Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Osteonecrosis results from cell death in components of bone: hematopoietic fat marrow and mineralized tissue.
Osteonecrosis in its prototypical form results from impairment of the blood supply to the femoral head; consequently, the terms avascular necrosis and aseptic necrosis are common synonyms for this disorder.
Clinical and histopathologic aspects of osteonecrosis can be late-stage manifestations of other conditions such as subchondral insufficiency fractures and rapidly progressive osteoarthritis.
The femoral head is the site most commonly and severely affected by osteonecrosis. However, osteonecrosis may also develop in other locations (including the distal femur, humeral head, and small bones of the wrist and foot).
Imaging findings are typical at the late stages of the disease, but radiographic findings can be normal at the early stages. Magnetic resonance imaging is especially useful in these stages.
Predisposing factors (mainly glucocorticoid and alcohol use) should be sought.
Osteonecrosis is a clinical syndrome in which regional death of the cellular elements of bone, typically in a subchondral location, may lead to trabecular and subchondral collapse with consequent pain, impairment, and long-term joint damage. Osteonecrosis can occur in a variety of clinical settings and skeletal sites, with or without apparent predisposing factors. Although a number of synonyms have historically been used to refer to this disorder (e.g., aseptic necrosis and avascular necrosis, bone infarct ), the commonalities among these suggest that osteonecrosis is the endpoint of a number of conditions that impair blood supply to regions of bone. However, within the clinical spectrum of osteonecrosis, distinct subsets exist with unique features that suggest rather specific causes (e.g., osteonecrosis of the jaw, transient osteoporosis of the hip in pregnancy, and subchondral insufficiency fracture in the knee). Clinicopathologic aspects of osteonecrosis can also manifest as a late stage of other conditions such as severe forms of osteoarthritis (OA). Diagnostic vigilance is required for osteonecrosis because it can be hard to detect radiographically, and early, appropriate, intervention is necessary.
Osteonecrosis is rare, and its occurrence is hard to estimate; analysis of two health record databases in the United Kingdom found age-adjusted annual incidence rates that had increased over time, reaching 3 per 100,000 per year by 2003. Overall, the incidence of osteonecrosis increased with age, but the gender ratio varied with age such that women 60 years of age and older had a higher incidence than men. A more recent retrospective cohort analysis of 3,338,463 adults followed in the Swedish national databases after 2005 examined incidence and risk factors for primary or secondary osteonecrosis at any skeletal site. The 10-year risk of osteonecrosis was 0.4%, and the incidence rate was 4.7 cases/10,000 person-years. The strongest risk factors for osteonecrosis were hip fracture (standardized incidence ratio 8.0), solid organ transplantation (SIR, 7.1), dialysis (SIR, 6.7), and osteomyelitis (SIR, 6.4). Osteonecrosis was approximately 10 times more common than a previous population-based studies have suggested. The strongest risk factors for osteonecrosis were dialysis, hip fracture, osteomyelitis, and solid organ transplantation, but only hip fractures have contributed substantially to the disease burden.
Osteonecrosis is associated with a variety of clinical conditions such as systemic lupus erythematosus (SLE) and chemical exposures such as glucocorticoid and alcohol use ( Box 208.1 ). Indeed, recent epidemiologic studies have tended to focus on specific subsets or exposures such as osteonecrosis associated with renal transplantation or intravenous administration of zoledronic acid. The factors that have received the most scrutiny include glucocorticoid exposure, alcohol use, SLE, coagulopathies, hemoglobinopathies, and bisphosphonate use. The most important of these are considered in detail below.
Alcohol use
Bisphosphonate use
Coagulopathies
Congenital dysplasia of the hip
Glucocorticoid use
Dysbaric osteonecrosis
Gaucher disease
Hemoglobinopathy
Hemophilic disorders
Human immunodeficiency virus infection
Hyperlipidemia
Legg-Calvé-Perthes disease
Pregnancy
Radiation
Slipped capital femoral epiphysis
Systemic lupus erythematosus
Trauma
Accumulated data from numerous clinical reports and risk factor studies provide strong evidence for a causal relationship between glucocorticoid exposure and the development of osteonecrosis. The range in reported incidence (or cumulative prevalence) rates of osteonecrosis among different cohorts taking glucocorticoids is very wide ranging from 0.6% to 38%, presumably in part explained because of varying exposures in terms of agent, dose, and duration, as well as factors such as age and underlying disorder (e.g., SLE). There is a general trend in the more robust analyses, toward increased risk of osteonecrosis with greater dose and duration of use, and also poorer course following hip-preserving interventions with continued use of glucocorticoids.
