Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The pancreas forms from two separate buds or anlagen , which later rotate and fuse. The ventral bud, which appears as a part of the developing hepatic duct, forms the posterior and inferior parts of the head and the uncinate process; the larger dorsal bud, which develops from the other side of the foregut and extends into the dorsal mesentery, forms the body, tail, and anterior part of the head. Abnormalities in this process result in annular pancreas and most types of heterotopic pancreas (see later). The pancreas is located in the retroperitoneum behind the stomach and transverse colon, and in front of the aorta and vena cava. It weighs on average between 40 and 180 g and measures 15–20 cm in length. It has no distinct capsule.
The normal adult pancreas has a dual composition. The exocrine component is made of lobular units of acini, which comprise 80% or more of the gland ( Fig. 22.1 ). They empty into ducts of increasingly larger size that finally merge into the main pancreatic duct (of Wirsung) and the accessory pancreatic duct (of Santorini). The duct of Wirsung ends in the papilla of Vater , usually in conjunction with the common bile duct (see also Chapter 21 ). The duct of Santorini ends separately in a minor duodenal papilla. Normally, the Wirsung and Santorini ducts are joined by numerous anastomotic connections; when this is not the case, and the duct of Santorini constitutes the principal drainage for the organ, the condition known as pancreas divisum results (see later).
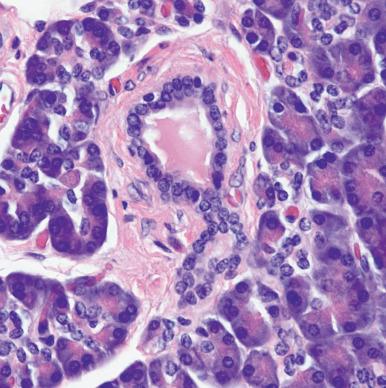
Acinar cells are large, pyramidally shaped, and highly polarized. The luminal border has prominent microvilli, the apical cytoplasm is filled with markedly eosinophilic periodic acid–Schiff (PAS)-positive zymogen granules, and the basilar cytoplasm is intensely basophilic because of the abundance of granular endoplasmic reticulum. Centroacinar cells have paler cytoplasm and oval nuclei; as their name indicates, they are located in the central portion of the acinus. They blend with the cells of the intercalated duct, which drains the acinus. Focal collections of centroacinar cells sometimes occur and should not be confused with Langerhans islets. The merging of intercalated ducts forms the intralobular ducts, which are lined by small cuboidal cells with pale cytoplasm. These are continued by the much larger interlobular ducts, which are lined by mucin-producing columnar cells. The microscopic composition of Wirsung and Santorini ducts is similar to that of the interlobular ducts, except for a greater representation of goblet cells.
The endocrine component of the pancreas is mainly represented by the Langerhans islets ( Fig. 22.2A and B ). These constitute 1%–2% of the adult pancreas but a much larger proportion (~20%) of the organ at the time of birth. Islets may be compact (90%) or diffuse; the latter are located in the portion of the pancreatic head derived from the ventral bud. Islets are not encapsulated and contain a rich capillary network. Their average diameter is 225 µm; individual variations in size exist, but any islet measuring over 400 µm should be regarded as abnormal. In adult humans, islets are composed of the following main cell types:
B cells . The insulin-secreting cells constitute two-thirds to three-fourths of the islet cell population and tend to group in center of the islet. B cells also secrete islet amyloid polypeptide (IAPP), a putative hormone that is thought to be co-released with insulin.
A cells . The glucagon-secreting cells make up one-fifth to one-fourth of the total islet cell population and are mainly located at their periphery.
D cells . The somatostatin-secreting cells are scattered throughout the islets and represent less than 10% of islet cells.
PP cells . The pancreatic polypeptide-secreting cells are scarce and usually present within diffuse islets .
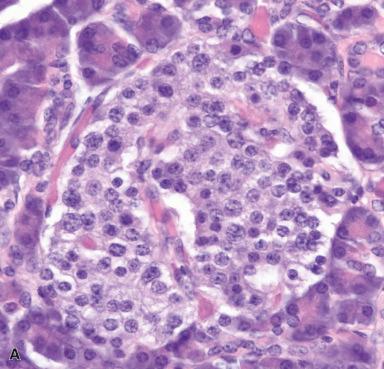
It should be pointed out that all of the endocrine cells present in the pancreas are of endodermal rather than neural crest derivation, as formerly believed. Islet cells mark with synaptophysin, chromogranin, and CD56 and are typically pancytokeratin negative.
