Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Perinatal stroke is a vascular event causing focal interruption of blood supply and can be categorized based on the vascular distribution of stroke (arterial or venous), age at the time of stroke, and age at presentation.
When a stroke is suspected in a neonate, neuroimaging is required for confirmation of diagnosis, followed by risk factor assessment and creation of a specific treatment plan.
In infants and neonates, cerebral sinus venous thrombosis usually presents with seizures and/or encephalopathy, and treatment varies from conservative neuromonitoring to anticoagulation.
Subdural and subarachnoid hemorrhages are both associated with vacuum/forceps-assisted deliveries and coagulopathy. Evaluation includes neuroimaging and monitoring, and outcomes vary based on location and size.
Vein of Galen malformation is the most common arteriovenous malformation of the newborn, often presenting with cardiac and/or neurologic complications. The clinical picture depends on the age at presentation.
Ischemic perinatal strokes (IPS) are focal or multifocal arterial or venous infarctions occurring between 20 weeks’ gestation and 28 days’ postnatal life and are confirmed by neuroimaging or neuropathologic studies. The reported incidence varies between 1 in 1600 and 1 in 5000 live births, with likely higher incidence given that most of the studies were retrospective and magnetic resonance imaging (MRI) was not routinely used. The IPS is responsible for one-third of term and late-preterm children affected with hemiplegic cerebral palsy (CP). IPS is slightly more common in males and non-Hispanic black ethnicity when compared to whites and occurs most often in the left middle cerebral artery (MCA) distribution, with the most affected region being the left cerebral hemisphere.
Risk factors for perinatal stroke include maternal primiparity, preeclampsia, prolonged rupture of membranes, chorioamnionitis, and cord anomalies. Presence of more than one of these risk factors can increase the probability of perinatal stroke to 1 in 200. Complicated deliveries involving emergency cesarean section or instrumentation have also been associated with IPS. Table 56.1 includes multiple proposed risk factors for perinatal stroke, mostly from studies presenting associations rather than causation. Most cases lack definitive causes.
| Maternal/Placental | Fetal/Neonatal |
|---|---|
|
|
Ischemic perinatal stroke is pathological or neuroradiological evidence of focal arterial or venous infarction that occurred in the perinatal period. The pathogenesis of IPS is not well understood. Physiologic changes in the mother during pregnancy may cause a hypercoagulable and prothrombotic state. Fetuses are also at increased risk for developing clots as physiologic polycythemia leads to hyperviscosity, and there is a depressing anticoagulant activity present. These factors, coupled with the placenta having areas of reduced blood flow, increase the proclivity for thrombotic generation on the fetal side of the placenta. These thrombi will travel via the umbilical vein and are poised to pass through the patent foramen ovale to enter the systemic and, most importantly, the cerebral arteries. Other fetal conditions leading to increased risk of perinatal stroke include twin pregnancies, twin-to-twin transfusion, arteriovenous malformations, prolonged neck traction, and cardiac defects. Perinatal arterial stroke (PAS) lesions are usually singular (70%), involving the anterior circulation (71%), posterior circulation (7%), or both (20%). Strokes are most commonly left-sided (51% of all strokes, 73% of all anterior strokes), with 9% occurring on the right and 20% showing bilateral distribution.
Classification of perinatal strokes can be categorized based on the vascular distribution of stroke (arterial or venous), age at the time of stroke, and age at presentation, with multiple authors using different terms to describe the IPS ( Table 56.2 ). Because the timing of the vascular event leading to IPS is almost always unknown, it has been suggested that the classification of IPS be based on the gestational or postnatal age at diagnosis.
