Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Multiple pathophysiologic mechanisms can present with pulmonary manifestations in term and preterm infants. The clinical picture is most commonly dominated by respiratory distress, which presents as tachypnea, grunting, flaring, retractions, cyanosis, and hypoxemia. However, apnea and hypoventilation are also common. In preterm infants, these manifestations are commonly associated with respiratory distress syndrome (RDS) as discussed in Chapter 64 . Nonpulmonary etiologies of respiratory distress include thermal instability, circulatory problems, cardiac disease, neuromuscular disorders, sepsis, anemia or polycythemia, and methemoglobinemia ( Box 66.1 ). This section presents an overview of the many other respiratory disorders that can affect preterm and term infants.
Central nervous system: asphyxia, hemorrhage, malformations, drugs, infection
Spinal cord: cord injury, spinal muscular atrophy
Nerves: phrenic nerve injury, cranial nerve palsy
Neuromuscular plate: myasthenia gravis
Muscular: dystrophies
Airway obstruction (see Chapter 68 )
Intrinsic: choanal atresia, floppy epiglottis, laryngeal web, cord paralysis, laryngospasm, malacia, tracheal stenosis
Extrinsic: tubes, secretions, Pierre Robin syndrome, macroglossia, goiter, vascular ring, cystic hygroma, mediastinal and cervical masses
Rib cage abnormalities
Thoracic dystrophies
Rickets and bone disease
Fractures
Pectus excavatum
Congenital eventration
Abdominal distention
Anemia
Polycythemia
Methemoglobinemia
Metabolic acidosis
Hypoglycemia
Hypocalcemia
Increased pulmonary flow
Patent ductus arteriosus
Ventricular septal defect
Transposition of the great arteries
Truncus arteriosus
Decreased pulmonary flow
Persistent pulmonary hypertension
Pulmonary atresia
Tetralogy of Fallot
Tricuspid atresia
Cardiomegaly
Tricuspid atresia
Ebstein anomaly
Left heart obstruction (e.g., coarctation, mitral atresia, total anomalous pulmonary venous return)
Hypotension
Sepsis
Pain
Hypothermia
Hyperthermia
Abnormal lung development varies from mild hypoplasia, which can either be a primary or secondary defect, to agenesis of a single lobe or whole lung. Agenesis of the lung is a rare congenital anomaly defined as the total absence of pulmonary parenchyma, its supporting vasculature, and bronchi after the bifurcation. Complete agenesis of the lung is usually unilateral (90%), although dysgenesis of the other lung has been reported. Pulmonary aplasia can be classified as a subset of pulmonary agenesis, defined by the presence of a blind-ending, rudimentary bronchus without associated lung parenchyma or pulmonary vasculature. It has been hypothesized that pulmonary agenesis is vascular in origin, most likely resulting from a disruption of the dorsal aortic arch blood flow during the fourth week of gestation. Although lung agenesis can be an isolated finding, in as many as 50%-76% cases it is associated with other congenital malformations, including urogenital, vertebral, cardiac, and gastrointestinal, as well as malformations of the first and second branchial arch derivatives and radial ray defects. As a result, pulmonary agenesis has been considered a subset of the VACTERL ( v ertebral, a nal atresia, c ardiac defect, t racheo- e sophageal fistula, r enal anomalies, l imb anomalies) sequence and Goldenhar syndrome. Malformations of the first and second branchial arches and/or radial ray malformations are the most common malformations associated with pulmonary agenesis, and all cases are ipsilateral to the pulmonary malformation. Those who do not have facial or radial ray anomalies appear to fit the VACTERL association.
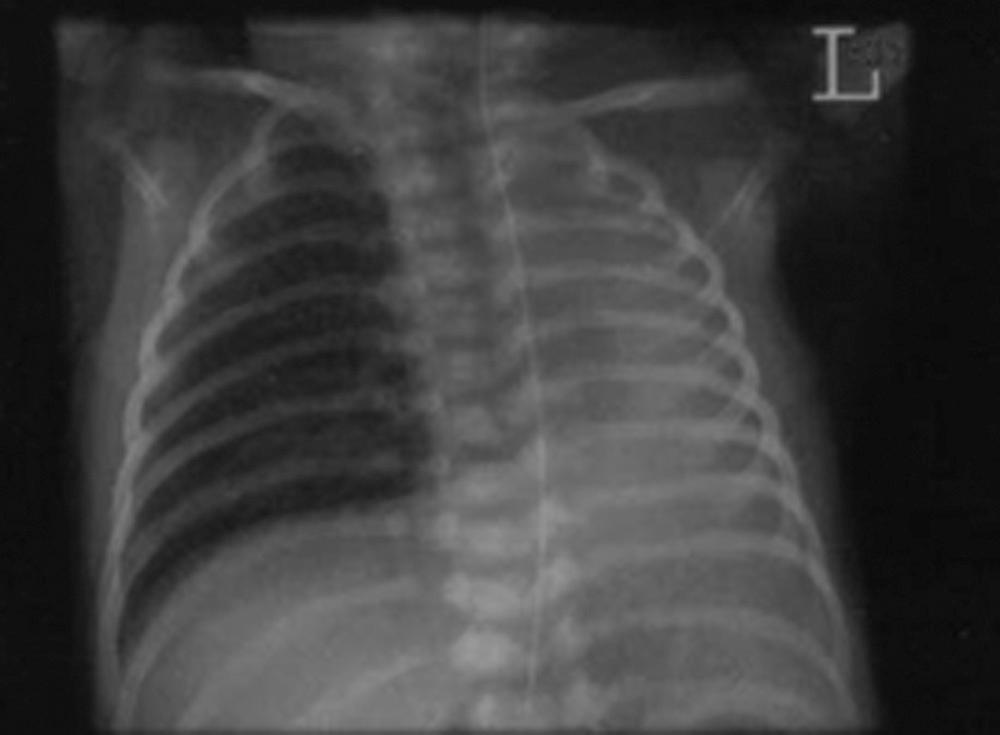
Diagnosis is confirmed on antenatal ultrasound by the presence of mediastinal shift in the absence of a diaphragmatic hernia. Antenatal echocardiogram reveals total absence of the pulmonary artery or one of its branches on the affected side. Magnetic resonance imaging (MRI) examination can confirm the diagnosis, evaluate the size of the remaining lung, and evaluate the presence of other congenital malformations. After delivery, diagnosis of infants with unilateral pulmonary agenesis can be suspected by decreased breath sounds and displacement of the mediastinum to the affected side. Some breath sounds, however, may be audible over the affected side if a portion of normal lung, which usually has undergone compensatory hypertrophy, has herniated across the midline into the affected hemithorax. The radiographic appearance of a radiopaque hemithorax helps confirm the diagnosis, and accompanying vertebral defects are not uncommon ( Fig. 66.1 ). Treatment is largely supportive, and the prognosis depends on the presence or absence of other anomalies; those with isolated pulmonary agenesis have better survival.