In addition to the Langerhans islets, endocrine cells can be found in connection with ducts and acini. Most of these cells are of Kulchitsky (serotonin-producing) and PP type. It should be noted that G (gastrin-producing) cells are not normally present in the adult pancreas, a remarkable fact considering that this organ is the most common site for the occurrence of G cell tumors/gastrinomas.
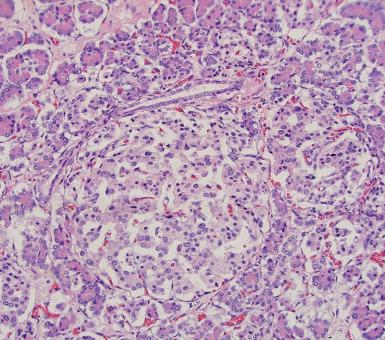
Nesidioblastosis is a clinically heterogeneous term that refers to pancreatic islet hypertrophy in the context of hyperinsulinemic hypoglycemia. It has been observed in infants with persistent neonatal hyperinsulinemic hypoglycemia, as well as infants of diabetic mothers, and has been associated with numerous conditions including Beckwith–Wiedemann syndrome and Zellweger syndrome. Morphologically, islets are in intimate association with ducts, with formation of so-called ductuloinsular complexes ( Fig. 22.3 ); islet cell hypertrophy and beta cells with pleomorphic nuclei may also be seen. Focal and diffuse forms of this condition have been described. The morphologic changes of nesidioblastosis can be seen in normal infants, as the endocrine component in neonates is more prominent than later in life. The morphologic changes have been occasionally reported in adult patients with persistent hyperinsulinemic hypoglycemia and have also been encountered in adults who have had bariatric surgery.
Complete pancreatic agenesis is an exceedingly rare lethal anomaly ; partial agenesis is also rare, and patients often present with diabetes. Annular pancreas is a rare embryologic abnormality in which the ventral primordium of the pancreas fails to rotate properly. It can be seen by itself or in combination with other congenital abnormalities of the gastrointestinal tract. It is more common in females and is associated with Down syndrome. Grossly, there is encirclement of the duodenum by pancreatic parenchyma, which may lead to constriction of the duodenal lumen. The duct in the annular pancreas originates anteriorly and courses to the right over the duodenum and then posteriorly and to the left behind the duodenum, passing near the common duct and finally joining the main pancreatic duct. These anatomic variations have to be kept in mind when surgery is contemplated. Microscopically, the annular pancreas contains a large number of PP cells in its many irregularly shaped islets, in keeping with its origin from the ventral bud (see previous section). Pancreatitis may develop in association with this anomaly. Pancreas divisum , or a congenitally separate pancreatic ductal system, occurs in approximately 10% of individuals. It has been claimed to predispose to the development of pancreatitis, but the evidence for this is less than conclusive.
Heterotopia of pancreatic tissue is a relatively frequent congenital anomaly. It is most common in the duodenum (particularly the second portion), stomach, and jejunum but also occurs in the ileum, Meckel diverticulum, gastric and intestinal diverticula, gallbladder and bile ducts, large bowel, spleen (usually within or immediately beneath the capsule), omentum, abdominal wall, and several other locations.
