Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Chronic pain is a complex, endless disturbance with unique qualities and psychiatric comorbidities. Traditional medical approaches to chronic pain emphasized diagnosis, distinguished between its psychologic and physiologic components, and sought its eradication. Currently, multidisciplinary pain teams focus on symptomatic management, reducing pain’s affective component (suffering), and restoring function. These teams see their role as pain management without holding out the expectation that they will eliminate pain.
As a member of a team or an individual treating physician, psychiatrists should be aware of common chronic pain syndromes and their underlying neuroanatomy, psychiatric comorbidity, and treatments involving long-term use of opioids (narcotics) and adjuvant medications (primarily nonanalgesic drugs, such as antidepressants and antiepileptics [AEDs]).
Nociceptive pain results from acute, ongoing tissue damage, such as trauma, metastasis to bone, dental infection, or disease of the viscera. It usually consists of dull aching pain at the site of the tissue damage. Diseased viscera, however, may also refer pain to another region, such as when gallbladder stones seem to produce pain in the scapula. Wherever the location, tissue damage triggers specific receptors (nociceptors) in the peripheral nervous system (PNS) and certain cranial nerves. Both peripheral and cranial nerves transmit the noxious stimuli to the central nervous system (CNS). Removing diseased tissue and other direct treatments reduce or eliminate nociceptive pain. Until the injury heals, analgesics reduce the pain.
Neuropathic pain, in contrast, results from pain signals transmitted by the nervous system, often without detectable tissue injury. It is a symptom of a wide variety of CNS and PNS disorders, including cranial neuropathies, mononeuropathy or polyneuropathy, brachial or lumbar plexus injury, lumbar spine disorders, complex regional pain syndrome, and thalamic injury.
Unlike the dull ache of nociceptive pain, neuropathic pain comprises electric, sharp, lancinating, or burning sensations. Also, in neuropathic pain, spontaneously occurring, painful paresthesias radiate not only throughout the distribution of an injured nerve or area, but also often well beyond. Another characteristic is that painful or even neutral stimuli elicit an intense, distorted, or exaggerated response— hyperalgesia or allodynia (see Chapter 5 ).
Physicians can rarely abolish neuropathic pain because they cannot remove or repair the injured nerves or other damaged tissue. Another reason is that nerve injury may eventually reorganize CNS pain perception through plasticity , the capacity of the CNS to reorganize its functions. Although plasticity is usually beneficial, in neuropathic pain it amplifies, distorts, and perpetuates pain. Whatever its source, neuropathic pain is common, chronic, disabling, and usually unrelenting. Moreover, it carries serious psychiatric comorbidity.
The Diagnostic and Statistical Manual of Mental Disorders, 5th Edition (DSM-5) categorizes pain somewhat differently. The former DSM diagnosis of Pain Disorder has now been subsumed into Somatic Symptom Disorder. In this diagnosis, pain represents the major focus of the clinical presentation and causes distress or functional impairment. It allows psychological factors to have an important role in the pain’s onset, severity, or maintenance. The diagnosis excludes pain better explained by a mood disorder, anxiety, or psychosis. The symptomatic state must last at least six months. It considers pain resulting from a neurologic or medical condition, such as headaches, peripheral neuropathy, or low back pain, to be a component of those conditions.
The DSM-5 allows the diagnosis of malingering when individuals knowingly falsely claim severe and prolonged symptoms, including pain. Incentives to malingering include financial expectations, freedom from work assignments, and seeking attention or retribution. Even though the proportion of all patients with chronic pain who are malingering may be less than 1%, their total number is quite large.
Probably no neurologic disorder has a greater psychiatric component than chronic pain. Pain management centers report as many as 50% of patients with chronic pain have dual diagnoses—particularly depression, but also somatoform and personality disorders, substance abuse, and posttraumatic stress disorders. Moreover, chronic pain is associated with drug and alcohol dependency, dysfunctional family relationships, and exaggeration of physical deficits.
