Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The kidneys develop from the intermediate mesoderm, which is located on each side of the embryo between the paraxial (somitic) and lateral plate mesoderm. After the fourth week, during which the embryo undergoes a complex folding process, the intermediate mesoderm forms a lateral nephrogenic cord and a medial genital (gonadal) ridge. The nephrogenic cord gives rise to three successive kidney precursors, while the genital ridge gives rise to the gonads.
The three kidney precursors—known as the pronephros, mesonephros, and metanephros, in order of appearance—develop in a cranial-to-caudal sequence along the nephrogenic cord. Although the pronephros and mesonephros completely regress in utero, they are nonetheless essential for the normal development of the metanephros, which becomes the definitive kidney. The pronephros and mesonephros can be viewed as intermediate structures in the historical evolution of the kidney because they have more important roles in organisms such as fish and amphibians.
Many signaling pathways have been found to play roles in the development of the kidneys, with the already expansive list growing on a regular basis. A detailed discussion of these pathways, however, is beyond the scope of this text, which will instead focus on the anatomic changes that occur during development.
At the start of the fourth week, the cervical portion of the nephrogenic cords undergoes mesenchymal-to-epithelial conversion to form the paired pronephric ducts, which grow in a caudal direction. A series of pronephric tubules appear medial to the ducts and connect them to the coelom, the precursor to the peritoneal space. These tubules constitute the paired pronephroi.
A glomerulus-like structure, known as the glomus, projects from the dorsal aorta into the coelom. The glomus produces filtrate, some of which enters the pronephric tubules and then passes into the pronephric ducts. The pronephric ducts, however, are blind-ended. Thus the pronephroi are not functional excretory organs during human development.
As the paired pronephric ducts continue to grow in a caudal direction, the pronephric tubules degenerate, becoming completely absent by day 25 of gestation. At the same time, however, the growing pronephric ducts continue to grow toward the caudal end of the embryo. These ducts, which become known as the mesonephric (wolffian) ducts, induce the formation of about 40 pairs of mesonephric tubules from the dorsolumbar region of the nephrogenic cords. These tubules constitute the paired mesonephroi.
Each mesonephric tubule receives filtrate from a glomerulus, which receives blood from a branch of the dorsal aorta and drains blood to the posterior cardinal vein. Some tubules drain urine into the mesonephric ducts.
Around the twenty-sixth day of gestation, the mesonephric ducts fuse with the cloaca, the precursor of the urinary bladder. At this point, the mesonephroi become functional excretory organs. The mesonephric tubules degenerate over subsequent months, however, and are almost completely absent by the fourth month of gestation. A small subset, however, persist into adulthood. In males, some of the most caudal tubules form the efferent ductules of the testes. Meanwhile, in females, some of the tubules form vestigial structures known as the epoophoron and paroophoron.
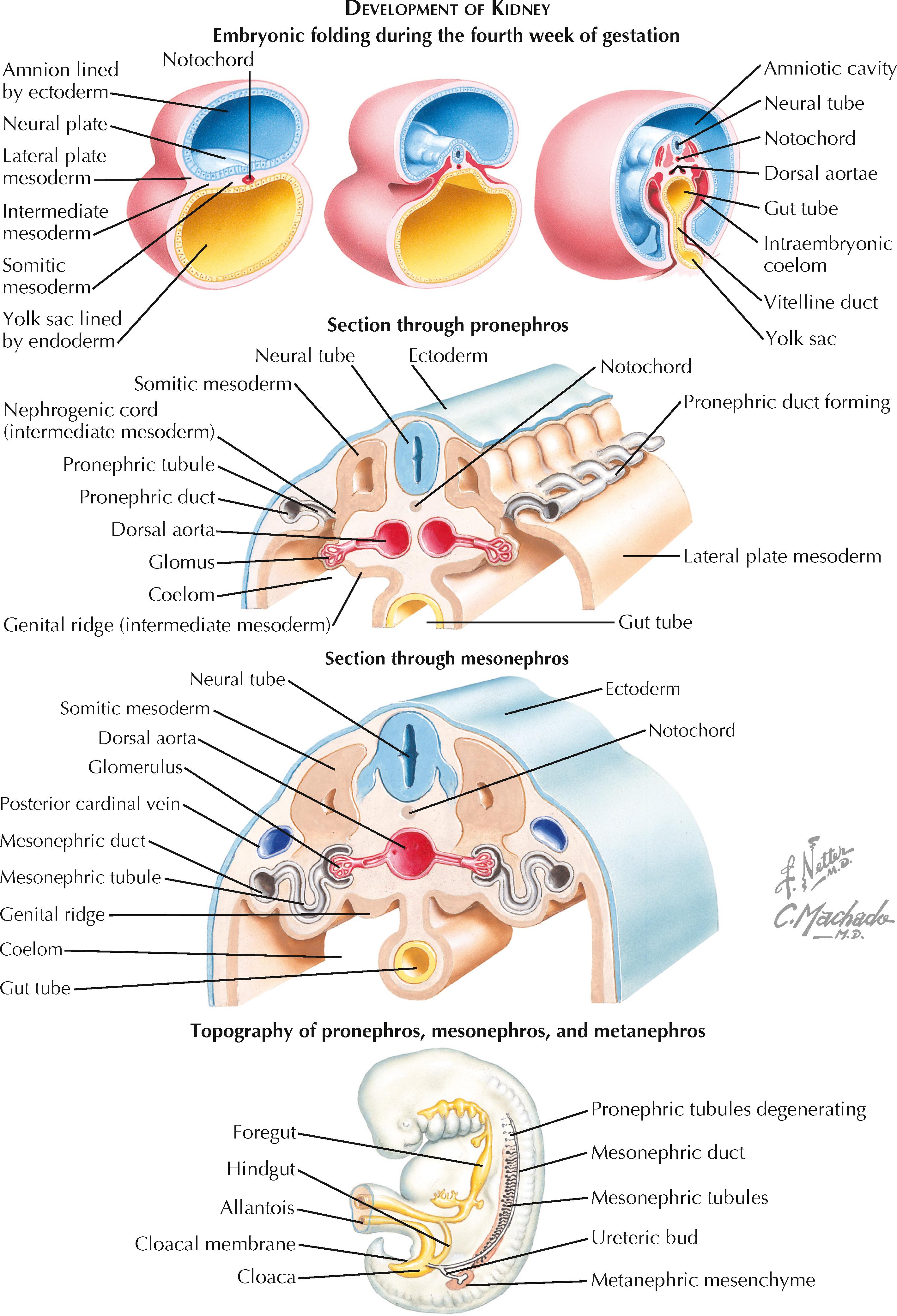
The paired metanephroi are the precursors of the definitive adult kidneys. They begin to form around the twenty-eighth day of gestation, shortly after the mesonephric ducts have fused with the cloaca. The caudal portion of each mesonephric duct sprouts a small diverticulum known as a ureteric bud. Each bud then grows toward a nearby mass of mesoderm known as the metanephric mesenchyme, which is located at the sacral end of the ipsilateral nephrogenic cord.
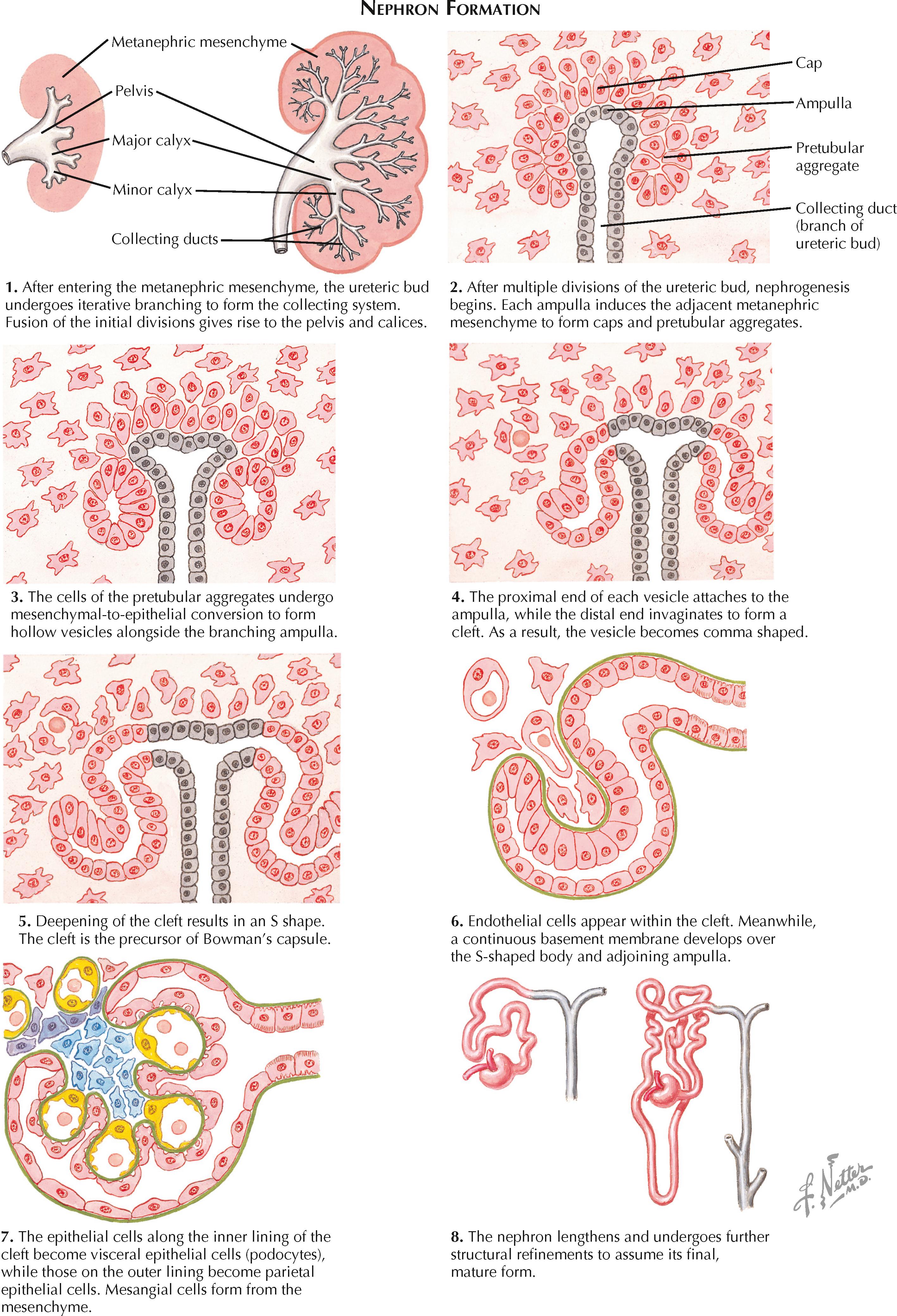
Once each ureteric bud enters its associated metanephric mesenchyme, it begins a process of iterative bifurcation that gives rise to the urine collecting system. The first eight bifurcations of the ureteric bud give rise to the renal pelvis, major calices, and minor calices. These initial divisions later fuse to a considerable extent, resulting in the definitive appearance of the pelvicaliceal system. The next dozen bifurcations give rise to the collecting duct system.
