Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Primary lung cancer is the leading cause of cancer-related mortality worldwide and accounts for more deaths than the next three malignancies combined—pancreatic, colorectal, and pancreatic cancer in men; breast, colorectal, and pancreatic cancer in women. In 2017, it was estimated that 222,500 new cases would be diagnosed, with 155,870 deaths expected from the disease. Lung cancers are generally divided into two types, non–small cell lung carcinoma (NSCLC), which accounts for 85% of all lung cancers, and small cell lung carcinoma (SCLC), which represents 15% of all lung cancers. NSCLC primarily includes adenocarcinoma and squamous cell carcinoma, as well as less common tumors such as large cell carcinoma, sarcomatoid carcinoma, and spindle cell sarcoma. SCLC is an aggressive malignancy and the most common primary pulmonary neuroendocrine neoplasm.
Diagnostic imaging plays a key role in the identification, characterization, and staging of lung cancer. Suspicious pulmonary lesions may be initially identified on chest radiography because the modality is widely available and relatively inexpensive. However, there are significant limitations of radiography, including the inability to detect small nodules and other abnormalities, poor delineation of primary tumor and nodal status, and the inability to evaluate for extrathoracic disease. It has been reported that approximately 12% to 30% of lung cancers are missed on chest radiography. Multidetector computed tomography (MDCT) is the primary imaging modality used to evaluate pulmonary nodules and masses, characterize primary tumors, and detect intrathoracic and extrathoracic lymphadenopathy and metastatic disease. Limitations of MDCT include inferiority to MRI in the visualization of mediastinal and chest wall invasion and inferiority to fluorodeoxyglucose (FDG)–positron emission tomography (PET)/CT in the evaluation of nodal and metastatic disease. MRI is not routinely used in the evaluation of lung cancer, but is complementary to conventional imaging techniques. Advantages of MRI include the ability to evaluate the status of the intrathoracic vasculature when there are contraindications to contrast-enhanced MDCT (e.g., renal failure, severe contrast allergy) and improved evaluation of the mediastinum and chest wall compared to MDCT. Limitations of MRI include inherent problems with imaging the chest, although improved MR techniques have resulted in decreased scan times and diminished respiratory and cardiac motion. PET/CT has demonstrated an improved ability to assess for intrathoracic and extrathoracic lymphadenopathy and metastatic disease, assess response to therapy, and detect residual and/or recurrent disease. Important restrictions of PET/CT include limited characterization of the primary tumor, limited evaluation of mediastinal and chest wall invasion, and false-positives and false-negatives, which can result in misinterpretation.
Adenocarcinoma is the most common subtype of NSCLC and represents a spectrum of malignant epithelial neoplasms characterized by glandular differentiation or mucus production. It is more common in women than in men and is associated with smoking in 46% of cases. Adenocarcinoma includes a wide variety of patterns and differentiation. An updated classification system was introduced in 2011 by a joint working group of the International Association for the Study of Lung Cancer (IASLC), American Thoracic Society (ATS), and European Respiratory Society (ERS) emphasizing the correlation among pathology, imaging, and clinical topics such as diagnosis, treatment, and prognosis ( Box 17.1 ). This new system eliminated the term bronchioloalveolar carcinoma (BAC) and grouped lesions into the following categories: (1) preinvasive lesions; (2) atypical adenomatous hyperplasia; (3) adenocarcinoma in situ; (4) minimally invasive adenocarcinoma; (5) invasive adenocarcinoma; (6) invasive adenocarcinoma; and (7) variants of invasive adenocarcinoma.
Preinvasive lesions
Atypical adenomatous hyperplasia
Adenocarcinoma in situ
Nonmucinous
Mucinous
Mixed nonmucinous/mucinous
Minimally invasive adenocarcinoma
Nonmucinous
Mucinous
Mixed nonmucinous/mucinous
Invasive adenocarcinoma
Lepidic predominant
Acinar predominant
Papillary predominant
Micropapillary predominant
Solid predominant with mucin production
Variants of invasive adenocarcinoma
Invasive mucinous adenocarcinoma
Colloid
Fetal
Enteric
ATS, American Thoracic Society; ERS, European Respiratory Society; IASLC, International Association for the Study of Lung Cancer.