Alcohol consumption is also considered to be an established risk factor for osteonecrosis. One case-control study demonstrated a dose–response relationship for alcohol in osteonecrosis, with risk ratios of 3, 10, and 18 for those regularly consuming less than 400, 400 to 1000, and more than 1000 mL/week or more, respectively, compared with nondrinkers. One theory suggests that altered fat metabolism secondary to alcohol use may lead to adipocyte hypertrophy and subsequent microvascular compression with resulting increased intraosseous pressure. Recently, it has also been determined that both alcohol and glucocorticoids have adverse effects on bone cell lineage by enhancing adipogenesis and inhibiting osteocytogenesis by marrow stem cells. Other studies point toward the direct cytotoxic effects of alcohol. Interestingly, there appear to be differences in the frequencies of alcohol-metabolizing enzyme gene polymorphisms among subpopulations of patients classified as having alcohol-related osteonecrosis.
In SLE, clinically apparent osteonecrosis is frequent, occurring in up to 15% of patients and an even higher prevalence when asymptomatic disease is detected using magnetic resonance imaging (MRI). Much of this risk can be explained by glucocorticoid treatment. However, attempts to determine whether other patient characteristics mediate this risk have produced inconsistent results. It is likely that multiple factors in patients with SLE contribute to the development of osteonecrosis, including procoagulant factors such as anticardiolipin antibodies.
Osteonecrosis appears to be associated with a range of coagulation disorders and with hemoglobinopathies, most notably sickle cell disease and sickle cell–hemoglobin C (hemoglobin SC) disease. Among patients with sickle cell disease, the prevalence of symptomatic osteonecrosis is 3% to 5% but rises to 10% to 41% when asymptomatic osteonecrosis is included. In sickle cell disease, the risk of osteonecrosis is related to the severity of the disease process, including the frequency of crises, hematocrit levels, and mean corpuscular volume. These clinical observations suggest that thrombosis and vasoocclusion participate in the development of osteonecrosis. Studies of hypofibrinolysis and thrombophilia in osteonecrosis have found increased plasminogen activator inhibitor (PAI) activity and polymorphisms of the PAI-1 gene in certain patients.
Osteonecrosis of the jaw is a rare disorder recognized in association with long-term use of bisphosphonates, but also with use of denosumab, antiangiogenic agents, and mTOR inhibitors and often in the setting of dental extraction, periodontal disease, and comorbidity. Many reported cases occurred in association with intravenous administration of bisphosphonates accompanying a chemotherapeutic regimen for multiple myeloma, breast cancer, or prostate cancer. In such patients treated with zoledronic acid or pamidronate (or both drugs), the cumulative prevalence is about 5%. The risk of osteonecrosis of the jaw related to oral bisphosphonate used for treatment of osteoporosis is much lower estimated at 0.01% to 0.04% but is related to increasing age and duration of use of bisphosphonate as well as dental extraction. A recent analysis of the U.K. National Health Service database found that ever-users of bisphosphonates had a sixfold increased risk for osteonecrosis of the jaw (adjusted RR = 6.1). The risk for osteonecrosis of the jaw was also increased in women with prior cancer who had not been exposed to bisphosphonates (RR = 3.4). The estimated absolute risk of hospital admission for osteonecrosis of the jaw over a 5-year period from age 70 to 74 in women without prior cancer was 0.09 per 1000 in never-users and 0.69 per 1000 in ever-users of oral bisphosphonates. In this U.K. population of postmenopausal women, use of oral bisphosphonates accounted for around one third of cases, with an excess risk of about 0.6/1000 users over 5 years. Risk for osteonecrosis of the jaw postdental extraction may also be increased in the presence of autoimmune disease (especially rheumatoid arthritis and Sjögren disease ). In the Japanese Osteoporosis Intervention Trial, the incidence of early stage osteonecrosis of the jaw was 6.1 per 1000 patient-years in the minodronic acid group and 3.4 in the raloxifene group.