The closest association exists between depression and chronic pain, which is almost invariably neuropathic. When present, depression lowers the threshold for pain, makes it refractory to treatment, and increases disability. In many patients, the causal relationship between pain and depression is unclear, but probably reciprocal. For example, painful injuries lead to depression, but certainly pre-existing depression, unhappiness at work, resentments, and other psychologic factors lead to chronic pain following injuries. With respect to chronic pain, major depression is more closely associated with the number of painful sites or painful conditions than the severity or duration of pain.
Physicians often refer patients with chronic pain for evaluation of depression, anxiety, posttraumatic stress disorder, or drug or alcohol abuse. Instead of waiting to be consulted as a last resort, psychiatrists should urge their colleagues to solicit consultations for patients (1) who have vegetative symptoms, regardless of the apparent connection to the pain, (2) whose pain or disability is refractory to medical treatment, (3) who seem to require excessive medication, (4) for psychopharmacology consultation, (5) for whom inpatient treatment or surgical procedures have been unsuccessful, or (6) who may have psychopathology that complicates their management.
Psychiatrists will, of course, be mindful of a patient’s pre-existing psychiatric disorder where pain is likely to be a symptom—especially depression, Somatic Symptom Disorder, Substance Use Disorders, or Personality Disorders. They should also appreciate that patients with dementia have diminished capacity to report pain and its characteristics. Moreover, pain and the medicines physicians use to treat it may impair patients’ ability to maintain a clear sensorium and to cooperate with evaluations.
When possible, psychiatrists should examine the physical location of a patient’s pain. An examination involving physical touch of the painful area, where appropriate, probably has therapeutic benefit, albeit the primitive one of “laying on of hands.” Also, in evaluating pain-related functional disability, psychiatrists should watch the patient sit, walk, and, if possible, use the affected part of the body. Many assessment scales have been developed for evaluation of pain, including evaluation of young children and patients with advanced dementia.
Ascending pathways rapidly bring information from the periphery to the brain. They identify a pain’s nature and location, arouse central mechanisms, and activate the limbic system. Analgesic pathways, originating in the brain and descending in the spinal cord, modulate pain perception. This neuroanatomy for pain and analgesia serves as the basis of many treatment strategies.
Painful conditions, like contusions and menstrual cramps, liberate inflammatory mediators including prostaglandins, arachidonic acid, and bradykinin that stimulate nociceptors. These mediators initiate pain transmission by triggering voltage-gated sodium channels in the PNS and CNS. Aspirin and non-steroidal anti-inflammatory agents (NSAIDs) block cyclooxygenase (COX), an enzyme crucial in the synthesis of prostaglandins, thereby reducing tissue inflammation and alleviating pain.Lidocaine, several AEDs, and tricyclic antidepressants (TCAs) also block these channels and reduce pain.
The PNS fibers enter the CNS at the spinal cord’s dorsal horn and, either immediately or after ascending a few segments, synapse in its substantia gelatinosa ( Fig. 14.1 ). At many of these synapses, the fibers release an 11-amino acid polypeptide, substance P , which constitutes the major neurotransmitter for pain at the spinal cord level.