As the collecting ducts are being formed, the surrounding metanephric mesenchyme differentiates into nephrons, each consisting of a glomerulus, proximal tubule, thin limb, distal tubule, and connecting tubule. The ends of these nephrons fuse with the developing collecting duct system. Throughout this process, the ureteric bud and metanephric mesenchyme provide essential inductive signals to one another. Thus, if either one is absent, the metanephros will not develop.
The first phase of nephron formation begins around the sixth or seventh week of gestation. The tips of the branching ureteric buds, known as ampullae, induce the condensation of adjacent mesenchymal cells. Some of the mesenchymal cells form caps over the ampullae, while others form clusters just lateral to the ampullae. The clusters, also known as pretubular aggregates, are the nephron precursors.
Each pretubular aggregate undergoes mesenchymal-to-epithelial conversion to form a hollow vesicle. The proximal end of each vesicle fuses with the adjacent ampulla, the precursor of a collecting duct. Meanwhile, the distal end of each vesicle invaginates to form a cleft, the precursor of Bowman's capsule. The formation and subsequent deepening of the cleft causes the vesicle to become a comma-shaped and then S-shaped body. The cells lining the inner part of the cleft are the precursors of the visceral epithelial cells (podocytes), while those lining the outer part are the precursors of the parietal epithelial cells.
In the S-shaped stage, endothelial cells appear within the cleft and become flattened and fenestrated. The developing podocytes send foot processes over the endothelial cells, and the podocyte basement membrane fuses with that of the endothelial cells, forming the three-layered glomerular basement membrane. Mesangial cell precursors, derived from the metanephric mesenchyme, enter the cleft and form the scaffolding for the developing glomerular capillaries. Throughout this process, the entire primitive nephron lengthens, giving rise to distinct proximal and distal segments.
The second phase of nephron formation begins at approximately the fourteenth week of gestation. During this phase, the ampullae grow outward toward the cortex without further division. As nephrons form adjacent to the growing ampullae in the manner described previously, older nephrons attach to the connecting tubules of newer nephrons rather than directly to the ampullae. This process gives rise to nephron arcades, all joined by a single connecting tubule to a collecting duct. These nephrons become the long-looped (juxtamedullary) nephrons in the mature kidney.
The third phase of nephron formation begins at approximately the twentieth week of gestation. During this phase, the ampullae continue to grow toward the cortex without further division; however, as new nephrons are formed, they retain their individual attachments to the collecting duct system. These nephrons become the short-looped (cortical) nephrons in the mature kidney.
After the thirty-sixth week of gestation, no new nephrons form, but the existing nephrons continue to undergo structural changes. For example, portions of the proximal and distal tubules become increasingly tortuous and convoluted, while the loops of Henle grow deeper into the medulla.
The metanephroi begin to produce urine at 9 weeks of gestation, even as active nephrogenesis is ongoing. Such urination becomes essential for maintaining a normal volume of amniotic fluid. The placenta, how ever, is the organ responsible for removing waste products from the fetus.
The fetal kidney has a lobated surface appearance, which can be attributed to condensations of metanephric mesenchyme around the initial branches of the ureteric buds. This surface lobation, however, usually disappears around 4 or 5 years of age, as additional tissue fills in the sulcated areas. If fetal lobation persists into adulthood, it is an inconsequential anatomic variant; however, such lobations may be sometimes mistaken for cortical scarring.
The urinary bladder develops from the cloaca, a primitive pouch that forms during the fourth week of gestation. At the beginning of the fourth week, the embryo remains a trilaminar structure consisting of ectoderm, mesoderm, and endoderm. The cloaca has not yet developed, but the cloacal membrane is visible as a small depression near the caudal end of the embryo. At this site, ectoderm from the neural plate merges with endoderm from the yolk sac, without an intervening layer of mesoderm.
During the fourth week the embryo undergoes a folding process, during which the yolk sac gives rise to the gut tube. The caudal part of the gut tube, known as the hindgut, terminates at the cloacal membrane. The most caudal end of the hindgut dilates to form the cloaca.
The cranioventral aspect of the cloaca is continuous with a narrow tube, known as the allantois, that extends into the connecting stalk. Meanwhile, the lateral walls of the cloaca receive the mesonephric (wolffian) ducts.
By the sixth week, a septum divides the cloaca into an anterior primitive urogenital sinus and posterior rectum. The exact mechanism of the septation process has long been a topic of active debate and investigation. Some have proposed that a septum oriented in the coronal plane descends through the cloaca in a cranial-to-caudal direction, while others have proposed that two lateral cloacal folds fuse in the midline to form a septum. Still others have proposed various combinations of the two previous theories. More recent investigations have rejected both of these theories, instead arguing that septation results from advancement of the dorsal cloaca toward the cloacal membrane as the embryo lengthens and rotates. During this process, the urorectal fold, located between the allantois and the hindgut, passively advances toward the cloacal membrane, causing effective septation. Subsequent apoptosis of the cloacal membrane establishes two distinct openings that lead to the primitive urogenital sinus and rectum. The tip of the septum lying between them gives rise to the perineal body.
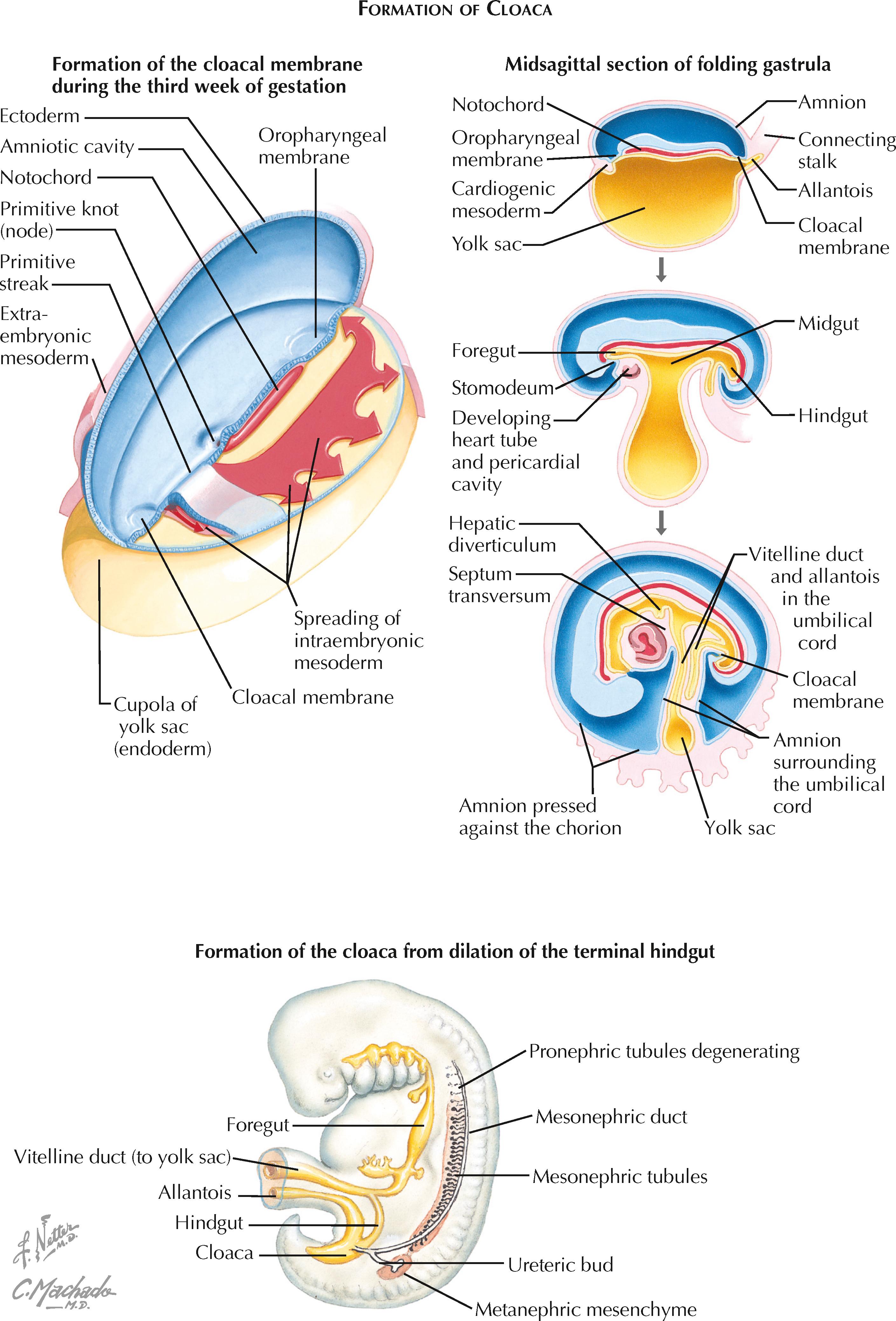
After cloacal septation is complete, the primitive urogenital sinus contains three major parts. The most caudal part is known as the definitive urogenital sinus, and it will become the penile and spongy urethra in males, or the vestibule of the vagina in females. The neck, located just proximal to the definitive urogenital sinus, will become the membranous and prostatic urethra in males, or the urethra in females. The bulging area proximal to the neck will become the urinary bladder in both sexes. The allantois, which connects the bladder to the umbilical cord, will regress to form the a thick, epithelial-lined tube known as the urachus, which in turn will further degenerate into a simple fibrous cord known as the median umbilical ligament.
