Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Ovarian cancer is the fifth most common cancer among women in the United States, accounting for one-fourth of all gynecologic cancers. It is the leading cause of death resulting from gynecologic cancer because it is difficult to detect before it disseminates.
Most ovarian neoplasms (80-85%) are derived from coelomic epithelium and are called epithelial carcinomas. The most common type of ovarian and fallopian tube cancer is serous adenocarcinoma. On the basis of relatively recent molecular, genetic, and pathologic data, many high-grade serous carcinomas formally designated as “ovarian cancers” have been shown to arise in the fimbrial end of the fallopian tubes.
Approximately 10% of epithelial ovarian cancers occur in women with a known hereditary predisposition.
The diagnosis of ovarian cancer requires a laparotomy or laparoscopy. In patients with no gross evidence of disease beyond the ovary and fallopian tube, the standard operation is total abdominal hysterectomy, bilateral salpingo-oophorectomy, infracolic omentectomy, and thorough surgical staging. In patients with advanced disease, cytoreductive surgery (“debulking”) is required. The objectives are to remove the primary tumor and all of the metastases, if possible.
Germ cell tumors of the ovary account for only about 2-3% of all ovarian malignancies, and they occur predominantly in young patients. They frequently produce either human chorionic gonadotropin or α-fetoprotein, which serve as tumor markers. The most common germ cell tumors are the dysgerminoma and immature teratoma.
Ovarian cancer is the fifth most common cancer among women in the United States, accounting for one-fourth of all gynecologic cancers. It is the leading cause of death from gynecologic cancer because it is difficult to detect before it disseminates. In 2013, 22,240 new cases and more than 14,030 deaths were expected from this disease. Most women with ovarian cancer are in their fifth or sixth decade of life.
Epithelial ovarian cancers have been thought to arise from a single layer of cells that covers the ovary or lines cysts immediately beneath the ovarian surface. There is now molecular, genetic, and pathologic evidence that as many as 60-80% of high-grade serous cancers actually arise in the fimbrial ends of the fallopian tubes. Peritoneal cancers arise in the tissues lining the peritoneal cavity, and most are histologically identical to serous carcinomas arising in the ovaries or fallopian tubes. Because the treatment of these malignancies is identical, they are now grouped together in the same International Federation of Gynecology and Obstetrics (FIGO) staging classification.
The cause of ovarian cancer is unknown. The patient characteristics found to be associated with an increased risk for epithelial ovarian cancer include white race; late age at menopause; family history of cancer of the ovary, breast, or bowel; and prolonged intervals of ovulation uninterrupted by pregnancy. There is an increased prevalence of ovarian cancer in nulliparous women and those who have been infertile.
The incidence of ovarian cancer varies in different geographic locations. Western countries, including the United States, have rates that are three to seven times higher than those in Japan. Second-generation Japanese immigrants to the United States have an incidence of ovarian cancer similar to that of American women. White Americans experience ovarian cancer about 1.5 times more frequently than do black Americans.
Approximately 10% of epithelial ovarian cancers occur in women with a hereditary predisposition. Although women with hereditary cancers may have two or more first-degree relatives on either the paternal or maternal side who have had breast or ovarian cancer, recent studies have shown that 30-40% of patients with a BRCA germline mutation have no family history of disease. It is recommended that all patients with a high-grade serous or endometrioid ovarian cancer be referred for genetic counseling and possible genetic testing. The pattern of inheritance is autosomal dominant. Breast cancers generally occur in young premenopausal women, whereas ovarian cancers occur at a median age of approximately 50 years.
The breast-ovarian cancer syndrome is caused by germline mutations in BRCA1, which is located on chromosome 17, or BRCA2, which is located on chromosome 13. Lynch II syndrome, a nonpolyposis colorectal cancer syndrome, is associated with mutations in the mismatch repair genes. Adenocarcinomas of the ovary, breast, colon, stomach, pancreas, and endometrium are seen in the families of these individuals.
The use of oral contraceptives has been found to protect against ovarian cancer, possibly because of suppression of ovulation. It has been postulated that incessant ovulation may predispose to malignant transformation in the ovary.