Pulmonary hypoplasia is the result of deficient or incomplete development of the lung parenchyma leading to decreased number of distal airways, alveoli, and associated pulmonary vessels. It can be classified as either primary , caused by intrinsic failure of normal lung development, or secondary , caused by a multitude of different pathologic processes that interfere with normal lung development. The pathophysiology of secondary pulmonary hypoplasia includes (1) oligohydramnios secondary to renal malformations, prolonged early amniotic fluid leak, placental abnormalities, or intrauterine growth restriction; (2) space-occupying lesions compressing the lungs and preventing normal growth as seen with congenital diaphragmatic hernia (which is thought to have some evidence of primary pulmonary hypoplasia as well), cystic lung disease, or cardiac malformations with extreme cardiomegaly (e.g., tricuspid atresia or Ebstein anomaly); and (3) absence or abnormal diaphragmatic activity (which is essential for lung development) resulting from a central or peripheral nervous system disorder or musculoskeletal disease, which can prevent chest wall expansion and breathing movements.
Diagnosis of pulmonary hypoplasia is made pathologically by measuring the lung-to-body ratio; however, this truly only captures those with lethal pulmonary hypoplasia. Methods to diagnose pulmonary hypoplasia antenatally in those fetuses at risk for developing pulmonary hypoplasia because of associated findings have been reported and include the use of prenatal ultrasound techniques, both two- and three-dimensional, as well as fetal MRI. Ultrasound measurement techniques include thoracic circumference (TC) corrected for gestational age or femur length, TC:abdominal circumference ratio, and thoracic:heart area. These techniques, however, measure the thoracic wall rather than lung parenchyma itself and have inadequate positive and negative predictive values to be a reliable tool for predicting pulmonary hypoplasia in isolation. More recently, measurement of lung volume using three-dimensional ultrasound has been reported, but even in the most experienced hands, 8%-48% of cases did not have a calculable lung volume on at least one side. MRI has shown promise in predicting pulmonary hypoplasia, especially in the setting of congenital diaphragmatic hernia (CDH), but seems to be no better for other causes of pulmonary hypoplasia. Pathologic examination of the hypoplastic lung can show a low ratio of lung to body weight, low DNA content, or decreased radial alveolar count. Peripheral bronchioles are decreased in number, as are the pulmonary arterioles, which often exhibit hypertrophy of medial smooth muscle, a predisposition to persistent pulmonary hypertension.
Secondary pulmonary hypoplasia is most commonly encountered in oligohydramnios and congenital diaphragmatic hernia (CDH). Survival of infants with pulmonary hypoplasia depends on the degree to which lung growth is restricted and the underlying cause of hypoplasia. It is not uncommon for these patients to present with severe respiratory distress associated with bilateral pneumothorax as well as hypoxia from both fixed and reactive pulmonary hypertension. Pulmonary hypoplasia is present in up to 33% of patients with oligohydramnios and can be associated with a high mortality rate (55%-100%) depending on the severity of hypoplasia (see Chapter 24 ). Patients with pulmonary hypoplasia secondary to prolonged premature rupture of membranes (PPROM) starting in the second trimester have been shown to have a better prognosis than initially expected. In four retrospective series of mid-trimester PPROM (18 0/7 to 24 6/7 weeks’ gestation), higher survival rates of 68%-73% to as high as 90% have been reported. This is thought to be secondary to improved obstetrical (prenatal steroids) and modern neonatal therapies. However, among survivors, 36%-46% had bronchopulmonary dysplasia. Both human and animal studies have shown that some of these infants who present with early severe respiratory failure consistent with pulmonary hypoplasia may benefit from inhaled nitric oxide (iNO). Supportive therapy with gentle ventilation is the mainstay of therapy. Serial amnioinfusion has been proposed as a potential therapy to mitigate the effects of oligohydramnios/anhydramnios and pulmonary hypoplasia caused by both PROM and renal abnormalities. When used in the setting of PROM, there is no improved survival, but success has been reported in a few case series when used for renal abnormalities. At this time, no randomized control trials exist to prove its efficacy in either case.
