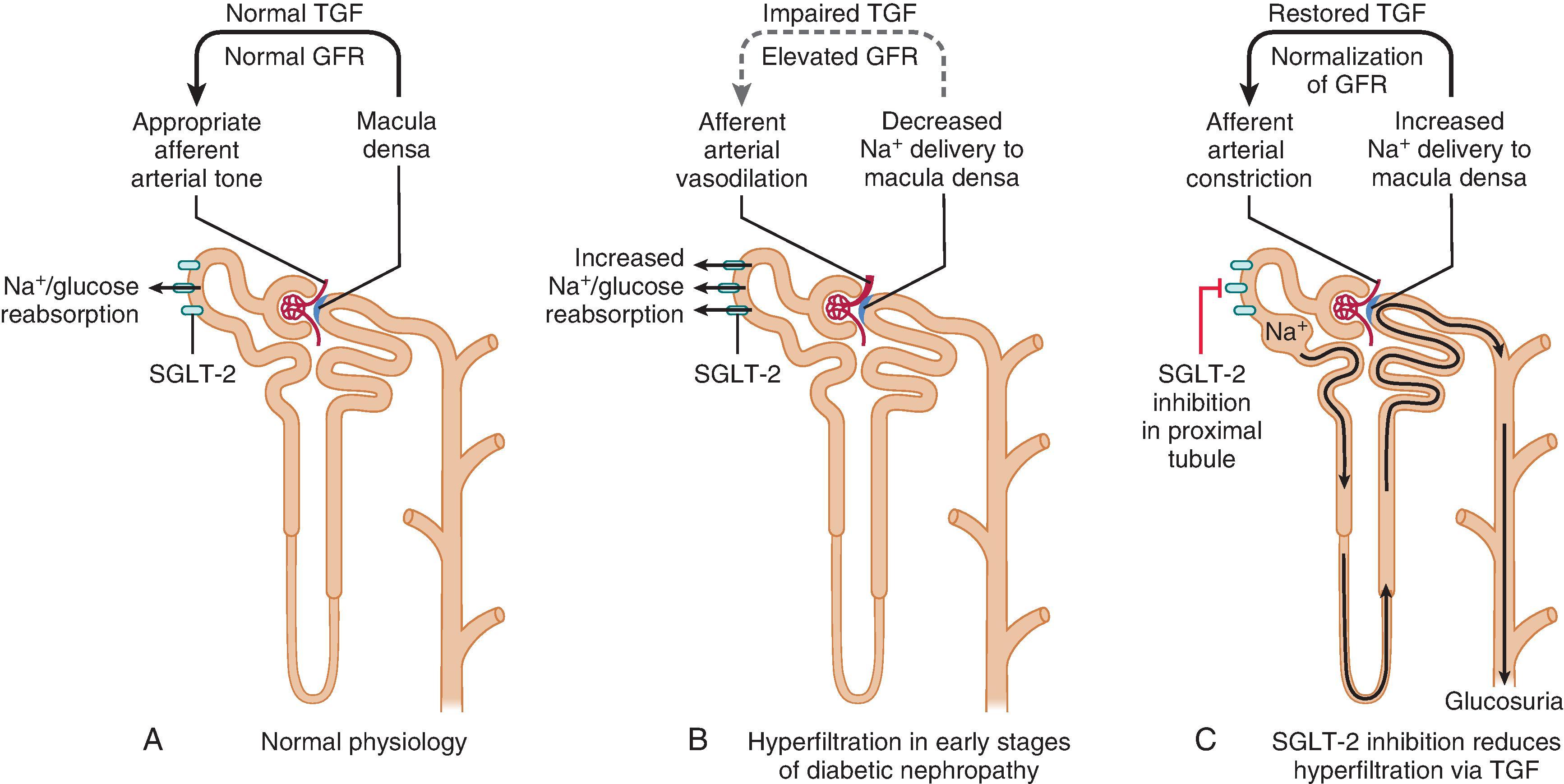
The acute effect of SGLT-2 on kidney function is likely to be primarily mediated by the tubuloglomerular feedback (TGF) mechanism ( Fig. 26.5 ). Increased bioactivity of SGLT-2 in patients with diabetes leads to increased sodium reabsorption at the proximal tubule and decreased distal sodium delivery to the macula densa, which is sensed as a reduction in effective circulating volume by the juxtaglomerular apparatus. This leads to downregulation of the TGF, vasodilation of the afferent renal arteriole, and attenuated hyperfiltration, at least in the setting of early T1D. Aside from attenuating TGF-mediated hyperfiltration, other direct kidney effects of SGLT-2 inhibition are being investigated, including suppression of proinflammatory and pro-fibrotic pathways and protection against kidney ischemia.