Patients with a known germline mutation (e.g., BRCA1 or BRCA2 mutation) may be offered prophylactic salpingo-oophorectomy once childbearing has been completed, and this operation is highly protective for ovarian and fallopian tube carcinomas. The risk of subsequent breast cancer is also significantly reduced in these women. There is still a small risk of peritoneal carcinoma after prophylactic salpingo-oophorectomy.
The results of some case-control studies suggest that the use of postmenopausal estrogen replacement therapy may increase the risk of ovarian cancer, but these data are controversial.
It has also been postulated that a causative agent could enter the peritoneal cavity through the lower genital tract. For example, the perineal use of asbestos-contaminated talc has been linked to the development of epithelial ovarian cancer. This possibility remains controversial, although tubal ligation and hysterectomy are both associated with a decreased risk of the disease.
Population screening for ovarian cancer is not feasible, because ultrasonography and available tumor markers, such as cancer antigen (CA)-125, lack specificity and sensitivity for early-stage disease. CA-125 is more useful in postmenopausal women because false-positive measurements occur commonly in premenopausal women in association with endometriosis, pelvic inflammatory disease, or uterine fibroids. Patients with a strong family history of epithelial ovarian cancer may benefit from surveillance with serial transvaginal ultrasonography and serum CA-125 level measurements.
Unfortunately, many patients in whom ovarian cancer develops have only nonspecific symptoms before dissemination takes place. In early-stage disease, vague abdominal pain or bloating is common, although symptoms of a mass compressing the bladder or rectum, such as urinary frequency or constipation, may bring the patient to a physician. Sometimes the patient complains of dyspareunia. Premenopausal women may experience menstrual irregularity. Only rarely does a patient present with acute symptoms, such as pain secondary to torsion, rupture, or intracystic hemorrhage.
In advanced-stage disease, patients most often present with abdominal pain or swelling. The latter is usually caused by ascites. Careful questioning usually reveals that there has been a history of vague abdominal symptoms, such as bloating, constipation, nausea, dyspepsia, anorexia, lethargy, or early satiety. Premenopausal patients may complain of irregular menses or heavy vaginal bleeding.
Clinically, patients with fallopian tube cancer can present with a vaginal discharge that is typically watery in nature, as well as with vaginal bleeding, pelvic pain, or some combination of symptoms. In postmenopausal patients, the vaginal discharge may be yellow, watery, and similar to that seen with a urinary fistula. A fallopian tube cancer should be suspected in a postmenopausal patient whose bleeding or abnormal cytologic findings are not explained by endometrial or endocervical curettage. In most patients, the diagnosis is not made preoperatively.
Peritoneal carcinomas usually present with abdominal swelling caused by ascites.
These malignancies are frequently misdiagnosed for several months because of the nonspecific nature of the symptoms. With ovarian cancer, a vaginal or rectal examination will usually reveal a solid, irregular, fixed pelvic mass; if combined with an upper abdominal mass, ascites, or both, the diagnosis is almost certain.
The diagnosis of ovarian, fallopian tube, and peritoneal cancer requires a laparotomy or laparoscopy. Routine preoperative hematologic and biochemical studies should be obtained, as should a chest radiograph. A pelvic and abdominal computed tomographic (CT) scan will exclude liver metastases and may demonstrate retroperitoneal lymphadenopathy.
Endometrial biopsy and endocervical curettage are necessary in patients with abnormal vaginal bleeding, because concurrent primary tumors occasionally occur in the ovary and endometrium. In the presence of ascites, abdominal paracentesis for cytologic evaluation should be performed to confirm the diagnosis of malignancy if neoadjuvant chemotherapy is planned. In patients with occult blood in the stool or significant intestinal symptoms, a barium enema or lower gastrointestinal endoscopy should be obtained to rule out a primary colonic cancer with ovarian metastasis. Similarly, an upper gastrointestinal endoscopy is important if significant gastric symptoms are present. Breast cancer may also metastasize to the ovaries, so bilateral mammograms should be obtained if there are any suspicious breast masses.