Congenital diaphragmatic hernia (CDH) results from a developmental defect during the formation of the diaphragm that permits abdominal contents to herniate into the thoracic cavity. In some cases, the herniated viscera are covered with a membranous sac. Defects of the diaphragm are classified according to the anatomic region of the diaphragm that is defective. The most common defect involves the posterolateral diaphragm (Bochdalek) and accounts for 70% of diaphragmatic hernias. Anterior diaphragm defects (Morgagni) account for 25%-30% of diaphragmatic hernias, and central diaphragmatic defects are rare, occurring in 2%-5% of cases. The incidence of CDH is 1 in 2000-3000 live births, and it occurs most commonly on the left side (85%), whereas bilateral hernias are rare (1%) and usually fatal. In CDH, there are fewer alveoli that have thickened septae, resulting in markedly diminished alveolar airspace and gas exchange surface area. Vascular development parallels that of the airways, thus there also are reduced number of vessels, adventitial thickening, medial muscle hyperplasia, and peripheral extension of the muscular layer into the smaller intra-acinary arterioles, resulting in a fixed increase in pulmonary vascular resistance. Both lungs are affected, the ipsilateral one more than the contralateral one. After delivery, these morphologic changes compromise effective gas exchange, resulting in respiratory failure and pulmonary hypertension.
To explain the severe respiratory failure at birth owing to pulmonary hypoplasia, a dual hit hypothesis has been proposed. Traditionally, it was thought that the pulmonary hypoplasia associated with CDH was caused by compression of the lungs by the herniated abdominal viscera during early development. However, as our understanding of the pathophysiology grew, it has been demonstrated that pulmonary development has already been disturbed prior to diaphragm development and before compression from abdominal organs can occur. Thus, the dual hit hypothesis has emerged to explain the severe pulmonary hypoplasia often seen in this condition. The first insult affects both lungs before diaphragm development is complete. The second insult affects the growth of the ipsilateral lung because of compression of that lung by the herniated viscera. The origin of the diaphragmatic defect lies in the disruption of the mesenchymal substrata of the pleuroperitoneal folds, which form the scaffold for the eventual migration of muscular precursor cells into the nascent diaphragm. In human embryology, this would point to a defect as early as 4-5 weeks’ gestation, long before the formation of the “muscular” diaphragm.
The pathogenesis of CDH and associated pulmonary hypoplasia are slowly being unraveled in part owing to animal models, which have confirmed that developmental regulation of the lung and diaphragm are controlled by some of the same genes. The nitrofen mouse model has proposed that CDH is a result of a disturbance in the retinoic acid pathway. A defect in the retinoid signaling pathway, specifically, inhibition of retinal dehydrogenase-2 (Raldh2), has been proposed as the likely etiology of the embryonic disruption resulting in CDH. However, despite convincing animal data, the use of nitrofen and retinol have not been linked to human CDH.
Congenital diaphragmatic hernia is either an isolated defect or can be associated with other congenital anomalies, including cardiac, urogenital, chromosomal, and musculoskeletal. Most reports estimate that 40%-60% of patients with CDH have non-hernia-related anomalies, designated CDH+. In a review of 3062 patients with CDH, the Congenital Diaphragmatic Hernia Study Group reported a 28% incidence of severe malformations (major cardiac, syndromal, and chromosomal disorders) in patients who did not undergo surgical repair secondary to unsalvageable hernia compared with 7% in repaired patients. In some of these CDH+ infants, the constellation of anomalies suggests a well-described genetic syndrome, the most frequent of which is Fryns syndrome, which has an autosomal recessive inheritance pattern. Another proportion of these CDH+ infants have been found to have chromosomal rearrangements including aneuploidy (trisomies 13, 18, 21, and Turner syndrome), segmental deletions, or duplications and rearrangements. Genetic analysis has identified a CDH critical region on chromosome 15q26, which codes for four genes involved in diaphragmatic morphogenesis. Microdeletions in this region have been described in several patients with nonisolated CDH and are associated with very high mortality. Other CDH “hot spots” include 1q41-q42, 3q22, 4p16, 8p23, 8q22, and 11p13. Haploinsufficiency or decreased expression of one or more genes encoded in these regions may cause or predispose to the development of CDH.
Recent advances in genetic diagnostic modalities along with the use of knockout mice have shed some light on specific genes that may be involved in the pathogenesis of CDH, including clues for both the diaphragmatic defect as well as the associated pulmonary hypoplasia. Several genes including Wilms tumor 1 ( Wt-1 ) and members of the sonic hedgehog ( Shh ) pathway ( Shh , Gli2 , Gli3 ) have been implicated in CDH. Wt-1- , Gli2 -, and Gli3 -null mice have all been shown to have diaphragmatic defects. Shh has been demonstrated to be downregulated in human hypoplastic lungs of CDH patients. Another growth factor–related gene, platelet-derived growth factor ( PDGFRα ), was demonstrated to play a role in posterolateral diaphragm and lung development, but the mice knockouts demonstrate features of Fryns syndrome; thus PDGFRα may be involved in nonisolated CDH. As advances are made in molecular genetics, more candidate genes are being discovered.