Proposed mechanisms for osteonecrosis of the jaw include susceptibility of bone to infection and necrosis caused by decreased bone turnover and direct toxicity of high-potency bisphosphonates to bone cells. There has been attention recently on the role of pathogenic bacteria in the progression of osteonecrosis of the jaw and the possibility of protection from normal indigenous microbiota. There have been cases of osteonecrosis of the jaw attributed to TNF-α inhibitors and rituximab, suggesting impairment of immune responsiveness as a possible contributor in this setting.
Necrosis of all cellular components of bone, both in hematopoietic fat marrow and mineralized tissue, is the hallmark of osteonecrosis. Death of the cellular components of bone results in destruction of the osseous components of bone with structural consequences ( Fig. 208.1 ).
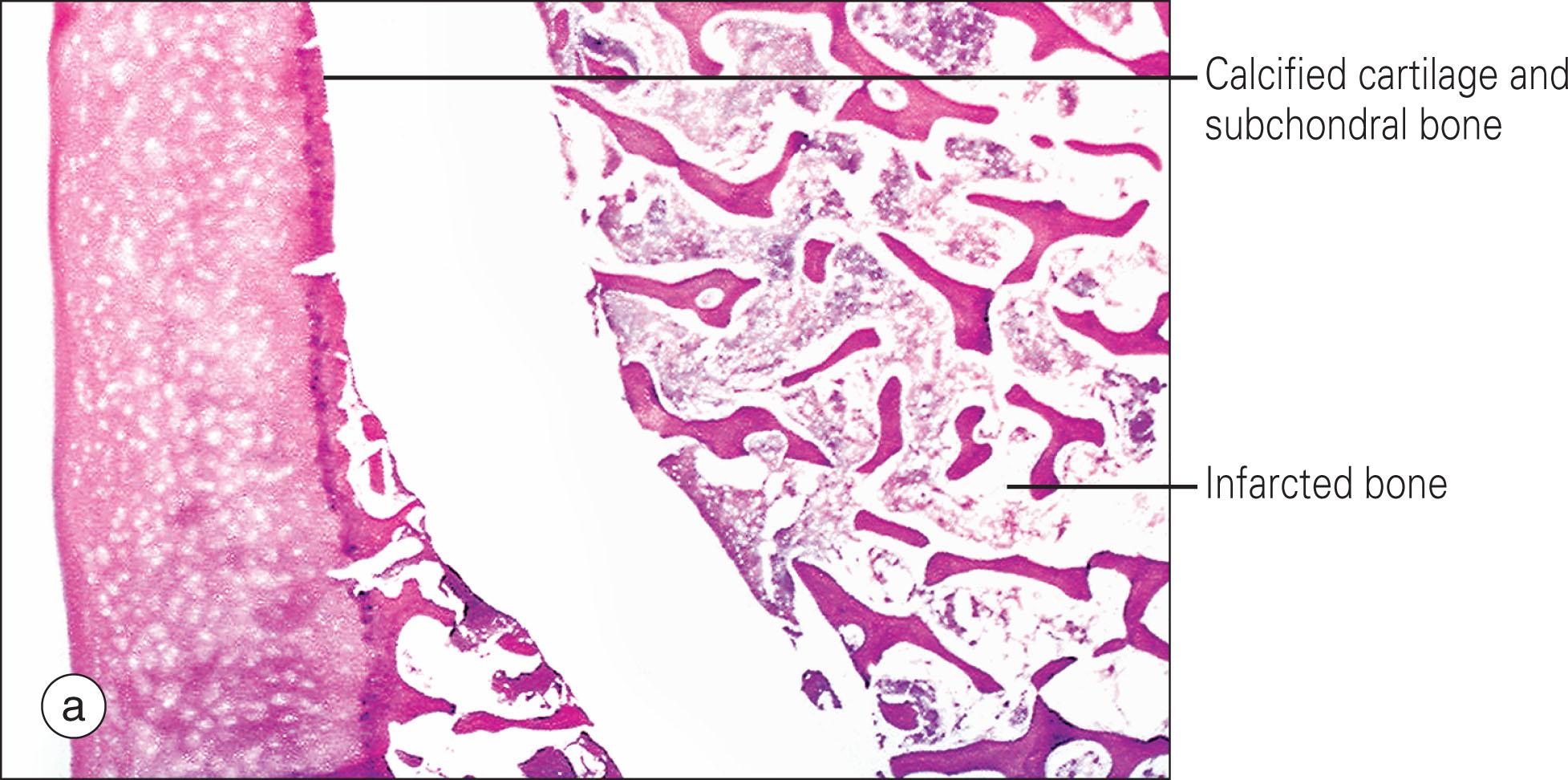
In the early (prenecrotic) stage, interstitial edema and plasmostasis are confined to the bone marrow, with occasional foam cells. The primary indication of necrosis of bone tissue is the disappearance of osteocytes, the cavities of which are empty and often enlarged. As the severity progresses, the medullary spaces become filled with necrotic tissue, which is subsequently associated with trabecular necrosis and emptying of the lacunae. The bone sequestrum is composed of dead trabeculae without osteocytes but maintained architecture. The bone marrow tissue exhibits disappearance of stem cells, adipocyte nucleolysis, and membrane loss, often without persistence of recognizable cell remnants. Subchondral fracture, when present, is usually located in the dead bone, typically somewhat parallel to the edges of the articular surface, which results in the characteristic radiologic “crescent sign” ( Fig. 208.2 ). Around the dead bone tissue is usually a zone of repair comprising fibrovascular proliferation, bone resorption, and new bone apposition on the remains of the old dead trabeculae.