During subsequent weeks, the definitive urogenital sinus continues to undergo structural changes as it becomes the mature bladder. By the tenth week, the endodermal cells become a single layer of cuboidal epithelium. Over subsequent weeks, additional cell layers appear, which begin to assume the characteristics of differentiated urothelial cells. Meanwhile, during the twelfth week, the surrounding splanchnopleuric mesoderm differentiates to form the detrusor muscle, which lines the urothelium. As bladder development proceeds, the mechanical distention associated with urine storage appears to be essential for the development of normal wall compliance.
The ureteric buds appear during the fifth week of gestation as small diverticula near the caudal ends of the mesonephric ducts (see Plate 2-1 ). They eventually give rise to the ureters, renal pelves, calices, and collecting ducts.
Although the ureteric buds originally drain into the mesonephric ducts, they are transferred to the future bladder in a process known as mesonephric duct exstrophy, which occurs during cloacal septation. In this process, the most caudal ends of the mesonephric ducts evert and balloon into the lumen of the urogenital sinus. Eventually, the eversion of each duct is extensive enough to bring the attached ureteric bud into the sinus. The buds then separate from the mesonephric ducts and fuse with the posterior wall of the urogenital sinus.
A ureteric bud with a more caudal position on the mesonephric duct will not be drawn far into the bladder during the exstrophy process, resulting in a more superior and lateral ureteric orifice, as well as a short course through the bladder wall. In contrast, a ureteric bud with a more cranial position on the mesonephric duct will be drawn deep into the bladder, resulting in a more inferior and medial ureteric orifice, as well as a longer intramural course.
Like the bladder, each ureter develops from a simple epithelial tube into a complex, multilayered structure containing urothelium, smooth muscle, and connective tissue. There is transient obliteration of the ureteral lumen during the sixth week of gestation. Recanalization quickly ensues, however, starting in the midureter region and progressing in both directions until the entire lumen is once again patent.
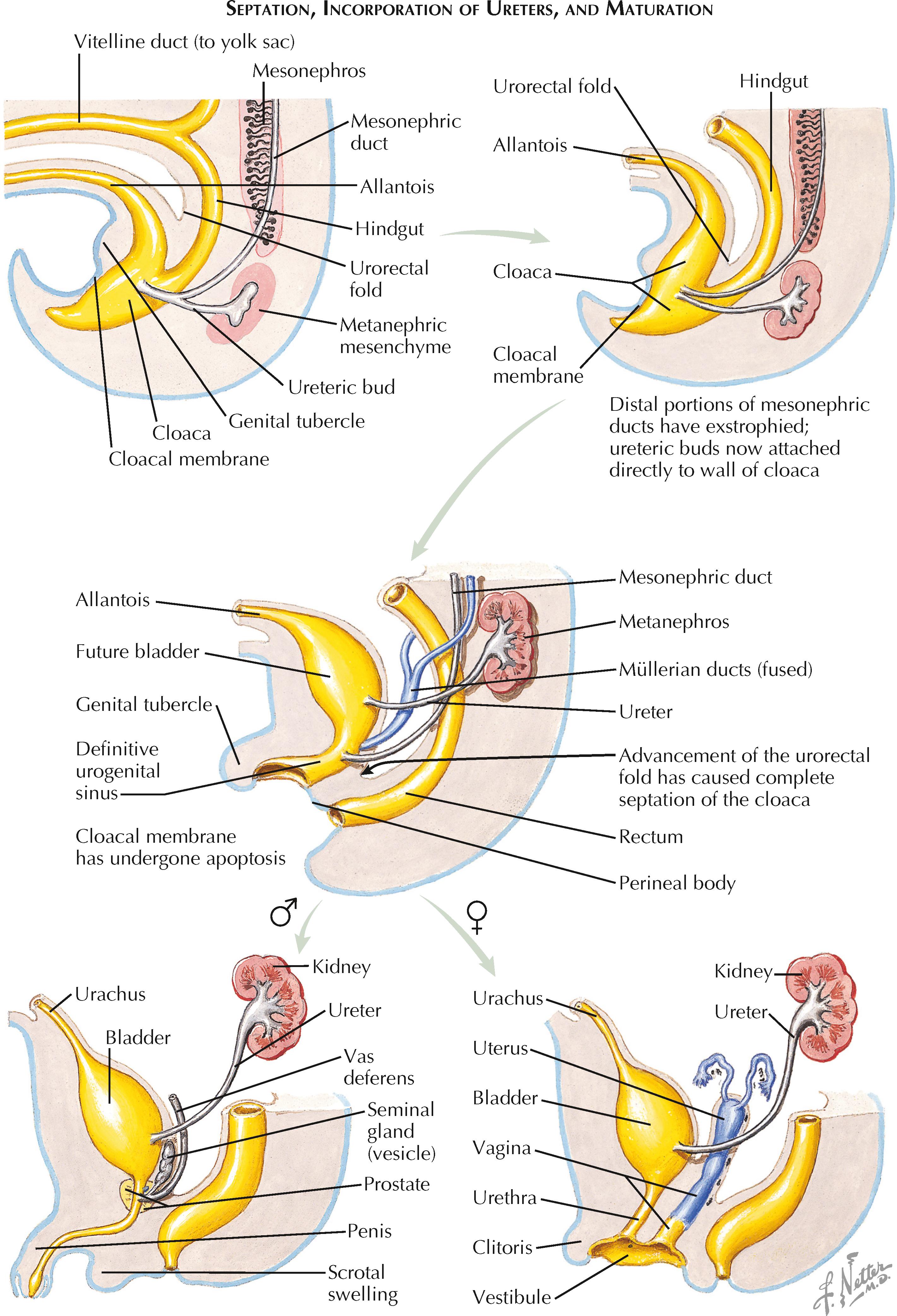
By the end of the exstrophy process, the mesonephric ducts terminate in the bladder medial and inferior to the future ureteric orifices. Although it was previously thought that the mesonephric ducts contributed to the formation of the trigone, this long-held view has recently been called into doubt.
In males, the mesonephric ducts become the ejaculatory ducts, vas deferens, seminal glands (vesicles), and epididymis. In females, in contrast, the mesonephric ducts largely degenerate, giving rise only to the vestigial structures known as the epoöphoron and paroöphoron. Instead, the paramesonephric (müllerian) ducts, which degenerate in males, are responsible for formation of the female reproductive tract. These ducts appear lateral to the mesonephric ducts during the sixth week, and in females they become the uterine (fallopian) tubes, uterus, and upper two thirds of the vagina.