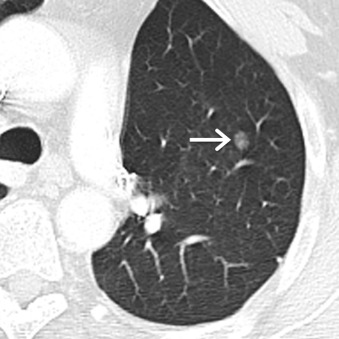
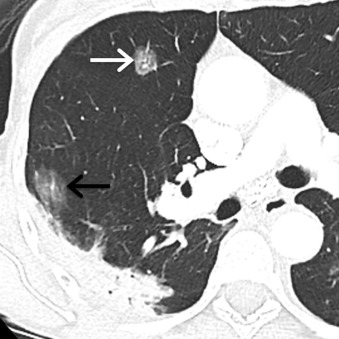
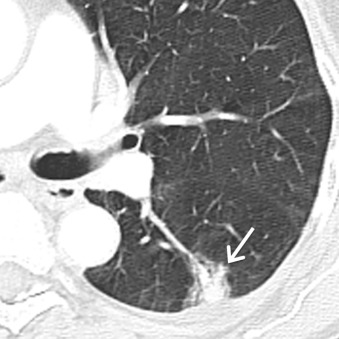
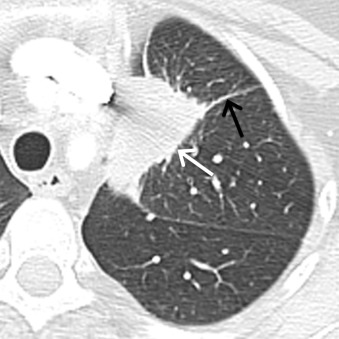
Atypical adenomatous hyperplasia (AAH) represents a small proliferation of atypical type II pneumocytes, Clara cells, and respiratory bronchioles measuring ≤5 mm. On MDCT, AAH manifests as a focal ground-glass nodule, typically measuring ≤5 mm, although they may range up to 12 mm in size ( Fig. 17.1 ). Adenocarcinoma in situ (AIS), formerly referred to as BAC, represents adenocarcinoma with pure lepidic growth, no evidence of nuclear atypia, and no evidence of stromal, vascular, or pleural invasion. Nonmucinous, mucinous, and mixed types have been described, although the nonmucinous type represents the majority of adenocarcinomas. On MDCT, AIS appears as a ground-glass, solid, or part solid nodule typically measuring ≤3 cm. These nodules may demonstrate slightly higher attenuation than AAH ( Fig. 17.2 ). Minimally invasive adenocarcinomas (MIAs) are characterized by lepidic predominant growth and invasion of ≤5 mm. On MDCT, MIAs usually manifest as a part solid nodule with a predominant ground-glass component and a solid component measuring ≤5 mm ( Fig. 17.3 ). Invasive adenocarcinomas are characterized by lepidic predominant spread and invasion of ≥5 mm. Acinar predominant, papillary predominant, micropapillary predominant, and solid predominant with mucin production types have been described. On MDCT, invasive adenocarcinomas manifest as a solid or part solid nodule, with a predominantly solid component ( Fig. 17.4 ). Local invasion of the pleura, chest wall, or diaphragm may be present. Other findings include air bronchograms, internal lucent regions, and masslike consolidation. Multiple pulmonary nodules, masses, and/or regions of consolidation may be present. PET/CT is most effective at staging and restaging aggressive tumors such as invasive adenocarcinomas. Other malignancies such as AIS and MIA may demonstrate little to no FDG uptake, which may be due to poor cellularity or slow cellular proliferation ( Fig. 17.5 ).