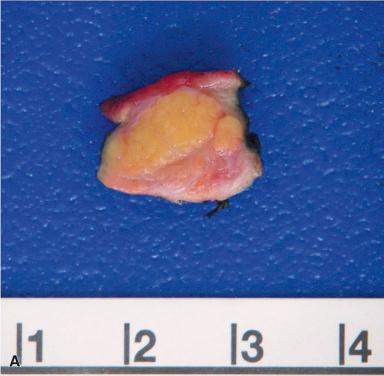
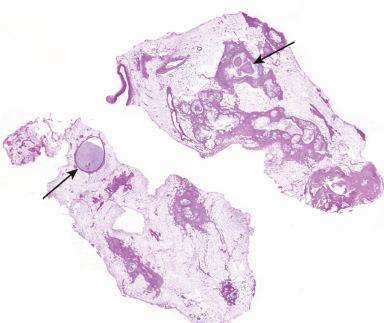
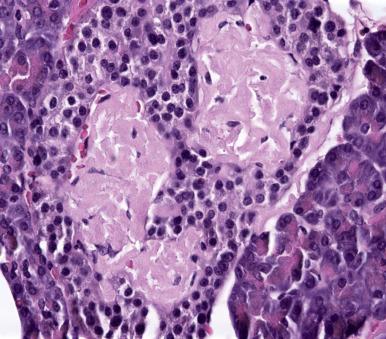
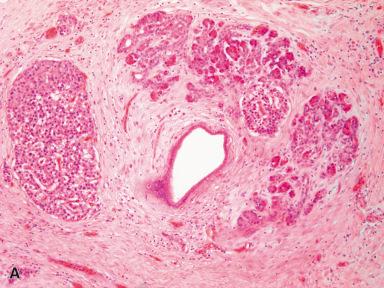
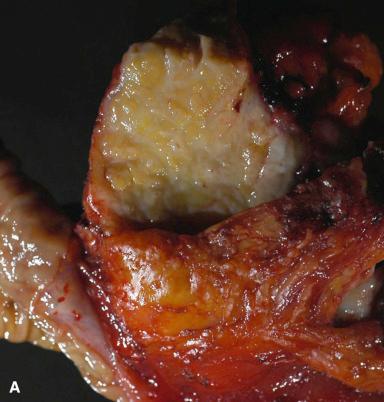
Grossly, the heterotopic tissue resembles normal pancreas. Firm, yellow, lobulated nodules measuring up to 4 cm are seen sharply circumscribed from the surrounding tissues ( Fig. 22.4A ). Central umbilication is often present in the cases located beneath mucosa, corresponding to a central duct that opens into the lumen ( Fig. 22.4B ); both the umbilication and the duct can be demonstrated radiographically and constitute an important diagnostic sign. Microscopically, acinar and ductal tissues are always present, whereas islet tissue is found in only one-third of the cases ( Fig. 22.4C and D ). In some cases, there is also a component of pyloric-type mucous glands. The islet cell component contains all the major types of endocrine cells, but their relative number varies a great deal from case to case. In most instances, the islets are rich in A cells and poor in PP cells (dorsal type), but in others the reverse is true (ventral type). True gastric heterotopic pancreas should be distinguished from pancreatic acinar metaplasia of the gastric or gastroesophageal junction mucosa.
Practically every pathologic change that occurs in the pancreas itself can occur in its heterotopic counterpart, including acute pancreatitis and neoplasms of either exocrine or endocrine type. Heterotopic pancreas is often asymptomatic, but in the stomach may cause hemorrhage, ulceration, or pyloric obstruction. When located in the area of the ampulla of Vater, it may result in obstructive jaundice. It has been postulated that some of the reported cases of intrasplenic mucinous cystadenomas and cystadenocarcinomas have arisen in heterotopic pancreatic tissue.
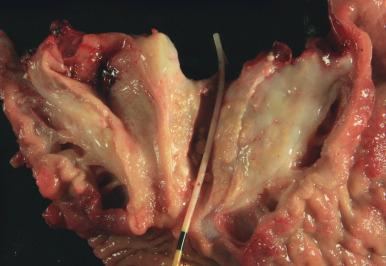
Cystic fibrosis is the most common inherited disorder among Caucasians and is due to genetic alterations in the cystic fibrosis transmembrane conductance regulator (CFTR) protein. The pancreas is the second most frequently affected organ after the lung; in fact, the disease was named for the cysts and fibrosis found in the pancreas in patients with this disease. The precipitation of secretions in the pancreatic ducts leads to obstruction and pancreatitis. Pancreata often show fatty replacement, as well as cyst formation, and the accumulation of inspissated eosinophilic secretions in dilated pancreatic ducts ( Fig. 22.5 ).
Both type 1 and type 2 diabetes may cause histologic changes in the pancreas. The pancreas decreases in size and weight as type 1 diabetes progresses, and islets show variability in size and shape, irregular shapes, reduced B cells, and patchy lymphocytic inflammation. Patients with type 2 diabetes also show reduction in number and density of islets, with reduction in A and B cells as well as islet amyloidosis ( Fig. 22.6 ).