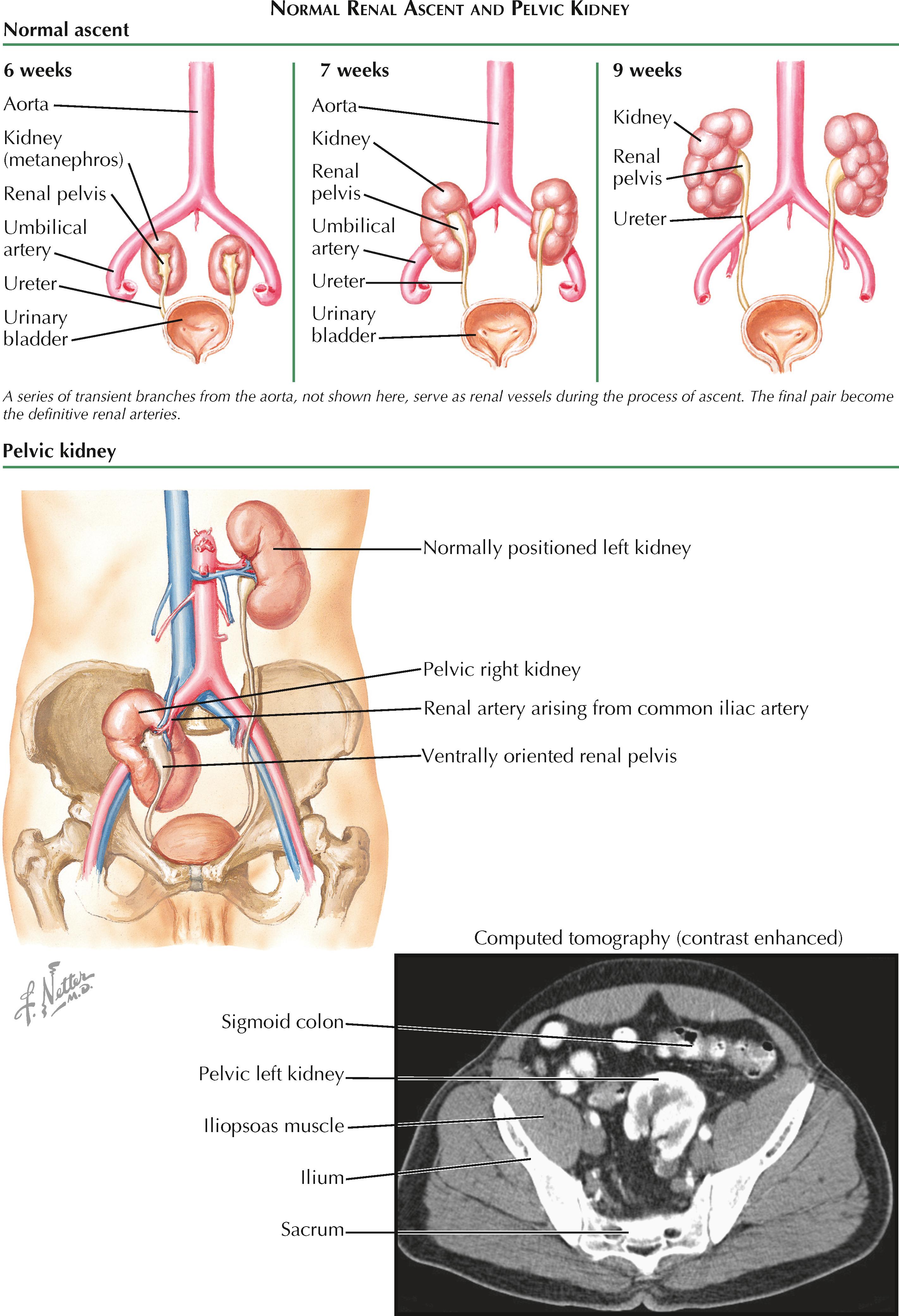
The adult kidneys are positioned in the lumbar retroperitoneum; however, their development begins in the sacral region of the fetus, where the paired metanephroi appear during the fifth week of development. Their change in position reflects a process known as renal ascent, which occurs during the sixth to ninth weeks of gestation. Although its exact mechanism is not well understood, it likely reflects rapid growth of the sacral end of the fetus, which leads to a change in the relative position of the kidneys.
As they ascend, the kidneys are vascularized by a series of transient branches from the dorsal aorta. In most individuals, all but the final pair of arterial branches degenerate, leaving one major renal artery extending to each kidney. In some individuals, however, the earlier branches of the aorta may fail to degenerate, resulting in aberrant persistence of an extrahilar (polar) artery. (This condition is so common that it is considered a normal anatomic variant, rather than a congenital defect, and is thus described in more detail in the section on normal renal vasculature. See Plate 1-12 .)
Renal ectopia results from abnormalities during the process of ascent. If a kidney fails to ascend at all, it is known as a pelvic kidney. If it undergoes incomplete ascent, it is known as a lumbar kidney. If it ascends too far and reaches the thorax, it is known as a thoracic kidney. Finally, if a kidney ascends to the contralateral side, it is known as a crossed ectopic kidney.
Pelvic kidney is the most common form of renal ectopia, with an estimated incidence of 1 in 2,200 to 3,000. A common view is that this pattern of ectopia represents persistence of fetal vasculature, which prevents the normal ascent of the kidney. Other possible causes, however, encompass intrinsic defects in the ureteric bed or metanephric mesenchyme.
The vessels that supply a pelvic kidney typically arise from the iliac vessels or the most inferior portion of the abdominal aorta. The ureter is short and often prone to reflux. The hilum may be directed ventrally, rather than medially, because of a failure of normal rotation (see Plate 2-7 ).
Most patients with pelvic kidneys are asymptomatic, and the abnormality is either incidentally noted or never discovered. A subset of individuals, however, may become symptomatic secondary to the development of an upper urinary tract obstruction, nephrolithiasis, or urinary tract infection. These sequelae occur if malrotation results in high insertion of the ureter or a vessel crossing the collecting system, since these can both cause urinary stasis and outflow obstruction.
Thus, patients with pelvic kidneys may occasionally have abdominal pain, hematuria, or a palpable abdominal mass. The pelvic kidney is then detected on further workup with ultrasound or computed tomography (CT). The treatment strategies for nephrolithiasis and ureteropelvic junction obstructions in patients with pelvic kidneys are largely the same as those used for patients with normally positioned kidneys; however, the abnormal course of the ureter may make ureteroscopy difficult, and there is a risk of damaging abnormally positioned vessels and nerves.
A pelvic kidney is more susceptible to injury from blunt trauma than a normally positioned kidney because the latter is (1) surrounded by a large, protective cushion of perinephric and retroperitoneal fat, (2) protected posteriorly by the ribs, and (3) located at a safe distance from the anterior abdominal wall and narrow pelvis. As a result, patients known to have pelvic kidneys should be encouraged to wear appropriate protection if they participate in contact sports.
Thoracic kidney is the rarest form of all renal ectopias, with an estimated incidence of 1 in 13,000 according to one autopsy series. Unlike pelvic kidneys, thoracic kidneys appear to be more common in males. An ectopic thoracic kidney may be located predominantly above or below the diaphragm. In either case, the intrathoracic portion passes through the lumbocostal triangle (foramen of Bochdalek) and is covered by a thin membrane of the diaphragm. As a result, the kidney does not reside within the pleural space; however, the adjacent region of the lung may be hypoplastic. Thoracic kidneys are more commonly seen on the left side, possibly because the liver blocks excessive ascent of the right kidney.
It is unclear why thoracic kidneys occur, but two possibilities include delayed closure of the diaphragm, as well as excessive and accelerated renal ascent.
The vessels that supply a thoracic kidney usually arise from the abdominal aorta at a higher position than normal. The ureter is appropriately increased in length and inserts normally into the bladder. Renal rotation is usually complete, and thus the renal pelvis has a normal medial orientation. Both the ureter and renal vessels pass through the lumbocostal triangle as they course from the kidney to the abdomen. The associated adrenal gland typically remains in its normal position but has been documented in some cases to be associated with the ectopic kidney.
Most thoracic kidneys are asymptomatic, causing neither respiratory nor urinary symptoms. Thus this abnormality often goes undetected unless a patient undergoes imaging for another unrelated reason.
Crossed ectopia of the kidney is an uncommon condition in which one or both kidneys are found on the contralateral side of the abdomen. The “crossing” of a kidney is evidenced by the path of its associated ureter, which crosses the midline to insert into the opposite side of the bladder.
The embryologic basis for crossed renal ectopia is not known. It has been speculated that during renal development, the ureteric bud may cross the midline to enter the contralateral metanephric mesenchyme. Others have suggested that abnormally positioned vessels, such as the umbilical artery, may obstruct the normal path of an ascending kidney, which then takes the path of least resistance to the contralateral side. Teratogens or genetic factors may also play a role.