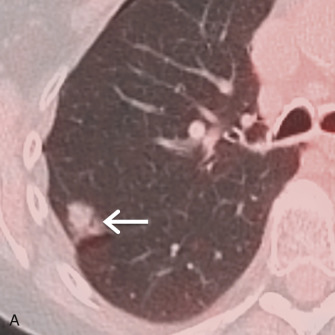
Squamous cell carcinoma (SCC) is the second most common subtype of NSCLC and accounts for 25% to 30% of all lung cancers. It is strongly associated with smoking, and 50% of tumors develop in current or former smokers. SCC is more common in men than women. Four histopathologic variants have been described—papillary, small cell, clear cell, and basaloid—and 7% to 10% of patients are asymptomatic. Clinical symptoms are more likely to be present when lesions are centrally located and in an advanced stage of disease. When present, symptoms most commonly reported include cough, dyspnea, hemoptysis, and fever. Paraneoplastic syndromes such as hypercalcemia due to secretion of a parathyroid hormone–like substance may be present. SCCs arising in the lung apices, also referred to as Pancoast tumors, can result in neuropathic pain due to involvement of the brachial plexus or Horner syndrome due to involvement of the sympathetic chain.
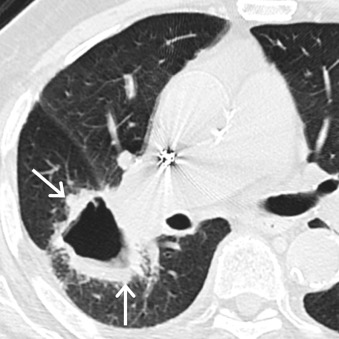
SCC typically arises from large bronchi in the central aspect of the chest. Thus, on MDCT, most SCCs manifest as a centrally located nodule or mass. An endobronchial component may be present, and postobstructive atelectasis and/or pneumonia may be visible. Alternatively, tumors may appear as a peripherally located nodule or mass, with lobulated or spiculated margins. Cavitation may be present and is more common in larger lesions ( Fig. 17.6 ). Central necrosis and wall thickening may be present. Calcification has been reported in 13% of lesions. On PET/CT, most tumors demonstrate FDG uptake greater than the mediastinal background. MRI can be used to assess for chest wall invasion and involvement of the brachial plexus in Pancoast tumors ( Fig. 17.7 ).
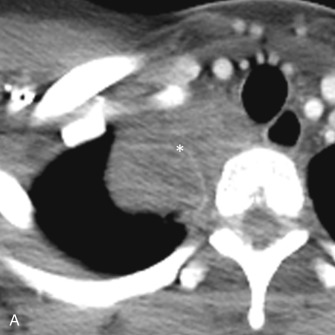
Large cell lung carcinoma (LCLC) represents a heterogeneous group of neoplasms that account for 5% to 10% of all lung cancers. LCLC encompasses several malignancies, including giant cell carcinoma, basaloid large cell carcinoma, clear cell carcinoma, lymphoepithelioma-like carcinoma, large cell carcinoma with rhabdoid phenotype, and large cell neuroendocrine carcinoma (LCNEC). LCLC has been described as a diagnosis of exclusion in that tumor cells lack histopathologic features that would otherwise classify the lesion as an SCLC or specific subtype of NSCLC. LCNEC is one of the most clinically significant LCLCs and represents a high-grade neuroendocrine malignancy characterized by neuroendocrine histologic features. Most LCNECs occur in men with a history of heavy smoking, and the mean age at the time of diagnosis is 65 years. Clinical symptoms include chest pain, hemoptysis, cough, and other symptoms such as dyspnea, weight loss, and fever. Unlike SCC, paraneoplastic syndromes are rare.