The main risk factors for acute pancreatitis are gallstones and alcohol abuse. Other factors include smoking (particularly when combined with alcohol use), type 2 diabetes, invasive pancreatobiliary procedures, and many drugs. Experimental work has been hampered by the fact that most laboratory animals do not suffer naturally from this illness.
Duct obstruction from any cause results in upstream blockage of pancreatic secretions, with resulting autodigestive injury and inflammation. Opie postulated an obstructive mechanism in a classic work published in 1901, after demonstrating in his famous case of pancreatitis the presence of a small stone lodged in the ampulla, which had converted the common bile duct and the main pancreatic duct (duct of Wirsung) into a “common channel.” The assumption was that the bile had passed from the common bile duct into the pancreatic duct and activated trypsinogen, thus beginning a series of events that included the digestion of the duct wall, adjacent parenchyma, and vessel walls by trypsin, as well as the splitting of fat and formation of calcium soaps by lipase. Fibrosis of the sphincter of Oddi or neoplasms in this region can induce a similar process, and cases of acute pancreatitis secondary to tumors of the ampullary area have been recorded. The difficulty with this theory is that the formation of a common channel with a stone impacted in the ampulla, or another clearly identifiable anatomic cause of obstruction, occurs in less than 5% of patients with acute pancreatitis. This discrepancy could be explained by postulating a transient blockage of the ampulla of Vater by a migrating gallstone, by biliary sludge, or by a spasm of the sphincter of Oddi, all of which could produce a common channel in the absence of an identifiable impacted stone. Acosta and Ledesma recovered gallstones in the feces of 34 of 36 patients with pancreatitis but in only 3 of 36 control subjects (patients with gallstones but no pancreatitis). Biliary sludge was found by Lee et al. in 23 of 31 patients with “id iopathic” acute pancreatitis, and sphincter spasm can be demonstrated in experimental animals and in humans under a variety of conditions. All these experiments demonstrate that a “common channel” may indeed occur under physiologic circumstances, and anomalous or variant pancreaticobiliary ductal anatomy may also contribute.
The relationship between alcohol and acute pancreatitis remains poorly understood, particularly as only a small percentage of alcohol users develop pancreatitis. Alcohol and its metabolites produce changes in acinar cells, which may promote activation of digestive enzymes and subsequent injury, but no specific predisposing or triggering factors have been identified. Regardless of the precise pathogenetic mechanism, it is a fact that the large majority of cases of acute pancreatitis are associated with biliary tract disease, alcohol use, and trauma.
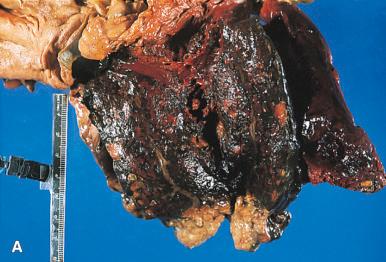
The gross changes in acute pancreatitis vary from a swollen and edematous but otherwise well-preserved organ to a barely recognizable hemorrhagic and necrotic mass of tissue ( Fig. 22.7A ). Yellow plaques and nodules representing fat necrosis are seen within the pancreas as well as throughout the mesenteric and peritoneal fat ( Fig. 22.7B and C ). The process sometimes spreads to the neighboring colon, and it may result in localized ileus, stenosis, perforation, fistulous formations, or ischemic necrosis.
Microscopically, early changes in the pancreas include acinar cell homogenization, ductal dilation with epithelial degeneration, diffuse interstitial edema, leukocytic infiltration, and fibroblastic reaction. If the disease progresses, extensive necrosis and hemorrhage of pancreatic and peripancreatic tissue supervene ( Fig. 22.7D ). The foci of peritoneal fat necrosis become almost immediately surrounded and infiltrated by neutrophils; this population later changes to foamy histiocytes and lymphocytes. Calcification occurs early and extensively in these areas. The pancreatic necrotic foci can undergo secondary infection; this condition, termed infected pancreatic necrosis , is currently regarded as the most common, most severe, and most lethal of the infectious complications of acute pancreatitis. Acute pancreatitis may evolve into a chronic form, and in some clinical circumstances (such as alcoholic pancreatitis) the acute and chronic phases probably represent a continuum.