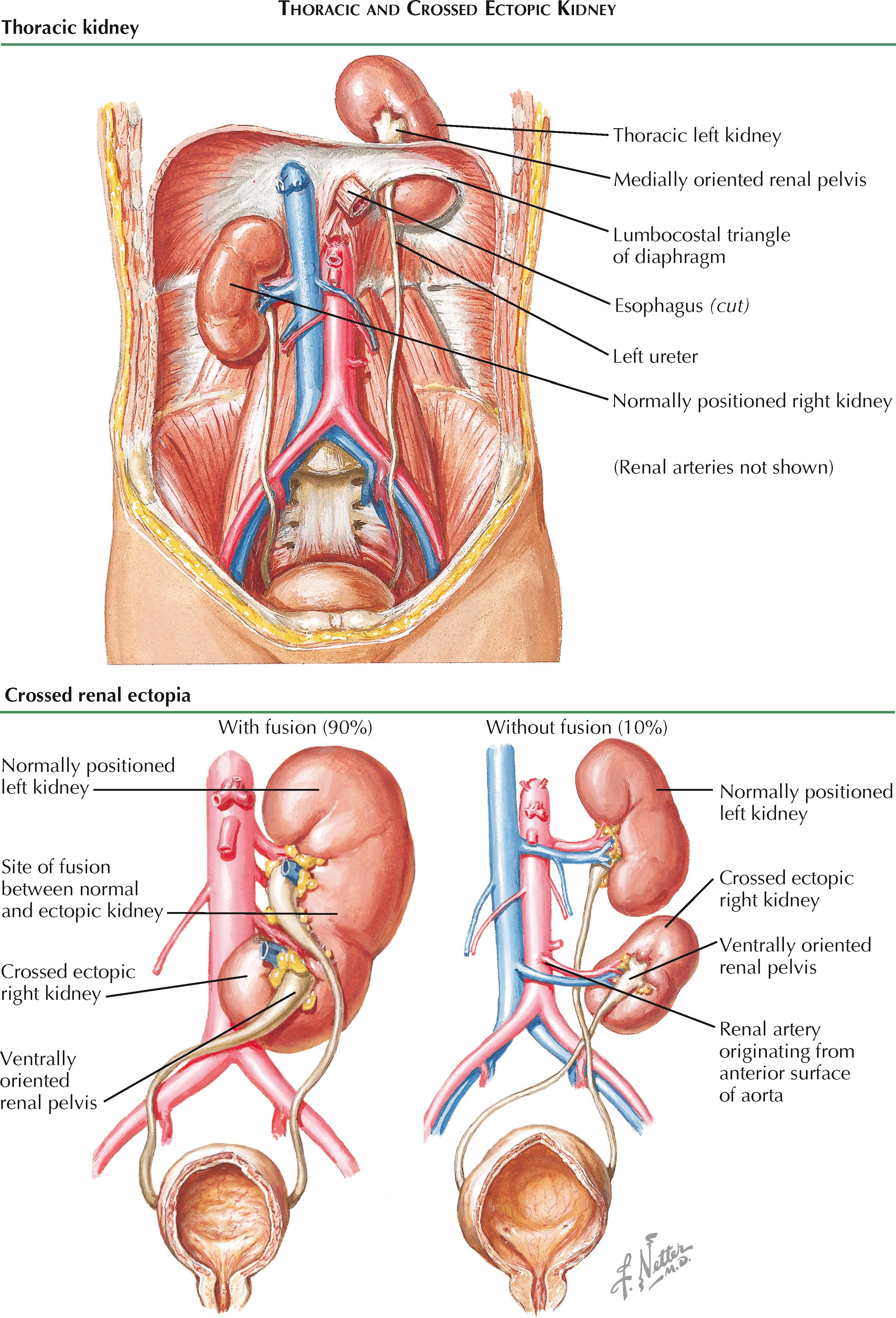
The crossed kidney generally lies caudal to the normally positioned kidney and has a ventrally oriented renal pelvis. In 90% of cases, the crossed kidney is fused with the inferior pole of the normally positioned kidney. In about 10% of cases, the two kidneys remain separate and distinct. The renal artery of the ectopic kidney may originate from the iliac artery or from either the lateral or anterior surfaces of the abdominal aorta.
Like pelvic kidneys, crossed ectopic kidneys are usually incidental, asymptomatic findings but may rarely occur with abdominal pain, hematuria, or other symptoms.
During their normal process of ascent (see Plate 2-5 ), the kidneys undergo 90 degrees of medial rotation, such that the renal pelvis is reoriented from its original ventral position to a final medial position. The mechanism of normal renal rotation is unknown but has been hypothesized to represent asymmetric branching of the ureteric bud in the metanephric mesenchyme. With an increased number of ventrolateral rather than dorsomedial ureteric bud branches, the metanephric mesenchyme would preferentially differentiate in a manner that could cause the appearance of rotation.
Renal malrotation is a rare phenomenon that may occur either in isolation or, more commonly, in combination with renal ectopia (see Plates 2-5 and 2-6 ). The true incidence is not well-characterized but, based on previous reports, is likely in the range of 1 : 500 to 1 : 1500 with an increased propensity among males.
It is unclear if renal malrotation represents abnormalities in the asymmetric branching process thought to underpin normal rotation, or whether it results from other factors. For example, it has been hypothesized that malrotation may occur if the ureteric bud inserts into an abnormal region of the metanephric mesenchyme. The association with renal ectopia suggests that certain processes may interrupt both normal ascent and rotation, or that ascent itself is important in some way for the normal process of rotation to occur.
In most cases of malrotation, the kidney fails to rotate at all, leaving the renal pelvis facing ventrally. Less frequently, the kidney may be only partially rotated, excessively rotated, or rotated in the wrong direction. Because the renal vessels are not believed to be responsible for malrotation, but rather twist around the kidneys as they rotate, their course offers a clue into the direction and degree of malrotation. For example, a kidney with a laterally directed renal pelvis may have undergone either 270 degrees of medial rotation or 90 degrees of lateral rotation. Likewise, a kidney with an ventrally directed renal pelvis may have undergone either no rotation at all or 365 degrees of rotation. In these cases, the path of the renal vessels allows one to make the distinction, as shown in the plate.
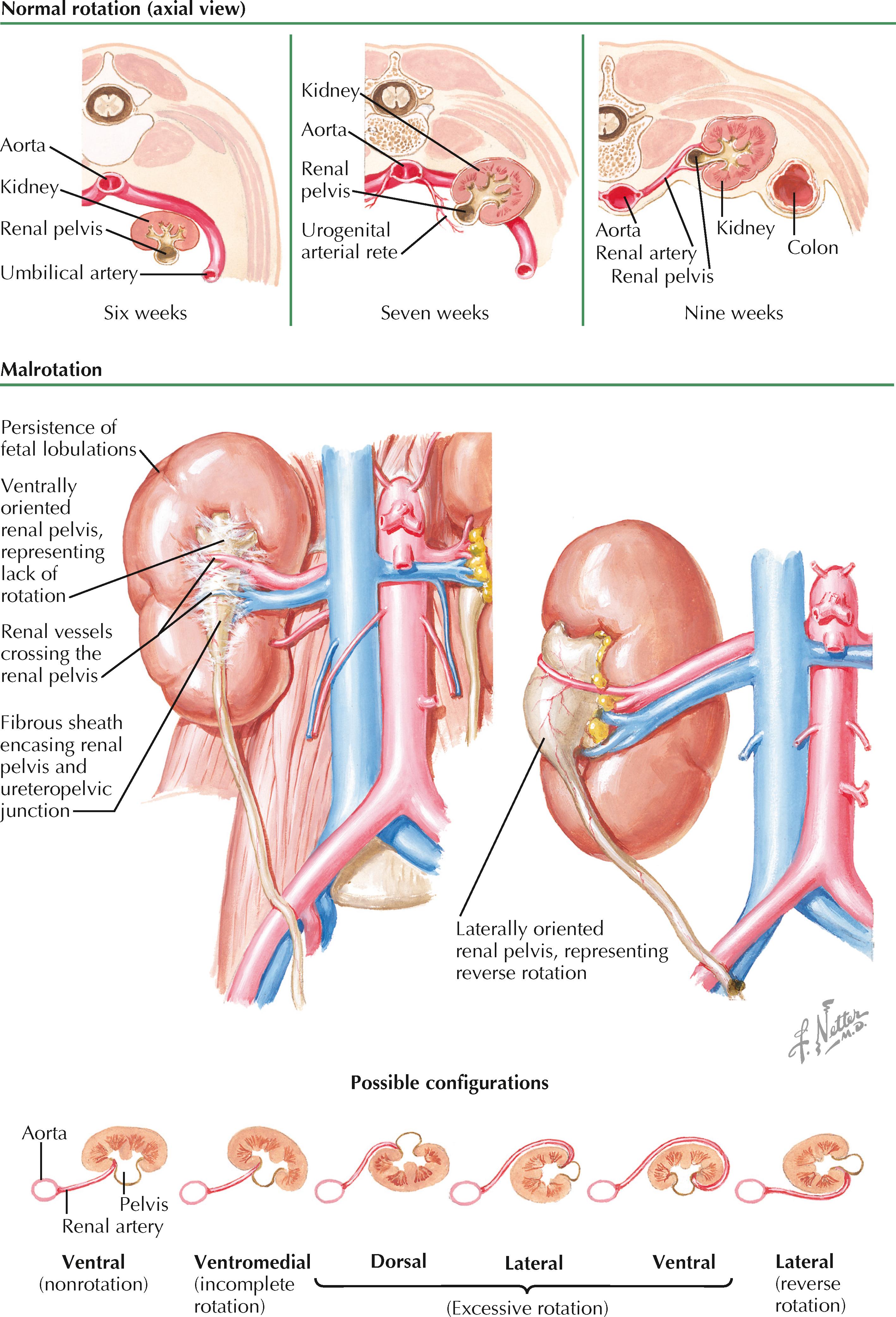
In addition to its association with ectopy, renal malrotation is usually associated with abnormalities in renal structure. For example, fetal lobulations are typically prominent over the gross surface. In addition, the renal pelvis is usually encased with an abnormally thick amount of fibrous tissue.