For all the reasons aforementioned, it is important to adequately evaluate for associated malformations in patients with CDH as well as genetic testing antenatally, if not then postnatally. About half of CDH cases are diagnosed antenatally. Antenatal diagnosis is associated with a poor prognosis, but the data suggest that infants with a prenatal diagnosis have a better chance of survival if they are born in a tertiary center with reported survival rates of isolated, operative CDH cases approaching 70%-90%. Several antenatal parameters have been evaluated for their ability to predict survival and morbidity in cases of isolated CDH. Most of these predictors rely on the indirect assessment of the size of the contralateral lung as a proxy for pulmonary hypoplasia. Methods used include lung area to head circumference ratio (LHR) between 22 and 28 weeks’ gestation, the presence of the liver in the chest (which has been thought to be most predictive), and estimated fetal lung volume by MRI. Because it has been reported that the LHR increases exponentially with gestational age, some experts have advocated that the LHR should be corrected for gestational age and have used an observed:expected LHR (O/E LHR) based on the LHR in the CDH patient compared with what is considered normal for that gestational age. When using the corrected LHR, the CDH registry has quoted the following numbers based on 184 fetuses with isolated left-sided CDH evaluated between 22 and 28 weeks’ gestation :
Fetuses with O/E LHR less than 15% have extreme pulmonary hypoplasia, with virtually no survivors.
Fetuses with O/E LHR between 15% and 25% have severe pulmonary hypoplasia, with a predicted survival of 20%. (Those with liver completely down in the abdomen fare better than those with liver herniated up into the chest.)
Fetuses with O/E LHR between 26% and 35% and those with O/E LHR between 36% and 45%, but liver in the chest, have moderate pulmonary hypoplasia, with expected survival between 30% and 60%.
Fetuses with O/E LHR between 36% and 45% with liver down and those with an O/E LHR greater than 45% have mild hypoplasia and are likely to survive (>75%).
More recently, authors have focused on the use of MRI to evaluate total lung volume (TLV), observed to expected TLV (O/E TLV), and the percentage of liver herniated into the chest (%LH) to predict survival and/or need for extracorporeal membrane oxygenation (ECMO) support. When using O/E TLV, thresholds of <25%, 25%-35%, and >35% were predictive of 0%-25%, 25%-69%, and 75%-89% survival respectively. And when combining o/e TLV and %LH, severe CDH defined as an O/E TLV <32% and %LH >21% carried a 50% mortality; mild CDH were those with an o/e TLV >32% and %LH <21% had only an 8% risk of mortality.
The ability of these parameters to predict survival has not been consistent. This is mostly secondary to the small number of patients in each series, challenges in measurement consistency among operators, lack of correlation with actual and functional lung volume and pulmonary vasculature size, inconsistent measures for survival or morbidity, and differences in postnatal management and survival. Nevertheless, these tools give clinicians some data to use when counseling families faced with the diagnosis of CDH.
The clinical presentation of patients with CDH can vary from asymptomatic in mild cases to severe respiratory failure at birth. Diagnosis should be suspected in previously undiagnosed patients by the presence of severe respiratory distress, cyanosis, scaphoid abdomen, and failure to improve with ventilation. Physical examination reveals absence of breath sounds on the affected side with displacement of heart sounds to the contralateral side, and occasionally bowel sounds can be heard over the thorax. Once the diagnosis is made or suspected, patients should be immediately intubated and an orogastric tube placed to evacuate the stomach. Aggressive ventilatory strategies should be avoided (see later). Chest radiograph shows the presence of bowel loops in the affected chest cavity with shifting of the heart to the contralateral side ( Fig. 66.2 ). If the stomach is included in the hernia, the tip of the orogastric tube will be seen within the thorax. The presence of liver in the chest is suspected by deviation of the umbilical venous line. Late presentation of Bochdalek hernia occurs in less than 3% of cases. These patients can be asymptomatic at birth and usually present later in life with respiratory or gastrointestinal symptoms. High index of suspicion is needed in these cases to prevent unwarranted and potentially dangerous interventions such as the insertion of a chest tube for suspected pleural effusion or pneumothorax. Diagnosis can be made after nasogastric tube insertion, contrast upper gastrointestinal study, or chest computed tomography (CT) scan. Prognosis for cases with late presentation is excellent once the correct diagnosis is made.

Improved survival has been reported using a consistent approach in the management of CDH that can be facilitated by the development of multidisciplinary standardized treatment guidelines, including input from neonatology, pediatric surgery, ECMO specialists, and respiratory therapy ( Box 66.2 ). Predetermined criteria for the use of ECMO and an underlying protect-the-lung strategy are essential components in the care of these infants and can be as important as the specific medical interventions chosen. Whereas animal studies have suggested lung immaturity and surfactant deficiency in models of CDH, the use of antenatal steroids and surfactant replacement has not been shown to be beneficial. A systematic review of strategies associated with improved survival among infants with CDH in 13 centers that cared for at least 20 patients and reported a survival rate of 75% or more has described multiple successful treatment strategies associated with this improved survival. Although these centers used different mechanical ventilation strategies, most of these targeted the use of gentle ventilation or permissive hypercapnia. The basic elements of this treatment strategy are:
Ventilation of the patient with low peak inspiratory pressures (PIP) to minimize lung injury. The goal of PIP is usually less than 25 cm H 2 O.
Accepting preductal saturations of greater than or equal to 85%, regardless of postductal saturation, and higher Pa co 2 levels of less than or equal to 65 with a pH of at least 7.25 as long as there is evidence of adequate tissue perfusion and oxygenation.
Instituting high-frequency oscillatory ventilation (HFOV) or high-frequency positive pressure ventilation once the preset limit failed to achieve adequate ventilation, although HFOV was used by some as the primary mode of ventilation.
Even though iNO might produce short-term benefits, the routine use of iNO is not supported by current data and might actually be associated with a worse outcome (see Chapter 70 ).