| Arterial Distribution | |||
|---|---|---|---|
| Age at Diagnosis | Terminology | Description | Typical Presentation |
| Fetal | Fetal Arterial Stroke | Arterial ischemic stroke found on prenatal imaging. | Incidental finding of diffusion restriction on fetal MRI. |
| Preterm | Perinatal/Neonatal Arterial Ischemic Stroke (P/NAIS) | Arterial ischemic stroke in infants < 35 weeks’ gestational age. | Incidental finding on cranial ultrasound; less likely to present with seizures. |
| Term | Perinatal/Neonatal Arterial Ischemic Stroke (P/NAIS) “Early PAS” |
Acute symptomatic neonatal arterial ischemic or fresh arterial strokes presenting in the first week of life. | Typically presents with seizures, apnea, or encephalopathy. |
| Term, 7–28 days | Perinatal/Neonatal Arterial Ischemic Stroke (P/NAIS) “Late PAS” |
Acute symptomatic neonatal arterial ischemic or fresh arterial stroke presenting during weeks 2–4 of life. | Typically presents with seizures or encephalopathy. |
| Infancy and/or later | Presumed Perinatal Ischemic Stroke (PPIS) | Arterial distribution stroke that presumably occurred between 20 weeks’ gestation and 28 days of life. | Normal neonatal course; present in infancy with hemiplegia, early handedness, or seizures. |
| Venous Distribution | |||
| Age at Diagnosis | Terminology | Description | Typical Presentation |
| Fetal | Fetal Venous Stroke | Venous distribution stroke is found in prenatal imaging, typically in the distribution of periventricular hemorrhagic infarction. | Incidental findings on fetal MRI or cranial ultrasound. |
| Preterm | Periventricular Hemorrhagic Infarction (PVHI) | Ischemic perinatal stroke in preterm infants often involves the periventricular region resulting in presumed venous infarction in infants born before 32 weeks. | Found on routine cranial ultrasound with associated intraventricular hemorrhage. Presentation ranges from subclinical to catastrophic. |
| Term | Cerebrosinovenous Thrombosis (CSVT) | Venous strokes in the term infant are secondary to partial or complete occlusion of a cranial venous sinus, deep cerebral vein, or smaller cortical veins. These may have a hemorrhagic component. Additionally, medullary vein thrombosis in term neonates may result in a periventricular infarct. | Can present with seizures; any term infant with intraventricular hemorrhage or temporal lobe hemorrhage should be evaluated further for CSVT. |
| Infancy and/or later | Presumed Perinatal Ischemic Stroke (PPIS) | “Fetal” stroke not diagnosed prenatally or asymptomatic perinatal stroke. PPIS often involves the periventricular region resulting in presumed venous infarction (PVI) in utero or in infants born prematurely. Criteria suggestive of PVI unilateral injury, focal periventricular encephalomalacia, internal capsule T2 prolongation, relative cortical sparing relative basal ganglia sparing, and remote hemorrhage. Medullary vein thrombosis is an additional venous etiology. |
Typically present in infancy with hemiplegia/early handedness or seizures. |
Diagnosing an infant with perinatal stroke is challenging in the newborn period. Most infants with PAS are asymptomatic at birth, and signs of acute illness are only seen in 25% of cases. Diffuse neurologic signs and symptoms are more common than focal signs, with the abnormal tone, apnea, and depressed level of consciousness more common than hemiparesis, which is usually absent or subtle in the neonate. Nonspecific symptoms include breathing and feeding difficulty. In the week following birth, most newborns with perinatal arterial ischemic stroke (PAIS) become symptomatic, with the most prevalent symptom being seizures (large range, up to 70% to 90%). Approximately 12% of infants with PAS present with recurrent focal seizures with typical onset at 12 to 48 hours of age. Typical presentation for different forms of perinatal stroke is presented in Table 56.2 .