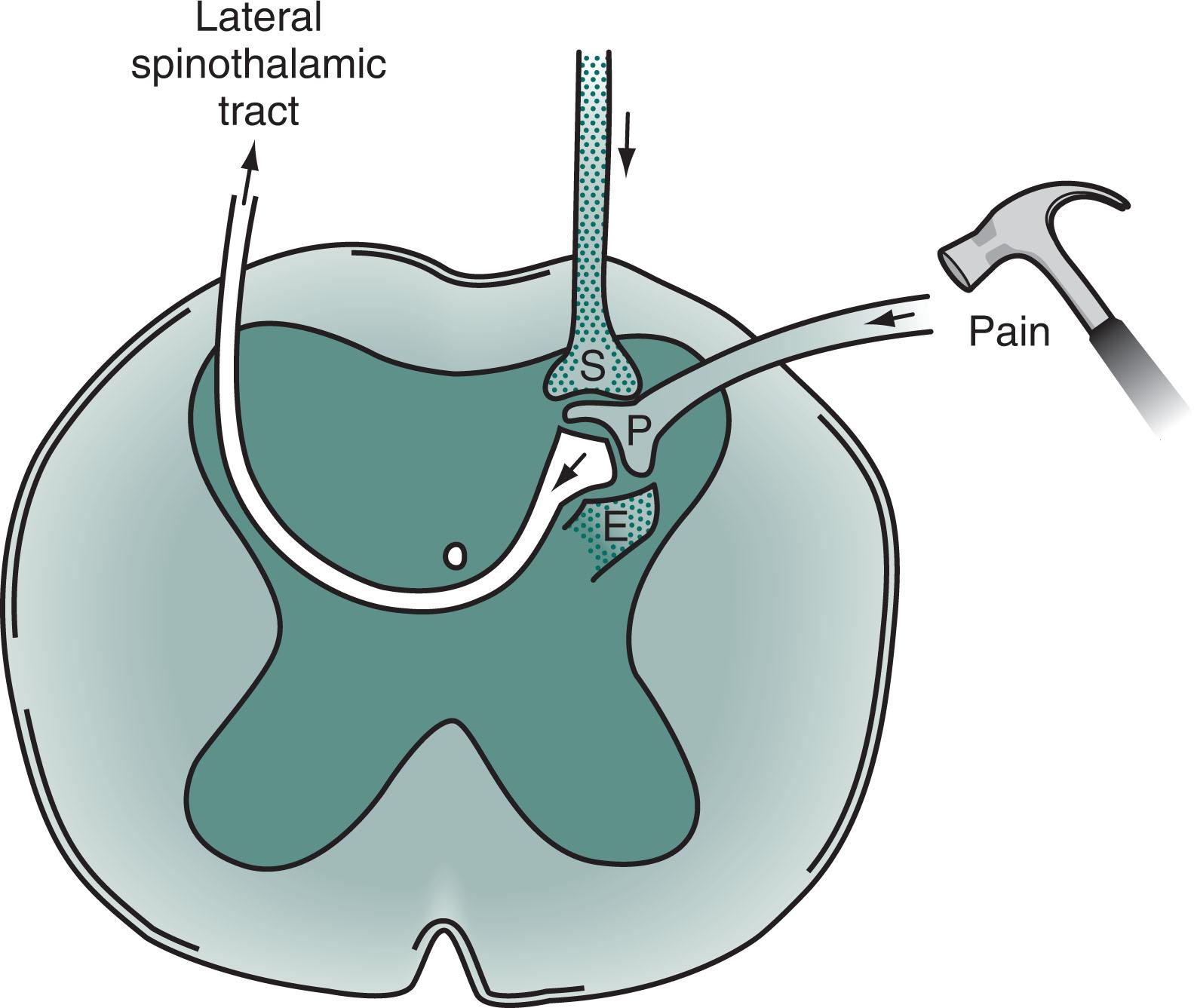
After the synapse, pain sensation ascends predominantly within the lateral spinothalamic tract to the brain (see Figs. 2.6 and 2.15 ). This crucial tract crosses from the substantia gelatinosa to the spinal cord’s other side and ascends contralateral to the injury to terminate in specific nuclei of the thalamus. Additional synapses relay the stimuli to the somatosensory cerebral cortex, enabling the individual to locate the pain.
In the spinohypothalamic tract , another ascending pain pathway, ipsilateral and contralateral fibers travel up the spinal cord and terminate directly in the hypothalamus. This pathway may explain pain-induced disturbances in temperature regulation, sleep, and other autonomic functions. The spinal cord also transmits pain in other, less well-defined ipsilateral and contralateral tracts.
In addition to relaying pain to the thalamus and hypothalamus, these tracts convey pain to the limbic system, reticular activating system, and other brainstem regions. These connections partially explain why individuals awaken when given a painful stimulus during sleep. It also accounts for chronic pain patients’ sleeplessness, loss of appetite, and a tendency to develop anxiety and mood disturbances. As a corollary, loss of this connection—from trauma, anoxia, or other insult—explains why patients who are unconscious cannot experience pain or suffer.