Pelvic ultrasonography, particularly transvaginal ultrasonography with or without color Doppler studies, may be useful for small (<8 cm) masses in premenopausal women. Masses that are predominantly solid or multilocular have a high probability of being neoplastic, whereas unilocular cystic masses are generally functional cysts. In postmenopausal women, ultrasonography may also be useful because small, unilocular cysts (<5 cm) that are stable are generally benign.
Several tumor markers have been investigated, but none has been consistently reliable. The tumor-associated antigen CA-125 is elevated in only about 50% of women with stage I ovarian cancer. When this assay is elevated, it is useful for monitoring the clinical course of the disease.
Ovarian and fallopian tube malignancies must be differentiated from benign neoplasms and functional cysts of the ovaries and fallopian tubes. In addition, a variety of gynecologic conditions can simulate a neoplastic process, including tubo-ovarian abscess, endometriosis, and a pedunculated uterine leiomyoma. Nongynecologic causes of pelvic tumor must also be excluded, such as an inflammatory or neoplastic disease of the colon, or a pelvic kidney.
Ovarian and fallopian tube cancers typically spread by exfoliating cells that disseminate and implant throughout the peritoneal cavity. The distribution of intraperitoneal metastases tends to follow the circulatory path of peritoneal fluid, so metastases are commonly seen on the posterior cul-de-sac, paracolic gutters, right hemidiaphragm, liver capsule, and omentum. Implants are also common on the bowel serosa and its mesenteries ( Figure 39-1 ). In general, they grow around the intestines, encasing them with tumor without invading the bowel lumen. Widespread bowel metastases can lead to a functional obstruction known as carcinomatous ileus.
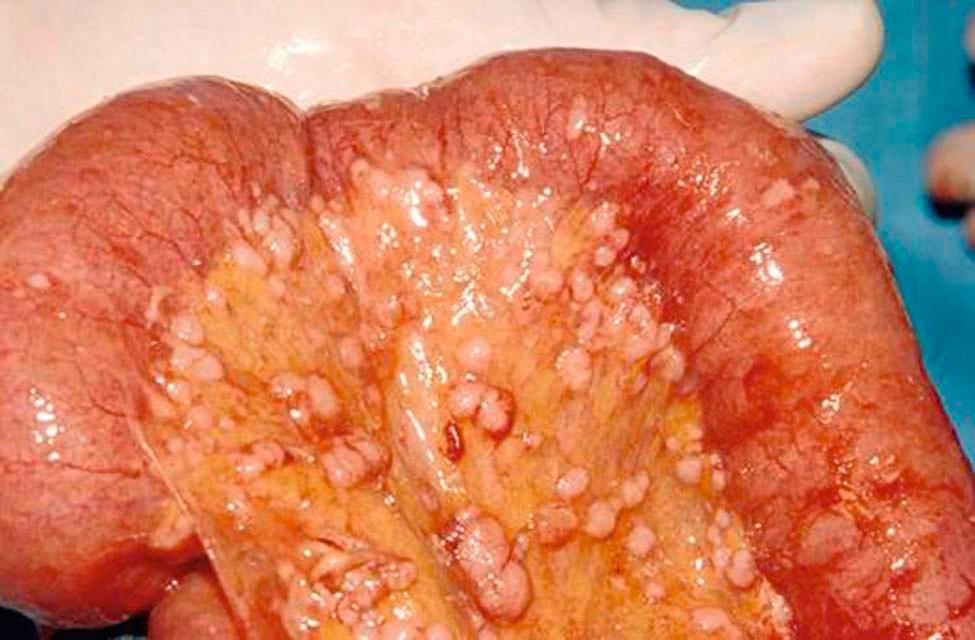
Lymphatic dissemination to the pelvic and paraaortic nodes is common, particularly in patients with advanced disease. Extensive blockage of the diaphragmatic lymphatics is at least partially responsible for the development of ascites. Hematogenous metastases are not common, and parenchymal metastases to the liver and lungs are seen in only about 2% of patients at initial presentation.