Four recent trials with SGLT-2 inhibition have demonstrated important cardiac benefits in patients with type 2 diabetes. Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes (EMPA-REG OUTCOME) and Canagliflozin and Cardiovascular and Renal Events in Type 2 Diabetes (CANVAS Program) included patients with type 2 diabetes and known atherosclerotic cardiovascular disease (ASCVD) in 100% and 66% of patients, respectively; each trial demonstrated a 14% reduction in the primary endpoint of major adverse cardiac event (MACE) outcome of cardiovascular death, non-fatal myocardial infarction, or non-fatal stroke in the group randomized to SGLT-2 inhibitors compared to placebo. Dapagliflozin and Cardiovascular Outcomes in Type 2 Diabetes (DECLARE TIMI-58) included patients with cardiac risk factors and only 40% of patients with a history of ASCVD, and demonstrated a 17% reduction in cardiovascular death or hospitalization for heart failure. Finally, in the Evaluation of Ertugliflozin Efficacy and Safety Cardiovascular Outcomes Trial (VERTIS CV), ertugliflozin demonstrated non-inferiority but did not impact MACE or CV death. Hospitalization for heart failure was, nevertheless, reduced by 30% in the cohort with established ASCVD, highlighting the consistency of the effect on this important clinical marker of benefit. Meta-analyzed data from the first three trials (excluding VERTIS CV) including over 34,000 patients revealed a reduction in the MACE outcome of 11% in patients with a history of ASCVD, and a 23% reduction in the risk of CV death or hospitalization for heart failure in patients with and without a history of ASCVD and heart failure.
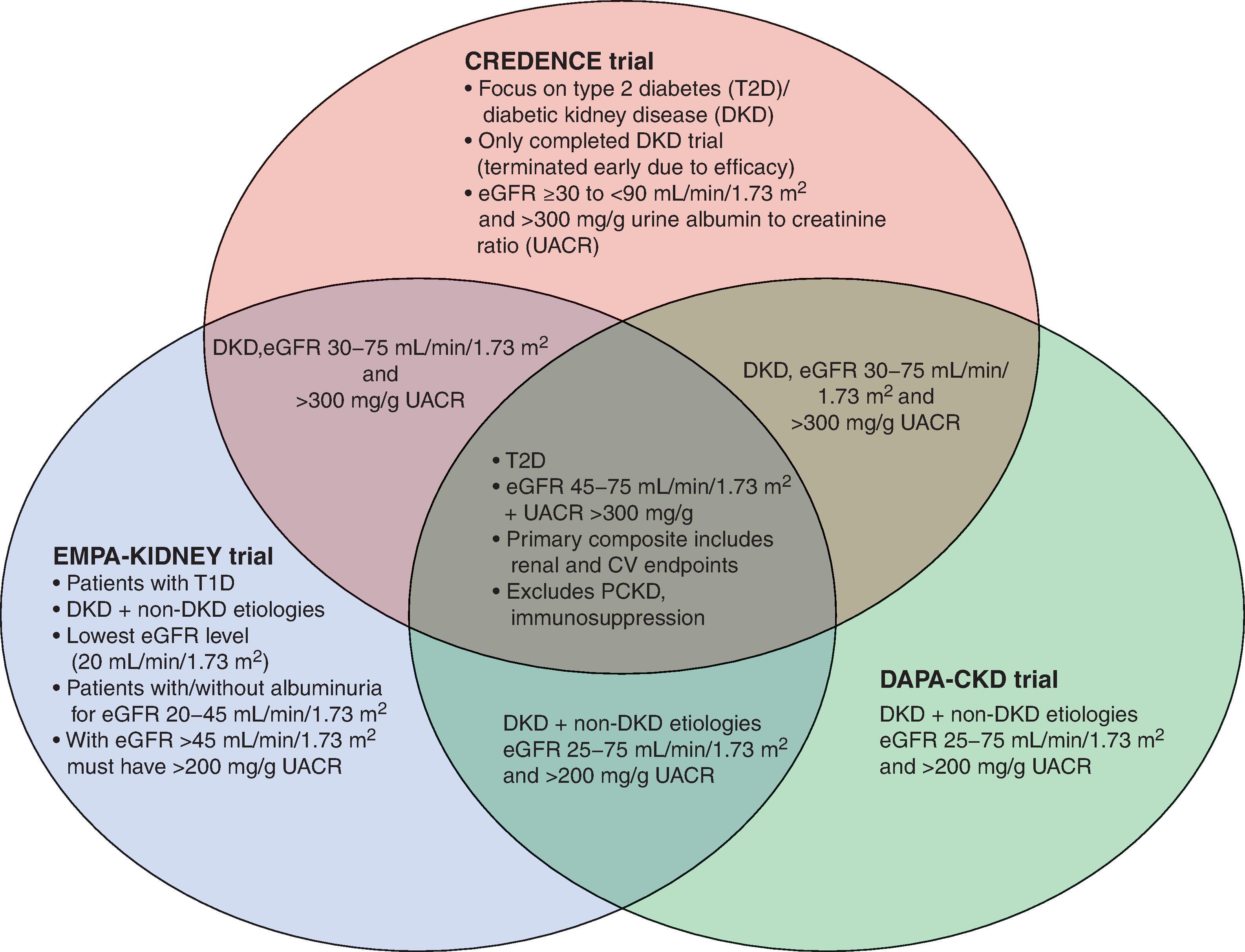
The abovementioned cardiovascular outcome trials demonstrated a kidney-protective role of SGLT-2 inhibition on top of usual therapy, with 80% of participants on renin-angiotensin-aldosterone system (RAAS) blockers. Patients randomized to SGLT-2 inhibitors had a 45%, 40%, 47%, and 35% reduction in the secondary composite renal endpoint (doubling of serum creatinine, 40% eGFR decline, end-stage kidney disease (ESKD), or renal death) in the EMPA-REG OUTCOME, CANVAS Program, DECLARE TIMI-58, and VERTIS CV trials, respectively. In the Canagliflozin and Renal Outcomes