Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
In addition to cardiac pacemakers and defibrillators, a number of noncardiac devices have been developed for electronic neuromodulation and drug delivery. Although these devices are placed by a variety of subspecialists for the treatment of chronic illnesses, if the devices malfunction, patients may arrive at the emergency department in an acute state, thereby necessitating intervention by the emergency provider.
External insulin infusion devices have become increasingly popular since their introduction in 1974. By 2007, there were more than 375,000 external insulin infusion pumps in use in the United States.
An external insulin infusion pump device consists of a portable, programmable infusion pump connected to a subcutaneously implanted catheter that is maintained in place with adhesive tape ( Fig. 70.1 ). The implanted catheter site varies, but it is commonly placed in the subcutaneous tissue of the abdomen in adults and the buttocks in young children. The thighs, hips, and upper part of the arms are other sites. It is recommended that the implanted catheter be replaced every 2 to 3 days.
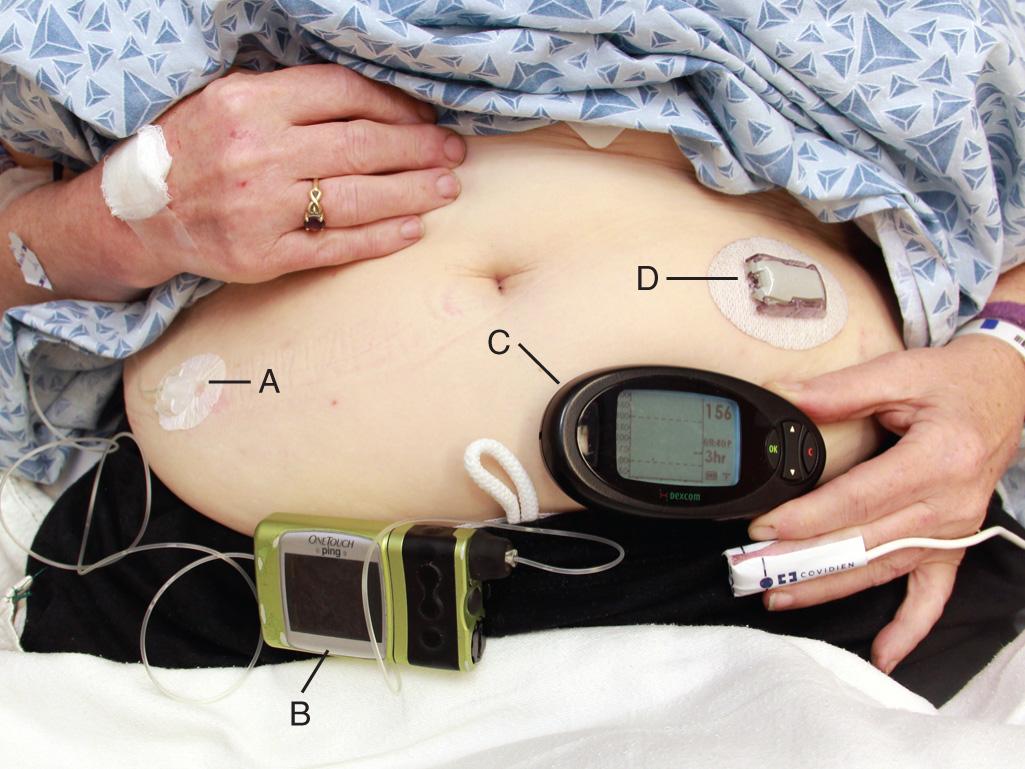
In 2010, the Food and Drug Administration (FDA) published a panel report that highlighted problems associated with insulin infusion devices. From the top five device manufacturers, the FDA noted 16,640 adverse events, including 310 deaths, 12,093 injuries, and 4294 malfunctions. Box 70.1 lists the most frequently reported problems with devices in descending frequency. Box 70.2 lists the most frequently reported patient-oriented adverse reactions, which include hyperglycemia and hospitalization. Similar findings were reported in a 2014 prospective study, in which the most clinically significant adverse events related to insulin infusion devices were hyperglycemia and ketosis.
Unknown (19.7%)
Replace (9%)
Audible alarm (6%)
Use-of-device issue (5%)
Device displays error message (4.8%)
Not applicable (4%)
Failure to deliver (3%)
No information (3%)
Repair (3%)
Self-activation or keying (1.8%)
Hyperglycemia (24.6%)
Hospitalization (21%)
Diabetic ketoacidosis (8%)
Treatment with medication (6%)
Blood glucose low (4.7%)
Therapy, nonsurgical management (4%)
No consequences to the patient (4%)
Unknown (3%)
Of the 310 deaths reportedly related to insulin infusion pumps in the FDA report, the vast majority of problems with the device were not known to the patient or providers at the time, and the root cause of failure of the device was not identified by the manufacturer. In 29 deaths, problems with the device that were identified included overinfusion, bent cannulas, disconnection, pump alarming, failure to deliver, suspected electromagnetic interference, and display failure.
If the device appears to be functioning normally, the American Association of Clinic Endocrinologists recommends that, should the patient be unable to manage his/ her own pump while in the emergency department (ED), “the specialist(s) responsible for the patient's ambulatory pump management should be contacted promptly to make decisions about infusion adjustments.” Moreover, device malfunction should be considered in patients with insulin infusion devices who present to the ED with symptoms that may be related to blood glucose. In addition to medical management, troubleshoot the device and remove it in cases of uncertainty or emergency.
To remove the catheter, simply peel off the adhesive and embedded catheter together to discontinue the flow of injected medication into the patient. There are a variety of proprietary pump manufacturers, each with their own device programming. Call the appropriate manufacturer to troubleshoot the device, visit the manufacturer's website for an online troubleshooting manual, or simply instruct the patient to discontinue use of the device and return to a standard calculated insulin schedule by manual injection until the device-related complication is investigated and resolved.
Intrathecal (spinal canal/subarachnoid space) drug delivery systems (IDDSs) have been in clinical use since the 1980s. The FDA approved the use of intrathecal baclofen in 1992 for severe spasticity secondary to spinal cord injury, multiple sclerosis, cerebral palsy, or stroke. Subsequently, the FDA approved the use of intrathecal morphine in 1995 and intrathecal ziconotide in 2004 for chronic pain refractory to traditional medical therapies. Ziconotide is a non-opioid calcium channel blocker, but its use may be complicated by confusion, somnolence, and other neurologic side effects. Although limited data demonstrate efficacy of intrathecal morphine and ziconotide in relieving refractory pain and efficacy of intrathecal baclofen in reducing spinal cord injury-induced spasticity, more quality clinical evidence is needed. IDDS devices are also used for the delivery of chemotherapeutic medications for specific oncologic conditions. Moreover, there are several off-label uses of various intrathecal medications. Pumps are often used to deliver analgesics to patients with intolerable side effects from oral or parenteral analgesics, such as nausea, pruritis, and cognitive dysfunction. Other medications used include bupivacaine, hydromorphone, fentanyl, sufentanil, clonidine, midazolam, and meperidine. Some patients will receive a combination of medications. There is minimal data comparing various medications and current regimens have been empirically derived.
The advantage of IDDSs for chronic pain is the ability to get medication directly to receptors in the spinal cord by placing the medication into the subarachnoid space. For intrathecal morphine infusions, approximately 1% of the total daily morphine dose is a standard starting point. Such small intrathecal doses reduce systemic concentrations and minimize side effects. Patients in chronic pain may also use oral analgesics on a rescue basis.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here