In most cases, malrotated kidneys cause no symptoms and are discovered only as incidental findings. In rare cases, however, patients may experience symptoms of upper tract obstruction, nephrolithiasis, or urinary tract infection. These occur if there is urinary stasis or outflow obstruction secondary to the fibrous encasing of the renal pelvis, a high insertion of the ureteropelvic junction, or obstruction of the renal pelvis by an overlying renal vessel. Such symptoms usually consist of nonspecific abdominal, flank, or back pain, and/or hematuria. The malrotation is then discovered on radiographic imaging of the abdomen. It is important to rule out the presence of a pelvic mass, which can rotate and displace the kidney from its normal position.
Most malrotated kidneys do not require definitive treatment. If significant symptoms persist, however, or if significant hydronephrosis is present, surgical repair of the renal pelvis and/or ureteropelvic junction may be necessary.
Renal agenesis is defined as the complete absence of one or both pairs of kidneys and ureters. It represents a failure of the ureteric bud and metanephric mesenchyme to engage in the process of reciprocal induction and differentiation required for metanephros formation. As a result, both the kidney and ureter fail to develop. Renal aplasia, in contrast, occurs when there is abnormal differentiation of the metanephric mesenchyme and ureteric bud that leads to involution of the kidney, but with persistence of a rudimentary collecting system.
Because numerous signaling molecules are involved in normal metanephros development, the range of genetic defects that may cause renal agenesis is vast and under active investigation. Recent evidence, however, suggests a prominent role for abnormalities in the GDNF-RET signaling cascade. GDNF (glial cell line-derived neurotrophic factor) is expressed in the masses of metanephric mesenchyme lying near the mesonephric (wolffian) ducts. It binds to RET, a receptor tyrosine kinase, in the mesonephric ducts and induces formation of the ureteric buds. Mutations in the genes encoding these proteins prevent formation of the ureteric buds and have been noted in fetuses with renal agenesis.
In some cases, male fetuses with renal agenesis still develop normal mesonephric duct derivatives (i.e., the vas deferens, seminal glands [vesicles], and epididymis), indicating that the underlying developmental defect affected only ureteric bud branching or metanephric induction. Fetuses with broader defects, in contrast, have likely sustained insults to the intermediate mesoderm, which gives rise to both the nephrogenic cords and genital ridge (see Plate 2-1 ).
Bilateral renal agenesis, defined as the complete absence of both kidneys and ureters, is a rare abnormality that occurs in approximately 1 : 5000 to 1 : 10,000 fetuses. Males are affected at least twice as often as females. There is evidence that a significant number of affected fetuses have abnormalities in signaling cascades important to renal development, such as the GDNF-RET pathway described above. A subset of affected fetuses, however, also have associated abnormalities that suggest a more wide-ranging defect in caudal development, such as sirenomelia (fused lower extremities, imperforate anus, renal agenesis, and abnormal or absent genitalia).
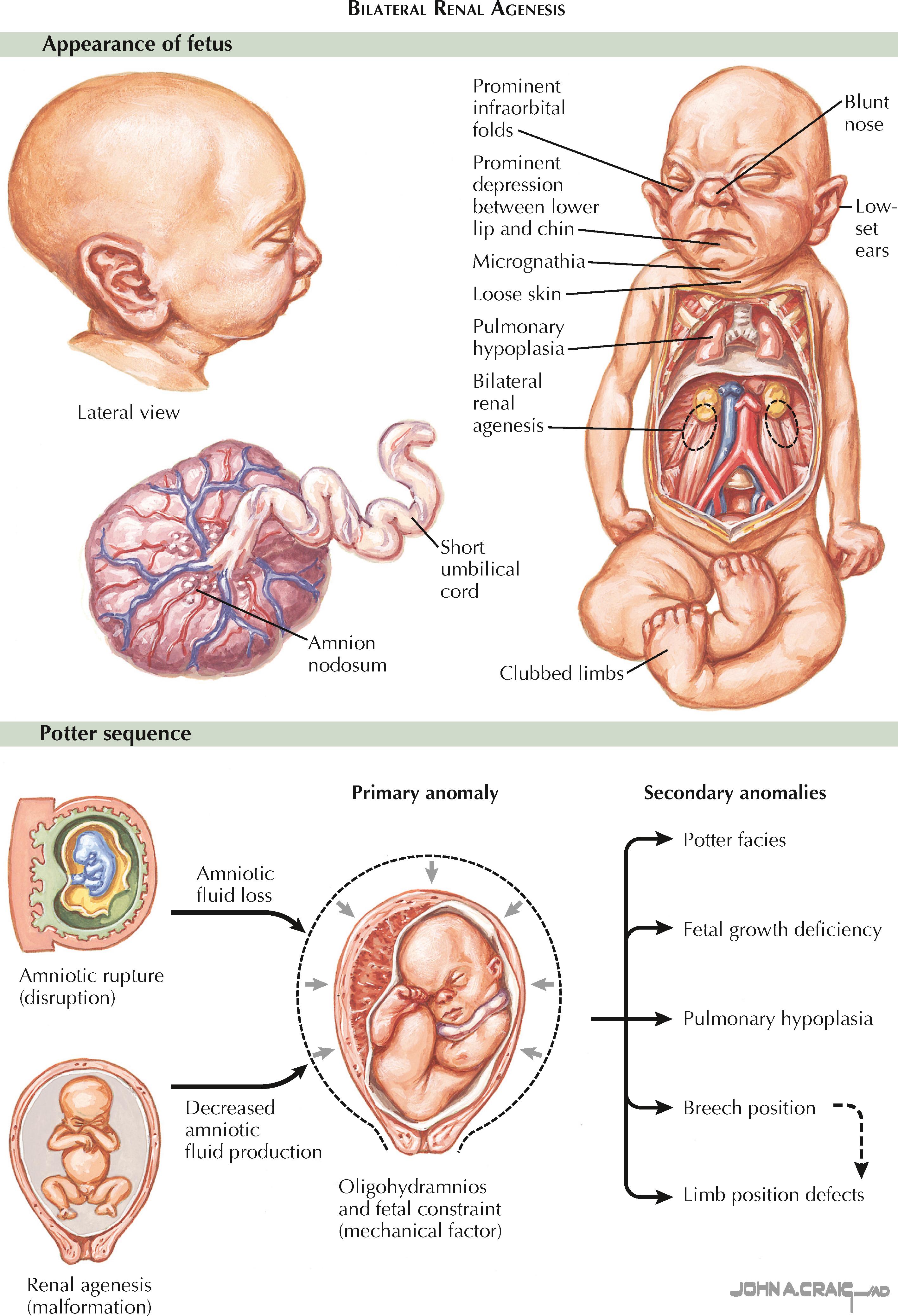
Bilateral renal agenesis is typically diagnosed using prenatal ultrasound. Normally the fetal kidneys can be visualized starting at approximately the twelfth week of gestation. In bilateral renal agenesis, however, no renal parenchyma can be visualized either in the normal renal fossae or other ectopic locations, such as the fetal pelvis or thorax. The fetal adrenal glands are in normal position but may appear less flattened because of the lack of normal compression from the kidneys. Finally, the fetal bladder appears empty, and the normal cycles of filling and emptying are not seen.
Severe oligohydramnios is a major consequence of bilateral renal agenesis and may be one of the more notable sonographic findings. Before 20 weeks of gestation, diffusion of fluid into the amnion produces a significant fraction of the amniotic fluid, which therefore may appear normal in volume even despite a lack of fetal renal function. After 20 weeks, however, the fetal kidneys are responsible for producing over 90% of the amniotic fluid. Severe oligohydramnios at this stage of development is therefore a very sensitive sign of bilateral agenesis. It is not, however, particularly specific, and other possible causes—including bilateral renal dysplasia, bilateral renal cystic disease, urinary outflow tract obstruction, premature rupture of membranes, and fetal demise—must be ruled out.
Severe oligohydramnios, irrespective of the cause, has numerous adverse effects on the fetus. First, the increase in intrauterine pressure results in physical compression of the growing fetus, which leads to a blunted nose; low-set, flattened ears that appear enlarged; micrognathia; prominent infraorbital folds; a prominent depression between the lower lip and chin; clubbed limbs; and dislocated hips. In addition, the lack of amniotic fluid causes abnormal development of the skin, which appears loose and excessively dry. Finally, the increased pressure on the fetal thorax and decline in circulating amniotic fluid leads to severe pulmonary hypoplasia. The cause-and-effect relationship between oligohydramnios and these various sequelae is known as the Potter sequence.
Bilateral renal agenesis is fatal. Forty percent of affected fetuses die in utero, and the remainder develop severe respiratory distress shortly after birth. Therefore, if the diagnosis is established using prenatal ultrasound, therapeutic abortion is typically recommended. Because most cases of bilateral renal agenesis are sporadic, the risk of recurrence in a subsequent pregnancy is low.
Unilateral renal agenesis, defined as the complete absence of one kidney and its associated ureter, occurs in approximately 1 : 1000 to 1 : 1200 individuals. Males are affected nearly twice as often as females. The underlying cause is thought to be abnormal interaction between a ureteric bud and its associated metanephric mesenchyme; however, it is unclear why some patients develop unilateral agenesis, whereas in others both sides are affected.
Like bilateral agenesis, unilateral renal agenesis is often discovered using prenatal ultrasound, which reveals an empty renal fossa without evidence of ectopic renal parenchyma, such as a pelvic kidney. Unlike in bilateral agenesis, however, urine production is normal, and therefore oligohydramnios does not occur. As a result, affected infants are typically born with a normal appearance and normal pulmonary function.