Death caused by ovarian cancer usually results from progressive encasement of abdominal organs, leading to anorexia, vomiting, and inanition. The bowel obstruction caused by tumor growth is often incomplete and intermittent and may last for several months before the patient's death.
The 2013 FIGO staging system for ovarian, fallopian tube, and peritoneal cancer is presented in Table 39-1 . Even though all macroscopic (visible) disease may appear to be confined to the ovaries and/or fallopian tubes at the time of laparotomy, microscopic spread may have already occurred; thus, patients must undergo a thorough “surgical staging.” Procedures necessary to stage these cancers are shown in Box 39-1 .
| International Federation of Gynecology and Obstetrics (FIGO) | Tumor, Node, Metastasis (TNM) | |
|---|---|---|
| Ov | Primary tumor, ovary | Tov |
| FT | Primary tumor, fallopian tube | Tft |
| P | Primary tumor, peritoneum | Tp |
| X | Primary tumor cannot be assessed | Tx |
| Designate histologic type: High-Grade Serous (HGS), Endometrioid (E), Clear Cell (CC), Mucinous (M), Low-Grade Serous (LG), Other or cannot be classified (O), Germ Cell (GC), Sex-Cord Stromal Cell Tumor (SC) |
||
| Stage I | Tumor confined to ovaries or fallopian tube(s) | T1 |
| IA | Tumor limited to one ovary (capsule intact) or fallopian tube No tumor on ovarian or fallopian tube surface No malignant cells in the ascites or peritoneal washings |
T1a |
| IB | Tumor limited to both ovaries (capsules intact) or fallopian tubes No tumor on ovarian or fallopian tube surface No malignant cells in the ascites or peritoneal washings |
T1b |
| IC | Tumor limited to one or both ovaries or fallopian tubes, with any of the following: | T1c |
| IC1 | Surgical spill intraoperatively | |
| IC2 | Capsule ruptured before surgery or tumor on ovarian or fallopian tube surface | |
| IC3 | Malignant cells in the ascites or peritoneal washings | |
| Stage II | Tumor involves one or both ovaries or fallopian tubes with pelvic extension (below pelvic brim) or peritoneal cancer (Tp) |
T2 |
| IIA | Extension and/or implants on the uterus and/or fallopian tubes/and/or ovaries | T2a |
| IIB | Extension to other pelvic intraperitoneal tissues | T2b |
| Stage III | Tumor involves one or both ovaries, fallopian tubes, or peritoneal cancer, with cytologically or histologically confirmed spread to the peritoneum outside the pelvis and/or metastasis to the retroperitoneal lymph nodes | T3 |
| IIIA | Metastasis to the retroperitoneal lymph nodes with or without microscopic peritoneal involvement beyond the pelvis | T1, T2, T3aN1 |
| IIIA1 | Positive retroperitoneal lymph nodes only (cytologically or histologically proven) | |
| IIIA1(i) | Metastasis ≤10 mm in greatest dimension | |
| IIIA1(ii) | Metastasis >10 mm in greatest dimension | |
| IIIA2 | Microscopic extrapelvic (above the pelvic brim) peritoneal involvement with or without positive retroperitoneal lymph nodes | T3a/T3aN1 |
| IIIB | Macroscopic peritoneal metastases beyond the pelvic brim ≤2 cm in greatest dimension, with or without metastasis to the retroperitoneal lymph nodes | T3b/T3bN1 |
| IIIC | Macroscopic peritoneal metastases beyond the pelvic brim >2 cm in greatest dimension, with or without metastases to the retroperitoneal nodes (Note 1) | T3c/T3cN1 |
| Stage IV | Distant metastasis excluding peritoneal metastases | Any T, Any N, M1 |
| IVA | Pleural effusion with positive cytology | |
| IVB | Metastases to extraabdominal organs (including inguinal lymph nodes and lymph nodes outside of abdominal cavity) | |
| Note 1: Includes extension of tumor to capsule of liver and spleen without parenchymal involvement of either organ | ||
| Note 2: Parenchymal metastases are stage IVB | ||
Additional notes:
|
||
* Procedures performed in patients with no visible evidence of metastatic disease.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here