On MDCT, LCNEC is usually indistinguishable from other lung cancers. Lesions usually appear as a large mass in the peripheral aspect of the chest, ranging in size from 13 to 92 mm ( Fig. 17.8 ). Only one-fifth of lesions are centrally located. Most tumors demonstrate lobular margins, although tumors with spiculated borders have been described. The attenuation of LCNEC has been described as similar to that of chest wall musculature. Following the administration of IV contrast, tumors demonstrate varying degrees of enhancement. Heterogeneous or peripheral enhancement may be seen in the setting of necrosis. Calcification is an infrequent finding and has been reported in less than 9% of cases. LCNEC typically demonstrates increased FDG uptake on PET/CT, and the modality is used for staging and restaging purposes.
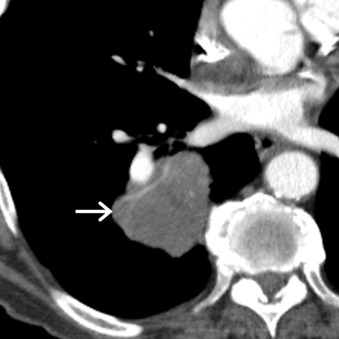
SCLCs account for approximately 13% to 15% of all lung cancers and are the most common primary pulmonary neuroendocrine neoplasm. SCLC has the strongest association with cigarette smoking, which is responsible for approximately 95% of cases of SCLC. In general, SCLC is much more aggressive than NSCLC and has a greater propensity for the early development of widespread metastases. SCLC has traditionally been staged with the Veterans Administration Lung Study Group (VALSG) systems, which divide SCLC into limited stage (LS) and extensive stage (ES) based on radiation treatment planning. However, the IASLC has recommended that the current seventh edition of the American Joint Committee on Cancer (AJCC) tumor-node-metastasis (TNM) staging system for lung cancer be used to stage SCLC.
Because 90% to 95% of SCLCs arise from lobar or main bronchi, most SCLCs are located within the central aspect of the chest and manifest as a mediastinal (92%) or hilar (84%) lymphadenopathy or pulmonary lesion ( Fig. 17.9 ). Postobstructive atelectasis and/or pneumonia may be seen in these patients. MDCT can be useful in revealing the presence and extent of mediastinal invasion. In 68% of patients, encasement of mediastinal structures such as the trachea, esophagus, heart, and vessels, including the superior vena cava, is evident ( Fig. 17.10 ). Intratumoral calcification has been reported in 23% of patients. A small group of patients with SCLC (<5%) may have a peripheral pulmonary nodule without associated lymphadenopathy. These peripherally located tumors usually manifest as well-defined nodules or masses that are homogeneous and demonstrate lobular margins and spiculation. Surrounding ground-glass opacities, representing edema or hemorrhage, may be identified. Unusual manifestations of SCLC on MDCT include consolidation, air space opacities, and lymphangitic carcinomatosis. On PET/CT, most SCLCs demonstrate markedly increased FDG uptake due to high metabolic activity. PET/CT has been shown to stage patients more accurately than conventional imaging and can be used for restaging purposes.
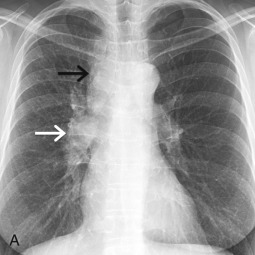
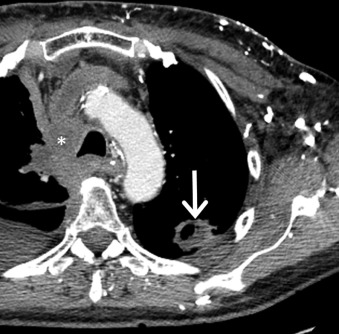
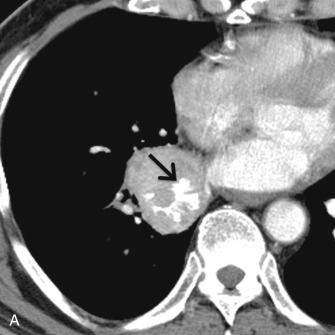
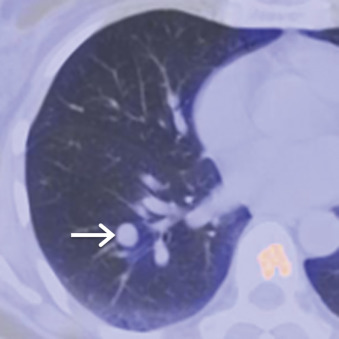
Carcinoid tumors are neuroendocrine neoplasms that are typically found in the gastrointestinal tract; however, 20% to 30% of all carcinoids arise from the respiratory tract, and these neoplasms represent 1% to 2% of all primary lung cancers. Carcinoid tumors are classified based on mitotic activity—typical (low grade) and atypical (intermediate grade). Most neoplasms are typical carcinoids, which are well-differentiated neoplasms with neuroendocrine features measuring ≥5 mm and exhibiting less than 2 mitoses/10 high-power field (HPF) or 2 mm 2 and no necrosis. Typical carcinoids are less aggressive, and 13% of patients present with lymph node involvement. Atypical carcinoid tumors account for 10% to 16% of carcinoids, are characterized by 2 to 10 mitoses/HPF or necrosis, and are more aggressive, and more patients (57%) present with lymph node involvement.