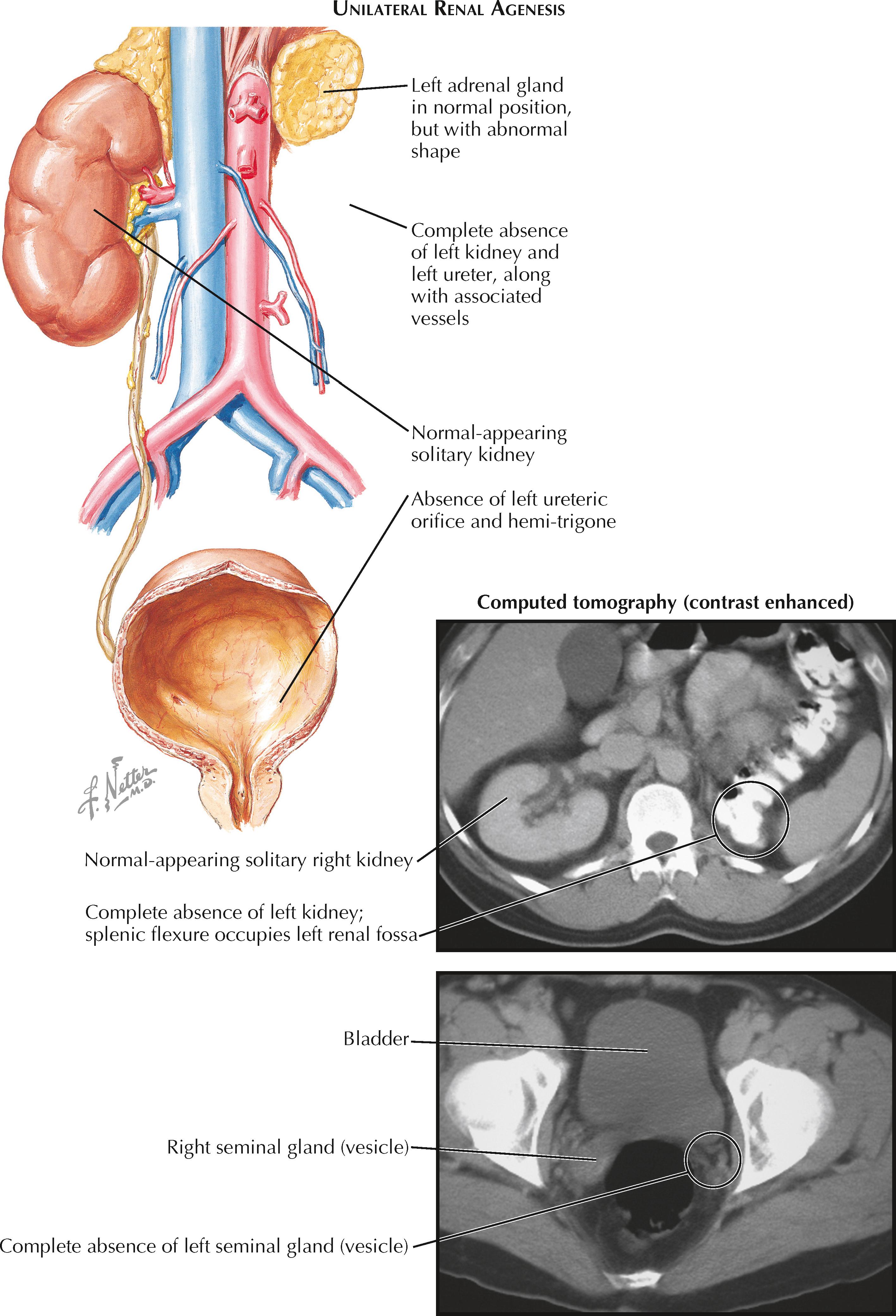
Many patients with unilateral renal agenesis have associated abnormalities in other organ systems. Indeed, the absence of a kidney may not even be discovered until an associated abnormality is investigated. Many of these abnormalities occur in structures derived from the mesonephric or paramesonephric ducts, suggesting a defect in the intermediate mesoderm early in development. In males, the ipsilateral mesonephric duct derivatives (vas deferens, seminal gland [vesicle], and epididymis) may be absent or rudimentary (i.e., a seminal gland [vesicle] cyst). Meanwhile, in females, a common associated anomaly is a unicornuate uterus, in which the side ipsilateral to the absent kidney is missing.
Associated abnormalities may also occur in the cardiovascular system (e.g., septal or valvular defects) or gastrointestinal systems (e.g., imperforate anus). Nearby musculoskeletal abnormalities may also be seen. In a smaller subset of patients, unilateral renal agenesis may be associated with a genetic syndrome that affects numerous organ systems, such as BOR (branchio-oto-renal) syndrome, Turner syndrome, Fanconi anemia, Kallmann syndrome, VACTERL (vertebral anomalies, anal atresia, cardiac defects, tracheoesophageal fistula, renal defects, limb defects), and others.
With a solitary kidney, renal function typically remains normal, although for unclear reasons there is an increased risk of vesicoureteral reflux, ureteropelvic junction obstruction, and ureterovesical junction obstruction. Later in life, some patients may develop renal insufficiency and proteinuria, likely secondary to hyperfiltration of the solitary kidney causing focal segmental glomerulosclerosis (see Plate 4-10 ). Their survival rate, however, appears to remain similar to that of normal individuals.
A supernumerary kidney is a very rare congenital anomaly. Unlike a kidney with a duplicated collecting system, which is far more common, a supernumerary kidney is a distinct mass of renal parenchyma with its own capsule, vessels, and collecting system. It is typically small and located just cephalad or caudal to the normally positioned kidney on the same side. Less commonly, it is located in a variety of other positions, such as the pelvis or midline. In some cases, the supernumerary kidney and normally positioned kidney may be loosely attached to each other by either fibrous tissue or a bridge of renal parenchyma.
In half of cases, the ureter associated with a supernumerary kidney fuses with that of the normally positioned ipsilateral kidney, as seen in the illustration; in the other half, the ureter has its own separate insertion into the bladder. In such cases, the Weigert-Meyer rule is usually obeyed, meaning that the ureter associated with the more caudally positioned kidney has an orifice located more superior and lateral than that of the cranially positioned kidney. The vessels to the supernumerary kidney usually originate from the aorta and inferior vena cava, although their origin is more variable with more caudally positioned kidneys.
The embryologic basis of the supernumerary kidney is not known but likely represents early division of the metanephric mesenchyme. A supernumerary kidney with a ureter that has its own insertion into the bladder likely reflects early division of the mesenchyme before insertion and branching of the ureteric bud (see Plate 2-2 ). The ureter to the supernumerary kidney probably represents a second ureteric bud that sprouted from the adjacent mesonephric duct, either by coincidence or as a direct effect of the divided mesenchyme. A supernumerary kidney with a ureter that fuses with that of the normal kidney likely reflects later division of the metanephric mesenchyme, perhaps in response to a ureteric bud that divided before insertion.
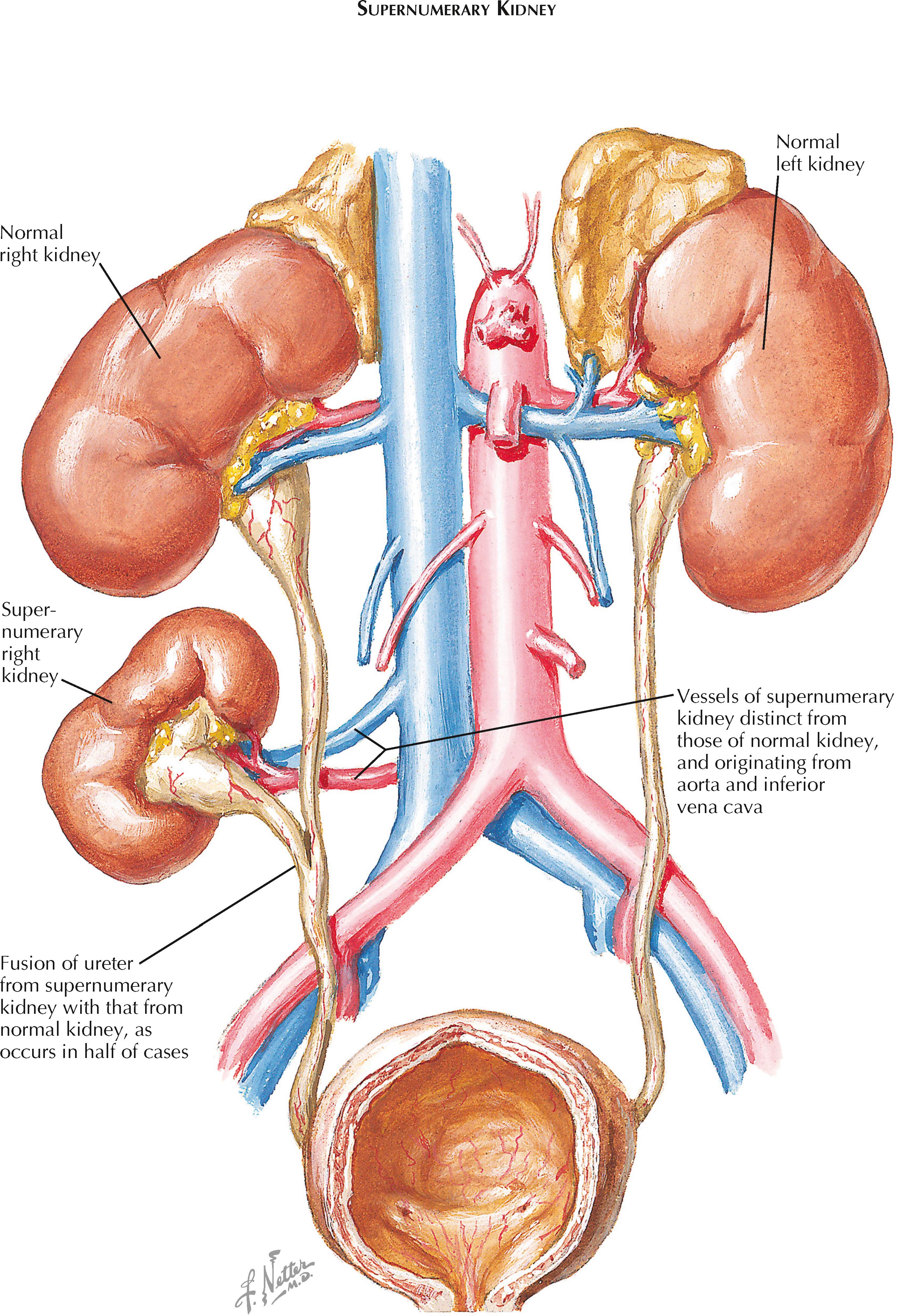
Supernumerary kidneys are often asymptomatic and do not affect overall renal function. Thus a significant number of such kidneys may never be discovered or may be noted only as incidental findings during the workup of another unrelated complaint. In some patients, however, supernumerary kidneys present as palpable abdominal masses or cause symptomatic nephrolithiasis or an upper urinary tract infection. Because of the rarity of this condition, affected patients are often not diagnosed until their fourth decade, if at all.