Patients typically present with abdominal pain and serum amylase and/or lipase levels elevated above three times normal. Of note, however, patients with severe pancreatitis may have only mild enzyme elevations, and elevated serum amylase can also be found with duodenal ulcer, volvulus, gangrenous cholecystitis, ruptured aortic aneurysm, and mesenteric thrombosis. Serum C-reactive protein is a reliable marker for the presence of pancreatic necrosis and may correlate with disease severity.
The overall mortality rate for acute pancreatitis is believed to be approximately 10%–20%, although both higher and lower mortality rates have been reported. Prognosis worsens with increased pancreatic necrosis. A number of classification schemes exist for stratifying severity of disease and prognosis. Early treatment consists of supportive care (particularly fluid resuscitation), pain management, and nutritional support. Endoscopic or surgical intervention may be necessary in patients with acute pancreatitis due to gallstones, and surgical necrosectomy may be required in patients with extensive pancreatic necrosis, particularly those who develop infected pancreatic necrosis, although many patients respond to antibiotics alone.
The term “chronic pancreatitis” implies a chronic fibroinflammatory state that ultimately leads to pancreatic insufficiency. In addition to alcohol, there are a wide variety of causes, including autoimmune, metabolic, hereditary, and anatomic etiologies. Similar to acute pancreatitis, the pathogenesis of chronic pancreatitis is not well understood, although there are a number of proposed theories. Some have proposed that alcohol predisposes patients to the formation of proteinaceous plugs and stones within the pancreatic ducts, leading to epithelial ulceration, scarring, and stricture formation. This theory has been questioned in the past, as only a minority of chronic pancreatitis patients have demonstrable stones or protein plugs within ducts, but has received renewed attention with the discovery of the link between chronic pancreatitis and the CFTR gene (see later). Others have proposed oxidative stress, direct toxic effects of alcohol, and the consequence of repeated bouts of acute pancreatitis as explanations for chronic pancreatitis. It is likely that diverse causes lead to the fibroinflammatory condition known as “chronic pancreatitis” through different pathways. Alcohol continues to be the most important risk factor, and smoking is also an important risk factor. Another contributing factor is prolonged ductal obstruction by tumor or stone (also known as chronic obstructive pancreatitis).
There is increasing evidence suggesting that a significant number of patients with idiopathic acute and chronic pancreatitis have underlying genetic alterations. Several of the identified mutations play a role in trypsinogen activation, pancreatic secretory function, or response to inflammation during pancreatic injury; affected genes include PRSS1, SPINK1, CTRC , and CFTR , as well as several others. However, although these genes have been linked to pancreatitis, penetrance is incomplete and the presence of mutation does not necessarily confer disease.
Clinically, chronic pancreatitis characteristically presents with abdominal pain, which can be very severe and often radiates to the mid-back or scapula. Later in the course of disease, patients may present with diabetes or malabsorption due to deficiency of both exocrine and endocrine function.
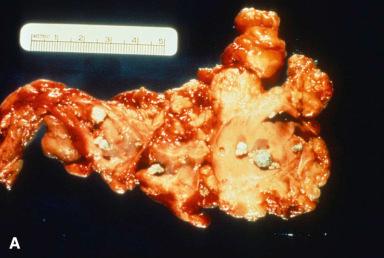
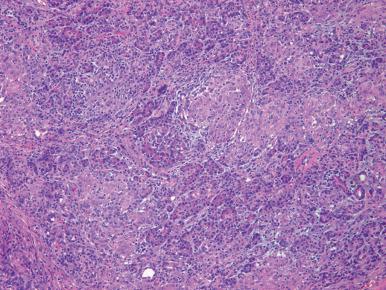
Approximately 80% of chronic pancreatitis cases are due to alcohol. The disease is characterized grossly by a nodular, firm, fibrotic, and misshapen pancreas that may be either enlarged or atrophic. The fibrosis typically affects the gland unevenly, leaving preserved architecture in some areas ( Fig. 22.8A–C ). Ducts embedded within the fibrosis may be irregular, stenotic, and/or dilated, and may contain calculi. Microscopically, the main features are fibrosis, initially interlobular, with chronic inflammation ( Fig. 22.9A and B ). As disease progresses, fibrosis increases, with loss of parenchyma, dilatation and distortion of ducts, and intraluminal eosinophilic mucoprotein plugs (which often calcify). When the calcification is very extensive, the term chronic calcifying pancreatitis has been used. The ductal epithelium may be hyperplastic or contain squamous metaplasia, and eventually the epithelium may be atrophic or completely denuded. Residual nerves and vessels may be prominent. The appearance of Langerhans islets in chronic pancreatitis varies considerably from case to case. Often, there is atrophy and distortion by the surrounding fibrotic tissue, with a relative decrease in the number of B cells. Conversely, other cases show a marked proliferation of islet cells, which may grow in cords and small clusters ( Fig. 22.9C ). Sometimes they may even simulate a malignant neoplasm ( Fig. 22.9D ). Concomitant hyperplasia of Brunner glands in the duodenum is a common finding in chronic pancreatitis.
