Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Previously known as congenital cystic adenomatoid malformation (CCAM)
A rare lesion caused by hamartomatous proliferation of the terminal bronchioles at the expense of alveolar development ▸ it is characterized by a multicystic mass of pulmonary tissue with proliferation of the bronchiolar structures
It is usually unilobar and communicates with a normal tracheobronchial tree ▸ it receives its blood supply from a normal pulmonary artery and vein ▸ it may compress the contralateral lung (resulting in hypoplasia)
Type 1 – macrocystic (65%): variable cysts with at least one dominant cyst present (up to 10 cm) ▸ this has a good prognosis (and an infrequent association with other congenital abnormalities)
Type 2 – microcystic (10–15%) : smaller more uniform cysts (up to 2 cm) ▸ congenital malformations are common (50%)
Type 3 – mixed (~8%) : solid-appearing microcysts (1.5 cm) with an associated mass effect (giving a ground-glass appearance on CT) ▸ it has a poor prognosis due to the associated congenital malformations and severe respiratory compromise ▸ nearly always mediastinal shift
Type 4 (10–15%): large cysts (up to 7 cm) ▸ may be a precursor of pleuropulmonary blastoma
Neonatal respiratory distress or it can present in older children ▸ symptoms are due to a combination of obstructive emphysema, mediastinal shift and infection
During the first few hours of life a CCAM will appear as a soft tissue mass (due to the retained lung fluid within it) which may cause mediastinal shift ▸ once this fluid has been reabsorbed it will be replaced by an air-filled cystic lesion
This can distinguish between a CCAM and a congenital diaphragmatic hernia
Echogenic soft tissue mass ▸ multiple variable size anechoic cysts or a homogeneous echogenic solid mass ▸ large lesions can cause mediastinal shift (± lung hypoplasia)
T2WI: hyperintense ▸ uni-/multilocular or solid ▸ over time the fluid can be replaced by air (± air–fluid levels)
CPAMs can communicate with the airways ▸ infected (30%) ▸ blood supply from the pulmonary artery with drainage via pulmonary veins