Development of the definitive adult kidney (metanephros) begins when the two ureteric buds invade the paired masses of metanephric mesenchyme (see Plate 2-2 ). Through a process known as branching morphogenesis, which depends on reciprocal signals between each ureteric bud and its associated mass of metanephric mesenchyme, the ureteric buds give rise to the ureters, renal pelves, calices, and collecting ducts, whereas the metanephric mesenchyme gives rise to nephrons.
Throughout this process, the two kidneys undergo separate but simultaneous development. As they undergo structural maturation, they also ascend in position (see Plate 2-5 ) from the sacral end of the fetus to the lumbar retroperitoneum.
Renal fusion can occur secondary to abnormalities in renal ascent, as in crossed renal ectopia (see Plate 2-6 ), or vice versa. In the former case, the superior pole of the crossed kidney ends up situated near the inferior pole of the normally positioned kidney, leading to fusion. In the latter case, a primary fusion event occurs, which then results in ectopia.
Horseshoe kidney, the most common type of renal fusion, occurs when an isthmus consisting of either fibrous tissue or functioning renal parenchyma connects the two kidneys in the midline. The overall incidence of this abnormality is estimated to about 1 : 600, with males affected twice as often as females. The horseshoe kidney is especially common in patients with chromosomal disorders, such as trisomy 18 and Turner syndrome.
The horseshoe kidney is thought to result from fusion of the two metanephroi during the sixth week of development, while they are still near one another at the sacral end of the fetus. It is believed that abnormal lateral flexion of the embryo may dislocate one kidney more medially, approximating it near the contralateral kidney and causing a fusion event. Ascent of the fused horseshoe kidney is prematurely terminated when the isthmus reaches the level of the inferior mesenteric artery, beyond which it is unable to cross.
The horseshoe kidney is typically situated in the lower lumbar region, below the normal position of the mature kidneys. The isthmus almost always connects the lower poles of the two fused kidneys, although in rare cases it may join the upper poles instead. The isthmus is usually situated anterior to the aorta and the inferior vena cava but may rarely be situated between these vessels or posterior to them both. Both renal pelves are usually oriented ventrally or ventromedially, secondary to a failure of rotation. The ureters insert normally into the bladder but are prone to reflux. In about 10% of patients, ureteral duplication is seen (see Plate 2-23 ). The renal vasculature is variable. The upper poles of each kidney are usually perfused by one or more ipsilateral branches of the aorta, whereas the lower poles and isthmus may receive their own branches from the aorta, iliac, or sacral arteries.
A horseshoe kidney rarely causes symptoms and is typically an incidental finding. A minority of patients, however, develop ureteropelvic junction obstructions, nephrolithiasis, or urinary tract infections. These complications may result from the abnormally high ureteropelvic junction or kinking of the ureters as they cross over the fused isthmus. In addition, some patients may experience traumatic injury to the isthmus due to its midline position anterior to the spine. A smaller subset of patients may present during childhood with Wilms tumor, as horseshoe kidney increases the risk.
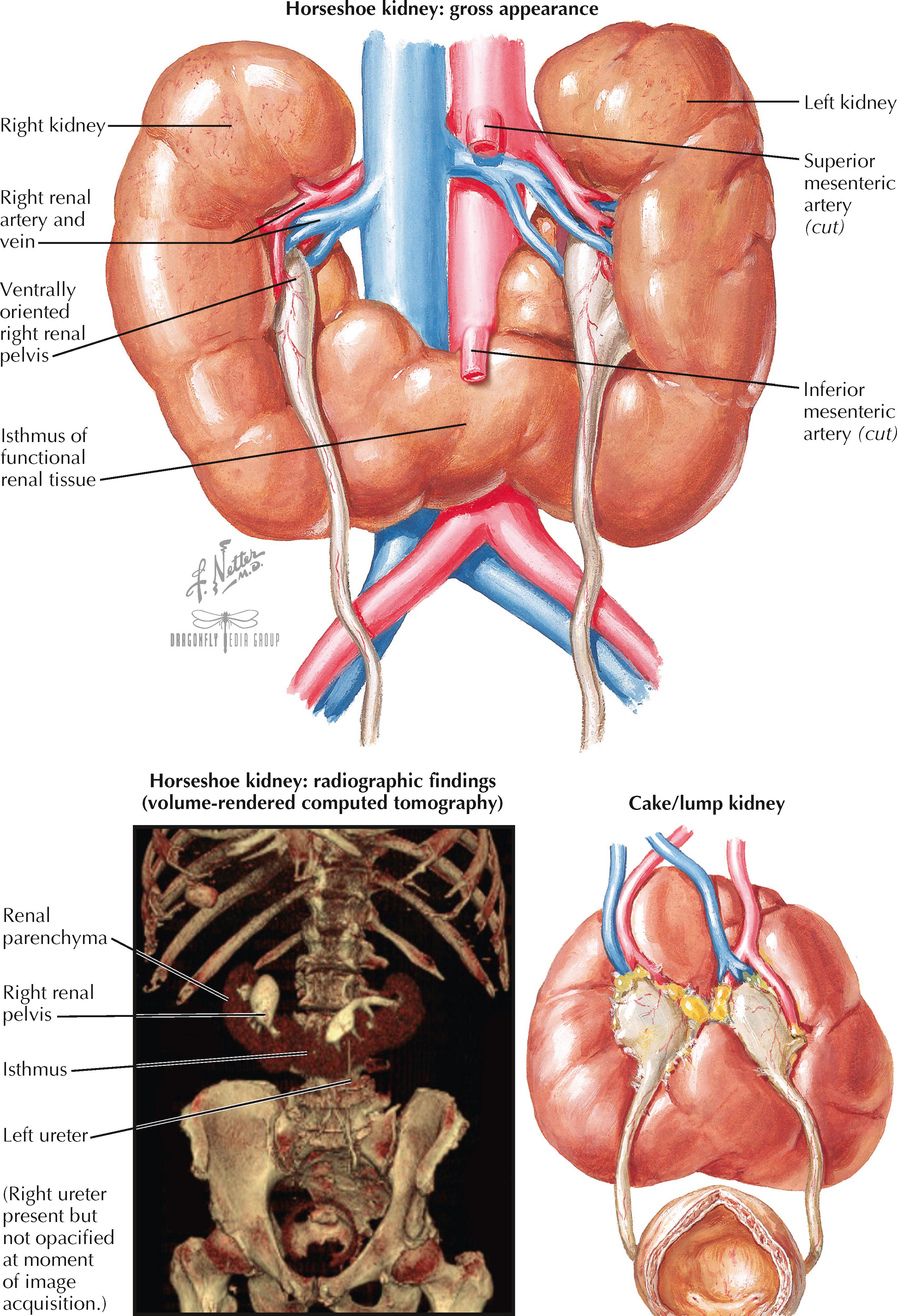
A small subset of patients with horseshoe kidney have concomitant abnormalities in other organ systems. Associated genital abnormalities include hypospadias and undescended testes in males, or vaginal septation and bicornuate uterus in females. Other associated abnormalities include neural tube defects and cardiac ventriculoseptal defects.
The “lump” or “cake” kidney is a renal fusion variant in which there is complete merging of the two kidneys, such that two separate masses are no longer distinguishable. This anomaly reflects a very early, complete fusion of the metanephroi. The symptoms, risks, and treatment options are largely the same as for horseshoe kidney.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here