Typical and atypical carcinoids demonstrate similar features on imaging studies. The most common finding on chest radiography is a well-defined hilar or perihilar nodule or mass. In general, carcinoids are more common in the right lung than in the left lung. The average size is 3 cm, with a range of 2 to 5 cm, although atypical carcinoids tend to be larger than typical carcinoids and are more likely to be located in the lung periphery. Associated findings, such as postobstructive atelectasis and/or pneumonia, may be present. MDCT usually demonstrates a soft tissue nodule that is often closely associated with an airway. In some cases, the tumor may be entirely endoluminal or partially endoluminal. Punctate, eccentric, or diffuse patterns of calcification have been reported in 30% of cases. Associated findings such as postobstructive atelectasis and/or pneumonia, mucoid impaction, bronchiectasis, or air trapping may be present. Carcinoid tumors may demonstrate enhancement following the administration of IV contrast ( Fig. 17.11 ). PET/CT is of limited utility in the evaluation of carcinoids because tumors may demonstrate little to no FDG uptake, and a high false-negative rate has been reported ( Fig. 17.12 ).
The IASLC has recommended that the current eighth edition of the AJCC TNM staging system be used to stage NSCLC, SCLC, and bronchopulmonary carcinoid. Traditionally, SCLC has been staged with the VALSG and modified VALSG systems, which divide SCLC into LS and ES based on radiation treatment planning.
Although it has been recommended that the TNM system replace the VALSG staging systems for the staging of SCLC, a brief review of the latter is beneficial because they are still commonly used in clinical practice. In the original VALSG system, LS-SCLC was characterized by tumor involvement limited to one hemithorax (with or without local extension) and no distant extrathoracic metastatic disease ( Box 17.2 ). Regional and ipsilateral supraclavicular lymph nodes were considered to be LS-SCLC if these groups could be included in a single safe and adequate radiation port. ES-SCLC applied to all other patients; this included specific features such as malignant pleural and pericardial effusions, contralateral hilar or supraclavicular lymph nodes, and metastatic disease that could not be treated in a single radiation port. In 1989, the IASLC proposed key changes to the VALSG system ( Box 17.3 ), the most significant of which included contralateral mediastinal and supraclavicular lymph nodes and ipsilateral pleural effusions (benign and malignant, regardless of cytology) into LS-SCLC. ES-SCLC disease was defined as disease beyond that of LS-SCLC. A retrospective study at one institution indicated that the IASLC system had better prognostic value than the VALSG system. In clinical practice, features of the VALSG and IASLC staging systems are often combined. For example, LS-SCLC can include contralateral mediastinal and ipsilateral supraclavicular lymph node involvement. However, certain findings, such as contralateral supraclavicular or hilar lymph node involvement, remain controversial, and clinical management is typically based on the ability to include these regions in a safe and adequate radiation port.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here