A good correlation exists between the severity of the histologic alterations and pancreatic exocrine function tests. Acinar necrosis and acute inflammation are seen more often in patients with persistent pain than in those who are free of pain. However, cases exist in which severe abdominal pain of pancreatic origin is accompanied by minimal morphologic changes.
Complications of chronic pancreatitis include both exocrine and endocrine insufficiency, stenosis of the common bile duct, pseudocysts, and pseudoaneurysm formation, which can lead to exsanguinating acute hemorrhage. A rare complication of both acute and chronic pancreatitis is the occurrence of widespread metastatic fat necrosis, presumably as a consequence of the liberation of lipase by the damaged organ. Subcutaneous tissue (particularly in the legs), mediastinum, pleura, pericardium, bone marrow, periarticular fat, and liver can be involved. Erythema nodosum–like lesions of the skin, polyarthropathy, and avascular bone necrosis are some of the alterations that may develop as a result.
Endoscopy is playing a larger role in the treatment of chronic pancreatitis, including ERCP for treatment of strictures and stone removal, and endoscopic ultrasound for the treatment of pancreatic fluid collections. Surgical therapy may involve pancreatic duct drainage, partial pancreatic resection, and near-total pancreatectomy with or without islet autotransplantation.
A great deal has been written recently on this disease, which is thought to have an autoimmune or hypersensitivity etiopathogenesis and shows good response to steroid therapy. It is subclassified into two types, which appear to be two distinctly different diseases. Occasional cases do not fit neatly into either of these two types but show intermediate histologic features.
Type 1 autoimmune pancreatitis is thought to be the pancreatic manifestation of a systemic fibroinflammatory disease that may involve a multitude of organs including the liver, gallbladder, biliary tree, lung, salivary gland, lacrimal duct, lymph node, kidney, and prostate. A unifying theme is increased IgG4+ plasma cells, typically more than 20/HPF, although this is not an entirely sensitive or specific finding in either tissue samples or serum measurement of IgG4. It occurs predominantly in middle-aged to elderly patients, with male predominance. Grossly, the pancreas is extremely firm, and the inflammatory process may form a mass lesion (most often in the pancreatic head) simulating pancreatic cancer clinically and intraoperatively ( Fig. 22.10A ). Microscopically, it is characterized by a dense lymphoplasmacytic infiltrate and fibrosis, hence the alternative term lymphoplasmacytic sclerosing pancreatitis ( Fig. 22.10B–D ) . The fibrosis may be storiform, with plasma cells infiltrating between the stromal fibers. The fibroinflammatory process may center around the ducts, the lobules, or both, leading some workers to subdivide the disease into ductocentric, lobulocentric, and mixed subypes. Non-necrotizing inflammation of veins and their surroundings (“venulitis” and “perivenulitis”) are often present. The inflammatory infiltrate can be patchy, rendering a biopsy diagnosis very difficult. When the fibroblastic component is very prominent, the condition can be confused with inflammatory myofibroblastic tumor. Increased numbers of IgG4-producing plasma cells may be found in ampullary/periampullary biopsies, as well as in pancreatic specimens, and may be of some diagnostic assistance.