in Type 2 Diabetes and Nephropathy (CREDENCE) trial, the first dedicated kidney outcome trial using an SGLT-2 inhibitor, the investigators focused on patients with type 2 diabetes on maximum tolerated RAAS blockade, with eGFR 30–90 mL/min/1.73 m² and albuminuria (urine albumin-to-creatinine ratio 300 to 5000 mg/g). A 30% reduction in the primary outcome of ESKD, doubling of serum creatinine, or kidney or cardiovascular death was seen in the group randomized to canagliflozin 100 mg versus placebo. Meta-analyzed data from EMPA-REG OUTCOME, the CANVAS program, DECLARE TIMI-58, and CREDENCE included over 38,723 participants with 335 kidney failure events and demonstrated a consistent 35% reduction of kidney failure with SGLT-2 inhibition. Renal benefits were observed across all studied GFR subgroups, down to GFR of 30 mL/min/1.73 m 2 and across albuminuria levels.
The Dapagliflozin And Prevention of Adverse outcomes in Chronic Kidney Disease (DAPA-CKD) trial enrolled patients with eGFR between 25 and 75 mL/min/1.73 m 2 and albuminuria (urine albumin-to-creatinine ratio 200 – 5000 mg/g), including patients with and without type 2 diabetes. DAPA-CKD was stopped early in March of 2020 due to overwhelming efficacy in the primary composite outcome of time to 50% eGFR decline, ESRD, and kidney or CV death, with similar results seen in participants with and without diabetes. The Study of Heart and Kidney Protection With Empagliflozin (EMPA-KIDNEY) is enrolling patients with eGFR as low as 20 mL/min/1.73 m 2 , with a range of albuminuria, who can be included depending on their baseline eGFR level ( Fig. 26.6 ). As of late 2021, this trial is ongoing. Based on this body of literature, SGLT-2 inhibitors are now recommended in clinical practice guidelines for secondary prevention of cardiovascular and/or kidney disease in patients with type 2 diabetes and albuminuria, with eGFR>30 mL/min/1.73 m². There are currently limited data around the use of SGLT-2 inhibitors in individuals with type 1 diabetes, and approval varies with jurisdiction and depends on other factors such as BMI, with significant care required to mitigate the known risk of (euglycemic) diabetic ketoacidosis in this population.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here