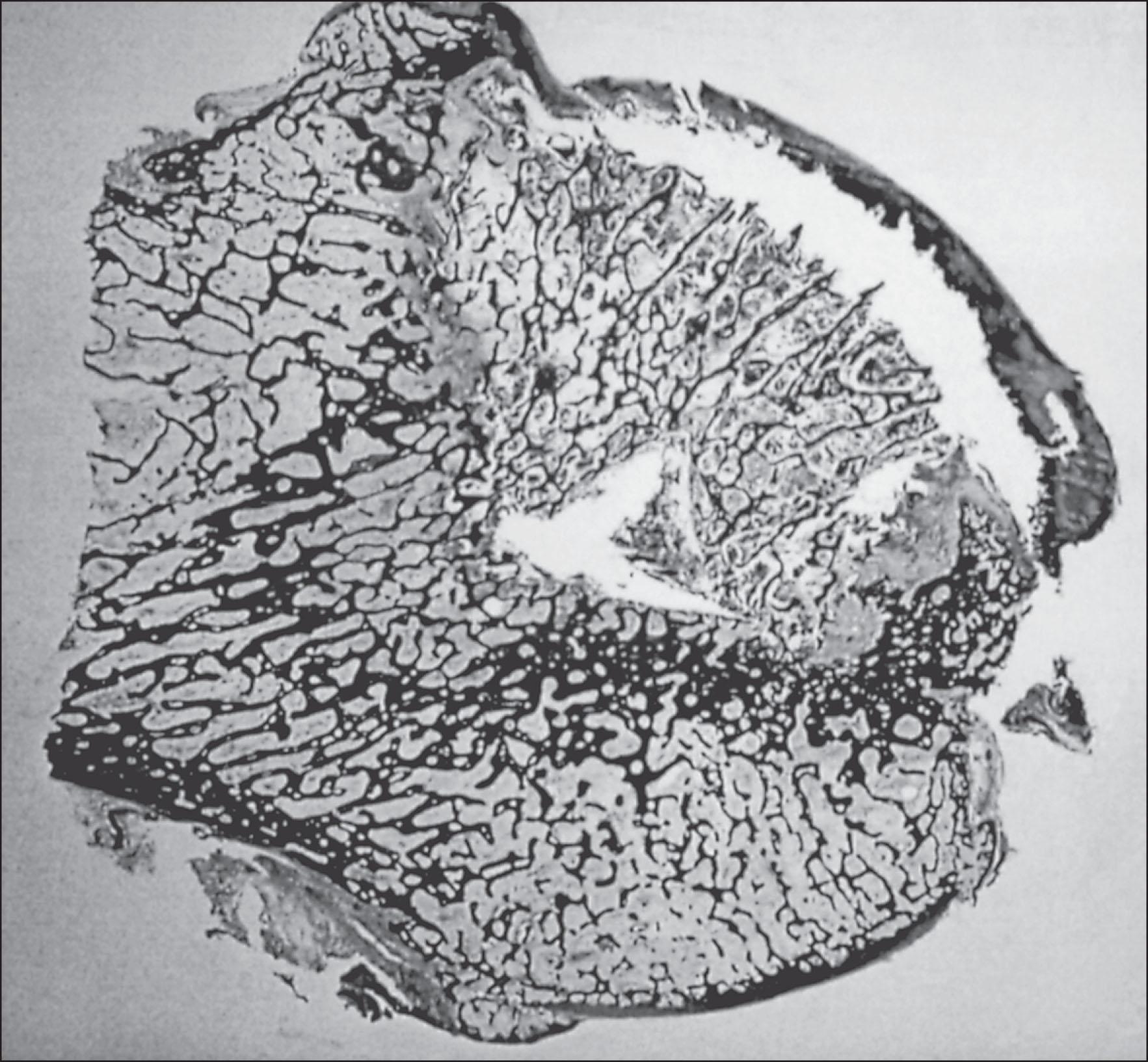
At a macroscopic level, osteonecrosis is regional and highly circumscribed. It can be confined to a medullary section of bone or involve a periarticular segment such as the subcortical femoral head. The biomechanical consequences of osteonecrosis are largely contingent on its anatomic location and extent of involvement. Areas confined to a medullary location can be asymptomatic, but involvement of the subcortex adjacent to a joint surface leads to articular collapse and joint destruction. For individuals with polyarticular osteonecrosis, as can be seen in sickle cell disease, the condition can have devastating functional consequences.
In its simplest characterization, focal bone death results from a compromised arterial blood supply, a paradigm best exemplified by the posttraumatic form of osteonecrosis. However, in the spontaneous forms of osteonecrosis, a pathologic basis for interruption of the blood supply is rarely clear. This conundrum has led to speculation about processes that could compromise the health of affected regions of bone and result from exposures known to be associated with osteonecrosis. Most of these involve a proposed vascular mechanism and include:
Disturbances of fat metabolism
Intravascular occlusion by coagulation, thrombi, fat emboli, or abnormally shaped red blood cells
Elevated intraosseous pressure
Intramedullary hemorrhage
Osteocytotoxicity
Other processes that might participate in the overall extent of destruction include the following:
The reparative response itself
Mechanical stress
Inhibition of angiogenesis
These different processes are considered below.
Evidence indicates that increases in the amount of fat within the femoral head elevate intracortical pressure and lead to sinusoidal collapse. Numerous studies have shown diverse effects of glucocorticoid exposure on lipid metabolism and on differentiation of bone marrow–derived stem cells favoring adipocytogenesis. In vivo studies have confirmed that glucocorticoids increase the fat content of the femoral head and that this increase correlates with an increase in