Assessment of the neonate with perinatal stroke includes neuroimaging (cranial ultrasound [CUS], head computed tomography [CT], brain MRI), electroencephalography, and echocardiogram to evaluate for congenital heart disease or intracardiac thrombus. Risk factors include a maternal history of autoimmune disorders, recurrent pregnancy loss, or thrombosis, and placental pathological examination and toxicology screens may also provide helpful information. Given that the most common presenting symptom is seizures, the work-up should begin with ruling out other etiologies of seizure such as hypoglycemia, hypocalcemia, electrolyte disorders, infection, and metabolic syndromes. In nonhemorrhagic stroke, MRI is considerably more sensitive than CUS or CT, making MRI the preferred imaging modality. MRI is the gold standard test for the detection of PAS. MRI sequences to be used include T1- and T2-weighted images, diffusion-weighted imaging (DWI), and MRI angiography, a technique that is helpful in detecting vascular stenosis, occlusion, or arteriopathy. Fig. 56.1 demonstrates an MRI of a term male with a PAS involving the right MCA. Of these sequences, the most sensitive is DWI performed in the first week of life. The area of infarction appears as a zone of high intensity on DWI and low intensity on the apparent diffusion coefficient (ADC) map. After the first week, the ischemic tissue appears to normalize, even though the area continues to be ischemic, a process known as pseudonormalization. Reduced contrast between cortex and white matter can be seen in the first 48 hours on T2-weighted imaging. Lower signal intensity will be seen on T1-weighted images. As time passes from the injury, cortical highlighting develops.
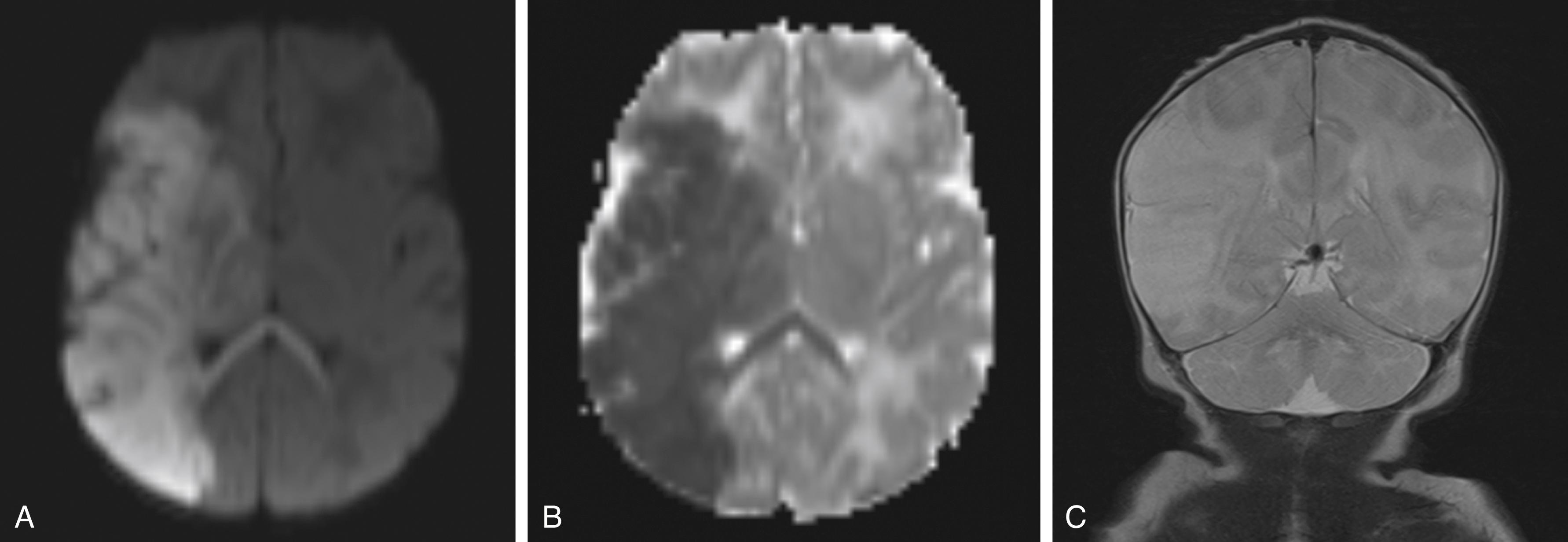
When a perinatal stroke is associated with a seizure, electroencephalography (EEG) could help localize the origin of the seizures and aid in the management of seizures. EEG is usually abnormal in infants with PAS, and the findings correlate with the location of infarction. Cranial US is widely available in most centers that do not have MRI capability. The sensitivity of CUS improves from 68% in the first few days to 87% in the first week. Because strokes may occur in the posterior circulation, posterior fontanelle imaging is necessary. Unlike MRI, CUS is operator dependent, and thus detection rates vary between centers. The use of CT is limited to situations when CUS and MRI are not available. CT is discouraged in neonates because of poor sensitivity and high radiation exposure.