Type 2 disease is also known as idiopathic duct-centric chronic pancreatitis or autoimmune pancreatitis with granulocytic epithelial lesions , and it is not associated with either elevated serum IgG4 or increased IgG4+ plasma cells, and thus some argue that it should not be called “autoimmune pancreatitis” at all. Approximately one-third of cases are associated with chronic idiopathic inflammatory bowel disease, but patients do not manifest a multiorgan fibroinflammatory process like that seen in type 1 disease. Patients are typically younger than in type 1 disease, and there is no male predominance. A lymphoplasmacytic infiltrate is common, but diffuse inflammation, obliterative phlebitis, and storiform fibrosis are less common than in type 1 disease. The most prominent feature is neutrophilic infiltration of the ductal epithelium (“granulocytic epithelial lesions”) ( Fig. 22.10E ), which may lead to duct destruction. As mentioned previously, there are only rare or absent IgG4-positive plasma cells.
This condition has also been described under a variety of names, including cystic dystrophy of heterotopic pancreas, pancreatic hamartoma of duodenum, paraduodenal wall cyst, or groove pancreatitis, the latter because it may involve the groove between the common bile duct, pancreas, and duodenum. The heterogeneity of names reflects various aspects of this disease. This form of chronic pancreatitis typically occurs in male alcoholics, and it is clinically significant because the dense fibrosis and irregular borders mimic neoplasia. Patients may present with signs and symptoms of duodenal obstruction.
The fibroinflammatory process is typically centered on the duodenal wall, which is thickened, fibrotic, and trabecular, in the region of the minor papilla ( Fig. 22.11 ). Sometimes a scar plate develops between the head of the pancreas and the duodenum. There are often numerous cysts, which may contain stones, fluid, or a grumous material, and which may be large enough to mimic intestinal duplication and/or compress the common bile duct. Microscopically, there is myoid proliferation of the duodenal wall in the area of the minor ampulla, with associated cystically dilated ducts and sometimes acinar tissue and islets (although pancreatic elements may be difficult to locate). The ducts contain inspissated secretions, which may rupture and induce a foreign body giant cell reaction. Neural hyperplasia and chronic inflammation may also be prominent. Although alcohol is clearly a risk factor, it is hypothesized that some anatomic variation in ductal anatomy, such as obstruction of an accessory pancreatic duct, makes this region susceptible to injury.
True idiopathic eosinophilic pancreatitis is extremely rare; numerous eosinophils in the pancreas are more likely to be secondary to autoimmune pancreatitis, allograft rejection, or histiocytosis X. Tropical pancreatitis , also known as tropical calculous pancreatitis or fibrocalculous pancreatic diabetes if patients are diabetic, is limited to Brazil and other countries close to the equator in central Africa and southern Asia. Although originally described in adolescents who were malnourished in childhood, more recent studies have emphasized that this disease can also occur in older patients, and a number of genetic abnormalities have also been described. Morphologic features are similar to alcoholic chronic pancreatitis. Obstructive chronic pancreatitis is the result of narrowing or occlusion of the pancreatic ducts, the most common causes being carcinoma and stones. Most of the stones are found in the duct of Wirsung within 2–4 cm of the ampulla of Vater and often restricted to a small portion of the gland. Hereditary pancreatitis is a rare autosomal dominant hereditary fibroinflammatory and necrotizing disease that begins in childhood or adolescence. It is the cause of only 1%–2% of patients with chronic pancreatitis. Clinically and pathologically it simulates chronic alcoholic pancreatitis, although some patients (particularly adults) show marked fatty replacement of the pancreas. The genetic alterations involve mutations in the PRSS1 (most common), SPINK1 , or CFTR genes.
Pancreatic abscesses are most often seen as complications of acute pancreatitis. They are bona fide infections, characterized by the presence of pus and the microbiologic identification of bacteria in over 90% of the cases. They have different presentation, clinical course, and outcome than infected pseudocysts and necrosis, from which they should be distinguished.
Although relatively rare, a number of viruses, bacteria, fungi, and parasites can cause pancreatitis, including mumps, adenovirus, CMV, Candida species, tuberculosis (which may mimic pancreatic adenocarcinoma radiographically), Clonorchis , Ascaris, and toxoplasmosis. Many of these cases, particularly adenovirus, CMV, and toxoplasmosis cases, are associated with immunocompromise.