Using ECMO as rescue therapy with variable indications in different centers, including persistent oxygenation index (OI) greater than 40, persistent hypoxemia, or failure of ventilatory management to support oxygenation, ventilation, or tissue perfusion (see Chapter 70 ).
Delaying surgical repair until physiologic stabilization and improvement of PPHN.
Surgical repair is delayed until the pulmonary hypertension is resolved or significantly improved such that, if need be, support may be escalated postoperatively if necessary. The procedure often involves either a primary closure of the diaphragmatic defect, or if too large, the use of a prosthetic patch. Recently, centers are reporting on thoracoscopic closure, but no trials comparing the methods exist.
Accept preductal saturations greater than or equal to 85%, Pa co 2 less than or equal to 65 mm Hg, and pH greater than or equal to 7.25.
Identify preset ventilatory limits that are not to be exceeded.
Use high-frequency oscillatory ventilation if conventional mechanical ventilation fails.
Use ECMO per preset criteria.
Delay surgery until persistent pulmonary hypertension improves.
Postnatal survival rate at tertiary centers has improved with reported rates of 70%-92%. However, the survival data might underestimate hidden mortality secondary to termination, stillborn, and referral pattern for outborn patients. With improvement in survival, there has been a focus on improving long-term morbidity of survivors. Infants born with CDH have multiple long-term morbidities affecting the pulmonary, gastrointestinal, neurologic, and skeletal systems. Respiratory complications include pulmonary vascular abnormalities presumably causing pulmonary hypertension, a higher incidence of obstructive airway disease, and a restrictive lung function pattern that continues into adulthood. Gastroesophageal reflux disease (GERD), sometimes in combination with failure to thrive, is a well-recognized complication in patients with CDH, and several patients require antireflux surgery. It is unknown whether GERD has an effect on pulmonary function in this population. Pulmonary hypoplasia and PPHN predispose children born with CDH to a high risk for hypoxemia, which may result in neurodevelopmental delay. It has been reported that infants with CDH are at higher risk to have neuromotor delay, hypotonia, and delayed language skills. There is also a high percentage of these infants with sensorineural hearing loss. Chest wall deformities and scoliosis are more common among CDH patients, although deformities are mild and surgery is rarely required. These data emphasize the need for a multidisciplinary team approach in the postoperative management and follow-up of all survivors of CDH. The American Academy of Pediatrics (AAP) section on surgery has provided a suggested schedule for follow-up for these children.
More recently, fetal therapies, namely fetoscopic tracheal occlusion (FETO) in the most severe cases (where survival was nil given prenatal predictors), have shown some promise in improving survival in those cases, including improved lung growth in utero, less pulmonary hypertension, and overall improved survival when compared to expectant management. However, some of these successes have come at a cost of increased preterm delivery, although with improved techniques, the rate of preterm birth is declining. There are several US centers trialing this approach now, and a large, international, multicenter, randomized control study is underway to evaluate not only FETO for severe CDH but also for moderately severe CDH and effectiveness performing fetoscopic tracheal occlusion at later gestational ages (30-32 weeks); this is the Tracheal Occlusion To Accelerate Lung Growth Trial.
Alveolar capillary dysplasia with misalignment of pulmonary veins (ACD/MPV) is a rare, often fatal pulmonary disease that presents in the newborn period, usually within the first 48 hours of life with severe hypoxemia and PPHN unresponsive to treatment. The true incidence of the disease is unknown, because the diagnosis is made by pathology, and if there is no autopsy or antemortem lung biopsy, the diagnosis cannot be made; thus, what is reported in the literature likely underestimates the true incidence of the disease. Histologically, ACD/MPV is characterized by paucity of capillaries proximal to the alveolar epithelium, anomalous distended pulmonary veins within the bronchovascular bundle (rather than within the interlobular septae), and immature alveolar development with medial thickening of small pulmonary arteries and muscularization of the arterioles, and in approximately one-third of cases, lymphangiectasis. These characteristic findings are diffuse in 85% of patients and patchy in the remainder. Although the mechanisms underlying the pathogenesis of ACD/MPV and its associated pulmonary hypertension are not fully understood, it has been suggested that it is related to a failure of fetal lung vascularization with capillary hypoplasia and discontinuity of these capillaries and pulmonary veins impairing pulmonary blood flow, as well as a component of reactive pulmonary vasoconstriction mediated by hypoxia. More recently, the role of prominent right-to-left intrapulmonary vascular shunt pathways have been identified and implicated in the severe hypoxia seen in these patients. Extrapulmonary findings are present in 50%-80% of cases, most commonly affecting the gastrointestinal, genitourinary, and cardiovascular systems. Although the disease is mostly sporadic, reports of multiple affected siblings in subsets of families suggest an autosomal recessive inheritance pattern. Mutations of the FOXF1 gene located on chromosome 16q24.1 have been identified in 70%-90% of patients with ACD/MPV, and more recently, there is evidence that genomic imprinting plays a role in the development of ACD/MPV. This may allow for prenatal genetic testing of high-risk families; however, it will not identify all cases of ACD.
Most patients with ACD/MPV present within the first few hours of life. These infants are usually born at term and have appropriate size and normal Apgar scores. Although these babies might be asymptomatic at delivery, respiratory distress, cyanosis, and hypoxemia progress quickly to respiratory failure in more than half of these patients within hours after delivery. About 14% of these patients do not present with symptoms until 2-6 weeks of life. Treatment is always unsatisfactory. Although a transient response to iNO might be observed, PPHN is not responsive to medical treatment, and the disease progression is that of a fulminant course and rapid progression to death, although there are reports of survival beyond the neonatal period. High index of suspicion and diagnostic lung biopsy are required to avoid the use of more invasive and futile treatments, including ECMO.