intraosseous pressure and decreased blood supply. Interestingly, this effect has been abrogated by the administration of clofibrate, a lipid-lowering agent. Steroids have also been shown to cause fat emboli in animal models. Taken together, these studies implicate an abnormality of fat metabolism or morphology that may compromise blood flow to the affected area through an increase in intraosseous pressure.
Abnormalities of lipid metabolism have also been observed in patients with osteonecrosis attributed to alcohol use. Patients with alcohol-associated osteonecrosis frequently have elevated serum cholesterol and triglyceride levels, and the cholesterol content of bone samples from patients with osteonecrosis has been found to be elevated. Abnormalities of trabecular fat cell size and bone marrow pressure have also been demonstrated in an alcoholic rabbit model. Alcohol also appears to induce differentiation of marrow stromal cells into adipocytes in a dose-dependent manner and reduces osteogenesis.
Intravascular coagulation of the intraosseous microcirculation, progressing to more generalized thrombosis, is a strong contender as a process leading to osteonecrosis. Many of the conditions reported to be associated with osteonecrosis have attributes capable of triggering intravascular coagulation. This could occur as a primary event (as in antiphospholipid antibody syndrome ) or as a secondary reaction to some other process (e.g., thrombosis secondary to fat embolism).
Also, numerous gene abnormalities or polymorphisms associated with hypercoagulable states occur with increased frequency in individuals with osteonecrosis, including the mutations factor V Leiden and prothrombin 20210 A and deficiency of protein C or S. Several studies of coagulation factors have found abnormalities relating to PAI in patients with osteonecrosis, which suggests reduced fibrinolysis as a potential risk factor. Such observations prompted a clinical trial of low-molecular-weight heparin, which slowed progression of osteonecrosis in patients with early-stage disease.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here