Many analgesic pathways interfere with pain transmission within the brain or spinal cord. Several pathways that originate in the frontal lobe and hypothalamus terminate in the gray matter surrounding the third ventricle and aqueduct of Sylvius ( periaqueductal gray matter ). They contain large amounts of endogenous opioids , which are powerful analgesics ( Box 14.1 ). Implanted electrodes that stimulate the periaqueductal gray matter area may provoke the release of endogenous opioids and thereby induce profound analgesia.
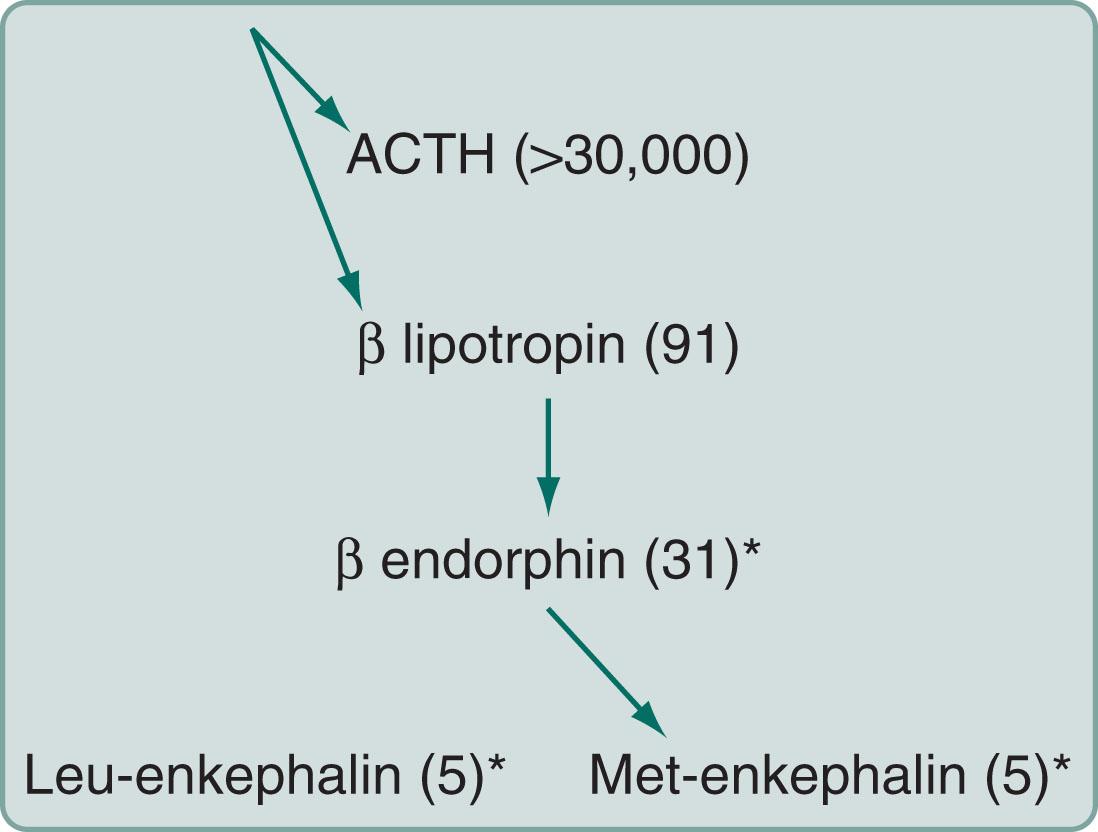
β-endorphin: An endogenous opioid concentrated in the pituitary gland and secreted with adrenocorticotropin (ACTH). It consists of amino acid numbers 61–91 of β-lipotropin and gives rise to the enkephalins (see Fig. 14.2 ).