The optimal thrombosis evaluation is currently debated, and laboratory results are usually inconclusive in neonates. Rates of possible thrombophilia as an underlying cause of PAIS vary in the literature up to as high as 68%. However, the International Pediatric Stroke Study found that only 19% of PAIS infants were diagnosed as having possible increased lipoprotein(a) level, methylene tetrahydrofolate reductase mutations, elevated β 2 -glycoprotein level, factor V Leiden, prothrombin gene 20210 A, low antithrombin III level, antiphospholipid antibodies, plasminogen activator inhibitor, or low protein S level. Routine thrombophilia testing of the neonate is not indicated without clinical suspicion.
Therapy for PAS is largely supportive and includes neuroprotective measures with avoidance of dehydration, hypoglycemia or hyponatremia, and maintenance of normothermia. Anticonvulsant therapy has the best evidence for use, but most infants will not require long-term therapy, and many experts increasingly suggest early discontinuation. Anticoagulation is currently debated in the literature, and the type of antithrombotic medication used varies from country to country. There is most consensus for the use of anticoagulant therapy in infants with congenital heart disease (CHD) and PAIS, as they are at increased risk of ongoing clot formation. Anticoagulation treatment should be considered in proven cardiac embolism, recurrent AIS, or prothrombotic state. Referral to physical, occupational, and speech therapy should be considered, as well as close follow-up after discharge.
In infants with acute PAS, EEG is a useful tool for prognostication. Specifically, abnormal background activity between 2 and 4 days of life can predict abnormal motor outcomes. Abnormal physical exams at discharge and seizures during the neonatal period are also predictive of long-term disability. Neurodevelopmental outcomes depend on the size and location of the stroke. The risk of recurrence and mortality in PAIS is very low. PAIS occurring in the MCA distribution may result in a hemiplegia rate of 50%. While most infants will be discharged seizure-free, up to 50% will later develop epilepsy or infantile spasms. Occurrence of more than ten neonatal seizures increases the risk of subsequent epilepsy 30 - fold. Cognitive and language deficits are more common in children with hemiplegia or epilepsy following PAS. Neuromotor impairment and hemiplegia are associated with PAS affecting basal ganglia, thalamus, or posterior limb of the internal capsule. In acute settings, motor outcomes may be predicted by the diffusion restriction within the corticospinal tract, while their asymmetry on MRI predicts the development of spastic hemiplegia. Language is affected, with about half of the cases having delays at 7 years. Visual function may be altered by PAS, and behavioral problems may also be seen.
Cerebral sinus venous thrombosis (CSVT) has a reported incidence of 0.6 to 12 per 100,000 live births. The wide range of reported incidence is likely due to variable awareness among clinicians and, therefore, variable use of neuroimaging to detect CSVT. The most commonly affected sinuses in neonates are the superior sagittal and the transverse sinuses. The transverse sinuses are more frequently involved in children older than 2 years of age (60% vs. 39%).
Impaired venous drainage from CSVT causes increased venous pressure that can result in increased capillary hydrostatic pressure. This elevated pressure leads to vasogenic edema and hemorrhagic infarction in the distribution of the cerebral sinus venous. When venous pressure is higher than arterial pressure, arterial flow decreases, and arterial ischemia and hemorrhagic infarction may occur. Maternal risk factors for CSVT include preeclampsia, chorioamnionitis, and gestational diabetes. Complicated delivery, meconium aspiration, and the need to be intubated have also been associated with CSVT. Similar to PAS, CHD is a major risk factor. Postnatal conditions such as meningitis, sepsis, dehydration, and ECLS are associated with CSVT.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here