Other inflammatory conditions include sarcoidosis , an exceptionally rare condition that is a manifestation of systemic disease ( Fig. 22.12 ) ; malakoplakia ; vasculitides, including Henoch–Schönlein purpura and polyarteritis nodosa ; and rheumatoid nodules, which have been described in the pancreas in an adult patient with longstanding rheumatoid arthritis.
In the past decade pancreatic transplants in the United States have declined by approximately 20%–30%, although the numbers continue to increase in other countries. The two main indications for pancreatic transplantation are chronic pancreatitis and insulin-resistant diabetes mellitus. At present, pancreatic graft survival rates are between 78% and 86%, depending on the type of transplant. The most common complication of pancreatic transplantation is graft thrombosis, which is believed to be related to multiple factors including vascular injury during procurement, preservation/perfusion variables, hypercoagulability, and decreased fibrinolysis found in diabetic recipients. The second most common complication is “graft pancreatitis,” which often occurs early in the post-transplant period and is also associated with reperfusion injury, donor factors, and cold ischemia time; early graft pancreatitis is associated with a high rate of graft loss. The two other important complications, which tend to occur at a later stage, are recurrence of the original disease and rejection. Biopsy evaluation of the pancreas allograft is crucial in establishing the cause of allograft dysfunction. Microscopic changes specific for rejection are vascular alterations such as “endothelialitis,” vasculitis, and obliterative endarteritis. Another common abnormality is a periductal lymphocytic or mixed infiltrate. Morphologic features found to correlate strongly with a negative outcome include moderate to severe inflammation of acinar tissue, acinar tissue loss and fibrosis, and vascular luminal narrowing. Vasculitis is a more aggressive and clinically more significant lesion than endothelialitis and defines the existence of severe rejection.
Transplanted pancreata show a significant increase in glucagon-producing islet cells and a corresponding decrease in insulin-producing cells, particularly in those that have rejection.
Recurrent diabetes in the graft occurs in approximately 6%–8% of patients and is characterized by selective loss of insulin-secreting cells and the presence of “isleitis”; the presence of circulating autoantibodies; and the presence of autoreactive T cells in the blood, graft, or peripancreatic lymph nodes.
Pseudocysts are the most common type of pancreatic cyst, comprising approximately 75% of pancreatic cystic lesions, and are related to acute or chronic pancreatitis, trauma, and, rarely, to neoplastic obstruction of large ducts. They can become very large and spread beyond the substance of the pancreas into the lesser peritoneal cavity, retroperitoneum, perirenal space, or subdiaphragmatic space. They range from 3 to 20 cm in size and are multiple in 10%–20% cases. Grossly, the wall is thick and irregular ( Fig. 22.13 ). The inner surface is ragged, and the intraluminal content is cloudy or bloody. Microscopically, the lack of epithelial lining is the main distinguishing feature between true cysts and cystic neoplasms, and any evidence of epithelium should prompt additional sections as cystic neoplasms with sloughed epithelial linings may mimic pseudocysts. Fibrin, inflammation, hemorrhage, and fibrosis are often prominent. The intraluminal fluid has a high amylase content. Communication with the ductal system may or may not be present.
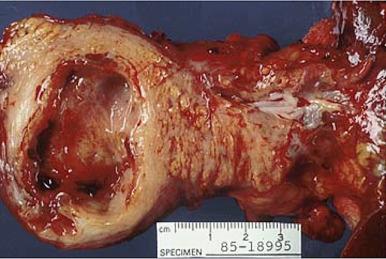
Complications of pancreatic pseudocyst include perforation, infection, and hemorrhage. The splenic artery is the most common source of the latter, which can be massive and result in sudden death.
Traditionally, internal drainage has been done by open or laparoscopic surgery in the form of transgastric cystogastrostomy or cystojejunostomy to a Roux-en-Y loop of jejunum, but endoscopic (transpapillary or transmural) drainage is becoming the preferred approach as the rate of pseudocyst resolution is comparable. External drainage is preferred for infected or ruptured pseudocysts. Pseudocysts in the tail of the pancreas (particularly if there is splenic vein involvement) and lesions that are anatomically not amenable to drainage should be surgically resected.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here