Congenital pulmonary lymphangiectasia (CPL) is a pulmonary disorder first described by Virchow in 1856, characterized by dilation of lymphatic vessels in multiple areas of the lungs. Most cases of CPL are sporadic with a predilection for male involvement (2 : 1). However, familial presentations have been described, suggesting an autosomal recessive inheritance pattern. The incidence is difficult to estimate as few cases are reported in the literature. Congenital pulmonary lymphangiectasia is classified as primary or secondary. Primary CPL can present as either a primary pulmonary developmental defect that can be localized or diffuse or as a part of a more generalized lymphatic developmental defect. Patients with generalized lymphangiectasia tend to have less severe pulmonary involvement. Secondary cases of CPL are often associated with cardiac malformations with obstructed pulmonary venous return, including obstructed total anomalous pulmonary venous return, hypoplastic left heart syndrome, and cor triatriatum. Congenital pulmonary lymphangiectasia has also been described in multiple syndromes, including Noonan, Down, and Ullrich-Turner. The characteristic pathologic finding of CPL is pulmonary lymphatic dilation in the subpleural, interlobar, perivascular, and peribronchial lymphatics. It may be associated with nonimmune hydrops fetalis and congenital chylothorax.
The etiology of CPL is not clear but is thought to be secondary to a failure of regression of lymphatics that normally occurs between 16 and 20 weeks’ gestation. Multiple genes have been found to be involved in lymphatic development, including FOXC2 , vascular endothelial growth factor 3, and integrin a9b1 genes. Mice homozygous for a null mutation in the integrin a9 subunit gene died of respiratory failure caused by bilateral chylothorax within 6-12 days after birth with pathologic features similar to those in CPL.
Patients with CPL usually present with intractable respiratory failure, cyanosis, and hypoxia associated with bilateral chylothoraces in the first few hours of life, although diagnosis can be delayed for several weeks in cases of unilobar involvement. Nonimmune hydrops is also a well-recognized presentation in patients with CPL. Examination of the pleural fluid shows characteristic findings of chylothorax, including a lymphocytosis and elevated triglycerides, although elevated triglycerides might be absent in nonfed infants (see later). Chest radiograph reveals hyperinflation of the lung with bilateral interstitial infiltrates and bilateral pleural effusions. High-resolution computed tomography demonstrates diffuse thickening of the peribronchovascular interstitium and the septa surrounding the lobules. Definitive diagnosis is made by lung biopsy showing the characteristic features: increased fibrous tissue with dilation of cystic lymphatic spaces and collapsed alveoli, although differentiation from lymphangiomatosis can be difficult.
Treatment is mostly supportive. Intubation and mechanical ventilation; drainage of pleural and peritoneal effusions; and correction of hypoxia, acidosis, and shock might be needed in the delivery room for stabilization. Persistent chylothorax might require chest tube placement. Nutritional therapy with medium-chain triglycerides and total parenteral nutrition has been successful in the treatment of CPL. Case reports of using octreotide and antiplasmin to treat CPL as well as intestinal lymphangiectasia have been reported with success. Pleurodesis with sclerosing agents has been used to treat persistent chylothoraces associated with the disease. More recently, reports of using lymphangiogram with ethiodized oil as a successful treatment modality have been reported. The prognosis appears to depend on the severity of symptoms in the immediate newborn period. Although traditionally thought to be fatal, there are reports of survival in some patients presenting with respiratory failure, chylothorax, and hydrops fetalis in the immediate neonatal period. Later presentation carries a better prognosis with the possibility of spontaneous resolution, although respiratory morbidity might be common.
Chylothorax is the accumulation of lymphatic fluid (chyle) in the pleural cavity. It is the most common cause of pleural effusion in neonates and can be primary (congenital) or secondary (acquired). It is a rare entity, with a reported incidence of 1 : 10,000 births, and affects males more than females (2 : 1). Congenital chylothorax, which accounts for less than 10% of all chylothoraces, may be associated with abnormalities of the lymphatic system; congenital malformations such as congenital heart disease or mediastinal malignancies; or chromosomal abnormalities such as trisomy 21, Noonan syndrome, or Turner syndrome. Secondary chylothoraces are most commonly associated with trauma during thoracic surgery but can also be the result of increased superior vena caval pressure caused by venous thrombosis.
Clinical presentation is that of respiratory distress secondary to lung compression, pulmonary hypoplasia, or symptoms of the underlying pulmonary or cardiac disease. Many cases of congenital chylothorax are diagnosed by prenatal ultrasound, and antenatal management consists of thoracocentesis or thoracoamniotic shunt placement to try to prevent the development of pulmonary hypoplasia. However, even with prenatal treatment, these infants can still present with significant respiratory distress at birth as fluid reaccumulates, requiring postnatal treatment.
Physical examination is significant for decreased breath sounds on the affected side with shifting of the cardiac apex to the contralateral side. Chest radiograph shows a pleural effusion, compression of the lung on the affected side, and displacement of the heart to the opposite side. Diagnosis is established by analysis of the pleural fluid. In neonates with established feedings, chylothorax appears milky in color; however, in nonfed neonates, it is clear. Buttiker and colleagues have proposed the following criteria for establishing the diagnosis of chylothorax: absolute cell count of greater than 1000/µL with a lymphocyte fraction of greater than 80% and triglyceride levels greater than 1.1 mmol/L.