There is a variable natural history and prognosis (larger lesions have a worse prognosis)
Lesions that are identified prenatally may involute in utero
Treatment: early surgical removal may be required if there is severe pulmonary compromise (and if the contralateral lung is not hypoplastic)
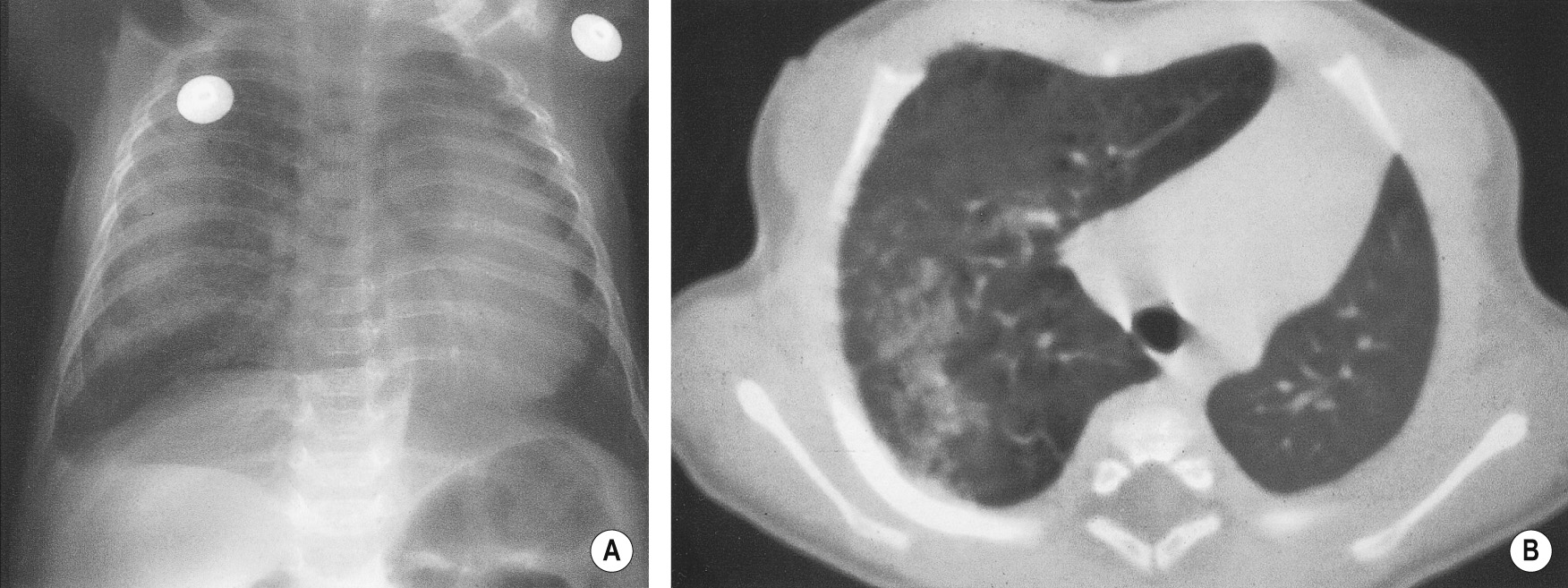
This anomaly shares some features with sequestration except:
The lung is normally connected to the bronchial tree
The vein draining the affected lobe (usually the right lower lobe) drains into the IVC or portal vein (rather than the left atrium)
There is usually an absent or small pulmonary artery perfusing the abnormal lung
The arterial supply is partly or wholly from the thoracic or abdominal aorta, or the coeliac axis
It may be asymptomatic or present with features of a left-to-right shunt
A small ipsilateral lung with ipsilateral mediastinal shift
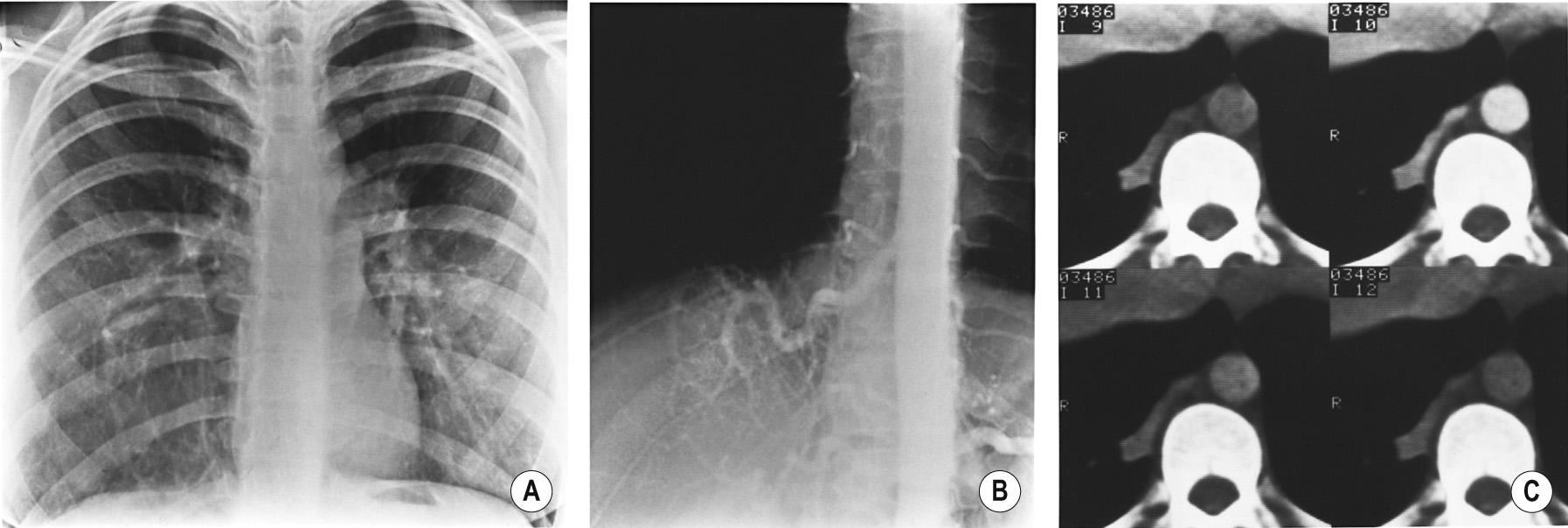
The abnormal pulmonary vein is seen draining down and enlarging towards the diaphragm in the shape of a ‘scimitar’ sword
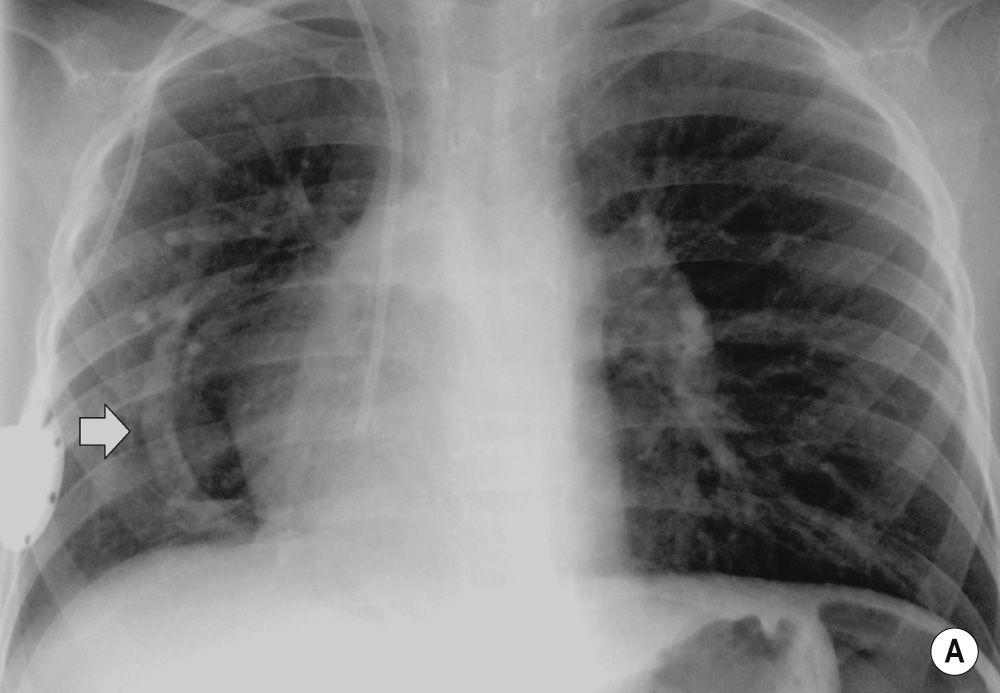