β-lipotropin : A 91-amino-acid polypeptide, which may be an ACTH fragment. It gives rise to β-endorphin but has no opioid activity itself (that is, β-lipotropin is not an endogenous opioid).
Dynorphin : An endogenous peptide opioid that binds to kappa opioid receptors.
Endogenous opioids : Polypeptides (amino acid chains) found within the CNS that create effects like those of morphine and other opioids. The effects of both endogenous and exogenous opioids are characteristically reversed by naloxone.
Endorphins : Endogenous morphine-like substances or opioid peptides. This term is virtually synonymous with endogenous opioids.
Enkephalins : Short (5-amino-acid) polypeptide endogenous opioids that include met-enkephalin and leu-enkephalin. They are found primarily in the amygdala, brainstem, and dorsal horn of the spinal cord.
Naloxone (Narcan): A pure opioid antagonist that reverses the effects of endogenous and exogenous opioids.
Substance P : An 11-amino-acid polypeptide that is probably the primary pain neurotransmitter at the first synapse of the primary afferent neuron in the spinal cord.
Similarly, short neurons located entirely within the spinal cord, interneurons , inhibit incoming PNS stimuli. These neurons release endogenous opioids and other neurotransmitters that reduce pain transmission.
Analgesic pathways also originate in the brainstem and descend in the spinal cord’s dorsolateral funiculus . They provide “descending analgesia” relief of pain by inhibiting both spinal cord synapses and their ascending pathways. Unlike most other analgesic pathways, they release serotonin .
Often called endorphins ( endogenous m orphine -like substances), endogenous opioids—endorphins, enkephalins, and dynorphins—are powerful analgesics composed of amino acid chains (polypeptides) and synthesized in the CNS ( Fig. 14.2 ). Endogenous opioids bind to receptors in the limbic system, periaqueductal gray matter, dorsal horn of the spinal cord, and other CNS sites. The “runner’s high” and the initial painlessness reported by wounded soldiers serve as examples of endorphins’ analgesic effects.
Synthetic (exogenous) opioids, particularly morphine and other medicines, are virtually identical to endogenous opioids. They bind to the same CNS receptors and produce the same effects—analgesia, mood elevation (euphoria), sedation, and respiratory depression. Naloxone (Narcan), an antagonist that competitively binds to the opiate receptor, reverses the effects of endogenous, as well as exogenous, opioids. Indeed, naloxone’s opioid antagonist effect is so characteristic that naloxone-reversibility serves as a criterion for ascertaining that opioid pathways mediate an analgesic’s effect.
Numerous analgesics, which can be administered through various routes, reduce tissue damage, interrupt pain transmission through peripheral or central pathways, or blunt pain’s impact on cerebral structure or function. The addition of psychologic treatment and physical therapy may further reduce pain and, as part of a complete care plan, decrease suffering, restore activities of daily living, and return control to patients. Because chronic pain remains notoriously refractory to conventional treatment, neurologists aim for pain management, not pain cure.
Aspirin, other salicylates, NSAIDs, steroids, and acetaminophen—non-opioid analgesics—inhibit prostaglandin synthesis at the injury ( Box 14.2 ). Through this mechanism, these medicines relieve acute and chronic pain of mild to moderate severity.
Acetaminophen (Tylenol and others)
Aspirin
Choline magnesium trisalicylate (Trilisate)
Diflunisal (Dolobid)
Nonsteroidal anti-inflammatory agents
Ibuprofen (Motrin, Advil)
Indomethacin (Indocin)
Ketorolac (Toradol)
Naproxen (Naprosyn)
Opioids
Non-opioid analgesics generally provide steady analgesia for weeks to months and avoid several potential problems. In particular, after completing a course of treatment, patients do not experience withdrawal symptoms. Also, except for the potential for high-dose steroids to cause steroid psychosis (see Chapter 15 ), these analgesics do not induce mood, cognitive, or thought disorders.