Optimal treatment for chylothoraces has not been defined but is mostly supportive while awaiting resolution of the effusion. Mechanical ventilation and drainage of the chylothorax might be needed in patients with large effusions, and nutritional support using total parenteral nutrition is essential. When feedings are started, formulas containing a high percentage of medium-chain triglycerides (MCTs) are recommended, because lymphatics are not needed for MCT absorption. In most cases, spontaneous resolution occurs within 4-6 weeks. Several treatment strategies have been described for cases with persistent chylothorax, including pleurodesis, ligation of the thoracic duct, and pleuroperitoneal shunt. Whereas povidone-iodine pleurodesis has been used successfully in persistent chylothorax, it has also been associated with renal failure. There is growing evidence from uncontrolled case studies suggesting a markedly positive effect of somatostatins, particularly octreotide, in the treatment of chylothorax with minimal side effects. In the absence of a controlled trial evaluating safety and efficacy, this therapy should be reserved for persistent and severe cases and not as first line of treatment. Case reports of successful treatment with oral sildenafil, perhaps by generation of new lymphatic vessels, have been reported. An emerging therapeutic modality is embolization of the thoracic duct with lipiodol; however, this procedure is in its infancy and will need further study before becoming a first-line therapy.
Congenital cystic lung disease encompasses a broad spectrum of rare, but clinically significant, developmental abnormalities that include congenital pulmonary airway malformation (CPAM; previously known as congenital cystic adenomatoid malformation CCAM), bronchopulmonary sequestration (BPS), bronchogenic cyst (BC), and congenital lobar emphysema (CLE). These lesions are rare with an estimated incidence of 1 per 11,000-35,000 births. They were originally thought to be separate entities; however, the description of coexistence of multiple lesions (bronchogenic cyst, CPAM, and extralobar BPS) in the same patient as well as reports of bronchial atresia in some specimens of all four entities suggests a common embryologic origin. These lesions can be identified on prenatal ultrasound, but the classification of the lesion should wait until postnatal examination and histology are available. These lesions should be followed closely in utero as some of these lesions can predispose the fetus to develop hydrops, and when this occurs in the second trimester, fetal intervention is warranted. Intervention includes repeated cyst aspiration, thoracoamniotic shunting, sclerotherapy, fetal surgery, or more recently, maternal betamethasone treatment. However, fetal intervention is relatively rarely needed, as many of these cystic lung lesions appear to regress in utero.
Congenital pulmonary airway malformation (CPAM) constitutes multiple different hamartomatous lesions arising from the abnormal branching of the immature bronchial tree. This entity was first described by Ch'in and Tang in 1949, and since then, the term has been evolving as our understanding of the entity improves. For the past 40 years, Stocker has classified the lesions into three types: I, II, and III. Stocker has since described two new types, 0 and 4, and has revised his original term congenital cystic adenomatoid malformation, and now refers to this entity of cystic lung lesions as congenital pulmonary airway malformations (CPAM) to reflect the site of suspected development of the malformation in the tracheobronchial tree, and that only three types (1, 2, and 3) are adenomatoid and only types 1, 2, and 4 are cystic. However, the term CCAM will still appear in the literature.
Congenital pulmonary airway malformation is the most common congenital cystic lung disease, occurring in 1 in 11,000-30,000 live births and affecting more males. Both lungs are affected equally, and the disease is most commonly unilobar with predilection for the lower lung lobes. Unlike BPS, it is connected to the tracheobronchial tree and has a pulmonary blood supply ( Table 66.1 ). Congenital pulmonary airway malformation develops during the pseudoglandular phase (7-17 weeks’ gestation) of fetal lung development. In addition to the classification system described in the preceding, some have suggested classifying the lesions, at least during the prenatal period, as microcystic versus macrocystic based on the gross anatomy and antenatal ultrasound appearance. Whereas the latter classification has poor correlation with histologic features, it has a much better prognostic value, with microcystic lesions (cysts <5 mm) having a poorer prognosis than macrocystic lesions (>5 mm).
| Congenital Pulmonary Airway Malformation | Bronchopulmonary Sequestration | |
|---|---|---|
| Classification | Types 0-4, microcystic and macrocystic | Intralobar and extralobar |
| Connection to tracheobronchial tree | Yes | No |
| Systemic blood supply | No | Yes |
| Associated malformation | Common | Less common |
| Location | Either lower lobe | Left lower lobe |
| Malignant transformation | Yes | Yes |
| Spontaneous regression of antenatally diagnosed cases | 15% | 75% |
Type 0 CPAM is the rarest form and arises from the trachea or bronchus and contains multiple small cysts. These infants also have other associated lesions, including cardiovascular anomalies, renal hypoplasia, and focal dermal hypoplasia. The lung itself is hypoplastic, weighing only 30%-50% of the expected weight. Microscopically, the tissue consists almost entirely of irregular bronchial-like structures lined by pseudostratified ciliated columnar epithelium surrounded by thick cartilage plates and bundles of smooth muscle fibers. Usually all lobes of the lung are involved; thus, this diagnosis is usually lethal.
Type 1 CPAM is the most common form, representing 60%-70% of all CPAMs. These lesions consist of large cysts (1-10 cm) surrounded by multiple small cysts that arise from the distal bronchus or proximal bronchiole and are rarely associated with other congenital malformations. These CPAMs can be large and can have significant mass effect in utero, which can lead to fetal hydrops and pulmonary hypoplasia. However, many of these cysts collapse as pregnancy progresses, allowing normal lung growth of the unaffected lobes. Radiographically, they appear as either a single or multiple air-filled or air/fluid-filled cysts in one or (much less frequently) multiple lobes. Depending on size, there can be flattening of the diaphragm, mediastinal shift, and compression of adjacent lung. Overall, these lesions have a good prognosis. However, a number of reports describe the occurrence of a bronchioloalveolar carcinoma, especially when the CPAM is not fully resected, with a malignant transformation risk of 1%.