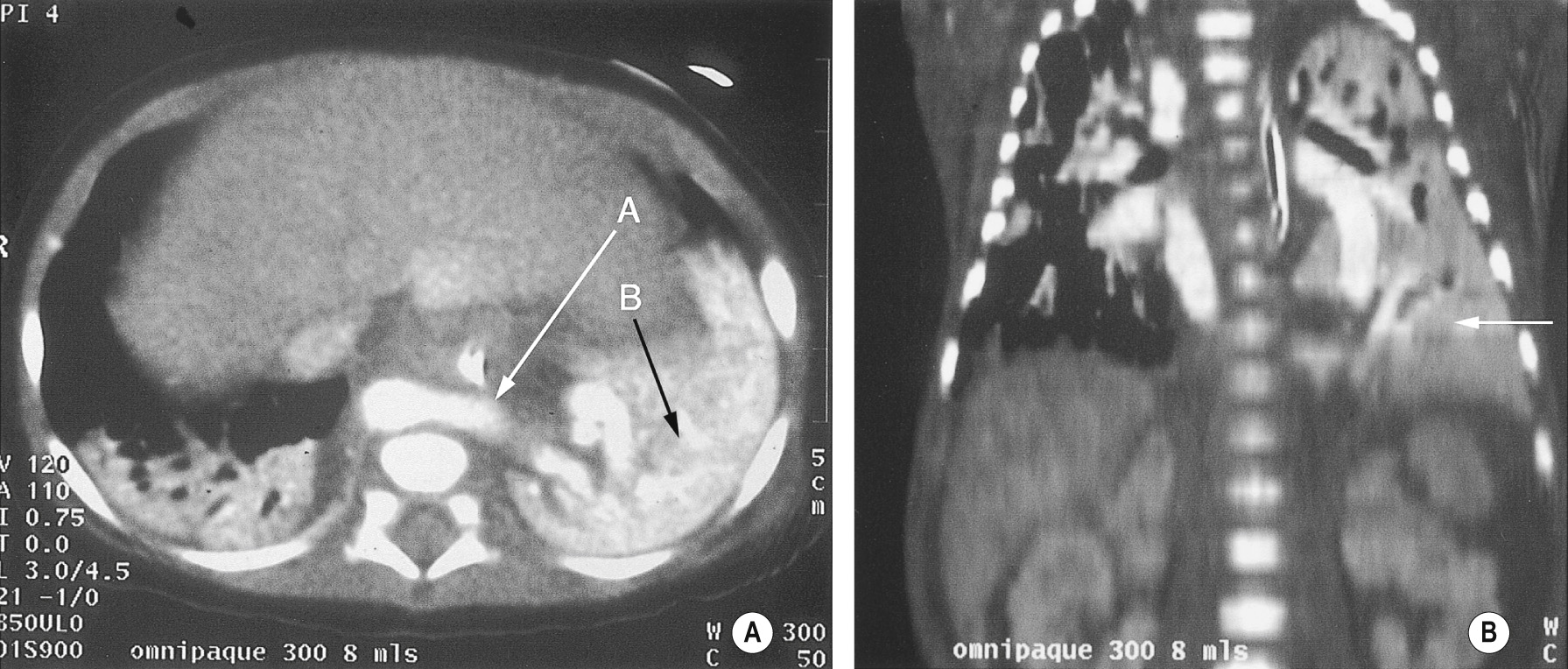
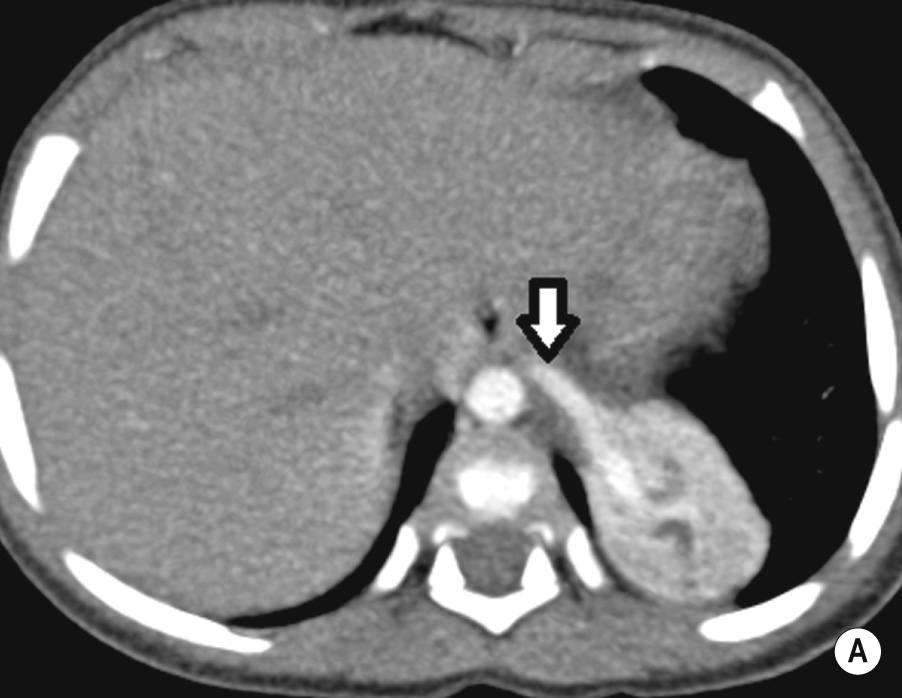
A congenital mass of aberrant pulmonary tissue that has no normal connection with the bronchial tree or pulmonary arteries ▸ lesions are defined as either intra- or extralobar
It derives its arterial supply from either the thoracic or abdominal aorta ▸ its venous drainage can either be via the pulmonary or systemic veins
It can present at birth or in an older child (depending on the type) with recurrent focal infections, bronchiectasis, haemoptysis or as an asymptomatic pulmonary mass
A solid well-defined highly echogenic mass ▸ the anomalous systemic arterial supply is difficult to visualize despite the availability of colour flow Doppler
A persistent multicystic basal opacity which is frequently left sided
This enables definition of the arterial and venous vascular anatomy
Solid, well-defined hyperintense T2WI mass
Extralobar sequestration can be located below the diaphragm and mimic a neuroblastoma or adrenal haemorrhage
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here