On the other hand, large doses of NSAIDs and aspirin can cause gastric irritation or hemorrhage, and prolonged use increases the risk of cardiovascular disease. In addition, although non-opioid analgesics provide dose-dependent pain relief, they do so only up to a point. Once these medicines provide their maximum pain relief, greater doses do not increase their benefit (the “ceiling effect”).
Non-opioid analgesics are more effective if patients take them on a prophylactic basis. For example, taking non-opioid analgesics prior to dental procedures or menses will avert much of the pain. They are also more effective if taken in a generous initial “loading” dose.
Although non-opioid analgesics alone offer minimal benefit in cases of neuropathic pain, they act synergistically when taken in combination with opioids. In other words, peripherally acting non-opioid analgesics enhance centrally acting opioid analgesics. For example, adding NSAIDs to morphine helps alleviate the pain of metastases to bone. Because non-opioid analgesics allow a smaller dose of opioids to be effective, they have an “opioid-sparing effect.”
With routine use, acetaminophen by itself or as an en-hancement to opioids provides effective, reasonably safe analgesia. Stern warnings about acetaminophen compounds, such as oxycodone-acetaminophen (Percocet) and hydrocodone-acetaminophen (Vicodin), as well as acetaminophen itself, causing liver damage are justifiable. Thus, individuals with alcoholism or liver disease, and those who might use it to commit suicide, should avoid acetaminophen treatment.
When chronic pain results from cancer, neurologists categorize it as “cancer pain” or “malignant pain,” but when it results from other conditions, it is categorized as “noncancer” pain. Opioids are unquestionably indicated for cancer pain (see Box 14.2 ). In addition, some studies suggest they are indicated, but with caution, for chronic noncancer pain syndromes.
Opioids have no ceiling effect. Greater doses or more potent preparations increase their analgesic effect. Adding NSAIDs or other non-opioid analgesics enhances opioids’ effect without risking their side effects. Conversely, when treating patients with noncancer pain who do not find relief with opioid treatment, physicians should slowly discontinue opioids rather than increasing the dosage.
Chronic pain patients may obtain opioid treatment from pills, transdermal patches, intranasal sprays, rectal suppositories, and epidural injections. An innovative technique, patient-controlled analgesia (PCA) , allows patients to regulate continual or intermittent intravenous opioid infusions. Through controls in the system, patients regulate the depth of analgesia without risking respiratory arrest. Even 6-year-old children can safely and effectively administer PCA.
Compared to older opioids, newer ones provide more rapid onset and longer duration of action. Some are long-acting because they are embedded in a matrix that slowly releases its medication.
Administering opioids by PCA, patches, or long-acting oral preparations on a regular prophylactic or time-dependent basis, such as every 2 to 4 hours, is more effective than administering them only at the onset of pain. Administering opioids only after pain has developed makes it more difficult to control, creates a pattern of “hills and valleys” (undertreatment alternating with overtreatment), prevents restful sleep, and increases side effects. Moreover, because patients, fearful about pain recurrence, develop anxiety and preoccupation with obtaining their medicines, they may seem to behave as addicts (see later).
Physicians who prescribe narcotics should select long-acting preparations, such as methadone or a transdermal opioid. Also, because various opioids affect different regions of the mu (μ) and related opioid receptors, physicians should vary the opioid for patients with intractable pain. However, due to the potential accumulation of large doses, transdermal and extended release oral opioid formulations carry a disproportionately greater risk of morbidity and mortality.
Physicians should also avoid changing from an oral to parenteral form of opioid at a given dose because the substitution will likely lead to an overdose. In the reverse situation, changing the same dose from an intramuscular or intravenous injection to pills is likely to produce under-treatment, which can cause withdrawal symptoms and recurrence of pain.
In an attempt to control indiscriminate prescription of opioids, the Food and Drug Administration (FDA) launched Risk Evaluation and Mitigation Strategies (REMS) for physicians who prescribe opioids. REMS will hopefully reduce inappropriate prescriptions, abuse, overdose, and other untoward effects. Additionally, physicians are accountable in many states with a requirement to check an electronic database of a patient’s controlled substance prescription history prior to writing a new prescription.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here