Type 2 CPAM accounts for 15% of all CPAMs and is the second most frequent type of CPAM. These malformations are of bronchiolar origin and consist of multiple small cysts (0.5-2 cm) and are often (≈50%) associated with other congenital anomalies, including renal agenesis/dysplasia, cardiovascular anomalies, congenital diaphragmatic hernia, and extralobar sequestrations. Radiographically, they are characterized by multiple small cysts that may not even be visible on chest x-ray. Prognosis is usually related to the severity of the associated anomalies.
Type 3 CPAM accounts for 5%-10% of all CPAMs. These are of bronchiolar/alveolar duct origin and almost exclusively seen in males. These lesions were the original congenital adenomatoid malformation described by Ch'in and Tang in 1949. They consist of multiple smaller cysts (rarely >0.2 cm) and appear as a solid mass that is associated with a significant risk of hydrops and polyhydramnios resulting from caval obstruction and cardiac compression secondary to mediastinal shift. This also leads to pulmonary hypoplasia, because unlike type 1 CPAMs, this lesion does not regress with progression of pregnancy. The extent of the pulmonary hypoplasia is the primary determinant of survival.
Finally, type 4 CPAM accounts for approximately 10% of CPAMs and is of distal acinar origin. The lesion primarily consists of large (up to 10 cm), air-filled, thin-walled cysts usually located at the lung periphery. This lesion can be asymptomatic at birth and presents from the neonatal period to 4 years of age. Often, this will be an incidental finding on an x-ray that was taken for other reasons, such as acute respiratory distress related to a tension pneumothorax, or pneumonia. This lesion can be confused with pleuropulmonary blastema; therefore, blastemas must be looked for histologically. Surgical resection of the lobe is accompanied by an excellent prognosis.
With improvement in prenatal imaging, most of these lesions are diagnosed prenatally, but some may not present until the postnatal period either as acute respiratory distress or as an incidental finding on a chest x-ray that was obtained for other reasons. Although the diagnosis of a congenital lung lesion is able to be made on prenatal ultrasound, it is difficult to distinguish CPAM from other cystic lung lesions. Adzick et al. proposed the classification of antenatal cystic lung lesions based on their appearance on ultrasound as either macrocystic (cysts ≥5 mm) or microcystic (cysts <5 mm).
Understanding of the natural history of CPAMs continues to evolve. Up to 15% of these lesions appear to “disappear” in the prenatal period, usually after 28 weeks’ gestation when the growth of these lesions tends to plateau; however, in most all cases, postnatal CT scan or fetal MRI will show persistence of the anomaly ( Fig. 66.3 ). The unpredictability of the in utero growth of these lesions requires careful follow-up. Midgestation, these lesions can grow quite rapidly, which can cause mediastinal shift, pulmonary hypoplasia, and impaired venous return leading to hydrops. It has been well established that the diagnosis of fetal hydrops associated with CPAM portends a poor prognosis, with mortality near 100%. In 2002, it was hypothesized that the volume of the lesion would predict whether hydrops would develop in the fetus. The CPAM volume ratio (CVR) was developed as a prognostic tool and is calculated by measuring the three dimensions of the lung lesion and dividing by the head circumference. A CVR greater than or equal to 1.6 at initial diagnosis was found to reliably predict a subgroup of fetuses at increased risk for developing fetal hydrops and is now used to evaluate which infants might benefit from in utero interventions. Fetal surgery using thoracoamniotic shunting or cyst aspiration has been successful for macrocystic lesions, with survival rates in hydropic fetuses of 50% and 69%, respectively. Microcystic lesions require open fetal surgery in the presence of hydrops with a survival rate of 52% (see Chapter 13 ). Patients who are not suitable for surgery benefit from antenatal steroids, which have been shown to decrease the size of microcystic CPAMs as well as resolve the hydrops in several case series. The ex utero intrapartum treatment (EXIT) procedure should be considered in patients with significant mediastinal shift and cardiac and lung compression at time of delivery. Recently, Ehrenberg-Buchner and colleagues report that the maximum and final CVR are independent predictors of respiratory morbidity at the time of delivery and need for neonatal intervention. According to their work, a maximum CVR >1 portends a 75% likelihood of having respiratory distress at birth; thus, these babies should be delivered at a tertiary care center. If the maximum CVR is <1, nearly 100% of those infants are asymptomatic at birth.

Postnatally, all CPAMs should have surgical evaluation. If the infant is symptomatic, surgical excision is indicated. However, there is still some debate over the appropriate management of those lesions that are asymptomatic at birth. All infants with CPAM should have a chest x-ray in the immediate neonatal period and chest CT scan at 4-6 weeks of age to evaluate the mass. There are justifications for prophylactic surgery: preventing chest infections and sepsis; preventing malignancy; early rather than delayed surgery may encourage compensatory lung growth; and reduction in postoperative complications (compared with emergency surgery). The majority opinion seems to favor elective resection at 2-6 months of age; however, this continues to be debated in the surgical arena. Following successful resection, the long-term functional outcome of children with CPAM is excellent, with no physical limitations or increased risk for infection.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here